Abstract
Chronic low-grade infection has been suggested to be associated with metabolic disorder such as diabetes. However, the molecular mechanism underlying this important association is largely unknown. The only clue established so far is that many subjects exhibit elevated levels of C-reactive protein as measured by highly sensitive assay. Here, we hypothesized that adipocyte–macrophage interaction plays a key role in amplifying such low grade infection to the level of influencing metabolic disorders. The presence of macrophages in abdominal adipose tissues was investigated by immunohistochemistry. To see whether molecules associated with acute phase protein, LPS signaling, and persistent recruitment of monocytes, are produced at higher amounts in adipocytes co-cultured with macrophages stimulated with low concentration of LPS (1 ng/ml), we measured serum amyloid A (SAA), LPS binding protein (LBP), soluble CD14 (sCD14), and RANTES levels in culture supernatant of co-cultures. Lastly, we investigated in vivo effect of low-grade LPS infusion on the production of these molecules using obese model mice. The macrophages were certainly identified in abdominal adipose tissues. Investigated molecules, especially LBP, SAA, and RANTES were produced at higher amounts in co-cultures stimulated with LPS compared with the cells without LPS. The ob/ob, and high-fat diet-induced obesity mice produced higher amounts of LBP, SAA, and RANTES one day after LPS infusion (1 ng/ml/g body weight) compared with ob/– and normal-fat fed control mice. Thus, adipocytes and infiltrated macrophages, and their interaction with low endotoxin stimulation appear to play an important role in amplifying and maintaining LPS-induced low-grade inflammation.
Keywords
Introduction
Chronic low-grade infections such as gut infection and/or periodontal disease have been suggested to play some important roles in the pathogenesis of metabolic disorders such as diabetes and subsequent cardiovascular risk.1,2 However, it is still unclear as to why such local low-grade infection-induced inflammation is amplified to the level of influencing systemic metabolic disorders. The only well-established clue so far is that most of these subjects exhibit elevated levels of the systemic acute phase inflammatory marker C-reactive protein as measured by highly sensitive assay (hsCRP). 3 In fact, elevated levels of hsCRP, although often for the most part in a healthy reference range, have been suggested to associate with the development of both diabetes and cardiovascular disorders.4,5 C-Reactive protein is generally believed to be produced by hepatocytes in response to IL-6. 6 Therefore, at least sufficient amount of IL-6 has to be produced as a consequence of these low-grade infections nearby the liver.
Recently, macrophages have been suggested to infiltrate into adipose tissue, to interact with adipocytes, thereby producing higher amounts of adipokines.7,8 In addition, free fatty acid (FFA) has been found to stimulate both cell types via TLR4. 9 However, TLR4 is also an important receptor for bacterial LPS, endotoxin. We previously established a co-culture system between adipocytes and macrophages, and examined IL-6 production when these cells were stimulated with low concentration of LPS (1 ng/ml). 10 We found that cultured adipocytes produced markedly elevated level of IL-6 (more than 100-fold elevation) as well as monocyte chemo-attractant protein-1 (MCP-1) when co-cultured with macrophages in the presence of LPS than co-cultured cells without LPS, and suggested that these phenomena might account for elevated levels of CRP in subjects with low-grade infectious diseases, as IL-6 may enter the liver via portal vein to stimulate hepatocytes to produce CRP. 11 Enhanced production of MCP-1 may further recruit macrophages into adipose tissues, as suggested by others. 12
We then analyzed and compared the gene expression profile in adipocytes co-cultured with macrophages in the presence or absence of low dose LPS (1 ng/ml) and found expression of many important genes associated with insulin resistance. Cardiovascular risk was impaired in LPS-treated adipocytes, such as elevated level of suppressor of cytokine signaling gene, plasminogen activator inhibitor-1 gene, and suppression of GLUT-4 gene expression. 11 However, since the suggested low-grade infectious diseases such as periodontal disease and/or gut infections are chronic conditions, we hypothesized that there must be a system amplifying persistent inflammation by Gram-negative bacterial infection, and ‘adipocyte–macrophage interaction’ may play a key role in this amplification. Especially, in cases of periodontal inflammation, where obesity, even the over-weight condition, appears to augment further the risk of severe periodontitis, we hypothesized adipose tissue might play an important role in amplifying low-grade infection-induced mild inflammation to the systemic level. 12 In this study, therefore, we wished to see the effects of ‘adipocyte–macrophage interaction’ on enhancing LPS-mediated signals and on inducing persistent recruitment of monocytes as well as enhanced production of inflammatory markers.
Materials and methods
Immunohistochemistry
In order to detect macrophages in adipose tissues of elderly human subjects, immunohistochemical analyses were performed with anti-human CD68 antibody (PG-M1; 1:100, DAKO, Glostrup, Denmark) using the ChemMate EnVision Detection Kit (DAKO) according to the manufacturer’s protocol, after deparaffinization and pretreatment with citric acid buffer (pH 6.0) for target retrieval. Biopsy samples were obtained from mesenteric adipose tissues of an autopsy case of malignant pleural mesothelioma (68-year-old, man) in agreement with the bereaved family. This was performed in Hyogo College of Medicine and the protocol was approved by Institutional Review Board. Before any autopsy, written informed consent was obtained from the bereaved family.
Cell culture
Mouse 3T3-L1 pre-adipocytes and mouse macrophage cell line RAW264.7 were used. Both cell lines were obtained from American Type Culture Collection (ATCC, Manassas, VA, USA). Pre-adipocytes were differentiated into mature adipocytes as described. 10 Co-culture was performed by using the Transwell system (Corning Inc., Acton, MA, USA) with 0.4 µm porous membrane to separate upper and lower chamber. Differentiated 3T3-L1 cells (1 × 105) were cultured in the lower chamber, while 5 × 104 RAW cells were cultured in the upper chamber as described in our previous study.
Measurement of secreted protein
We previously performed gene expression analyses in 3T3-L1 adipocytes co-cultured with RAW macrophages with or without LPS stimulation, and our previous study demonstrated enhanced SAA gene expression in adipocytes with LPS stimulation as compared with the cells without LPS stimulation. 11 Additionally, we also observed enhanced gene expression associated with LPS signaling such as LBP, CD14, TLR2, and TLR4. 11 Furthermore, we also found markedly enhanced gene expression encoding RANTES, another important chemokine for monocytes. 11 Therefore, in this study, we looked for SAA, LBP, sCD14, and RANTES as secreted and/or shed proteins. We measured the concentration of these proteins in culture supernatants of co-cultures. The cells were stimulated with 1 ng/ml of Escherichia coli LPS for 4, 8, 12 and 24 h, and culture supernatant was collected at each time point. The SAA, LBP, sCD14 concentrations in the culture supernatants were measured by using commercial immunoassay (mouse SAA, LBP, sCD14, and RANTES ELISA kits; Endogen Inc., Woburn, MA, USA). As a control, each cell was cultured independently.
Preparation of model animals
Eleven-week-old ob/ob (B6.V-Lepob/J) mice and ob/– mice as control mice were used as the genetically obese model. As diet-induced obesity model, high fat-diet-induced obesity mice (C57BL/6J) and normal-fat fed control mice were prepared. Both mice lines were generated in a commercial laboratory (Charles River Japan, Shizuoka, Japan). For LPS infusion, we injected 50 ng E. coli LPS into circulation of ob/ob mice and its control via tail vein, while 30 ng LPS was used for high-fat diet induced obese model mice and its control. The average weight of 11-week-old ob/ob mice and high-fat diet-induced obese mice was usually 50 g and 30 g, respectively, and we employed these concentrations so that the final LPS concentration in these model animals would be 1 ng/ml/g body weight. All animals were handled in strict accordance with good animal practice as defined by the guideline of Ministry of Education, Culture, Sports, Science and Technology of Japan, and the protocol was approved by Hiroshima University Ethical Committee on Animal Experiments.
Measurement of serum proteins
Mice were sacrificed 1, 2, and 4 d after LPS injection. Blood samples were taken by heart puncture from each mouse including LPS untreated mice. Serum was separated by centrifugation, and LBP, SAA, and RANTES concentrations were measured by ELISA as described above.
Statistical analysis
Statistical analyses for determining significant differences in the production of each cytokine, according to culture conditions, and the serum cytokine concentration, according to LPS injection or not, were performed using the Student t-test.
Results
Identification of macrophages in human adipose tissue
Since previous studies including ours had suggested that macrophages infiltrated into adipose tissue plays an important role in amplifying low-grade inflammatory reactions, we first wished to confirm the presence of macrophages in biopsy sample of abdominal visceral, but not subcutaneous fat tissues in human. The result is shown in Figure 1. We confirmed the presence of CD68-positive macrophages in adipose tissues.
Immunohistochemical observation of macrophages in adipose tissue section. CD68 positive cells were stained as described in Materials and methods. Positively stained macrophages are indicated by the arrow.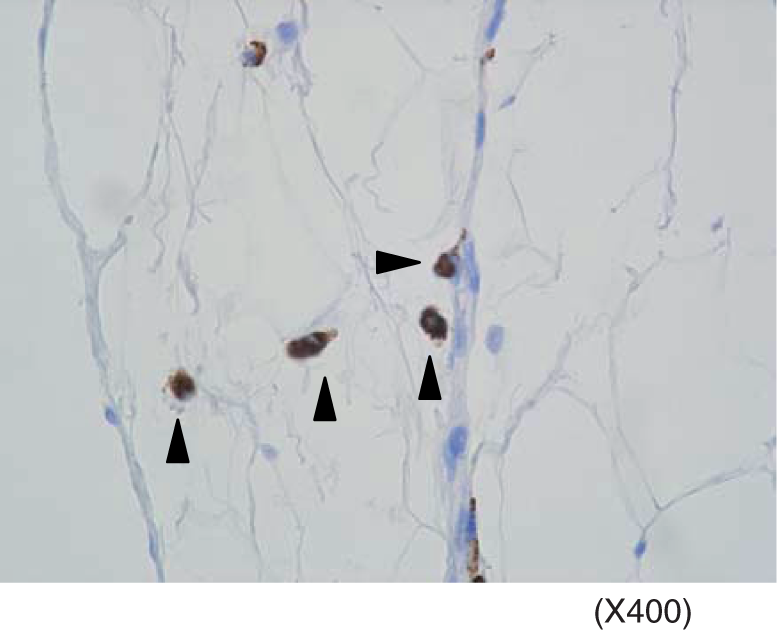
Adipocytes secrete higher amount of inflammatory marker, SAA when co-cultured with macrophages in the presence of LPS
Since our previous study demonstrated enhanced gene expression of SAA in adipocytes co-cultured with macrophages stimulated by LPS than the cells without LPS, and its relative expression was 16.1, 22.8, 19.7 and 38.1 times higher at 4, 8, 12 and 24 h after LPS stimulation, we examined SAA protein expression as a representative of inflammatory markers in co-cultures with or without LPS, and in each cell culture. The results are shown in Figure 2. As expected, major source of SAA appeared to be adipocytes, as RAW cells produced very low amounts of SAA regardless of the presence or absence of LPS even after 24 h culture. In co-cultures stimulated with 1 ng/ml of LPS, SAA production was higher than adipocyte culture alone as well as co-cultures without LPS, indicating that SAA production is up-regulated with low LPS stimulation in adipose tissues with macrophage infiltration.
Serum amyloid A production in co-culture of 3T3-L1 adipocytes and RAW macrophages, 3T3-L1 adipocytes, and RAW cells. SAA production was measured in co-cultured cells (left), 3T3-L1 adipocytes,(right), and RAW macrophages (right). Closed box indicates the data with cells without LPS stimulation, while open box indicates the data with the cells stimulate by LPS. *P < 0.05 by Student t-test.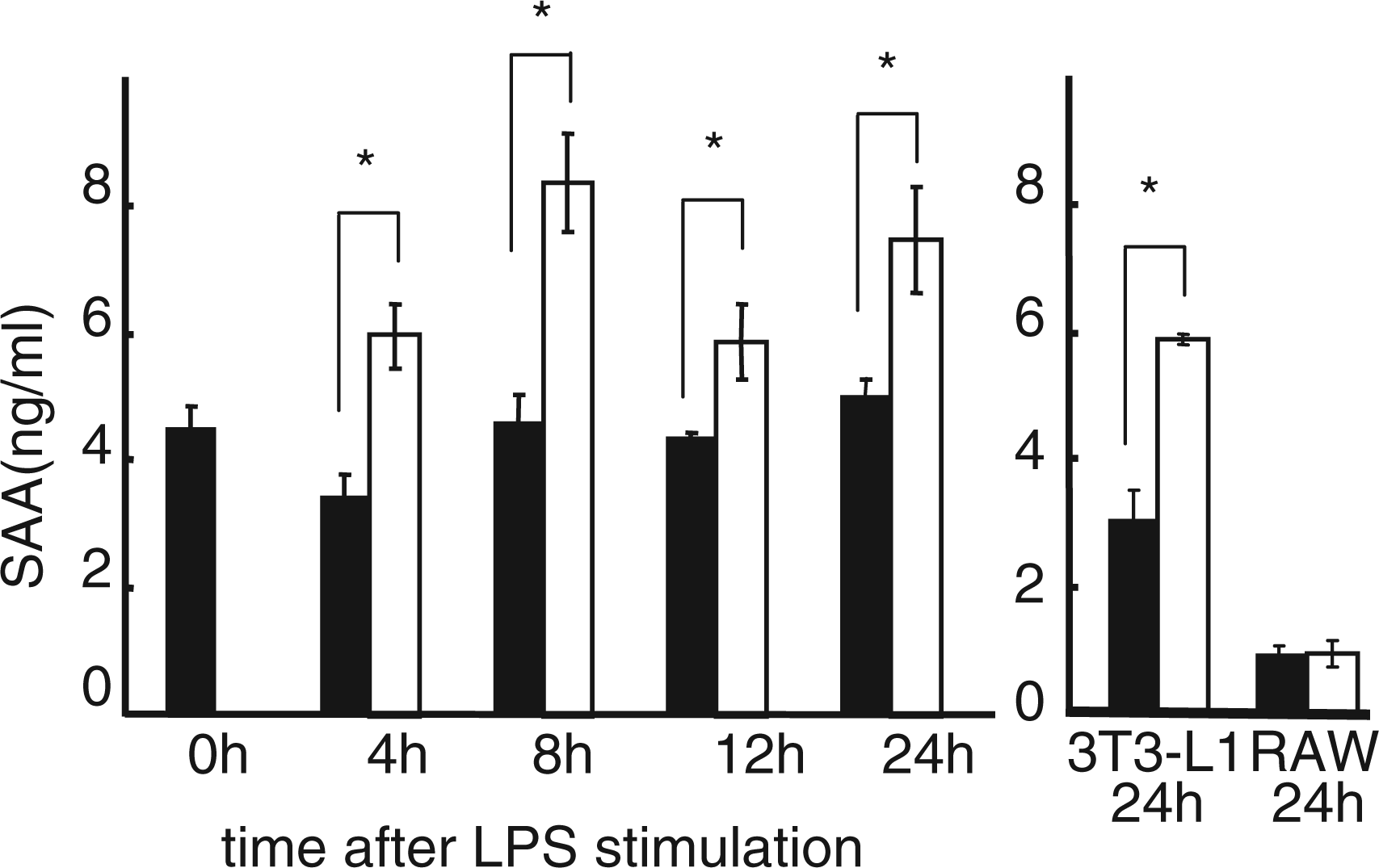
Adipocytes secrete higher amounts of LBP and sCD14 when co-cultured with macrophages in the presence of LPS
It is well-known that LPS signaling requires co-effector molecules such as LPS binding proteins and/or CD14 molecules for transducing signals into the cells via TLR4, and our previous study demonstrated elevated gene expression encoding LBP, CD14, and TLR2 in adipocytes co-cultured with macrophages in the presence of LPS than the cells without LPS (6.0, 11.0, 20.0, and 16.6 times higher expression at 4, 8, 12, and 24 h after LPS stimulation for LBP; 3.9, 3.5, 5.6, 7.7 times higher expression for CD14; 12.6, 10.8, 8.4, 3.3 times higher expression for TLR2). Therefore, we next investigated protein expression of LBP and sCD14 in co-cultured cells and in each cell culture with or without LPS. The results are summarised in Figure 3. As demonstrated, adipocytes produced markedly elevated amounts of LBP when co-cultured with macrophages in the presence of LPS. We also tried to detect sCD14 in culture supernatants. Although in most cases, the amounts of sCD14 were below detection limit, we could detect elevated concentration of sCD14 in co-cultures after 24 h stimulation with LPS. Since sCD14 is a shed form of membrane bound CD14, we speculate that detectable amounts of sCD14 are only released into culture supernatants of co-cultures in 24 h after LPS stimulation.
Lipopolysaccharide-binding protein (A) and sCD14 (B) production in co-culture of 3T3-L1 adipocytes and RAW macrophages, 3T3-L1 adipocytes, and RAW cells. (A) LBP production was measured in co-cultured cells (left), 3T3-L1 adipocytes,(right), and RAW macrophages (right). (B) sCD14 was only detected in co-cultures 24 h after LPS stimulation. Closed box indicates the data with cells without LPS stimulation, while open box indicates the data with the cells stimulate by LPS. *P < 0.05 by Student t-test.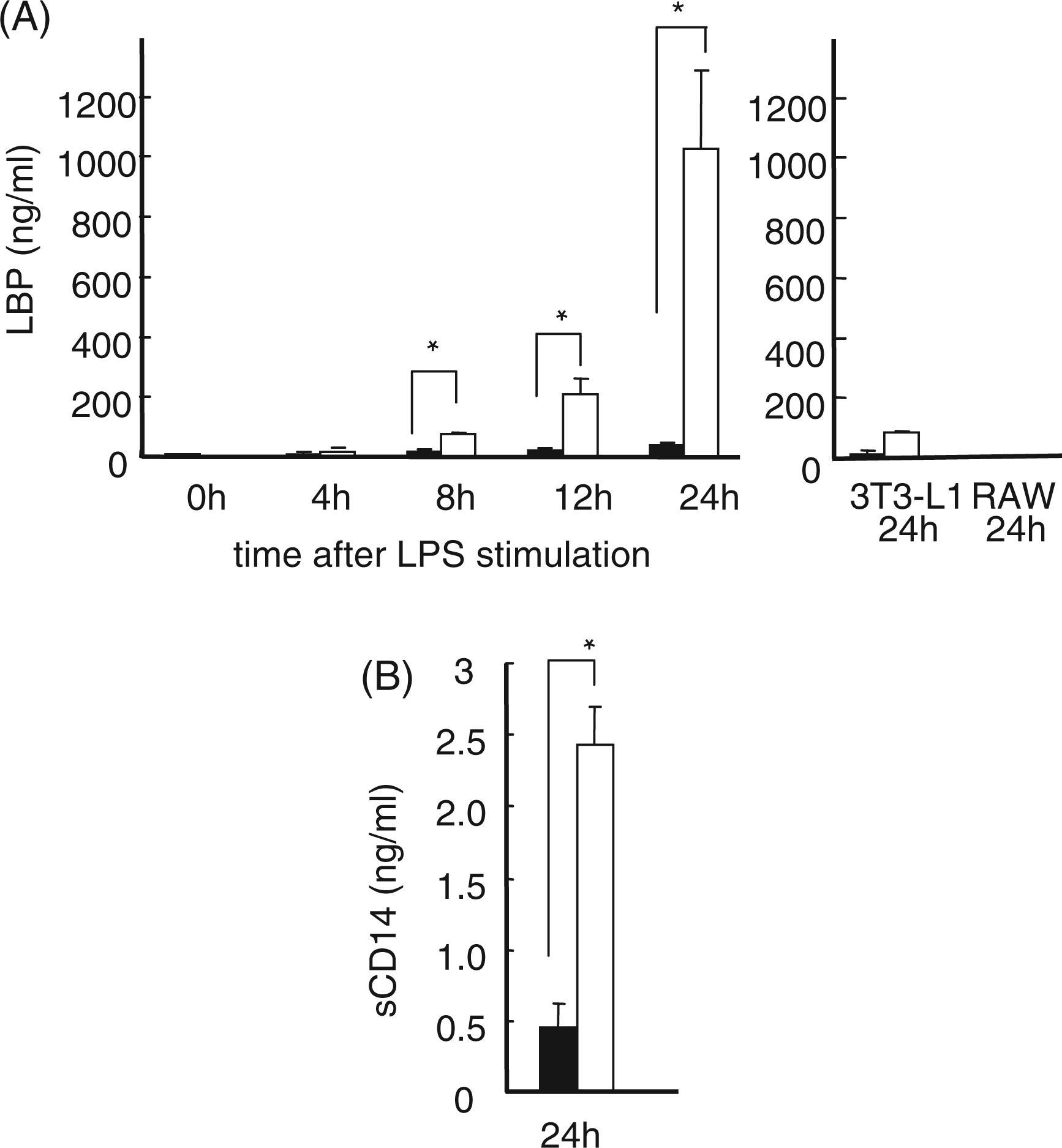
Adipocytes secrete higher amounts of RANTES when co-cultured with macrophages in the presence of LPS
It has been demonstrated that both macrophage infiltration and the presence of TLR4 ligands are essential for enhancing adipose tissue-originated inflammation to the systemically detectable levels. In this connection, we previously reported that MCP-1 expression was up-regulated in adipocytes co-cultured with macrophages in the presence of LPS. Besides MCP-1, we also noted marked up-regulation of the genes encoding RANTES in adipocytes co-cultured with macrophages in the presence of LPS than the cells without LPS stimulation (32.4, 123.2, 155.4, and 36.9 times higher expression at 4, 8, 12, and 24 h after LPS stimulation). Therefore, we next measured the time course RANTES production in co-cultures as well as each cell culture. The results are shown in Figure 4. Unlike IL-6, MCP-1, SAA, and LBP, RANTES appeared to be produced by both adipocytes and macrophages. However, clearly, co-cultures up-regulated RANTES production in the presence of LPS. This up-regulation was not observed in co-cultures without LPS.
RANTES production in co-culture of 3T3-L1 adipocytes and RAW macrophages, 3T3-L1 adipocytes, and RAW cells. RANTES production was measured in co-cultured cells (left), 3T3-L1 adipocytes,(right), and RAW macrophages (right). Closed box indicates the data with cells without LPS stimulation, while open box indicates the data with the cells stimulate by LPS. *P < 0.05 by Student t-test.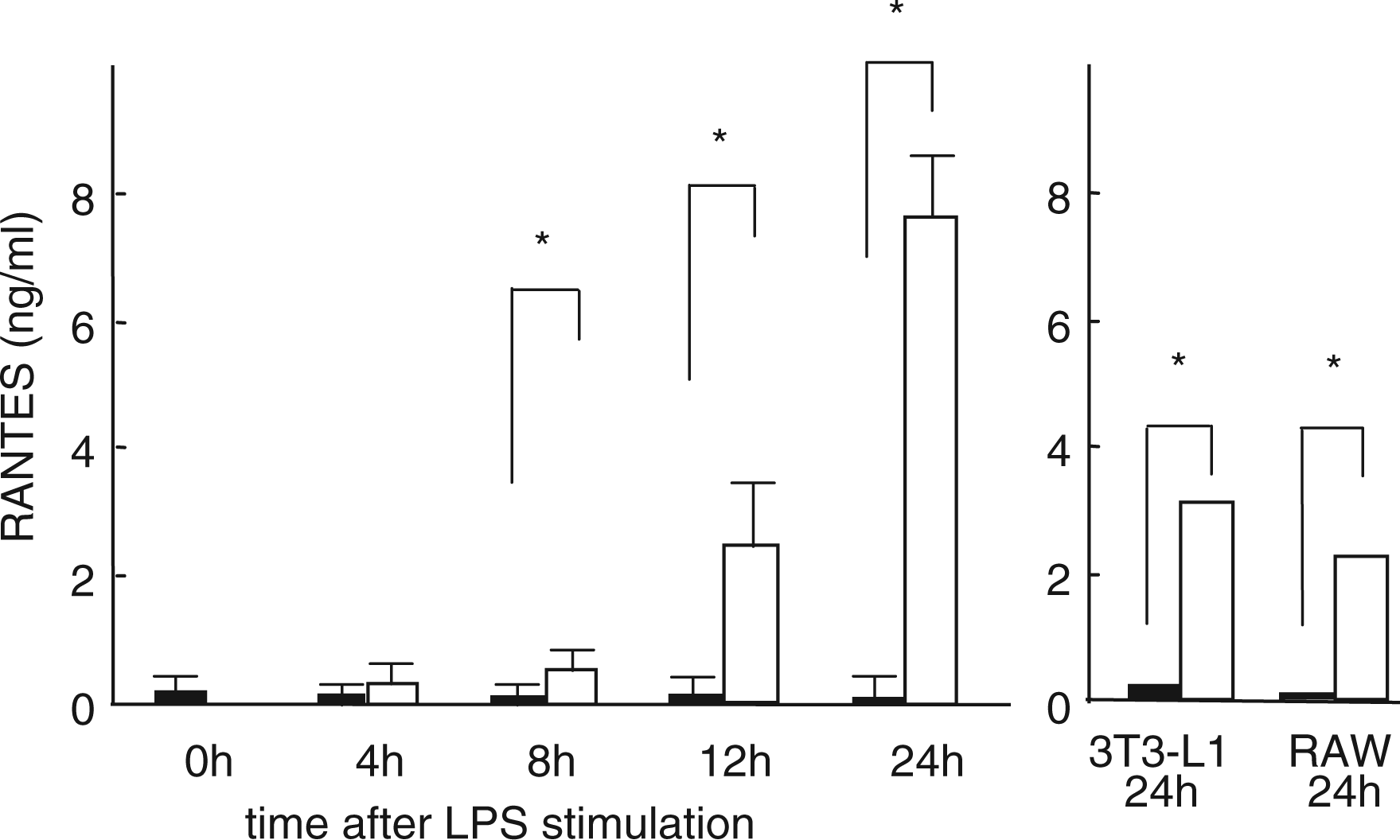
Obese model mice produce higher amounts of LBP, SAA, and RANTES by low-dose LPS challenge
Finally, we wished to confirm our current in vitro data in in vivo situation by using animal models. We used two lines of obese model mice, genetically obese mice (ob/ob and its control, ob/–) and diet-induced acquired obese model mice (high fat diet-induced obese mice and control normal fat-fed mice). The results are shown in Figure 5. In both models, low dose single LPS injection up-regulated circulating LBP, SAA, and RANTES protein concentration one day after LPS injection. This up-regulation was significantly higher than that of control mice. Since our LPS infusion was transient, all cytokine levels returned to basal level 2 d after LPS infusion.
Lipopolysaccharide-binding protein (A and B), SAA (C and D), and RANTES (E and F) concentration in genetically obese mice (A, C, and E) and diet-induced obese mice (B, D, and F) after LPS injection. Sera were obtained 0, 1, 2, 4 d after LPS injection as described in Materials and methods. Solid line indicates the data with obese (either ob/ob or high fat-induced obese) mice, while dotted line indicates the data with control normal weight (ob/– or normal diet-fed) mice. *P < 0.05 by Student t-test.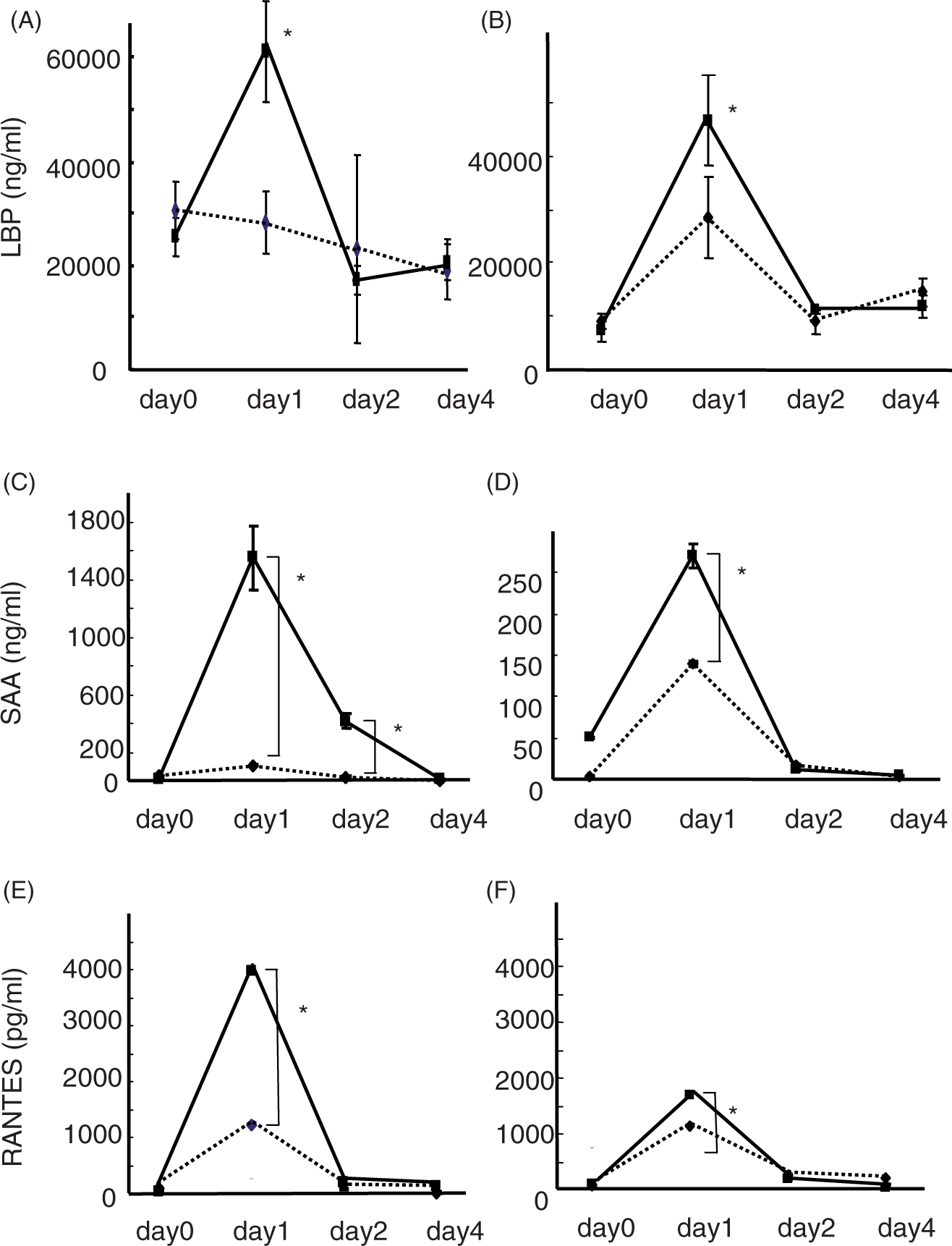
Discussion
It is well-established that mild elevation of CRP predicts the future development of myocardial infarction.4,5,13 C-Reactive protein is known to be produced by hepatocytes in response to IL-6. 6 In this connection, we previously reported that cultured adipocytes when co-cultured with macrophages produce markedly elevated IL-6 in response to low-dose bacterial endotoxin. 10 These results strongly indicate that IL-6 produced by ‘adipocyte–macrophage interaction’ through LPS signals enters the liver via portal vein and stimulates hepatocytes to synthesize CRP. In fact, we also confirmed in the current study the presence of CD68-positive macrophages in aged human abdominal adipose tissues by immunohistochemistry. Although several previous studies have shown the presence of macrophages in human subcutaneous adipose tissues,14,15 very few studies actually confirmed the presence of macrophages in human visceral fat tissues. In our previous study, markedly elevated MCP-1 production by adipocytes co-cultured with macrophages in the presence of LPS was also noted. 10 Elevated MCP-1 may further recruit monocyte/macrophage lineage into adipose tissues. 16 In this study, we also confirmed markedly elevated RANTES production both in vitro and in vivo study using co-culture system and obese model animals, both of which are challenged by bacterial endotoxin. RANTES has been suggested to recruit monocytes as well as T-lymphocytes.17,18 It also promotes adherence of monocytes to endothelial cells. 19 Taken together, RANTES may also support the recruitment of monocytes into adipose tissues. Interestingly, a recent study suggested the presence of T-lymphocytes in adipose tissues, 20 – 22 and RANTES may also play a role in recruiting T lymphocytes into adipose tissues.
Besides RANTES, it appears that adipocytes are a major source of both LBP and SAA based on the current study. As far as we know, this is the first report demonstrating the production of LBP and CD14 by adipocytes. Lipopolysaccharide-binding protein is an essential co-molecule transducing LPS signals into the cell. 23 Although CD14 and its soluble form (sCD14) may not be essential components for LPS signaling, previous study had clearly shown that the presences of these molecules further augments LPS signaling. 23 Therefore, elevated LBP and sCD14 may be one of the important phenomena accounting for persistent low-grade inflammation in subjects with low-level Gram-negative bacterial infection. Although precise serum LPS concentration in such subjects with severe periodontitis and/or gut infection is not well-confirmed yet due possibly to the existence of lipoproteins bound to LPS, previous study has indicated that, in case of severe periodontitis subjects, serum LPS concentration appeared to be almost 0.87–1.0 ng/ml. 24
The SAA is known to be one of the acute phase proteins like CRP. 6 In rodent models, it is well known that CRP is not as sensitive a marker as SAA, while in humans both CRP and SAA act as sensitive markers of acute inflammation. In the rodent model, we found that SAA is produced at higher amounts in obese mice, as adipose tissue itself appears to produce higher amounts of SAA when stimulated by LPS with increased macrophage infiltration as demonstrated in this study. Serum amyloid A has been suggested to be associated with enhanced lipolysis as well as reduced nitric oxide synthesis.25,26 A recent report indicated significant association between clinical severity of periodontitis and serum triglyceride level. 27 Furthermore, as mentioned above, obesity and/or overweight are associated with periodontitis. 12 Triglycerides are mainly synthesized in the liver from FFA following lipolysis. Thus, observed high triglycerides in severe periodontitis subjects may also indicate enhanced lipolysis in such subjects.27,28 Interestingly, elevated SAA in association with periodontitis has recently been reported, although this report did not indicate the source of SAA in such periodontitis patients. 29
Conclusions
This study suggests that ‘adipocyte–macrophage interaction’ may mediate LPS-induced low-grade inflammation and further up-regulates inflammatory responses to the systemic level. The hypothetical role of the molecules closed up in our current study is summarized in Figure 6. We believe this is an important underlying mechanism as to why local, low-grade infection is up-regulated to the level of systemic low-grade inflammation.
Schematic illustration of the roles of each molecule in enhanced inflammatory changes in adipose tissues stimulated by LPS. Lipopolysaccharide from gut infection or periodontal infection activates adipose tissues to produce higher amounts of LBP, sCD14, which act to amplify inflammatory stimuli. RANTES and MCP-1 may further recruit macrophages into adipose tissues. Inflammatory markers such as CRP and SAA are also produced at high level. Serum amyloid A is directly produced by adipocytes. ALL these changes contribute to prolong low-grade inflammation.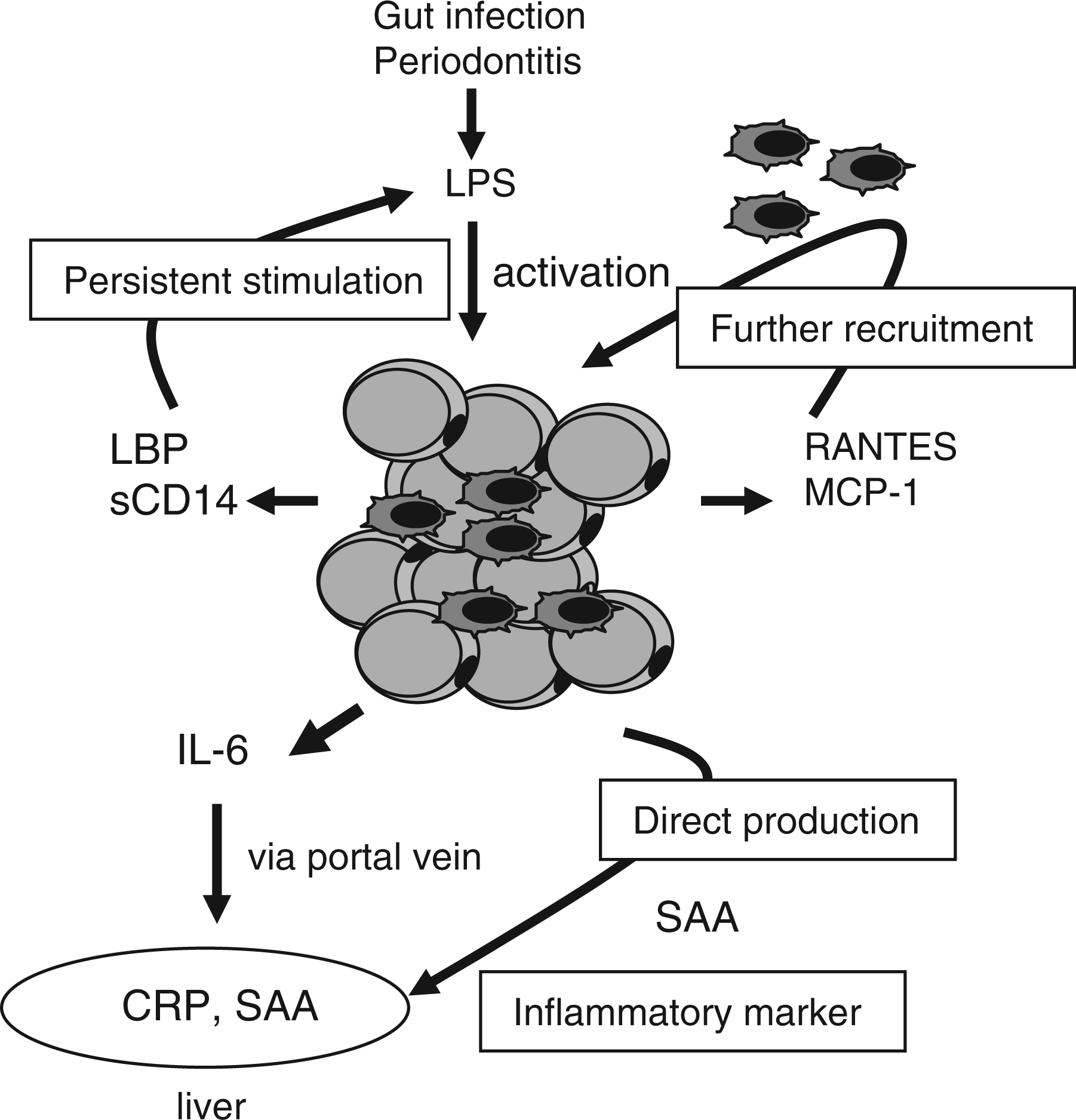
Footnotes
Acknowledgements
This work was supported, in part, by a Grant-in-Aid (No. 21390556, 22792086, 22390401) from the Japan Society for the Promotion of Science and from the Academic Frontier Project for Private Universities: matching fund subsidy from the Ministry of Education, Culture, Sports, Science and Technology, 2007–2011.
The authors declare there is no conflict of interest associated with this study.
