Abstract
The Berrettini branch is at risk of iatrogenic injury during carpal tunnel surgery. We describe a systematic ultrasound method to visualize this branch, ensuring its preservation during ultrasound-guided percutaneous release.
Keywords
The Berrettini branch is a superficial palmar communicating nerve between the median and ulnar nerves (Ferrari and Gilbert, 1991). With a prevalence of 81–100% in anatomical dissections it is considered a permanent connection and not an anatomic variation. Ferrari and Gilbert (1991) have classified it into four types (Figure 1). Type III, the most frequent configuration according to Stančić et al. (1999), is distal to the transverse carpal ligament (TCL), with a proximodistal oblique trajectory from the ulnar nerve to the third common digital nerve of the median nerve. Type II Berrettini branch is parallel and very close (<4 mm) to the distal margin of the TCL, and thus at risk of iatrogenic injury during carpal tunnel release (CTR). Type I starts crossing over the TCL following an oblique trajectory and may be at risk if the TCL incision is performed too close to the hook of hamate or in case of a hypoplastic hook of hamate. Transection of the Berrettini branch results in disabling neuropathic pain with dysesthesia affecting the ulnar side of the middle finger and the radial half of the ring finger (Ferrari and Gilbert, 1991; Stančić et al., 1999). The complication is underdiagnosed and frequently confused with complex regional pain syndrome.
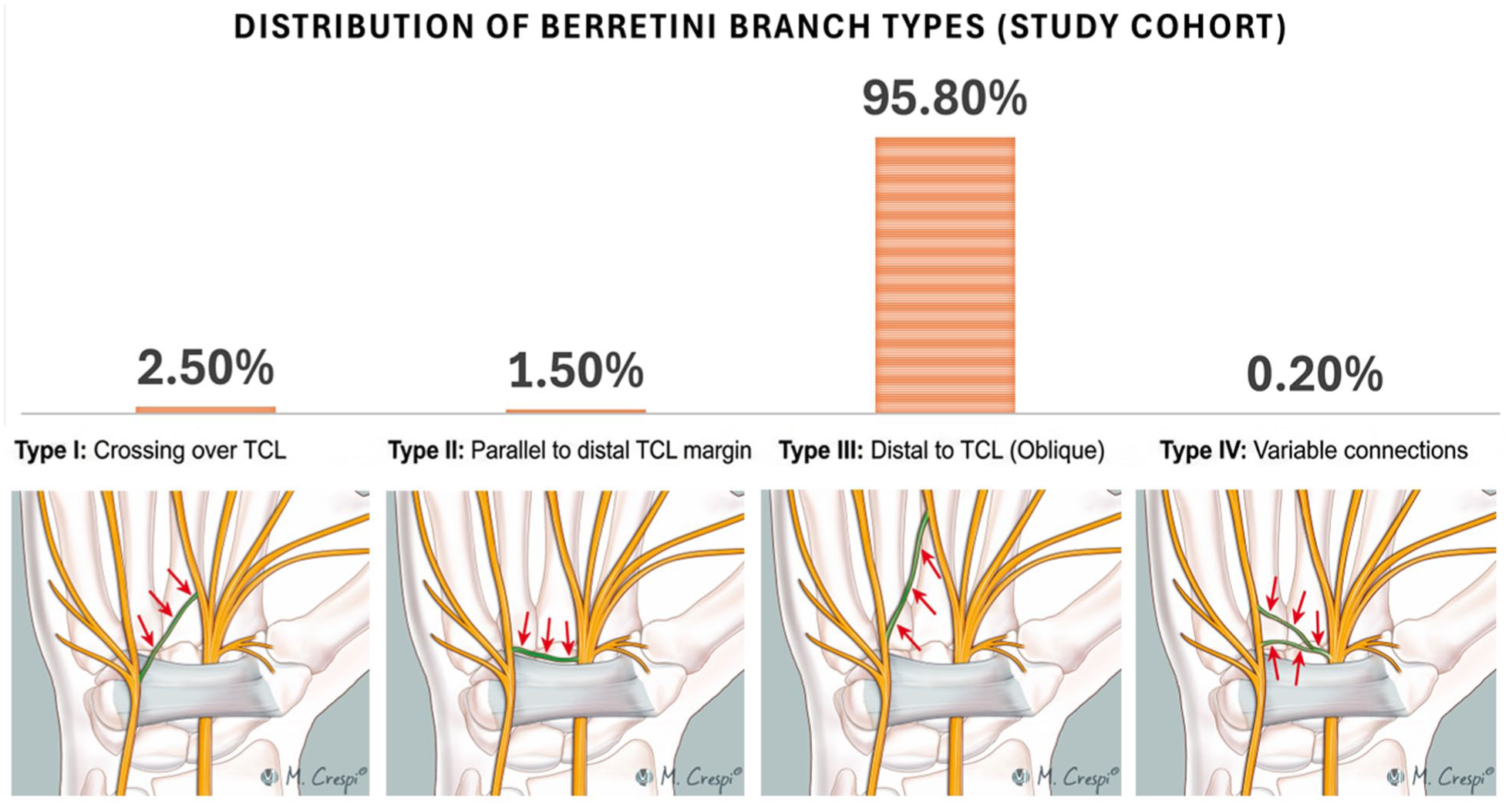
Distribution of Berrettini branch types identified in the study cohort (top), with their anatomical course (bottom). The bar chart shows the relative prevalence of the four configurations described by Ferrari and Gilbert (1991): type I crossing over the transverse carpal ligament (TCL); type II running parallel and close to the distal margin of the TCL; type III located distal to the TCL with an oblique trajectory; and type IV with variable connections. Red arrows indicate the course of the Berrettini branch.
As ultrasound-guided carpal tunnel release becomes more popular, it is necessary to mitigate the risk of iatrogenic injury by systematically visualizing the Berrettini branch before TCL release. Dukan et al. (2021), in a study on eight fresh anatomical specimens, reported the potential value of high-resolution sonography to trace this nerve. After cadaveric dissections and correlation with sonographic imaging, the senior author (FM) of this article has developed the following method that he uses before and during CTR (Figure 2). The location and type of Berrettini branch is routinely assessed, using a 4–12 MHz linear or matrix probe (Logiq, General Electric, Chicago, IL, USA) (occasionally, with higher frequency, up to 22 MHz, for better visualization). The probe is aligned along the ulnar origin of the superficial palmar arch. Close to the distal ulnar artery, beneath the pulsating artery, the superficial distal branch of the ulnar nerve is found with its characteristic fascicular nerve echostructure. From there, the Berrettini branch is progressively traced, up to its junction with the terminal branches of the median nerve. The Berrettini branch can then be classified as type I, II/IV or III (the exceptional type IV is difficult to distinguish from type II on ultrasound). In cases of a type II/IV configuration, the release of the TCL is performed with extra caution. It is safe in this circumstance to avoid using a push-type or a hook-type carpal tunnel knife or a thread. The CT Sono-Instrument® (Spirecut SA, Muttenz, Switzerland) allows progressive sectioning of the TCL. Designed for percutaneous use, its cutting extremity, with its echogenic lateral flange, is well visualized under ultrasound guidance and enables controlled release, with an oblique cut if necessary, to preserve the branch (Moungondo et al., 2024).
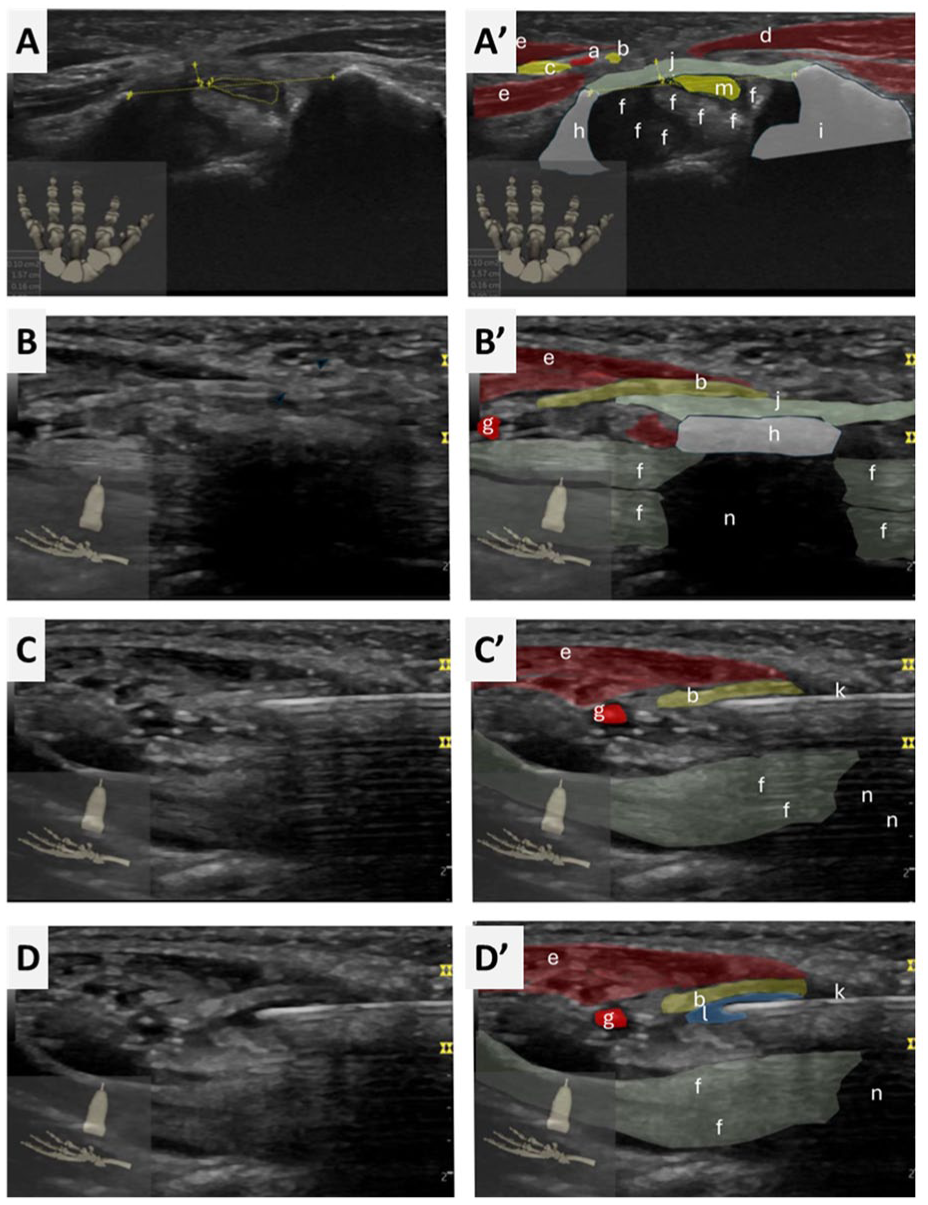
Example of a type I Berrettini branch as seen in short axis ultrasound view (A and A′), and in long axis views (B, B′, C, C′, D, D′). Each left image is repeated on the right with colour highlights of important anatomical structures. The Berrettini branch (b) is enhanced and mobilized by xylocaine injection just under and just distal to the transverse carpal ligament. In this specific case the branch is radial to the hamate hook, hanging just over the transverse carpal ligament, and the release needs to be performed more radially (a, ulnar artery; b, Berrettini branch; c, ulnar nerve; d, thenar muscles; e, hypothenar muscles; f, flexor tendons; g, palmar arch; h, hook of hamate; i, tubercle of trapezium; j, transverse carpal ligament; k, puncture needle; l, injected fluid; m, median nerve; n, hook of hamate shadow cone).
Since 2021, the senior author has routinely used this approach in a continuous series of 857 patients undergoing percutaneous carpal tunnel release. In this cohort, a Berrettini branch was identified in all patients, with 1.7% (15 patients) presenting the high-risk type II/IV configuration (Figure 1). No patient reported postoperative symptoms suggestive of a Berrettini branch injury, nor was any case suspected of postoperative complex regional pain syndrome.
In conclusion, the Berrettini branch is at risk of iatrogenic injury during CTR. Modern ultrasound visualization enhances safety by enabling precise localization of this tiny nerve. Caution is mandatory, particularly in cases of type II configuration. Adaptation of surgical technique may minimize the likelihood of nerve injury.
Footnotes
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Fabian Moungondo receives royalties from Spirecut. Frédéric Schuind holds shares in Spirecut. Georgia Antoniou has no conflicts of interest to declare.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
Ethical approval was not sought for this observational technical report, as it describes a surgical approach routinely performed in clinical practice. The study was completed in accordance with the Helsinki Declaration as revised in 2013.
Informed consent
Informed consent was not required because no patient-identifiable data are presented.
