Abstract
Introduction:
Amyloid deposition in the tenosynovium and transverse carpal ligament, and particularly cardiac involvement, are rare in patients with carpal tunnel syndrome; however, the prevalence may be underreported. This prospective study assessed the prevalence of amyloid deposits in the synovium and transverse carpal ligament and whether dual-site sampling improved detection. Risk factors for a positive test and asymptomatic cardiac involvement were also investigated.
Methods:
In a prospective cohort of 100 consecutive patients undergoing carpal tunnel release, intraoperative biopsies were taken from the tenosynovium and the transverse carpal ligament and examined using Congo red and immunohistochemistry staining. Concordance between biopsy sites was assessed, and independent predictors for a positive test were evaluated by multivariable logistic regression. Amyloid-positive patients underwent cardiac assessments, including cardiac biomarkers and 99mTc-pyrophosphate scintigraphy.
Results:
Amyloid deposition was present in 62 patients. Concordance between biopsy sites was 91%. Older age, male sex, bilateral carpal tunnel syndrome, multiple trigger fingers, and spinal stenosis independently predicted tissue amyloid. Asymptomatic cardiac amyloid deposition was identified in eight patients: three fulfilled criteria for cardiac amyloidosis and five demonstrated scintigraphic uptake with preserved function.
Conclusion:
Amyloid deposition in carpal tunnel syndrome was common in our study population, and subclinical cardiac involvement was not rare. Dual-site biopsy improved detection compared with the single-site sampling, and we recommend this routinely during carpal tunnel release. If not feasible, we recommend prioritizing older or male patients and those with bilateral carpal tunnel syndrome, multiple trigger fingers or spinal stenosis. Multiple trigger fingers may indicate a systemic process and high amyloid/cardiac risk.
Level of Evidence:
III
Keywords
Introduction
Systemic amyloidosis is characterized by the abnormal folding of amyloid protein as well as its deposition in tissues and organs, resulting in various clinical manifestations such as neuropathy, nephropathy and cardiomyopathy (Buxbaum et al., 2024; Koike and Katsuno, 2020; Nader et al., 2022). Cardiac amyloid deposition may cause severe complications, including restrictive cardiomyopathy, heart failure and conduction disturbances. The two main types of protein that lead to cardiac amyloidosis are amyloid light chain (AL) and amyloid transthyretin (ATTR) (Vranian et al., 2015). Patients with cardiac amyloidosis have a poor prognosis, with a median survival of less than 6 months from the onset of heart failure symptoms in untreated AL amyloidosis (Grogan et al., 2017; Grogan and Dispenzieri, 2015) and 2.6–3.6 years in ATTR amyloidosis (Lane et al., 2019). Notably, deposition of amyloid protein may occur in musculoskeletal tissues 5–10 years before the onset of cardiac symptoms (Sperry et al., 2018; Yamada et al., 2020).
The prevalence of amyloid deposits in the tenosynovium of patients with carpal tunnel syndrome (CTS) is reported to range from 2 to 37% (Bastian, 1974; Sperry et al., 2018; Takashio et al., 2023), with 0–2% showing cardiac involvement (Hansen et al., 2020; Sperry et al., 2018). This may indicate underdiagnosis of cardiac amyloidosis.
The reported risk factors for amyloid deposition include bilateral CTS, male sex, older age, cardiovascular disease, biceps tendon rupture, lumbar spinal stenosis, valvular heart disease, hearing loss and family history (Brunet et al., 2024; Donnelly et al., 2019). In contrast, metabolic syndrome (Balci and Utku, 2007) and diabetes (Argirò et al., 2025), which are associated with CTS, and musculoskeletal conditions such as rotator cuff tears (Yamada et al., 2024) and trigger fingers (Sperry et al., 2021), have not been thoroughly investigated as potential risk factors for amyloid deposition. Previous studies have linked trigger fingers to amyloid deposition (Gray et al., 2025; Sood and Lipira, 2022; Sperry et al., 2021). Whether multiple trigger fingers independently predict tissue amyloid in a prospective, fully adjusted cohort has not been firmly established. Furthermore, the optimal tissue sample (the tenosynovium, transverse carpal ligament (TCL) or their combination) for the biopsy in diagnosing amyloid deposition lacks consensus (Nakamichi and Tachibana, 1998; Ozdag et al., 2024). A standardized protocol for biopsy would aid in the early detection of systemic amyloidosis.
The aims of this study were to determine the prevalence of amyloid deposits in the tenosynovium and TCL of patients with CTS. We hypothesized that performing biopsies at both sites would improve the detection of amyloid. We also assessed risk factors for the presence of amyloid and cardiac involvement. We expected that multiple trigger fingers might serve as a predictor for amyloid positivity.
Methods
The institutional review board of our hospital approved this study. We reviewed the clinical data from 100 consecutive patients with CTS who were included in a prospective cohort study at our hospital and underwent carpal tunnel release (CTR) between April 2020 and April 2024. The eligibility criteria excluded patients receiving maintenance dialysis and those with pre-existing cardiac amyloidosis before CTR. No patients met these criteria. The diagnosis of CTS was determined through clinical examination and confirmed by nerve conduction studies. We explained to patients before CTR that amyloidosis screening would be performed by intraoperative biopsy, and for tissue-positive cases, further evaluations would follow. All participants provided written informed consent. All surgeries were performed by the first author.
All patients underwent mini-open CTR using an upper arm tourniquet and local anaesthetic. During CTR, 5 mm2 of TCL and a limited sample of peritendinous tenosynovium from within the carpal tunnel were harvested for histopathology. During a pilot phase involving seven patients, the two tissues were submitted together as a single specimen. However, we found that combining the tissues made it difficult to determine which tissue (tenosynovium or TCL) contained Congo red-positive amyloid deposits. Subsequently, the protocol was refined, and for the remaining 93 patients, tenosynovium and TCL were submitted separately in formalin for pathological analysis, processed for Congo red staining and examined under a polarized light microscope. Therefore, the initial seven pilot cases were excluded from analyses examining whether separate tenosynovium and TCL sampling increased the amyloid-detection rate. However, these seven cases were included in all other analyses. Positive biopsy samples were further subtyped in another institution. The amyloid subtype was determined by immunohistochemistry using primary antibodies against a part of transthyretin and the constant regions of immunoglobulin light chains (κ and λ). Transthyretin positivity indicated ATTR-type amyloidosis. Monoclonal immunoglobulin light chains cause AL amyloidosis; therefore, κ/λ positivity was interpreted as AL-type (Aκ/Aλ) amyloidosis (Gustavsson et al., 1994; Hoshii et al., 2006).
Patients were prospectively enrolled. Information about risk factors for amyloid deposition was obtained by interviewing the patient at enrolment and by retrospectively reviewing of the medical files. The risk factors included bilateral CTS, trigger finger (Sperry et al., 2021), multiple trigger fingers (Gray et al., 2025), diabetes (Argirò et al., 2025), cardiac conditions (such as heart failure (Sperry et al., 2018), valvular heart disease (Brunet et al., 2024), and arrhythmia (Donnelly et al., 2019)), symptomatic spinal stenosis (Yanagisawa et al., 2015), prior rotator cuff repair (Yamada et al., 2024) and treated hyperlipidaemia (Balci and Utku, 2007). Basic demographic information was also collected. A ‘trigger finger’ was defined as any stenosing flexor tenosynovitis affecting a single digit at any time in the patient’s history. ‘Multiple trigger fingers’ was defined as two or more affected digits occurring either concurrently or at different times.
All patients with confirmed ATTR-type amyloidosis were referred to the cardiology department for primary cardiac evaluation and management. Genetic counselling and TTR gene testing were offered to all ATTR-positive patients who provided consent. Cardiac function was assessed using biomarkers, including high-sensitivity cardiac troponin T and N-terminal pro-B-type natriuretic peptide and by echocardiography. Myocardial deposition was assessed using 99mTc-pyrophosphate scintigraphy, performed using Symbia Intevo two dual-headed single-photon emission computed tomography with low-energy, high-resolution collimators (Siemens Healthineers, Erlangen, Germany).
Patients with AL-type amyloidosis, associated with clonal plasma-cell disorders such as multiple myeloma and light-chain monoclonal gammopathy of undetermined significance, were referred first to the haematology department for assessment, including serum and urine immunofixation and serum-free light-chain assays, to confirm systemic AL amyloidosis. In AL cases, the cardiology department was consulted when clinical findings or investigations suggested cardiac involvement.
Statistical methods
Age was not normally distributed according to the Shapiro–Wilk test (p = 0.004); therefore, group differences were analysed with the Mann–Whitney U-test. Owing to small sample sizes in certain subgroups, categorical variables (sex, bilateral CTS, trigger finger, multiple trigger fingers, diabetes, cardiac disease, spinal stenosis, rotator cuff repair and hyperlipidaemia) were examined using Fisher’s exact test. To account for multiple comparisons, a multivariate logistic regression model was constructed incorporating a forced-entry method for clinically relevant variables that reached statistical significance in univariate analyses (p < 0.10). This approach helped mitigate the risk of type-I errors. Statistical significance was set at p < 0.05. A post hoc power analysis was conducted to assess the statistical power of our multivariate logistic regression analyses.
Results
Figure 1 is a flow chart summarizing the movement of 100 consecutive patients through each diagnostic step from biopsy to cardiac evaluation. In April 2024, 100 patients were enrolled. They had a median age of 74 years (IQR 68 to 80) and 34% were men. Amyloid deposits were detected in 62 patients. Subtyping identified 61 cases of ATTR and one case of Aκ amyloid. In the Aκ case, tissue staining was κ-positive and TTR-negative, consistent with AL-type deposits. The patient underwent haematological assessment, and normal serum and urine studies excluded systemic AL amyloidosis. Of the 61 ATTR-positive patients, 59 consented to genetic testing, which revealed one patient with the hereditary V122I variant. Two patients declined genetic testing.
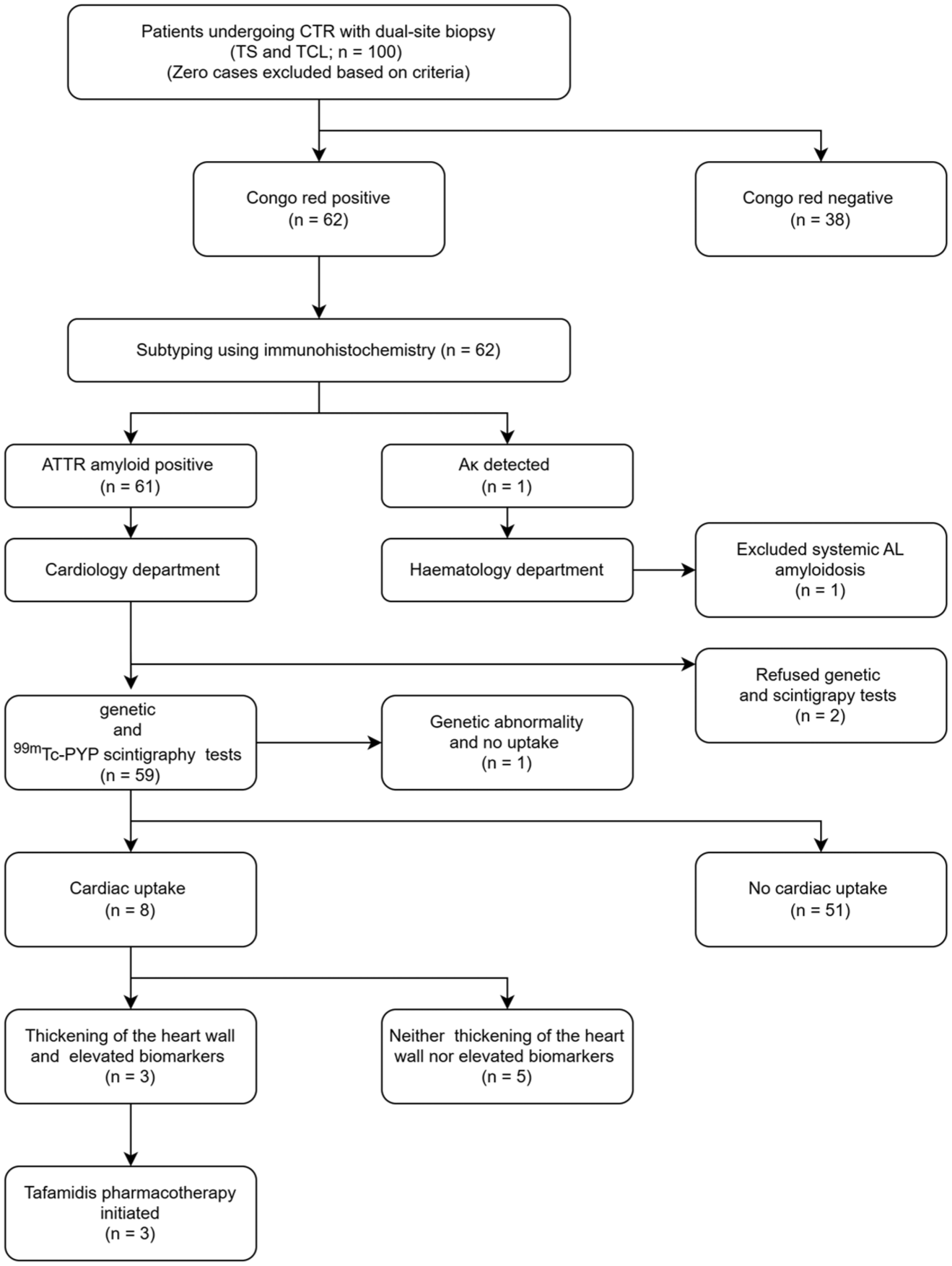
Flowchart of amyloid detection and outcomes in patients with carpal tunnel syndrome. CTR: Carpal tunnel release; TS: tenosynovium; TCL: transverse carpal ligament; ATTR: amyloid transthyretin; AL: light chains; Aκ: kappa light chain amyloid; 99mTc-PYP: technetium-99m pyrophosphate.
Table 1 shows the distribution of positive and negative results at each biopsy site, demonstrating a concordance of 91% between tenosynovial and TCL biopsies in 85 of 93 cases.
The number of amyloid-positive and negative results and concordance between tenosynovium and transverse carpal ligament biopsies of 93 patients undergoing carpal tunnel release.

Patients with amyloid-positive findings were more likely to be older and men, and to exhibit bilateral CTS, multiple trigger fingers, cardiac disease and spinal stenosis (Table 2). In a subsequent multivariate model adjusting for these five variables, excluding cardiac disease, all remained significantly associated with amyloid positivity in both the tenosynovium and TCL groups (Table 3).
Clinical characteristics of amyloid-positive and amyloid-negative patients with carpal tunnel syndrome.
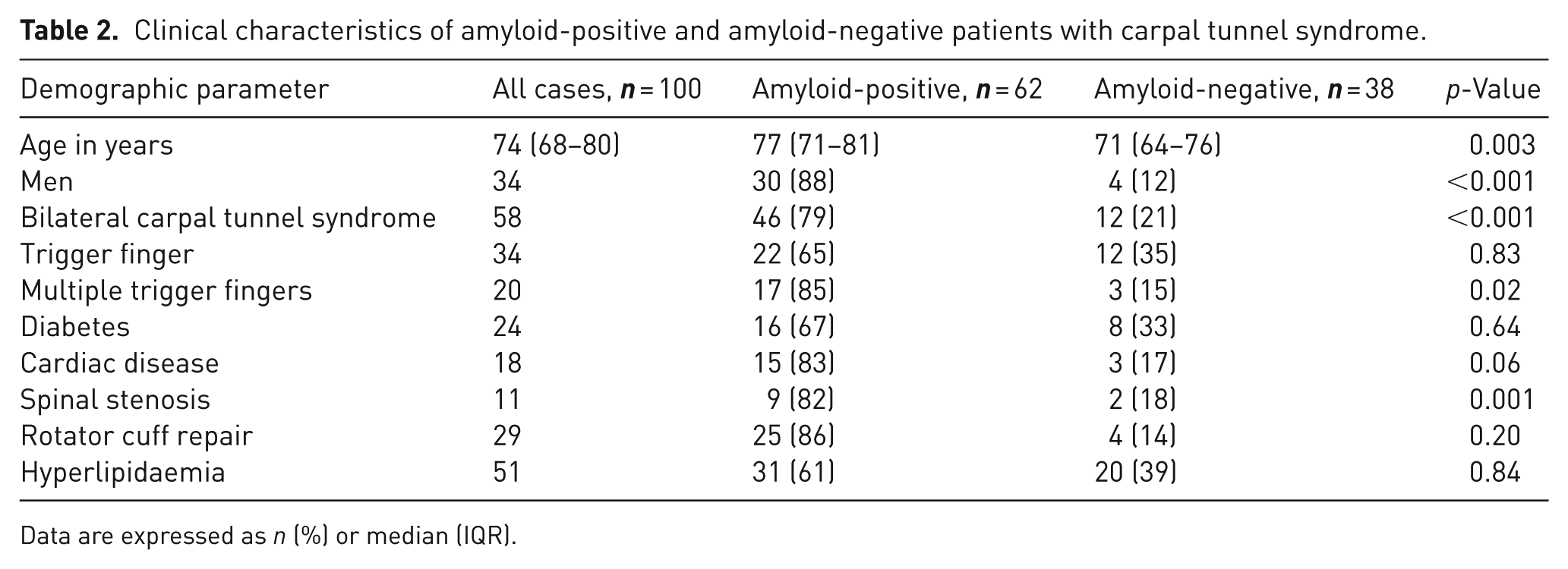
Data are expressed as n (%) or median (IQR).
Multivariate model for predicting the diagnosis of amyloid deposition in patients with carpal tunnel syndrome.
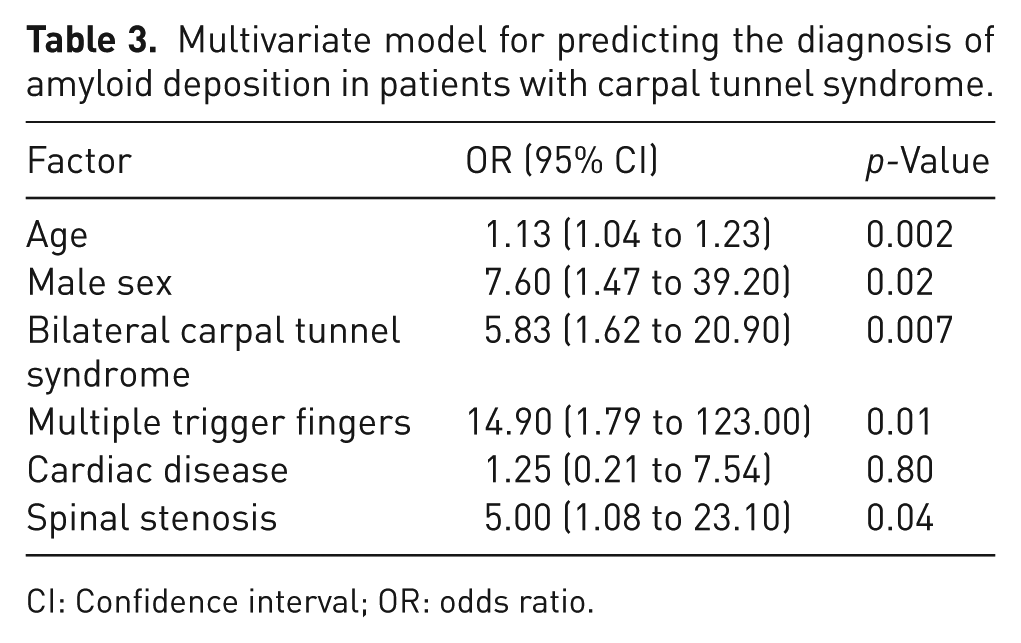
CI: Confidence interval; OR: odds ratio.
A post hoc power analysis was conducted for the binary covariates included in the logistic regression model: male sex, bilateral CTS, multiple trigger fingers and lumbar spinal stenosis. Our study, involving 100 patients and an observed amyloid positivity rate of 62%, achieved adequate power (>80%) to detect large effect sizes (odds ratios ⩾ 3.0) for binary predictors with prevalence rates of ⩾20%. This calculation did not include the continuous predictor (age).
Cardiac scintigraphy was performed in 59 out of 61 amyloid-positive patients (97%), with two patients declining the procedure. This comprehensive cardiac evaluation revealed asymptomatic cardiac amyloid deposition in eight patients (13% of amyloid-positive cases), including four men and four women, with a median age of 77 years (IQR 75–79). These patients exhibited grade 2 or 3 uptake on 99mTc-pyrophosphate scintigraphy, indicative of cardiac amyloid deposition. Notably, three of these patients had an interventricular septum thickness of ⩾12 mm, which fulfils the echocardiographic criterion for cardiac hypertrophy in cardiac amyloidosis according to the Japanese Circulation Society 2020 guidelines (Kitaoka et al., 2020). In addition, these patients had elevated biomarker levels and were subsequently placed on pharmacotherapy with tafamidis, a stabilizer of transthyretin approved for the treatment of ATTR cardiac amyloidosis. The remaining five patients exhibited scintigraphic evidence of cardiac amyloid deposition without any signs of cardiac dysfunction, suggesting the presence of pre-clinical cardiac involvement. The eight patients were diagnosed with cardiac amyloidosis.
Discussion
A prevalence of amyloid positivity of 62% was found in the study of 100 patients undergoing CTR. Biopsying both the TCL and tenosynovium improved detection compared with the efficiency of biopsying either site alone (91% concordance; four positives would have been missed by sampling only one site). Multivariable analysis showed that older age, male sex, bilateral CTS, multiple trigger fingers and spinal stenosis independently predicted amyloid positivity. Subsequent screening revealed previously undiagnosed cardiac involvement in eight patients, three of whom met the diagnostic criteria and started therapy.
The amyloid-positivity prevalence of 62% in our study was considerably higher than that reported in most previous studies, which ranged from 2 to 37% (Bastian, 1974; Sperry et al., 2018; Takashio et al., 2023). Carpal tunnel syndrome-related amyloid deposition may be underestimated. Inter-centre differences in diagnostic yield are well recognized, reflecting variation in biopsy site selection and sampling, tissue processing, optimization of Congo red staining with polarization microscopy, immunohistochemical antibody performance and interpretation, and access to mass-spectrometry-based typing (Benson et al., 2022). In addition, previous reports have noted a relatively high prevalence of amyloid deposits among Japanese populations. Sekijima et al. (2011) reported 34% prevalence in older Japanese patients with CTS, Sugiura et al. (2021) found 34% in their Japanese cohort, and Takashio et al. (2023) reported 37% prevalence, suggesting the possibility of local or ethnic influences.
Our dual-biopsy approach from both the tenosynovium and TCL revealed a 9% discordance. Ozdag et al. (2024) reported a 7% discordance rate between the two sites in 148 cases, which is comparable with our findings. Elzinga et al. (2023) found 100% concordance between tenosynovium and TCL biopsies in 18 cases with confirmed or suspected amyloidosis. Nakamichi and Tachibana (1998) obtained biopsies from both sites but did not report specific concordance rates between tenosynovium and TCL. Our series demonstrated a similar concordance of 91%. Four amyloid-positive cases would have been missed by sampling the tenosynovium alone and another four by sampling only the TCL (overall discordance 9%) (Table 1). However, despite the relatively low discordance rate, relying solely on a single biopsy site could reduce the amyloid-detection rate owing to the limited tissue samples available, potentially leading to underdiagnosis of fatal systemic amyloidosis.
In our study, older age, male sex, bilateral CTS, multiple trigger digits and spinal stenosis were identified as independent risk factors for amyloid deposition. Using a nationwide database, Sood and Lipira (2022) reported that patients who underwent surgery for both trigger digit release and CTR had a higher subsequent incidence of amyloidosis than did those who underwent trigger digit release or CTR alone. In contrast, prospective biopsy studies showed 24% amyloid positivity with multiple trigger fingers even without CTS (Gray et al., 2025) and 2% with a single trigger finger (Sperry et al., 2021). The nationwide database analysis by Sood and Lipira (2022) had limitations inherent to claims-based analyses. The dataset offered limited clinical details and was susceptible to miscoding; thus, unmeasured or mismeasured factors may have resulted in residual confounding despite covariate adjustment. Gray et al. (2025) and Sperry et al. (2021) did not consider multivariate adjustment. Our prospectively collected dataset was analysed with adjusted logistic regression models. Consistent with the multivariate analysis by Brunet et al. (2024), we confirmed that trigger finger was a risk factor, but only when multiple digits were involved. This supports a dose–response relationship that may indicate systemic organ amyloid accumulation. Although bilateral CTS was also statistically significant in the multivariate analysis, its screening value was modest because most patients manifest bilateral disease on careful assessment (Padua et al., 1998). Our study could identify multiple trigger fingers as an independent predictor, serving as a practical clinical marker of increased amyloid risk.
The prevalence of cardiac involvement in our study was 8%, which was markedly higher than that observed in other studies where the incidence was 0–2% (Hansen et al., 2020; Sperry et al., 2018). One reason for this discrepancy may be that previous studies performed cardiac scintigraphy on only a limited number of patients, even when amyloid positivity was detected in the synovium or ligament. In contrast, our study performed cardiac scintigraphy in nearly all patients with amyloid-positive findings. Specifically, eight patients showed positive uptake on 99mTc-pyrophosphate scintigraphy. Of these, three patients were diagnosed with cardiac amyloidosis based on elevated cardiac biomarkers and clinical assessment of cardiac function. The remaining five patients showed scintigraphic evidence of cardiac amyloid deposition without signs of cardiac dysfunction. This raises the possibility that these patients may develop cardiac dysfunction in the future despite currently having preserved cardiac function.
This study has some limitations. Although 100 patients were enrolled, this study took approximately 4 years to complete. This extended period was impacted by the coronavirus (COVID-19) pandemic, during which patients hesitated to seek medical care. Although we determined a pragmatic sample size of 100 patients, anticipating a potential decline in our annual CTR volume owing to the pandemic, we recognize that this approach may have limited the statistical power to detect small-to-moderate effect sizes in our multivariate analyses. Additionally, our results were obtained from a single institution, which limits its external validity and may introduce selection bias that could not be fully controlled. Another limitation is the lack of a formal cost–benefit or cost-effectiveness analysis for dual biopsies. Additionally, we performed only a cross-sectional analysis, leaving the long-term progression of subclinical or pre-clinical amyloidosis detected in patients with CTS uncertain. Although three patients with confirmed cardiac involvement began tafamidis-based therapy, the remaining five demonstrated myocardial uptake on scintigraphy and had normal cardiac biomarkers (high-sensitivity cardiac troponin T and N-terminal pro-B-type natriuretic peptide) levels within institutional reference ranges. These patients require prolonged follow-up to determine whether they may eventually develop symptomatic cardiac amyloidosis. Such prospective longitudinal data would provide important insights into whether routine screening at the time of CTR effectively impacts morbidity and mortality.
For hand surgeons, we recommend routine dual-site biopsy during CTR. Where this is not feasible, older patients, men and patients with bilateral CTS, multiple trigger fingers or spinal stenosis should be prioritized.
Footnotes
Acknowledgements
We thank all patients who participated and allowed their data to be anonymously used for the present study. In addition, we acknowledge Hiroo Kawano, MD, PhD, of Yamaguchi University Hospital, for contributing to the genetic tests and diagnosis. We would like to thank Editage (![]() ) for the English-language editing.
) for the English-language editing.
Writing assistance: The authors used ChatGPT v4 (OpenAI, San Francisco, USA) to generate an initial draft of the Introduction and to refine grammar and style throughout the manuscript. All AI-generated text was critically reviewed and edited by the authors to ensure accuracy and originality.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
Ethical approval of this study was obtained from the Research Ethics Board of the Saiseikai Yamaguchi General Hospital (R2-7).
Informed consent
Written and verbal informed consent was obtained from all participants before the study.
Data availability
The data supporting the findings of this study are not publicly available owing to ethical restrictions and concerns regarding patient confidentiality. However, de-identified data or summary results can be obtained from the corresponding author upon reasonable request, provided the request complies with ethical guidelines and institutional review board policies.
