Abstract
Carpal dislocations result from sequential disruption of the complex relationship between the bones and ligaments of the wrist. Injuries to the carpus occur via predictable mechanisms, an understanding of which is critical to identify and treat these frequently missed patterns of injury and to avoid the sequela of chronic instability. Lunate dislocations are by far the most common, but isolated dislocation of other carpal bones can also occur. Open reduction and internal fixation still remains the gold standard for treatment regardless of the debate around the specific approaches. These high-energy injuries are associated with significant long-term morbidity even when identified promptly and appropriately treated. This review will focus on the evaluation and management of common forms of carpal dislocations.
Introduction
While injuries to the carpal bones and ligaments are uncommon, recognizing and properly treating them is important to prevent pain and degenerative changes. Due to the complex nature of the carpal bones and ligaments, carpal ligamentous injuries, carpal dislocations and even fractures are often missed during initial presentation (Bentley et al., 2022; Herzberg et al., 1993). Many patients may not re-present with symptoms until degeneration and arthritis have already occurred.
Anatomy
The carpal bones are held together by the metacarpal bones and by a complex series of ligaments connecting them to each other and to the radius and ulna. These ligaments are crucial for carpal stability by preventing translation, rotation and angulation. The palmar ligaments can be thought of as two inverted ‘V-shaped’ structures. The proximal ‘V’ ligaments act as a weightbearing structure and include the radiolunotriquetral, ulnotriquetral and ulnolunate ligaments, which anchor the lunate and triquetrum to the wrist. The lunate is also anchored by strong ligaments to the surrounding proximal carpal row by the scapholunate and lunotriquetral ligaments. The distal ‘V’ includes key ligaments connecting the radius, ulna and proximal carpal row to the distal carpal row, primarily by the radioscaphocapitate and ulnocapitate ligaments. In addition, the scaphotrapeziotrapezoid ligament is an intrinsic structure that influences scaphoid biomechanics. Between the two palmar Vs is the space of Poirier, an interval of weak ligamentous support located at the capitate-lunate articulation and the carpal tunnel floor (Figure 1).
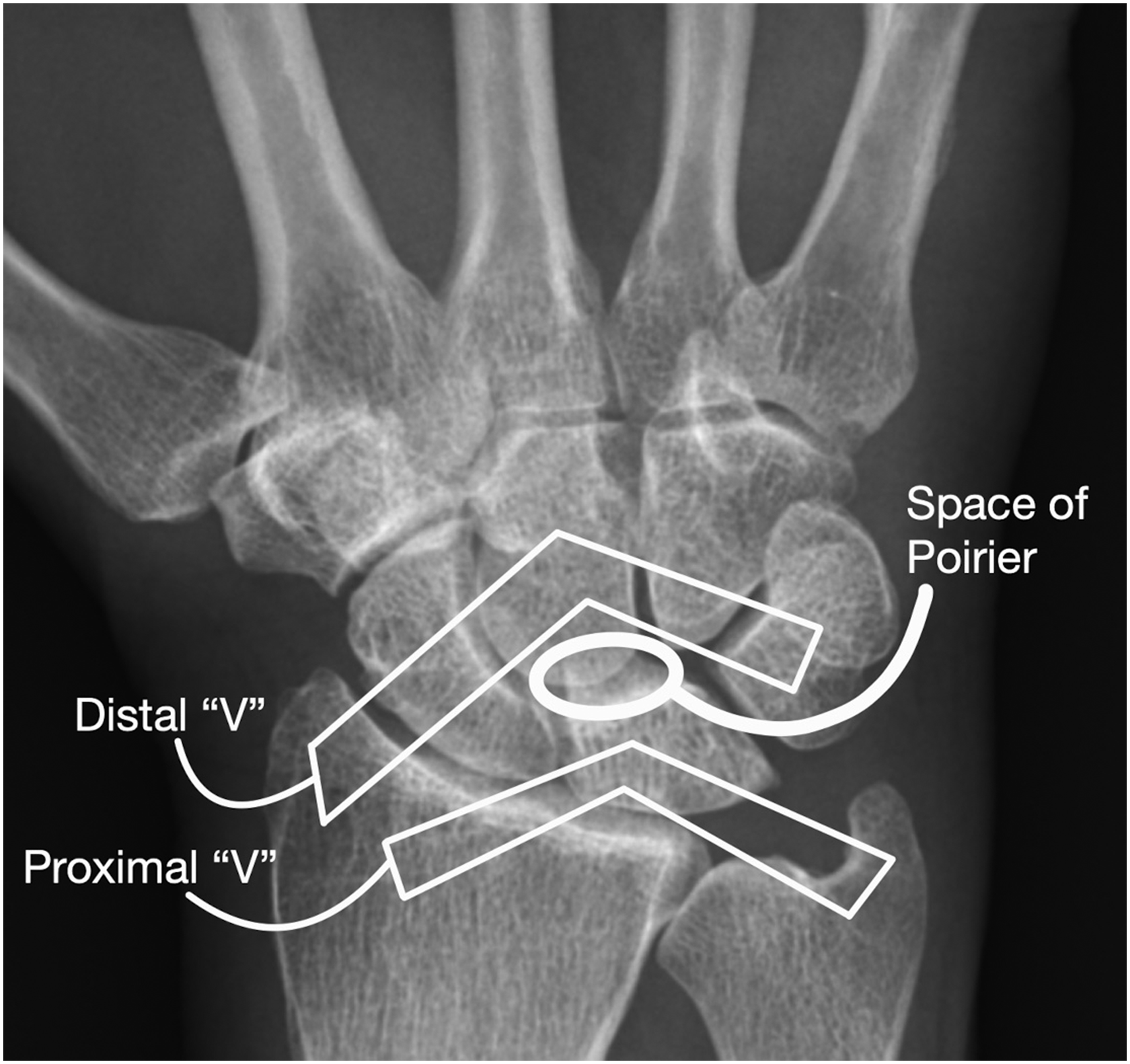
Posteroanterior view of the hand with a conceptual graphic of the extrinsic carpal ligaments that form two inverted ‘Vs’ with the space of Poirier between them.
On the dorsal side, the dorsal radiotriquetral and dorsal scaphotriquetral ligament also form a V-shaped structure that converges on the triquetrum. This structure limits intercarpal supination and palmar flexion, while also supporting the lunate dorsally (Sennwald and Segmuller, 1986). The scapholunate (SL) articulation is stabilized by the SL ligament complex comprising of primary and secondary stabilizers. The scapholunate interosseous ligament (SLIL) acts as a primary stabilizer while the dorsal intercarpal, dorsal radiocarpal, scaphotrapeziotrapezoid, radioscaphocapitate and radiolunate ligaments function as secondary stabilizers (Short et al., 2002). This complex can be divided into three components: volar, proximal and dorsal. The dorsal component is the thickest and is crucial to the stability of the SL articulation, especially against translation (Mitsuyasu et al., 2004). The volar aspect stabilizes against rotation. Similarly, the lunotriquetral (LT) articulation is also stabilized by a ligamentous complex, with palmar, proximal and dorsal components. Unlike the SL ligamentous complex, the volar component is the thickest and strongest, preventing volar translation, while the dorsal aspect acts as a rotational constraint. The primary stabilizer is the lunotriquetral interosseous ligament (LTIL) while the secondary stabilizers are the dorsal radiocarpal and volar-sided radiolunate ligaments and scaphotrapeziotrapezoid ligament (Horii et al., 1991). The LT ligamentous complex is much stronger than that of the SL complex, hence why these injuries are less common.
Perilunate injuries: perilunate dislocation and lunate dislocation
Anatomy and pathology
The pathomechanical force of perilunate injuries, including perilunate dislocation and lunate dislocation, is that of wrist extension, ulnar deviation and intercarpal supination (Kapoor et al., 2020; Mayfield et al., 1980). This leads to a disturbance of the carpal ligaments from radial to ulnar, with the first stage being disruption of the scapholunate articulation, namely injury to the scapholunate and radioscaphocapitate ligaments. The second stage is disruption of the lunocapitate articulation, followed by the third stage being disruption of the lunotriquetral articulation with failure of the lunotriquetral ligament. Finally, the dorsal radiotriquetral and scaphotriquetral ligaments can also be disrupted during intercarpal supination. The result of this circumferential ligamentous disruption of the lunate can lead to lunate rotation and dislocation through the space of Poirier. The space of Poirier is weaker in wrist extension and with disruption of the dorsal radiocarpal ligaments, the lunate can dislocate volarly into this space. As a consequence, in lunate dislocations, there may be impingement of median nerve within the carpal tunnel. In perilunate dislocations, through the same mechanism, the weaker ligaments within the space of Poirier permit the proximal carpal row to dorsally dislocate from the intact lunate (Kapoor et al., 2020; Mayfield et al., 1980). On rare occasions, the lunate can dislocate dorsally or the carpus can dislocate volarly around the lunate (Herzberg et al., 1993).
Imaging
Radiographs must be scrutinized for evidence of carpal subluxation or dislocation that may be evidenced by disruption of Gilula’s arcs on the posteroanterior (PA) view (Gilula et al., 1984) (Figure 2). Other signs of perilunate injury include an abnormal overlap of the carpal bones, loss of carpal height and ulnocarpal translation of the wrist. defined as greater than 50% uncovering of the lunate. In a lunate dislocation, the lunate may appear as triangular in shape or as a ‘piece of pie’ sign (Propp and Chin, 1989). On the lateral view, there will be loss of collinearity between the capitate, lunate and distal radius. A scapholunate angle greater than 70° indicates disruption of the SLIL. For lunate dislocations, a ‘spilled teacup sign’ is present when the lunate is dislocated from lunate fossa and tilts volarly (Green and O’Brien, 1980) (Figure 3). In a perilunate dislocation, the lunate stays in position on the radius while the surrounding carpus dislocates, often dorsally (Figure 4). Traction views can be helpful to better delineate the pattern of injury and assess for associated fractures.
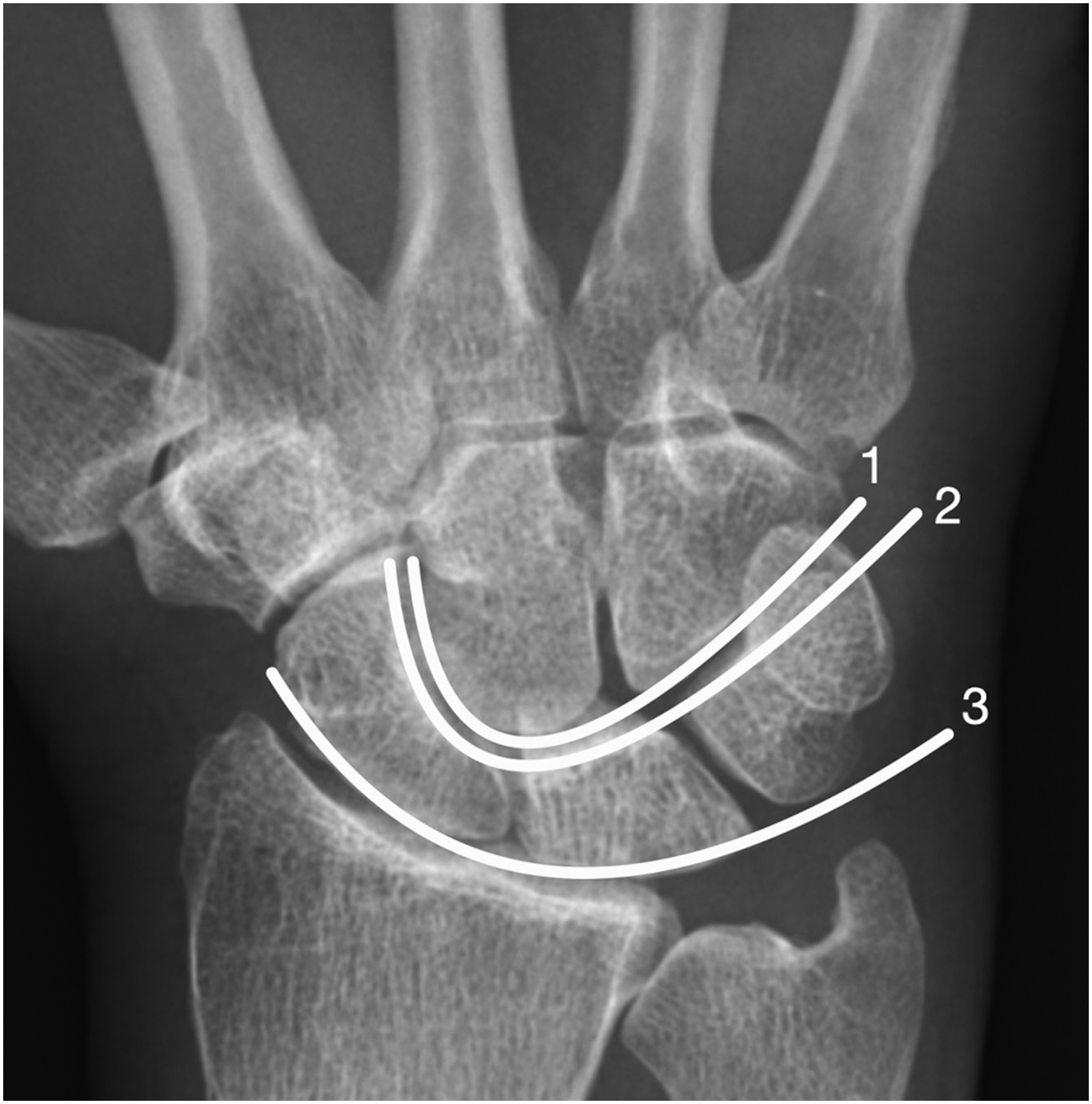
Posteroanterior view of the hand showing Gilula’s three arcs: (1) proximal convexity along the scaphoid, lunate and triquetrum; (2) distal concavity along the scaphoid, lunate and triquetrum; and (3) proximal convexity along the capitate and hamate.
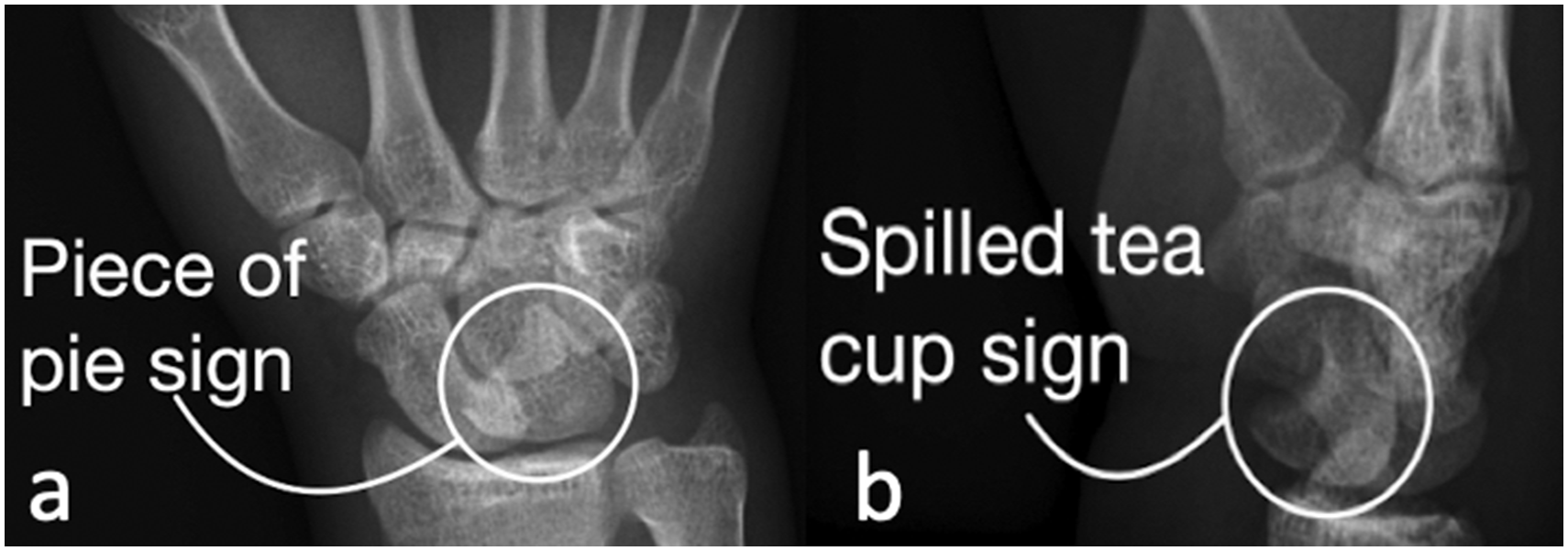
(a) Posteroanterior radiographs of volar lunate dislocation with a ‘piece of pie’ sign; (b) lateral radiograph showing ‘spilled teacup’ sign.
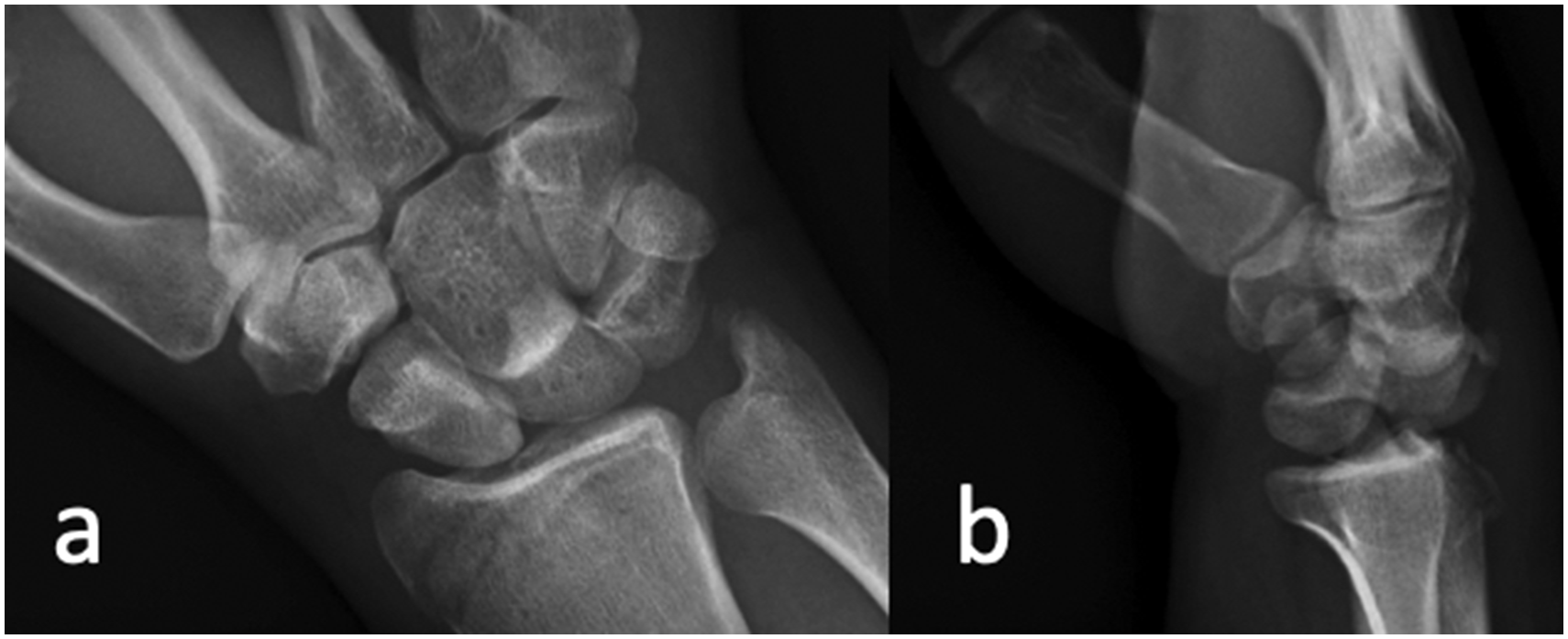
(a) Posteroanterior and (b) lateral radiographs of a dorsal perilunate dislocation.
Classification
A recent review by Liechti et al. (2023) found the high majority of these injuries affected men (94%), primarily after high-energy falls on an outstretched hand, followed by motor vehicle accidents and sports injuries. The mean age of injured patients was 30 years. The vast majority of perilunate dislocations and lunate dislocations occurs secondary to dorsal displacement of the carpus. However, there are reports of volar dislocation with forced hyperflexion and supination of the wrist (Masmejean et al., 1998). Greater arc injuries are classified based on the fractured bone using the suffix trans-. Fractures of the scaphoid, capitate, lunate, triquetrum and radial styloid have all been reported with the scaphoid being the most frequently fractured carpal bone with this injury pattern (Herzberg et al., 1993; Scalcione et al., 2014). Lesser arc injuries are purely ligamentous (Figure 5). Herzberg et al. (1993) classified perilunate injuries into two types that can help inform initial closed management. In stage I, the lunate remains in place in the lunate fossa of the radius, whereas in stage II, the lunate itself is subluxated or dislocated with respect to the radius. They subclassified stage II into dorsal or palmar, depending on the direction of displacement of the capitate in relation to lunate. Herzberg (2000) later subclassified stage II into stage IIA where the lunate dislocates and rotates less than 90°, and stage IIB where the lunate dislocates and rotates more than 90°.
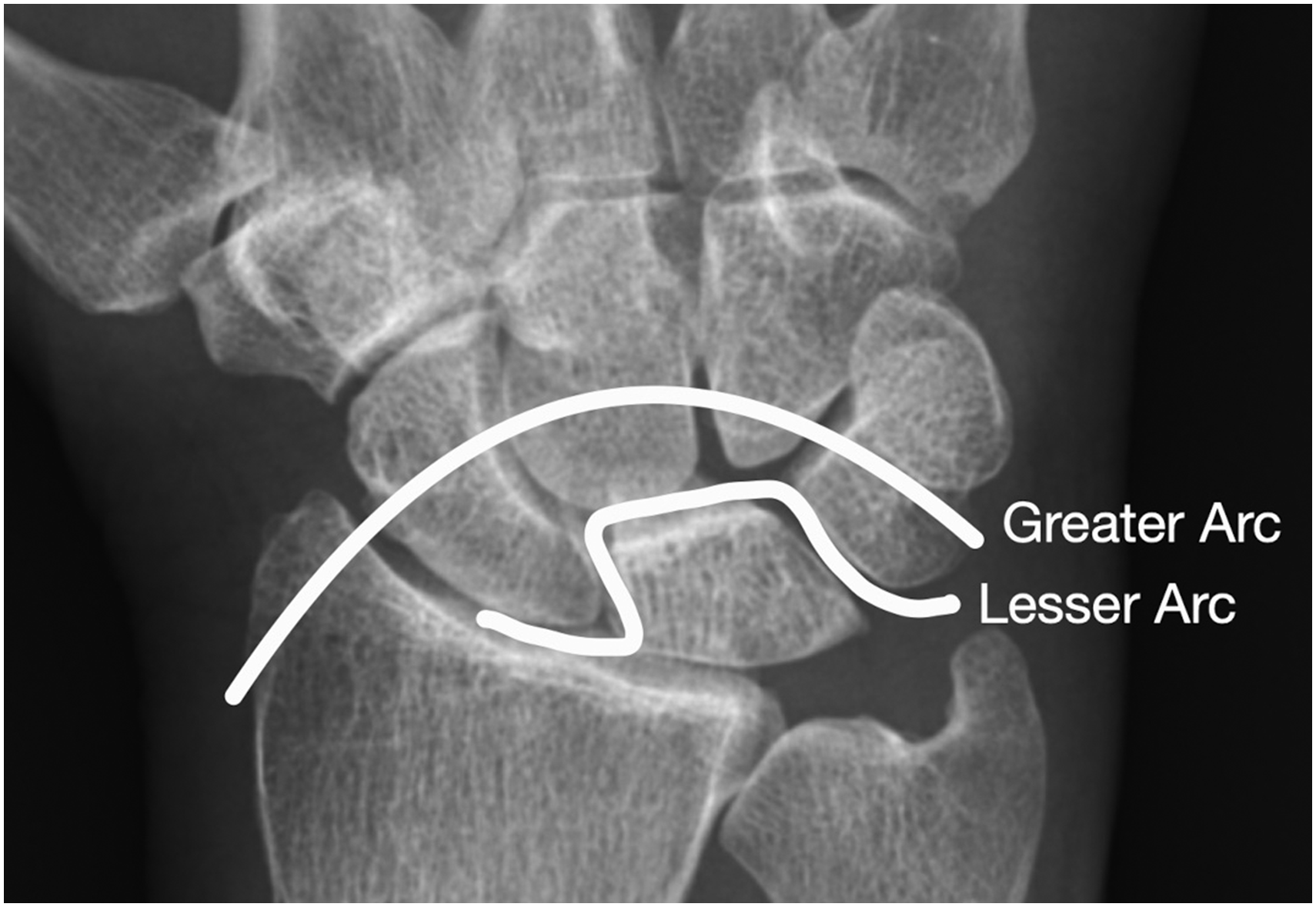
Posteroanterior view of the hand outlining the greater and lesser arcs.
Management of acute injuries
Management of perilunate injuries depends on the chronicity of the injury as chronic injuries are unlikely to be reducible in a closed manner. Herzberg et al. (1993) showed that injuries treated within 7 to 45 days had similar outcomes to those treated within 7 days, whereas treatment after 45 days had significantly worse outcomes. For acute injuries, closed reduction of all Herzberg stage I and IIA injuries should be attempted in the emergency department (Herzberg, 2000). Some authors recommend against attempting closed manipulation of Herzberg IIB injuries as a reduction is less likely to be successful due to soft tissue interposition and there is a greater risk of iatrogenic injury to the remaining ligamentous attachments (Herzberg et al., 1993, 2000; Scalcione et al., 2014). Up to one-third of patients may present with median nerve symptoms, either from direct trauma, soft tissue compression, impingement by the volar aspect of the lunate or hematoma within the carpal tunnel. The presence of median nerve symptoms should prompt urgent reduction and serial examinations to monitor for acute carpal tunnel syndrome, which is characterized by progressively worsening hypoesthesia (Kardashian et al., 2011; Liu et al., 2017). After successful reduction, the wrist should be immobilized in a splint. Surgery may be delayed several days to allow for resolution of the swelling. If there is no fracture, definitive management options include closed reduction and immobilization, closed reduction with percutaneous pinning (CRPP) with or without arthroscopic assistance, or open reduction and internal fixation (ORIF). Closed reduction and splinting have universally poor outcomes, with more than half failing to maintain reduction and suffering from the sequela of chronic instability (Apergis et al., 1997; Cooney et al., 1987; Frane and Goldenberg, 2022). Closed reduction without fixation should only be pursued as a last resort, and serial radiographic monitoring is critical. CRPP requires an anatomic reduction to succeed because malreduction prevents apposition of the torn ligaments resulting in non-healing and persistent SL or LT instability. Recent studies of arthroscopic-assisted closed reduction and pinning have shown promising results comparable to ORIF (Herzberg et al., 2015; Liu et al., 2017).
ORIF is the gold standard in perilunate injuries, both in purely ligamentous and in those with an associated fracture (Herzberg et al., 1993; Liechti et al., 2023; Minami and Kaneda, 1993; Trumble and Verheyden, 2004).
Surgical approach and repair
ORIF may be accomplished via a dorsal, volar or combined approach (Frane and Goldenberg, 2022; Hildebrand et al., 2000; Melone et al., 2000; Sotereanos et al., 1997; Trumble and Verheyden, 2004). A dorsal approach allows direct repair of the stout dorsal portion of the SLIL as well as excellent visualization of the proximal carpal row and midcarpal joints but does not allow repair of the stout volar LTIL. A volar approach can be extended to decompress the carpal tunnel, can assist with reduction of the volarly dislocated lunate and can allow for direct repair of the LTIL. Some authors advocate a combined approach to allow repair of both the SLIL and LTIL. Herzberg (2000) recommended using a single approach for stage I and IIA injuries, but a combined dorsal-palmar approach for stage IIB injuries. A recent large systemic review by Liechti et al. (2023) found that the combined dorsal-palmar approach was the most commonly used (49%). However, they also found a higher overall complication rate for the combined dorsal-palmar approach (17%) compared to an isolated dorsal approach (8%). The most common complications noted were nonunion and loss of reduction. There was a higher complication rate for ORIF (17%) than arthroscopic-assisted procedures (5%), primarily from loss of reduction seen with the open procedures (11% compared to 0% in arthroscopic- assisted procedures). Functional and patient-reported outcomes were comparable between all procedure types. The authors postulate the denervation effect from both the severity of injury and combined dorsal-palmar approach may reduce pain and help patients maintain wrist function despite the development of arthritis (Liechti et al., 2023). On the other hand, Kremer et al. (2010) found better patient-reported outcomes for isolated volar and dorsal approaches compared to a combined dorsal-palmar approach. Similarly, Oh et al. (2017) found better patient-reported outcomes for the less invasive arthroscopic surgery compared to ORIF. Some believe the higher rate of nonunion, loss of reduction and possible worse functional outcomes with more invasive approaches may be due to devascularization and iatrogenic damage to the surrounding extrinsic ligaments (Liechti et al., 2023). For patients with persistent medial nerve symptoms, a carpal tunnel release should be performed. Carpal fractures are fixed with K-wires or headless screws. The SL and LT intervals should be reduced and secured using either K-wires or temporary screws and the midcarpal joint also secured temporarily, with the proximal carpal row aligned with the distal row (Souer et al., 2007; Trumble and Verheyden, 2004). The SLIL should be repaired at a minimum with some authors advocating for dorsal or volar repair of the LTIL as well (Knoll et al., 2005; Minami and Kaneda, 1993). In patients with trans-scaphoid perilunate fracture dislocations, the SLIL should still be assessed to ensure it has not been disrupted as there have been reports of concurrent scaphoid fracture and SLIL disruption (Liu et al., 2017). Arthroscopy can be used as an adjunct for debridement and confirmation of reduction. Ulnocarpal translation is an emerging area of interest as Song et al. (2009) noted 80% of patients had greater than 50% uncovering of the lunate post reduction. A significant improvement in ulnocarpal translation with temporary pinning of the lunate to the radius was also noted. After ORIF, patients are immobilized for 8 to 12 weeks before pins are removed.
Management of chronic perilunate injuries
Herzberg (2008) demonstrated that injuries presenting more than 45 days from injury had significantly worse outcomes. These delayed presentations may be better suited for salvage procedures, such as proximal row carpectomy or wrist fusion (Herzberg, 2008; van der Oest et al., 2022).
Outcomes and complications
A high rate of post-traumatic degenerative changes can be expected, with 70% of patients having radiographic evidence of arthrosis at a mean follow-up of 18 years. However, this finding did not appear to correlate strongly with functional outcomes (Krief et al., 2015). Median nerve symptoms and reduced sensory function may persist in 18% of patients years after initial injury (Dunn et al., 2018; Liechti et al., 2023). Manual laborers and those injured at work tend to have poorer outcomes and are less likely to return to their previous employment (Kremer et al., 2010; Lebot et al., 2021). Complications after surgery occur in about 15% of patients, with the majority due to nonunion or loss of reduction. Implant failure, tendon complications, complex regional pain syndrome and infection are rare and occur in less than 2% of patients (Liechti et al., 2023).
Pisiform dislocation
Anatomy and pathology
The pisiform is embedded within the tendon of the flexor carpi ulnaris and acts as a sesamoid bone. Its ligamentous attachments are the pisohamate, pisometacarpal and ulnar pisotriquetral (PT) ligaments. Although rare, injury to this ligamentous complex may cause pisiform dislocation. The pisiform plays a role in medial column stability by preventing subluxation of the triquetrum and by acting as a fulcrum for the flexion of the wrist. Dislocation and subsequent instability at the PT articulation can predispose to degenerative changes and arthrosis. Acute injury may result from a direct pisiform injury during a fall on an outstretched hand causing ligament displacement, or from indirect wrist hyperextension causing pisohamate or ulnar PT ligament elongation. Chronic PT ligament complex injury can result from acute pisiform dislocation or repetitive recreational occupational activities involving wrist flexion and extension. During extension, the pisiform translates tightly against the triquetrum, and during flexion, it moves away from the triquetrum (Moojen et al., 2001; Rayan, 2005).
Imaging
Semi-supinated views of the wrist help evaluate the PT articulation and can be performed in flexion, extension and neutral with the wrist in 30° of supination (Jameson et al., 2002). Pisiform ligament complex injury can be defined radiographically as a widening of the joint space greater than 4 mm, an angle greater than 20° between the articular surfaces of the pisiform and triquetrum, or displacement greater than 15% in the proximal-distal plane (Moraux et al., 2014). Pisiform ligament complex injury can be classified into three stages based on clinical and radiographic findings. Stage I is defined as clinical instability without radiographic changes, stage II as instability with altered radiographic parameters and stage III as frank dislocation of the pisiform (Rayan, 2005). Fluoroscopy can help to obtain a dynamic assessment of the PT articulation. Computed tomography and magnetic resonance imaging can also be helpful adjuncts to identify associated fractures and assess the integrity of the ligaments.
Management
Patients with acute pisiform dislocation should be treated with reduction and immobilization. In patients with chronic instability after dislocation or repetitive use, a trial of non-operative management is reasonable with activity modification, immobilization, non-steroidal anti-inflammatory drugs (NSAIDs) and steroid injection (Paley et al., 1987; Rayan, 2005). However, the response rate has been reported as only about 35% (Palmieri, 1982). For cases refractory to conservative management, excision of the pisiform serves as the mainstay, with multiple studies showing good pain relief without significant losses in range of motion or grip strength (Campion et al., 2014; Lautenbach et al., 2013). Patients with symptoms of ulnar nerve compression should also undergo release of Guyon’s canal concurrently (Carroll and Coyle, 1985). PT arthrodesis is an alternative with early studies showing some promise for patients who require forceful wrist flexion (Singer et al., 2011).
Triquetral-hamate dislocation
Anatomy and pathology
The triquetrum and hamate are connected by the triquetrohamate (TH) ligaments, both volarly and dorsally, as well as the medial TH ligament, also known as the ulnar arm of the arcuate ligament. The medial TH ligament limits widening of the TH joint during radial deviation of the wrist and potential dislocation. Injury to this complex ligament is rare and all triquetral dislocations have been reported in men in the setting of high-energy trauma (Braig et al., 2014). Untreated injuries or those with resulting instability typically present with pain over the ulnar and dorsal aspects of the wrist with activities that require forceful radial and ulnar deviation of the wrist. Patients are frequently asymptomatic with day-to-day activities and are more likely to complain of the pain interfering with their professional or recreational activities (Zancolli, 2001).
Imaging
Stress radiographs should be performed and compared to the contralateral wrist. This is accomplished by pronating the wrist and forearm 10° to 20° and taking PA radiographs with the wrist in neutral, forced radial deviation and forced ulnar deviation. Injury may be evidenced by opening of the angle between the articular surface of the triquetrum and the hamate or widening of the medial side of the TH joint relative to the contralateral side (Garth et al., 1985; Zancolli, 2001). Disruption of Gilula’s arcs may also be present with dislocation. Magnetic resonance imaging will show disruption of both the medial TH ligament and the floor of the extensor carpi ulnaris (ECU) sheath.
Management
Acute dislocations should be reduced and immobilized. Injuries left untreated or for those that develop resulting instability, first-line treatment is conservative with immobilization, NSAIDs, physical therapy and steroid injections. When considering surgical intervention, other forms of carpal dislocation or instability should be concurrently addressed if they are present as patients with associated conditions have been shown to have a less satisfactory outcome with surgical management than those with isolated triquetral-hamate injury (Zancolli, 2001). Stabilization of the TH articulation is accomplished through reconstruction of the medial TH ligament and the floor of the sixth dorsal compartment by imbrication with the wrist in 10° of ulnar deviation. This repair may then be reinforced by using a portion of the ECU tendon with its distal insertion left intact. In a series of eight patients undergoing surgical management, all five patients with isolated triquetral-hamate instability reported excellent outcomes with no pain and return to full professional activities (Zancolli, 2001).
Summary
Carpal dislocations are high-energy injuries that are the result of sequential ligamentous failure. An understanding of carpal anatomy and biomechanics is necessary in identifying these injuries and treatment. Perilunate and lunate dislocation results from a complex circumferential pattern of injury in four stages. Less common patterns include dislocation of the pisiform and, rarer even, other isolated carpal bone dislocations. Prompt identification of injury with stabilization and repair of the supporting ligamentous structures are crucial. These high-energy injuries are associated with significant long-term morbidity even when identified promptly and appropriately treated.
Footnotes
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: AG serves as an educational consultant for Skeletal Dynamics. The remaining authors declare no potential conflicts of interest.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
