Abstract
Soft tissue reconstruction of the upper extremity requires consideration of wound bed status, varied anatomic composition, future function and tissue match, whether in thickness, pliability or involvement of other tissue components. Of the options available, microvascular free tissue flaps allow maximal customizability with the avoidance of long-term donor site morbidity. Free tissue transfers have evolved, given increased surgical proficiency, from direct vessel-based flaps to septocutaneous vessel-based flaps, to musculocutaneous perforator flaps, and most recently to free-style free flaps. With increases in technical complexity come limitless alternatives. We documented the progression of free flap upper extremity reconstruction in recreating form and function of the upper extremity. The foundations laid should allow surgeons the freedom and versatility to choose the most faithful restoration of the defect and produce the best functional and aesthetic results.
Introduction
The upper extremity is a complex marriage of anatomy, function and aesthetics. It is uniquely challenging to reconstruct. The soft tissue envelope varies in composition, thickness, durability, adherence and even adnexal organs, depending on the location. Delicate vital structures such as nerves, joints and tendons lie just deep to the surface, requiring robust vascularized soft tissue coverage.
Extensive options are available, from secondary intention, local-regional flaps, pedicled transfers, to free flaps. In the modern era of reconstructive microsurgery, the reconstructive ladder should be no more than a general guideline. Instead, liberal adaptation of the reconstructive escalator can optimize the best coverage, reconstruction and function. Adjacent tissue rearrangement using local flaps could be plagued by inadequate coverage, poor donor appearance and risk of contracture from restricted positioning. Thus, over the years, we have focused on the progression of free tissue transfer.
Free microvascular transfer has an extensive history in upper extremity reconstruction (Fang and Chung, 2014). Originating in 1965, Yang et al. (1977) performed the first series of free toe to thumb transfers, followed closely by Cobbett in 1968 (Cobbett, 1969). Harii et al. (1974) presented the first fasciocutaneous groin flap to the hand in 1973. Ikuta et al. (1976) published the first free functioning gracilis muscle to bilateral forearms for Volkmann’s contracture. Free tissue transfer techniques transformed over the last several decades, mainly due to enhanced understanding of the vasculature and development of dissection techniques and instruments.
In this review article, we summarize the progression of free flap upper extremity reconstruction from our institutional experience at Chang Gung Memorial Hospital, in recreating form and function of the upper extremity. The foundations laid should allow surgeons the freedom and versatility to choose the most faithful restoration of the defect and produce the best functional and aesthetic results.
Evolution and nomenclature of flaps
Cutaneous flaps are defined by their anatomic blood supply (Wallace et al., 2009). These include direct cutaneous vessels, septocutaneous vessels and musculocutaneous perforators (Figure 1). Knowledge of the vascular anatomy allows some degree of planning for flap harvest, with anticipated more skill and experience required when dissecting a musculocutaneous perforator over a direct cutaneous vessel flap, for example. With the advancement of microsurgical techniques, however, focus has shifted from the ease of flap harvest to the selection of an ideal flap to recreate an aesthetic and functional upper extremity.
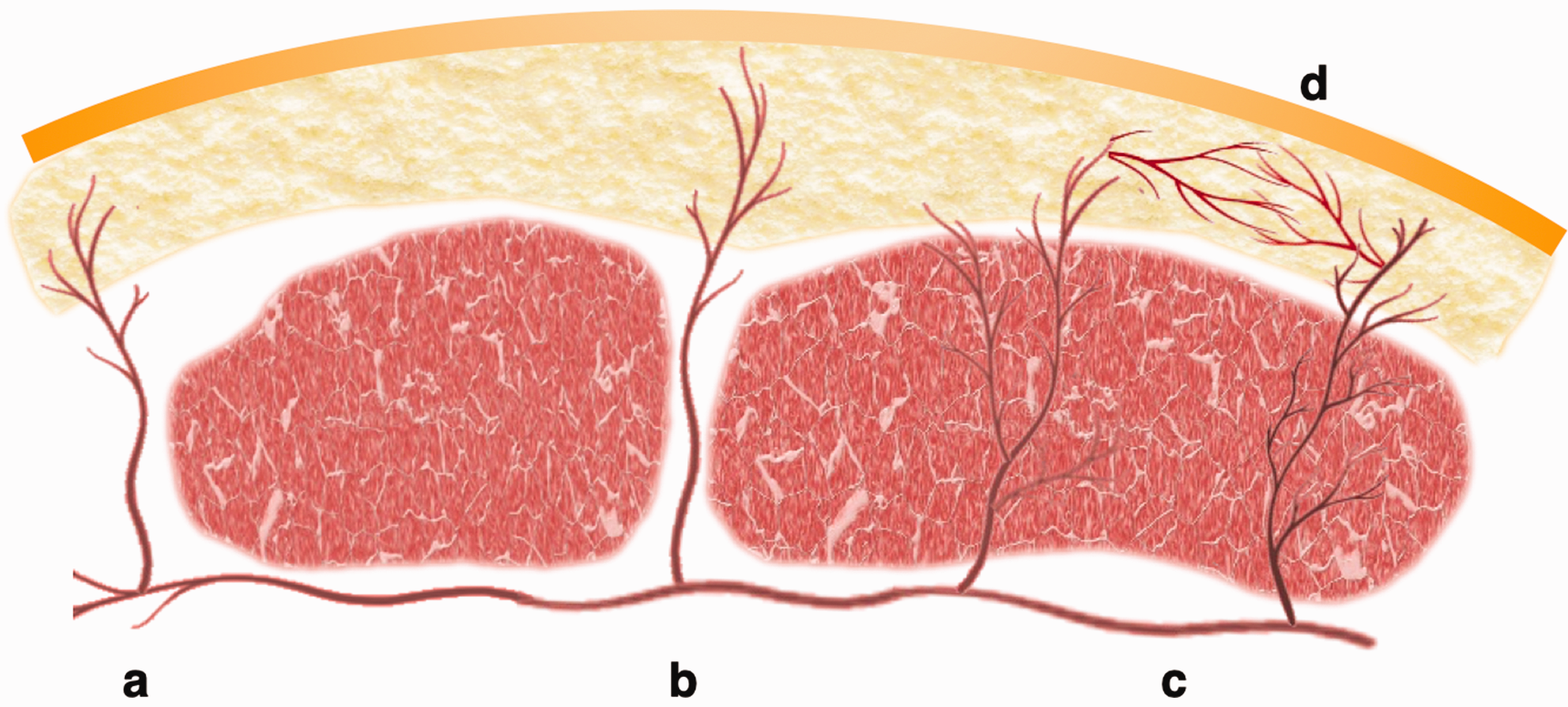
Vasculature of flaps. (a) Direct vessels pierce the deep fascia without traversing any structures. (b) Septocutaneous vessels travel along the muscle septae before piercing the deep fascia. (c) Musculocutaneous perforators penetrate the muscle before entering deep fascia and (d) Perforators are connected to other perforators via suprafascial choke vessels.
Direct vessel-based flaps
Early free flap harvest relied upon direct vessels that pierce the deep fascia without traversing other structures, allowing for a relatively easy and straightforward dissection. The free groin flap, considered the first free tissue transfer, was an early option for resurfacing sizeable wounds but is inaptly thick for more distal coverage. Modification to the superficial circumflex iliac artery perforator permits harvest as thin as 3 mm, allowing for customized flap thickness for contouring for the distal upper extremity, such as the dorsal hand or forearm (Chen et al., 2019; Wagner et al., 2020).
If pursuing mobile fascial reconstruction, particularly important in the dorsal hand, the free temporal fascia flap, based on the superficial temporal artery, is an excellent option for ultrathin coverage promoting underlying tendon excursion (Figure 2) (Carty et al., 2010; Müller-Seubert et al., 2021). Caution is warranted, however, in cases indicating secondary re-elevation for tendon reconstruction, for which such a delicate flap likely will not tolerate. While dissection of direct vessels is relatively uncomplicated, skin flaps meeting these criteria are limited in number compared to those requiring intra-muscular dissection.
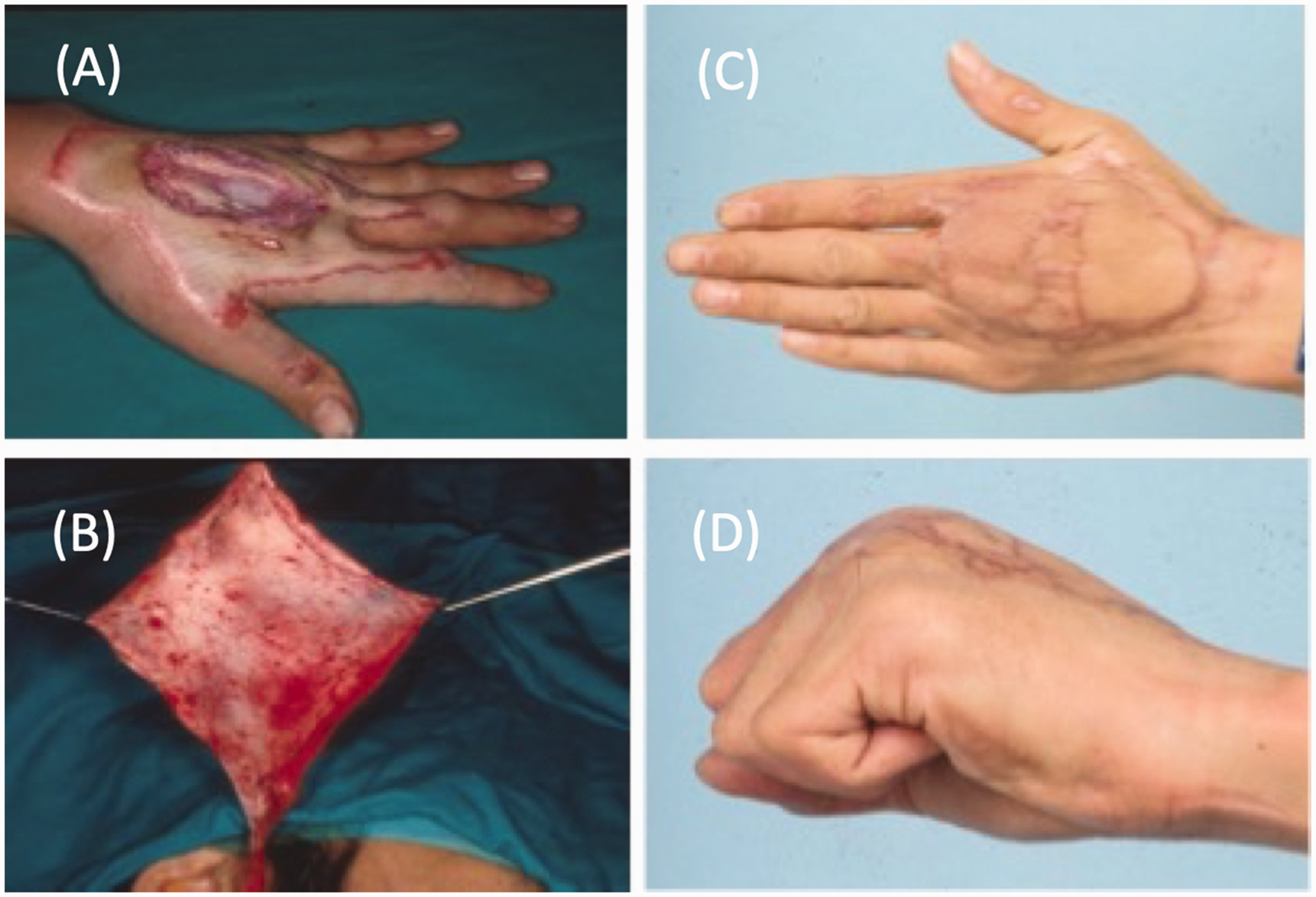
Free temporal fascia flap for dorsal hand coverage. (a) Left dorsal hand defect with exposed extensor tendons. (b) Thin temporal fascia flap raised on the superficial temporal vessels. (c) Long-term follow-up with appropriate cosmesis of the dorsal hand and (d) Good excursion and gliding of the extensor tendons beneath the fascia flap.
Septocutaneous vessel-based skin flaps
Septocutaneous vessel-based flaps, commonly pursued before the perforator flap era, similarly allow for a straightforward harvest. For example, the lateral arm flap provides consistent septal perforators from the posterior radial collateral vessels (Figure 3) (Ye et al., 2019). Limitations of this flap include a modest size, relatively short pedicle and excessive bulk, making it unsuitable for distal forearm or hand resurfacing. Surgeons must be prudent of using another part of the ipsilateral upper extremity as a donor site, given the conspicuous scarring and preclusion of a two-team approach. Thus, we consider the lateral arm flap as a secondary option in our institution and often a relative contraindication to upper extremity reconstruction.
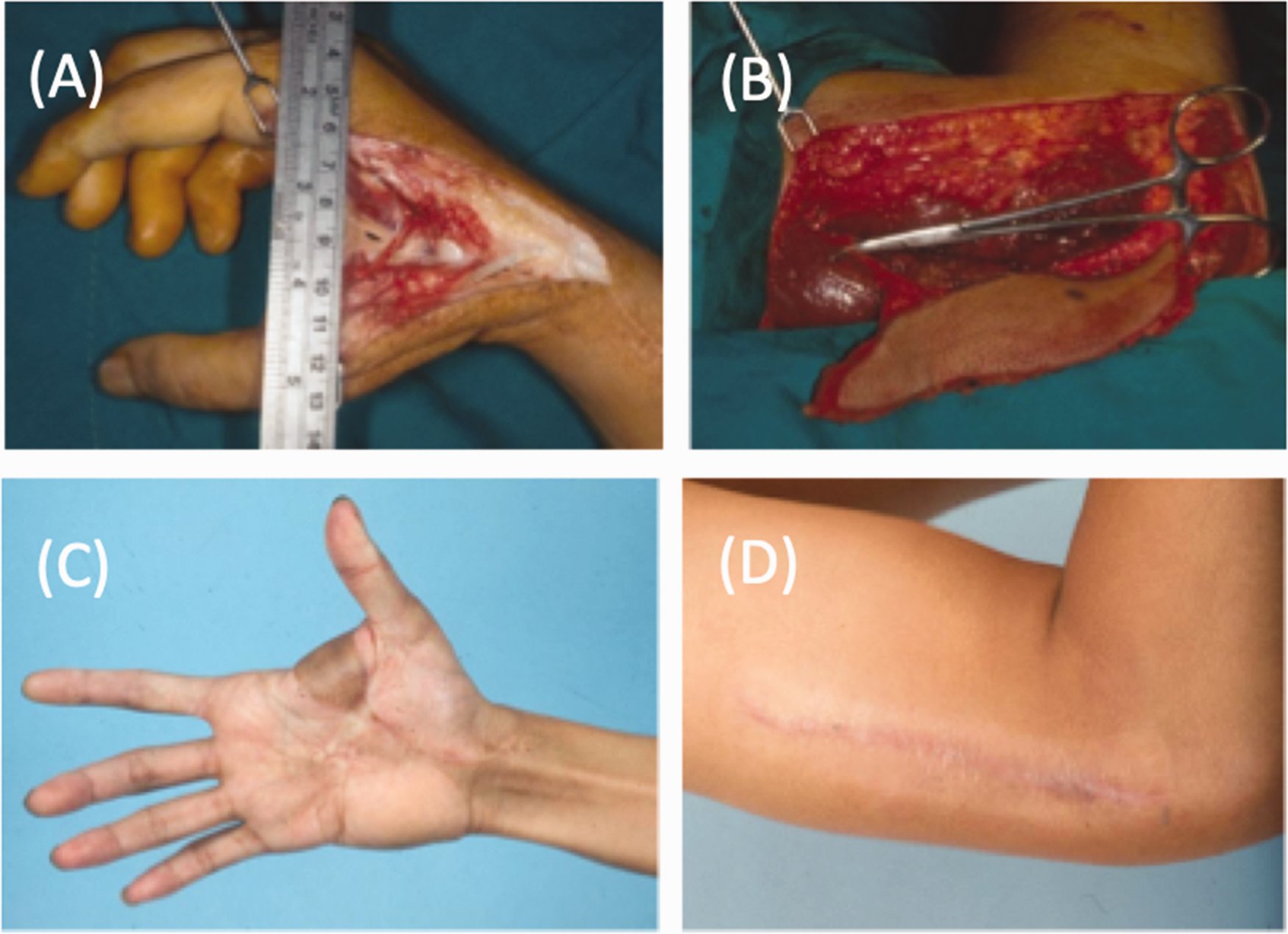
The lateral arm flap for reconstruction of a first webspace defect. (a) A large defect involving the dorsal and volar first webspace, requiring pliable coverage. (b) The lateral arm flap harvested on the posterior radial collateral artery. (c) Follow-up with excellent first webspace mobility and (d) Visible scar at the donor site, making this donor site less desirable.
The versatile free fibular osteoseptocutaneous flap is commonly used to reconstruct upper extremity defects involving long bone, skin and soft tissue bulk (Figure 4). In the upper extremity, free vascularized fibula flaps are perhaps best known for primary oncologic reconstruction of the humerus, radius and ulna (Claxton et al., 2020; Landau et al., 2018). Inset can be accomplished in a flow-through fashion, allowing revascularization of the distal extremity while simultaneously replacing all constituents of the resected tissue. Given its segmental blood supply, the fibula can alternatively be adapted for recreating multiple metacarpal bones or phalanges (Simsek et al., 2012).
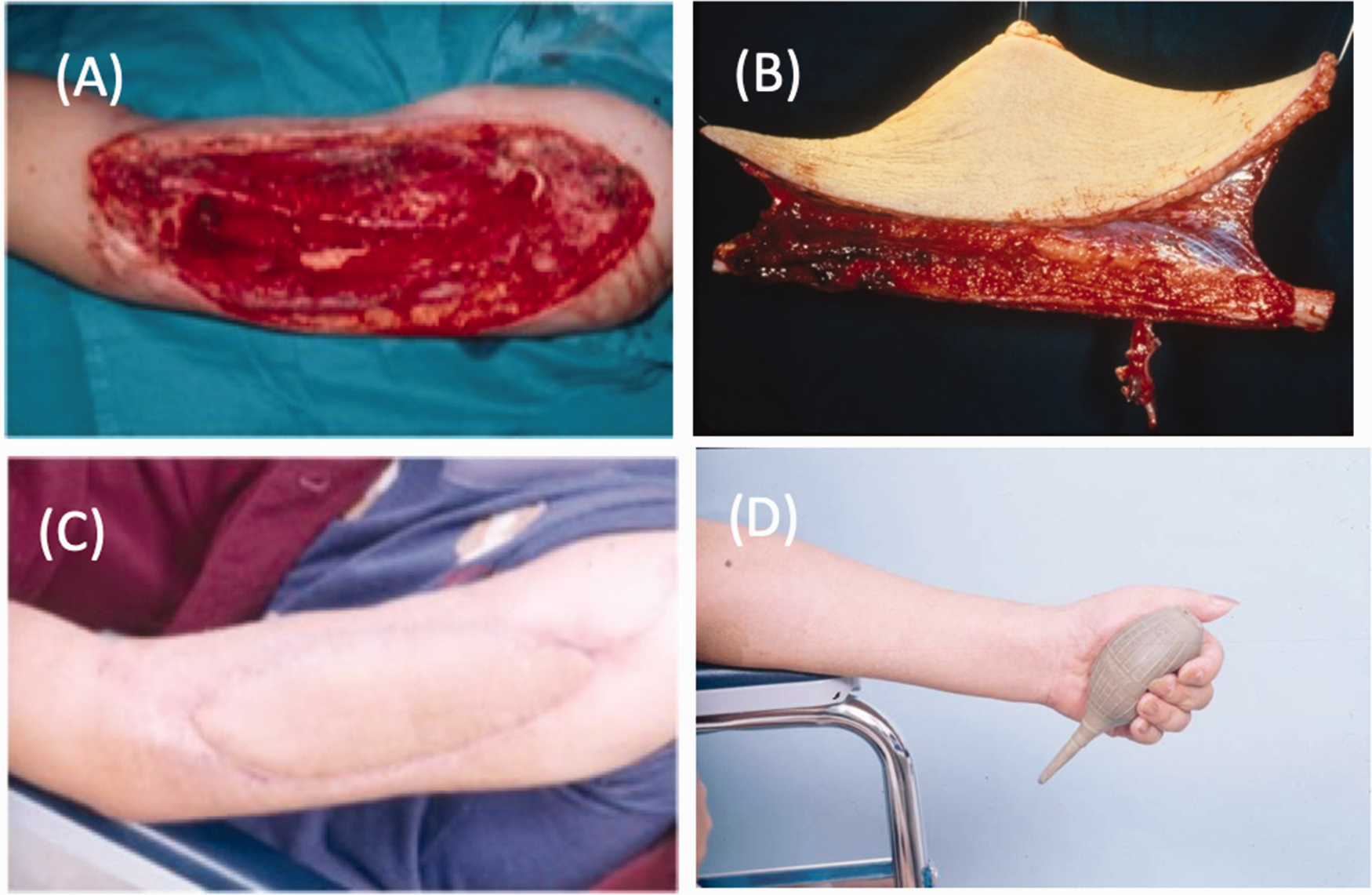
Free osteoseptocutaneous fibula flap for reconstruction of a compound humerus defect. (a) Large left upper arm defect including soft tissue, muscle and humerus bone. (b) Free osteoseptocutaneous fibular harvested with a skin paddle and (c, d) Long-term follow-up after coverage with good skin and dead space approximation.
Perforator-based free tissue transfer
There are various definitions of what a perforator flap is. At Chang Gung Memorial Hospital, we define perforator flaps as those requiring intramuscular dissections in order to isolate the cutaneous vessels that penetrate muscle, in addition to piercing fascia before entering the skin (Wei et al., 2001). The advent and understanding of the perforator flap ushered in a new era, and as proficiency in intra-muscular vessel dissection became commonplace, a profound number of novel flaps began to emerge.
For example, knowledge of the circumflex femoral vessel topography allows elevation of the anterolateral/anteromedial thigh, vastus lateralis, tensor fascia lata, rectus femoris and iliac crest independently and collaboratively. Given a consistent pedicle, flap size up to 450 cm2 and versatility, the anterolateral thigh (ALT) free transfer is considered an ideal flap for soft tissue coverage (Chang-Cheng Chang et al., 2016; Suondoh et al., 2020; Yang et al., 2020). What makes this flap indispensable is its customizable size, thickness, composition and pedicle configuration, allowing contour of almost any defect to be reconstructed (Ellabban et al., 2021). ALT flaps can also be inset in a flow-through fashion for sizeable wounds, as a chimeric flap for multiple areas or as a composite flap including muscle and bone (Figures 5 and 6) (Hsiao et al., 2013; Yang et al., 2020).
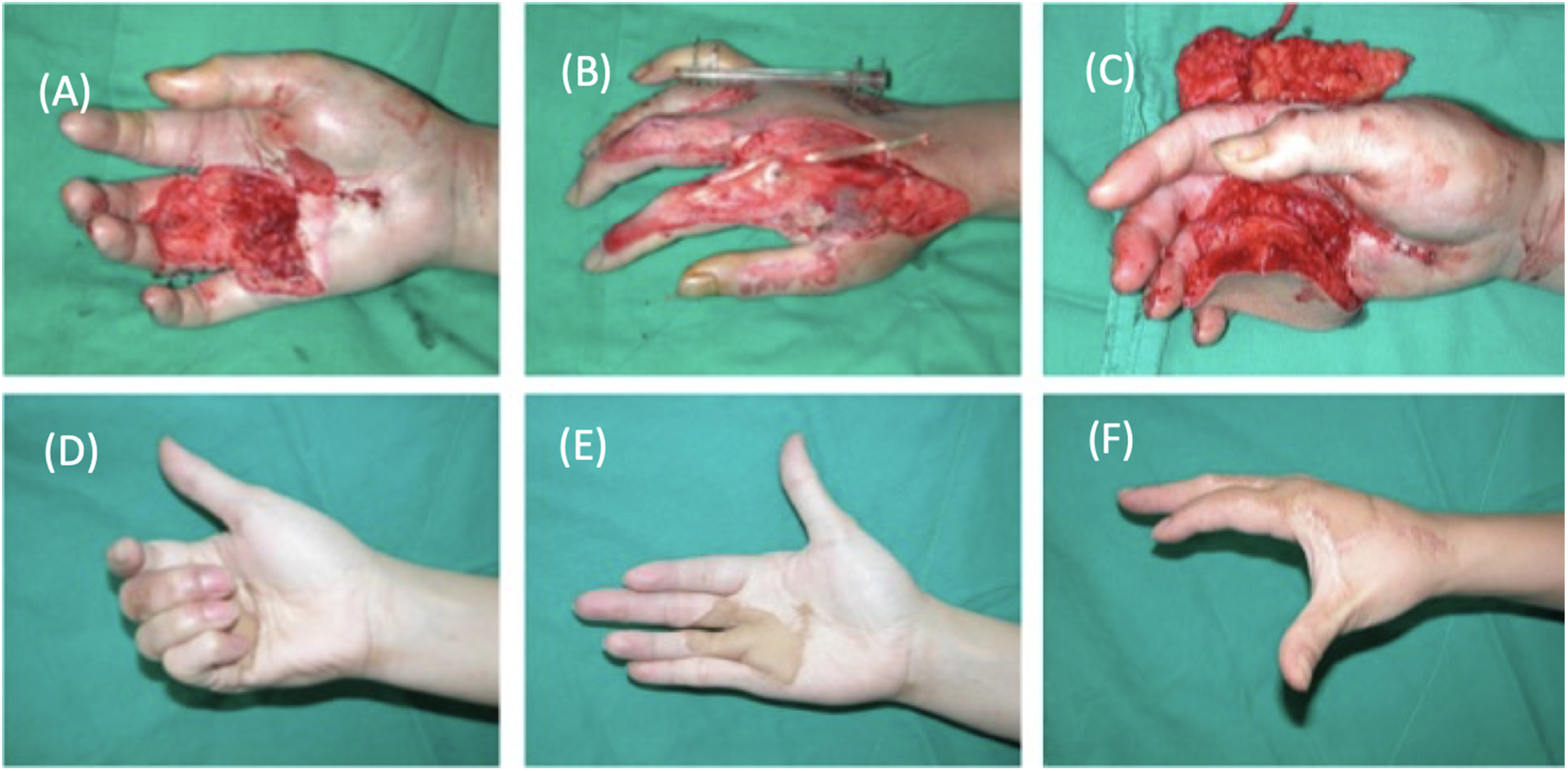
Chimeric anterolateral thigh flap for reconstruction of a composite dorsal and volar hand injury. (a, b) Volar and dorsal hand coverage defect with exposed tendons, muscles and neurovascular structures. (c) Inset of a thinned chimeric anterolateral thigh flap in both the dorsal and volar components and (d–f) Good contour and vascularized coverage of a fully functional hand without contractures.
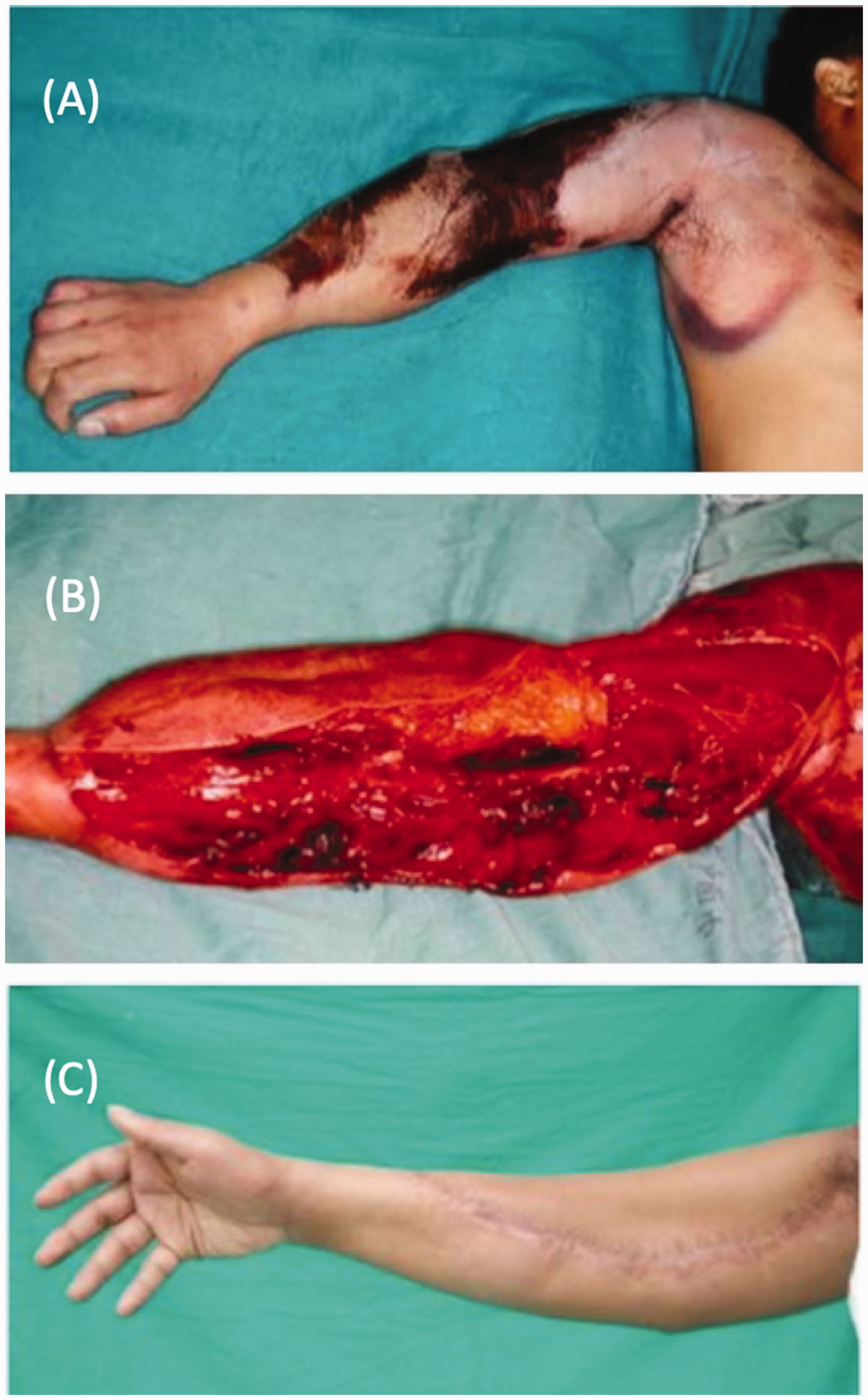
Flow-through anterolateral thigh flap for upper arm devascularizing injury. (a) Significant crush and burn injury involving the upper arm and forearm. (b) After debridement including brachial artery resection, the vessel was immediately reconstructed with a flow-through anterolateral thigh flap with the lateral circumflex femoral artery as the new vascular conduit and (c) Long-term postoperative follow-up with vessel vascularized, functional limb.
More recent developments of the perforator flap family include the thin medial sural artery perforator flap and the profunda artery perforator flap, commonly used for resurfacing the hand and upper arm, respectively (Daar et al., 2019; Deek et al., 2020).
The thoracodorsal system (Hattori et al., 2020; Lin et al., 2006) similarly bears origin to the cutaneous thoracodorsal perforators, latissimus dorsi, serratus anterior, rib and scapular bone. By using microsurgical dissection techniques, the thoracodorsal perforator flap can be thinned subdermally to a thickness of 3 mm for distal extremity coverage (Hattori et al., 2020).
Free-style free tissue transfer
It seems the superlative flap is one that can be tailored to the exact components, thickness, pliability and vessel size necessary, while at the same time reducing donor site morbidity and allowing for faithful reconstruction of the deficit tissues. As our technique for perforator dissection progressed, so did the search for such customization. The free-style free flap thus represents a natural evolution from the perforator flap concept.
In 1975, Taylor and Daniel performed the first retrograde dissection by identifying perforators through the gastrocnemius and tracing them towards the sural artery (Taylor and Daniel, 1975). Koshima et al. (1998) were the first to propose raising a perforator flap without a specified pedicle. In 2003, Wei and Mardini (2003, 2004) developed the free-style free flap technique, which takes advantage of intra-muscular dissection of any surface of the body unfamiliar to the surgeon
Free-style perforator dissection involves a retrograde flap harvest technique; first identifying the desired tissue from the ideal donor site based on skin quality, thickness and colour match, then locating a perforator with Doppler ultrasound, followed by vessel dissection to the desired length and vessel size, including submillimetre diameters. While free-style flaps are often indicated for smaller defects, dimensions have been reported up to 187 cm2 and pedicle length up to 12 cm (Wei and Mardini, 2004). At the recipient site, unnamed vessels can similarly be explored for anastomosis. Thus, surgeons forgo traditional anatomic limitations, and the microsurgeon is free from the constraint of a previous known vascular pedicle needed for conventional free flaps. Instead, flaps based on any skin vessel, named or unnamed, can be relocated to any suitable vessel at the recipient site, as defined by the ‘perforator-to-perforator’ concept.
Such a free-style free tissue transfer technique permits unparalleled adaptation of soft tissue transfer, particularly important in the upper extremity where defects involve multiple anatomic regions with varied reconstructive requirements, such as the need for a thin, pliable flap (Figure 7). In addition, distant donor sites can be chosen to allow for a two-team approach. Indications include small- to moderate-sized defects, which do not require a long pedicle. While seemingly complicated, only two techniques are required: (1) the ability to map cutaneous vessels with Doppler ultrasound; and (2) the ability to perform intramuscular dissection of perforators. With the aforementioned skills, one can harvest a flap from anywhere and in any form, dependent upon the choice of skin vessel.
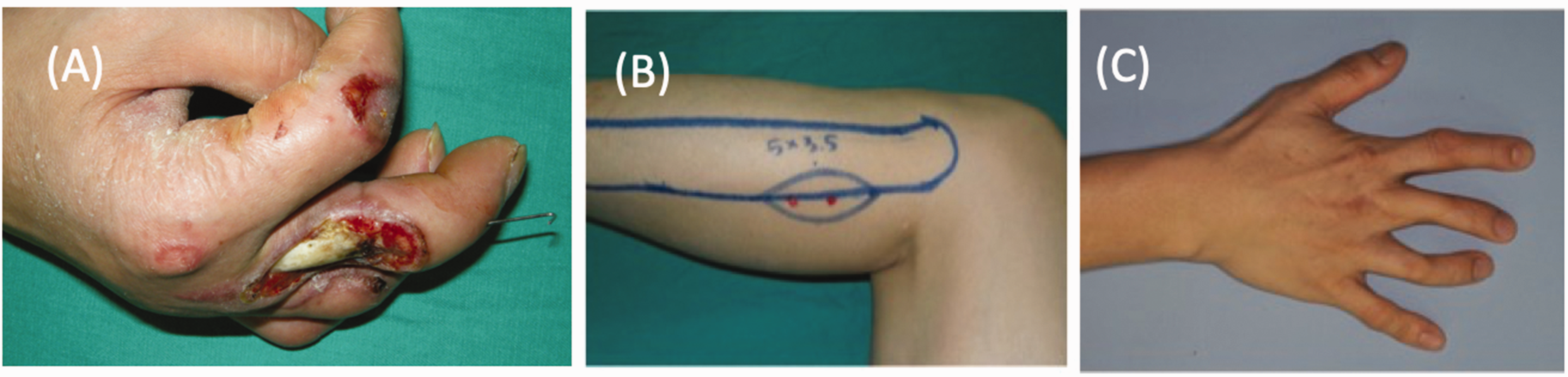
Free-style free flap for reconstruction of a dorsal finger defect. (a) Right dorsal middle finger defect with exposed tendons. (b) Perforators identified with Doppler ultrasound, over an anatomically unfamiliar donor site in the left lateral lower leg and (c) Intraoperative identification of the perforator. (d) Long-term follow-up with good results.
Considerations when choosing a flap in the upper extremity
While we focus on flap vascular types in this discussion, flaps are ultimately selected based on the anatomic restoration needed. The upper extremity is an exposed and aesthetically prominent region. Colour and skin quality mismatch should be avoided, particularly in non-Caucasian patients. For this reason, and the abundance of other suitable donor anatomy, we choose to avoid the upper extremity as a donor site, as previously mentioned.
The glabrous skin of the palm and ventral fingers should be matched with comparable tissues from the plantar regions of the foot. A unique option for finger pulp reconstruction is the free glabrous skin flap from the toe, replacing like with like, with emphasis on a stable and powerful pinch (Figure 8) (Kwon et al., 2021). The medial plantar septocutaneous vessel-based flap also provides elegant glabrous resurfacing of the palm, allowing for robust, shear-resistant tissue ((Troisi et al., 2019).
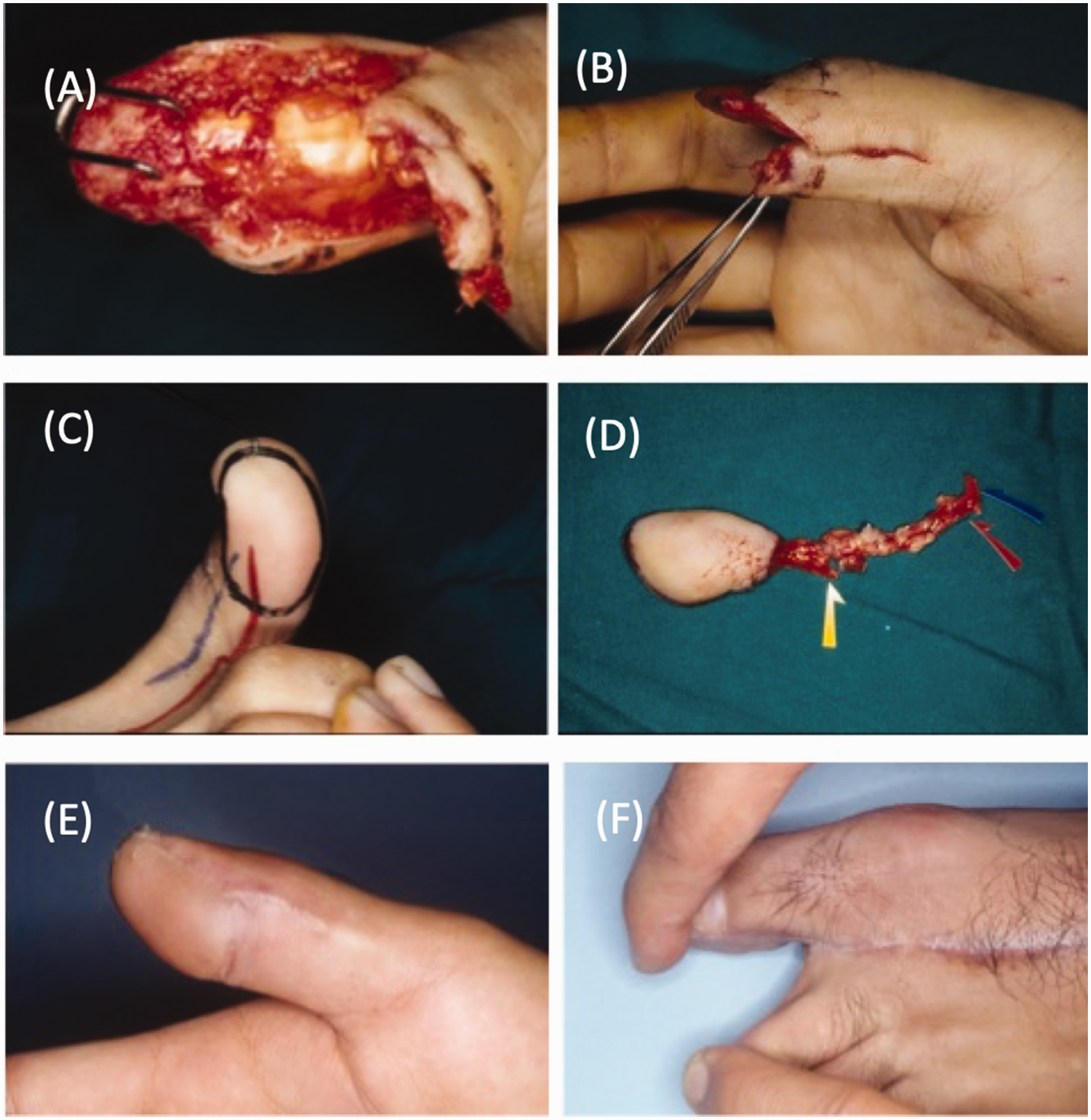
Free glabrous first toe flap for thumb pulp reconstruction. (a) Thumb pulp defect with exposed bone. (b) Thumb pulp defect could not be resurfaced with advanced Moberg flap. (c) Planned glabrous toe flap with direct digital vessel perforators. (d) Glabrous toe flap raised with the digital nerve, artery and vein, respectively. (e) Long-term appearance of the sensate glabrous skin flap on the thumb pulp and (f) Donor site with minimal aesthetic or functional deficit.
Injured webspaces of the hand can lead to limited range of motion, severe contractures or even syndactylization. Arterialized venous flaps are common for single webspace contractures but are plagued with increased complication rates. The advent of shunt-restriction optimized the peripheral perfusion through the venous flaps and has led to decreasing rates of congestion, making this a reliable, thin and versatile alternative (Lam et al., 2013; Lin et al., 2016; 2010).
Equivalent toe webspace tissue can be transferred on the dorsal metatarsal artery for multiple webspace coverage benefiting from healthy vascularized tissue (Figure 9). The dorsal hand is draped with particularly fragile thin skin devoid of subcutaneous tissue bulk. As previously mentioned, the direct temporal fascia flap is an option for unrestricted tendon gliding but is disadvantaged by the need for a separate skin graft and poor quality for future re-elevation. Although fasciocutaneous flaps have historically been considered bulky, both the anterolateral thigh flap and the superficial circumflex iliac artery perforator flap could be significantly thinned to provide excellent coverage (Chen et al., 2019; Mekky, 2018). Fasciocutaneous flaps comprise both soft tissue coverage and fascial gliding for tendon excursion, allowing safe defect coverage and tendon reconstruction simultaneously.
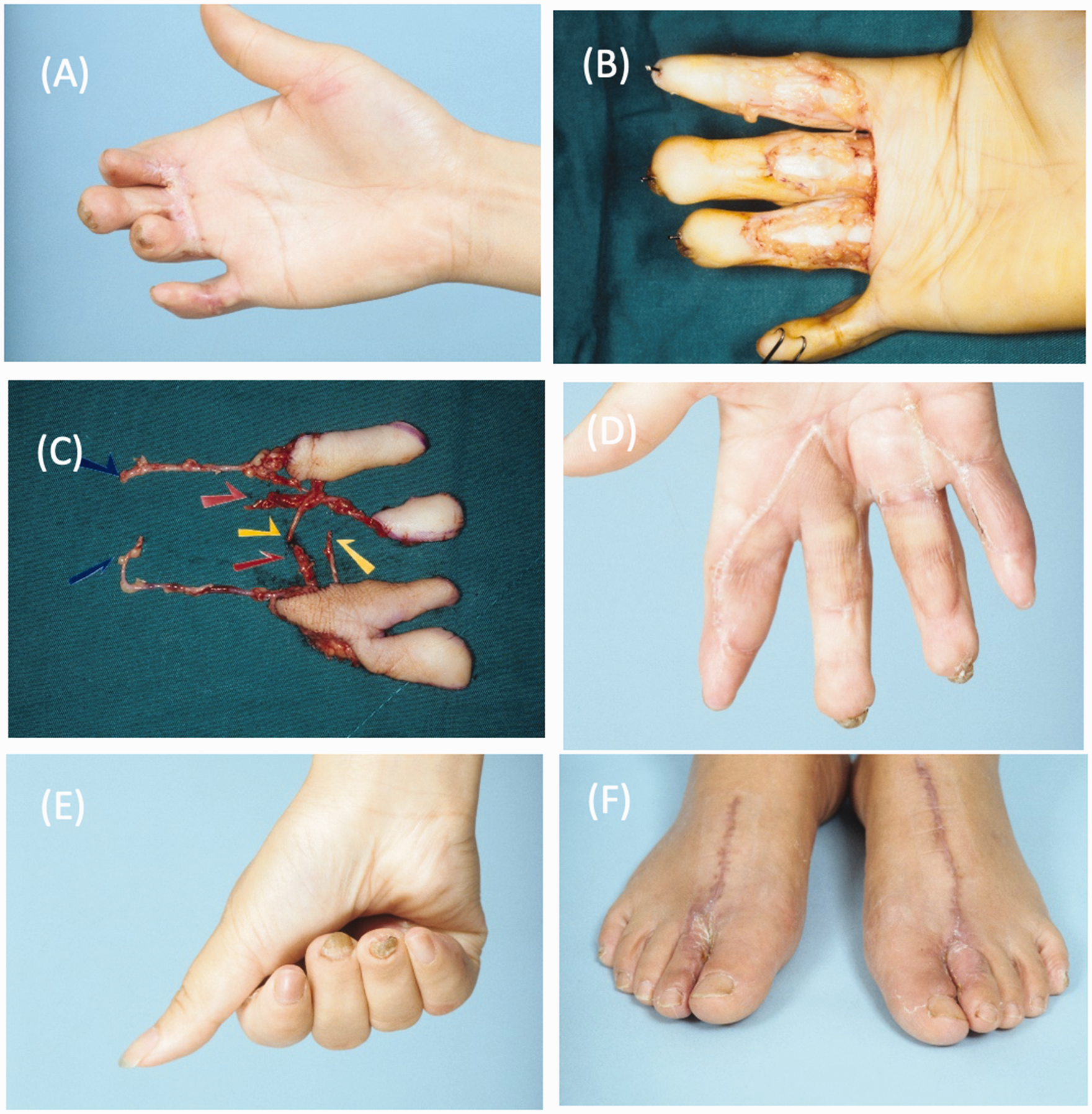
Post-traumatic amputation and syndactyly reconstructed with surgical release and bilateral foot webspace flaps. (a) Post-traumatic palmar defect with scar syndactyly between the index, middle and ring fingers. (b) Webspaces and volar skin defects after release. (c) Bilateral first webspace flap from the feet, harvested for reconstruction and (d–f) Appearance and function of the reconstructed hand and donor sites.
Although soft tissue restoration is emphasized in this article, bony defects are often encountered in upper extremity injuries. Dependent on the size and mechanism of injury, options for bone reconstruction range from bone grafting, membrane induction, secondary distraction, pedicled bone flaps, to free osseous flaps. Special mention is made for the increasingly popular medial femoral condyle flap, based on the descending genicular artery (Figure 10) (Zhou et al., 2021). This versatile flap provides sizeable corticocancellous bone as well as the harvest of cartilage to provide an articular surface suitable for carpal bone or intercondylar defects (Hsu et al., 2017). If soft tissue is desired, a skin paddle can be harvested from direct branches from the proximal saphenous artery, or distal septal vessels or intramuscular perforators arising from the vastus medialis and, if needed, a segment of the vastus medialis can be included. As such, this flap is rising to the forefront of bony hand reconstruction.
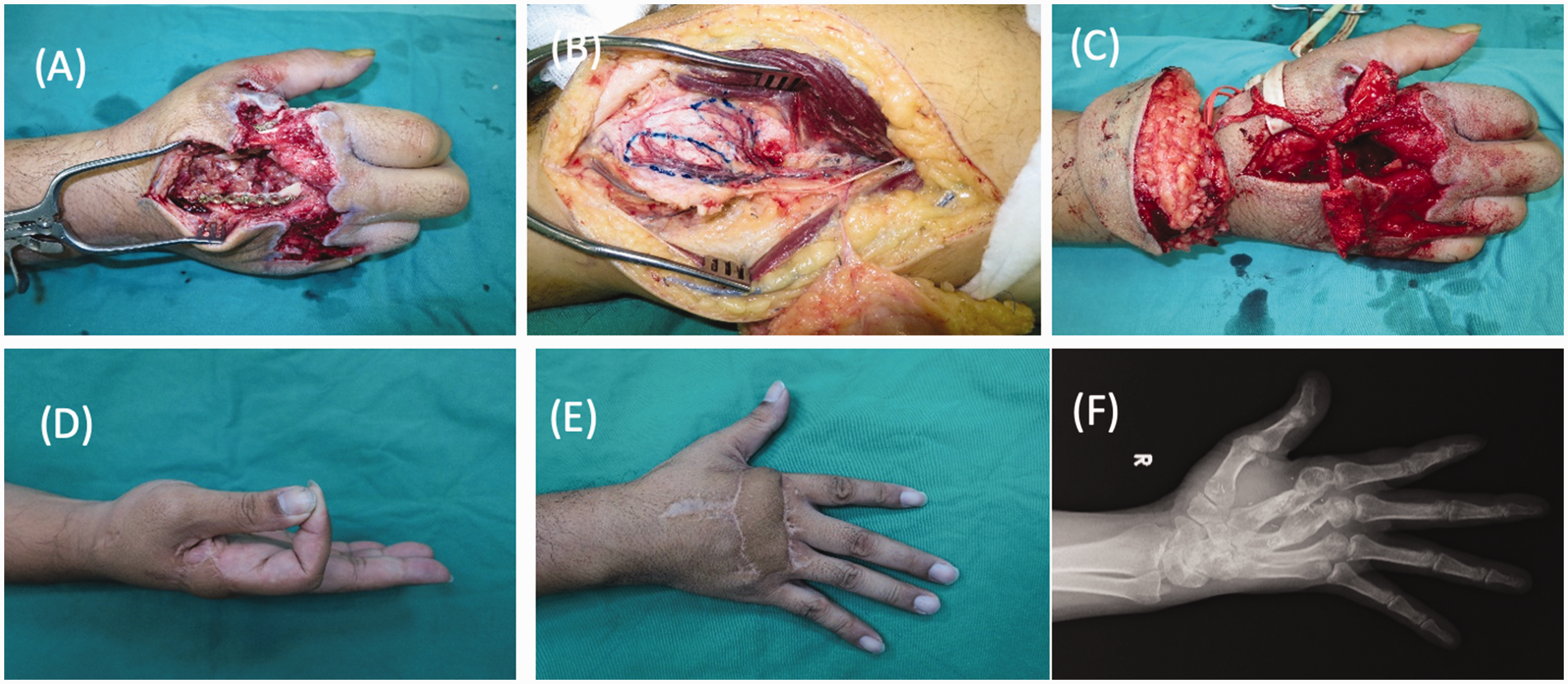
A medial femoral condyle bone harvested for with skin flap. (a) Preoperatively, a dorsal hand defect with second and third metacarpal bone loss. (b) Chimeric medial femoral condyle osteocutaneous flap (two separate condyle bones) designed. (c) Inset of chimeric medial femoral condyle flap and (d–f) Appearance and radiological image at 33-month follow-up.
Management of the recipient bed
The recipient wound is as important, if not more so, than the choice of flap donor. It is crucial for the surgeon to recognize tissue status: (1) viable versus non-viable; (2) severely contaminated versus manageable contamination; and (3) expendable tissues, such as muscles with redundant functions, versus non-expendable tissues, such as major nerves. Irreversibly contaminated wounds can fester severe infections that will compromise even the most robust reconstructions. Clear identification of expendable structures will allow aggressive debridement. Tissue viability, and therefore the size of the defect, may not be immediately apparent. If needed, the use of biologic dressings, such as porcine skin or Integra (Integra LifeSciences, Hudson, NH, USA) may salvage borderline structures, and at least allow for declaration of non-viable underlying tissues (Reynolds et al., 2018; Wang et al., 2020).
Occasionally, the area in need of resurfacing may be too extensive for adequate coverage, such as in multiple finger degloving injuries. Surgeons must begin with the end in mind—to recognize what can be reconstructed at a delayed stage, sacrifice non-vital areas and consider the utility of heterotopic transfers. A detailed description of toe-to-hand transfers is beyond the scope of this article, but suffice to say, microsurgeons must remember this option in their armamentarium when considering structures to be sacrificed (Figure 11). Non-expendable anatomy, such as the metacarpal phalangeal or interphalangeal joints, are prioritized for coverage. Early coverage of even grossly non-viable major nerves may aid in future neurotization, maintaining the remaining structural continuity for proper tension and length of second-stage nerve grafts.
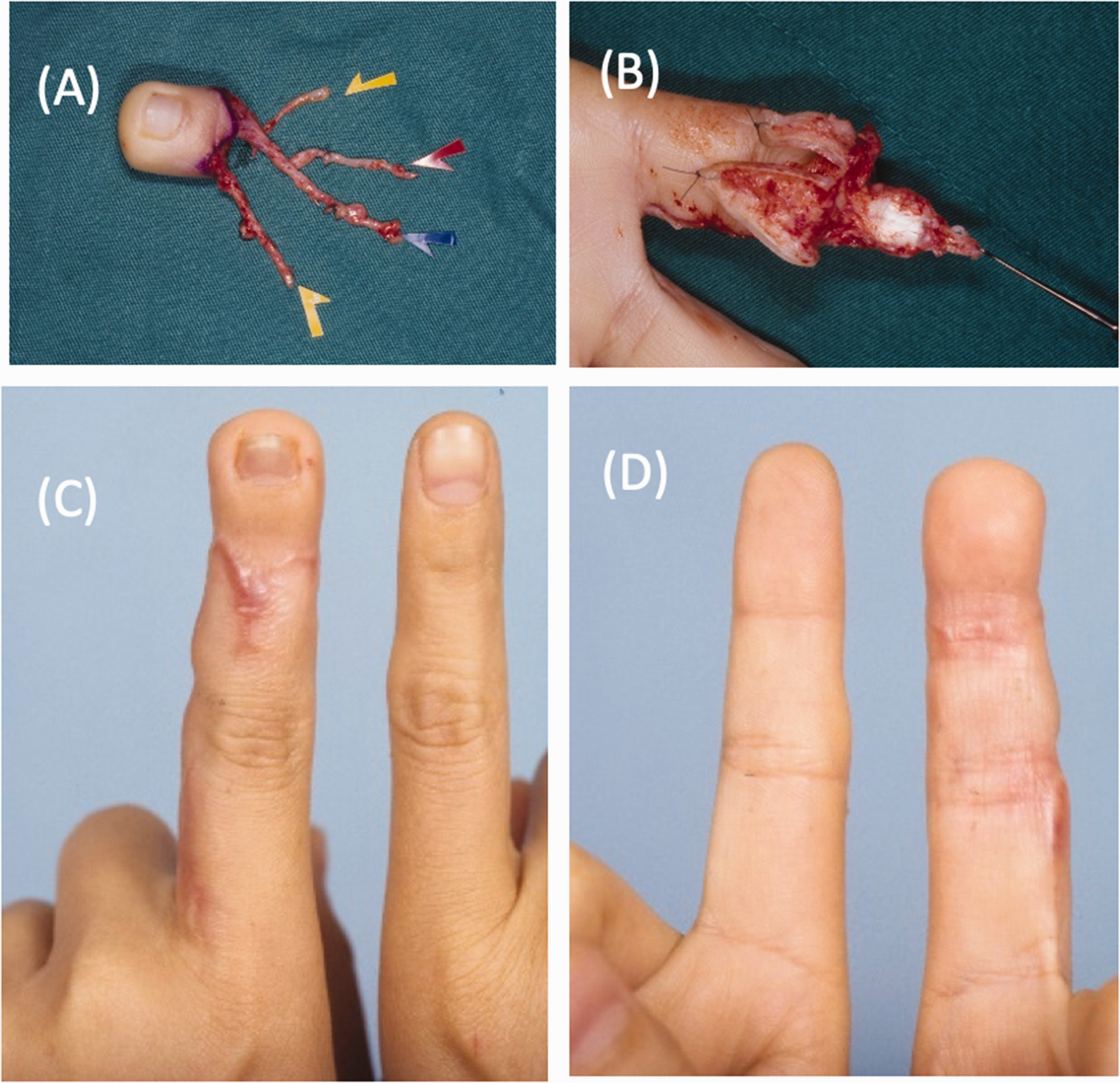
Distal index finger amputation reconstructed with a second toe wrap-around flap. (a) Loss of nail and pulp in distal index finger. (b) Second toe wrap-around flap harvested for reconstruction. (c, d) Appearance after second toe wrap-around transfer reconstruction.
Final considerations
Given our immense choice of free tissue transfers, made possibly infinite with the inclusion of free-style flaps, there should be little excuse for selecting an inappropriate flap reconstruction nowadays. Similarly, we should also be able to optimize the morbidity of the donor site, even as we consider the defect to be reconstructed. Several considerations should be emphasized:
The goal is not just to heal the wound, but to maintain maximal function, with secondary reconstruction in mind. A good choice of flap accelerates the rehabilitation process and reduces the need for further surgeries and inpatient encounters. The lower extremity has emerged as an ideal donor site for the upper extremity, boasting similar tissue types, a multitude of flap options, inconspicuous donor appearance and facility of a two-team approach. The upper extremity, on the whole, should be avoided as a donor site. While the posterior interosseous, lateral arm and radial forearm flap were initially popular options, poor, visible upper extremity scarring precludes their use if other suitable locales are present. Co-surgeons should consider distant donor sites that allow two teams to work simultaneously. Local-regional flaps were once the traditional preference for upper extremity reconstruction, citing ease of elevation, but offer subpar donor scars, coverage dimensions and limited adaptability. As we hone our abilities in microvascular free tissue transfer, we now consider free flaps as first-line options for upper extremity resurfacing. While we favour thin coverage for the distal upper extremity, fragile flaps may not tolerate multiple attempts at re-elevation during future procedure, such as tendon or bony reconstruction. In addition, adequate durability can aid early mobilization. Particular to the upper extremity, where defects are often small, there are abundant donor sites limited only by the presence of Doppler ultrasound detection. As microsurgeons are becoming comfortable with submillimetre anastomoses, flaps can be designed with shorter, smaller pedicles, in any location previously unfamiliar to the surgeon. Thus, we believe free-style free flaps will forge the future of upper extremity resurfacing.
Conclusion
Our institutional experience in the evolution of flap anatomy and dissection techniques has convinced us of the prudency in the selection of reconstructive options, whether that be local, regional or distant flaps, especially for defects in the upper limb. As such, we advocate for microsurgical training for all hand surgeons, to harness the ability to perform any form of reconstruction. Our knowledge of upper limb anatomy and the ability to appreciate the demands of its form and function mean that we have a responsibility to optimize reconstruction for our patients while minimizing donor defects. As our technical capabilities continue to evolve, we can look forward to a focus on the true goal of reconstructive surgery: recreating the limb before the defect.
Footnotes
Declaration of conflicting interests
The authors declare no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Informed consent
Written informed consent was obtained from the patients for their anonymized information to be published in this article.
