Abstract
The anconeus nerve is the longest branch of the radial nerve and suitable as a donor for the neurotization of the axillary nerve. The aim of this study was to map its topographical course with reference to palpable, anatomical landmarks. The anconeus nerve was followed in 15 cadaveric specimens from its origin to its entry to the anconeus. It runs between the lateral and the medial head of the triceps before entering the medial head and running intramuscularly further distal. Exiting the muscle, it lies on the periosteum and the articular capsule of the elbow, before entering the anconeus muscle. Two types of anconeus nerve in relation to branches innervating triceps were found: nine nerves also innervated the lateral triceps head, while the other six only contributed two branches to its innervation. The course of the anconeus nerve is important for harvesting as a donor nerve and to protect the nerve in surgical elbow approaches.
Keywords
Introduction
The anconeus nerve is the longest branch of the radial in the upper arm. The course of this nerve and its branching pattern have been described by a few authors (Ahn et al., 2011; Bertelli and Ghizoni 2014; Bertelli et al., 2007; Gerwin et al., 1996). Reports of the exact trifurcation point of the radial nerve (and so the origin of the anconeus nerve) have been variable. Gerwin et al. (1996) described the trifurcation point at the lateral aspect of humeral diaphysis, while Özer et al. (2006) described it at the posterior aspect of the humerus, but medial to the humeral diaphysis. Thereafter, the anconeus nerve travels distally along the posterior aspect of humerus to innervate the medial head of triceps and anconeus muscle.
The precise anatomy of the anconeus nerve is an important subject for study, first to protect it during surgical approaches to the elbow (O'Driscoll, 2000; Özer et al., 2005; Sun et al., 2015) and second because it may be a potential donor for transfer to other peripheral nerves of the upper extremity (Bertelli and Ghizoni, 2014; Bertelli et al., 2007). In the previous descriptions, we have not seen clear topographic relationships between the nerve and applicable bony landmarks, and therefore saw the necessity to review the course of the anconeus nerve. In addition, precise topographical mapping of the course of the nerve could answer the current controversies. The aim of this study was to take a precise look at the anconeus nerve, including its origin and relationship to muscular branches to the triceps, and to map the topographical course in relation to clear anatomical landmarks.
Methods
We performed anatomical dissections of 15 fresh frozen upper limb specimens from eight cadavers. The cadavers were placed in prone position and the dissection was started from the posterior aspect of the arms using magnification loupes (4.5×). After removal of skin and subcutaneous tissues, the radial nerve was visualized between the lateral and long heads of triceps (Figure 1). The nerve was followed distally until the anconeus nerve was discernible and started to separate from the radial nerve, without proximal intraneural dissection. This point was defined as the apparent origin of the anconeus nerve, and the position was recorded in relation to the intercondylar line. The anconeus nerve was traced distally from its originating point on its course between the lateral-long head and medial head, piercing the medial head of the triceps up to its entry to the anconeus (Figure 2). Nerve branches innervating the medial head of triceps were visualized and branches to the lateral head of the triceps muscle were recorded.
Anatomical dissection of the lateral aspect of a right arm and elbow showing the radial nerve (white star) and the anconeus nerve (yellow star). Critical passage of the anconeus nerve (coloured in yellow) proximal to its entry to the anconeus muscle (yellow star). Note the close contact with the elbow joint capsule (yellow cross) and the periosteoum of the distal humerus (green cross).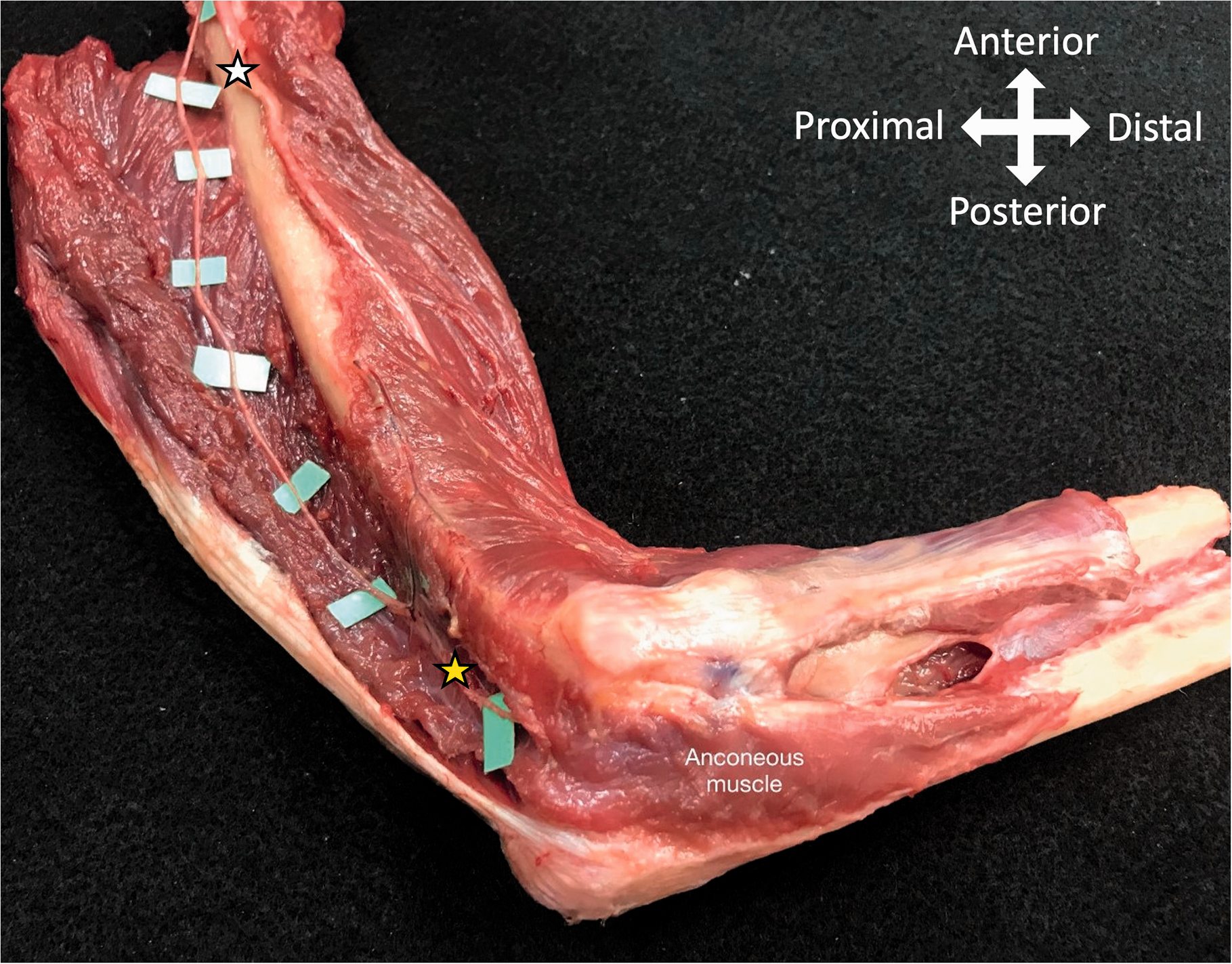
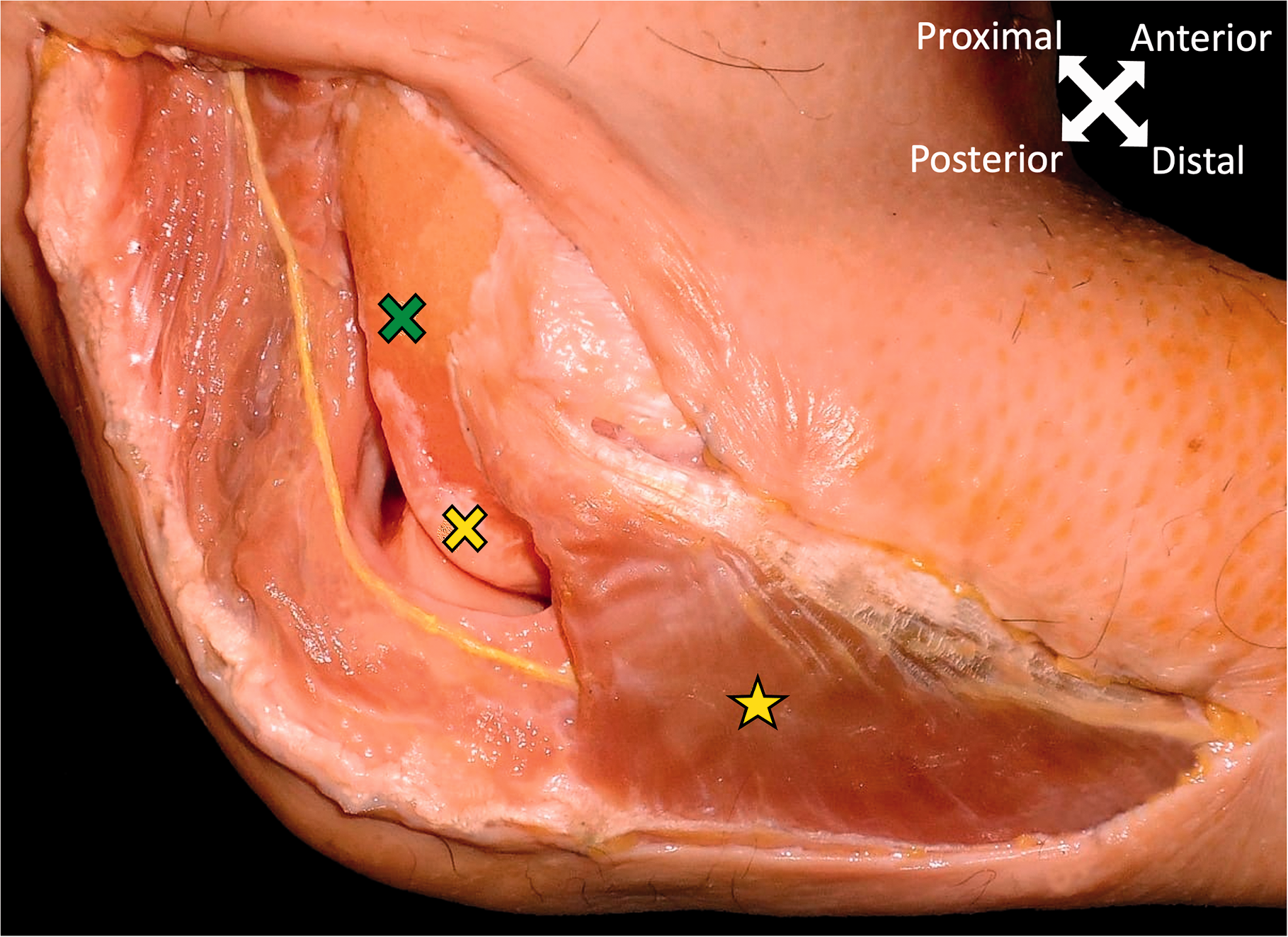
Some easily discernible bony landmarks (tip of the olecranon, medial and lateral epicondyle) were defined, and measurements were performed, including the intercondylar distance and the distance between the lateral epicondyle and the tip of the olecranon. Then the relations between the topographic course of the anconeus nerve and chosen landmarks were assessed. The diameter of the nerve was measured at its apparent origin and at the point of its muscular insertion. Measurements were made using a calliper, with the elbows flexed at 90° and the forearm in neutral rotation. The intersection points of the anconeus nerve and the intercondylar line and the line between the tip of the olecranon and the lateral epicondyle were calculated and reported as a ratio.
Results
Fifteen fresh frozen elbows from eight cadavers (two male, six female) with a mean age of 77 years (range 64–91) were used for this study. In general, the anconeus nerve separated from the radial nerve (Figure 3), with an initial mean diameter of 1.5 mm (standard deviation (SD): 0.2; range 1.1–1.8), a mean of 164 mm (SD: 15; range 142–195) proximal to the lateral epicondyle (Table S1), on the postero-medial side of the humerus. The nerve ran between the lateral and the medial head of the triceps muscle, before entering the medial head at a mean of 102 mm (SD: 24; range 69–147) proximal to the intercondylar line and running intramuscularly towards the distal humerus. Exiting the muscle for a short distance, the nerve lay on the periosteum of the distal humerus and the dorsolateral aspect of the elbows’ joint capsules, before entering the anconeus muscle with a mean diameter of 0.5 mm (SD:0.1; range 0.3–0.8) (Figure 2). Entering the anconeus muscle, the nerve passes the line between the olecranon and the lateral epicondyle at a mean of 57% (SD 6) measured from the olecranon (Figure 4, Table S2).
Anatomical dissection of the posterior aspect of the arm, showing the relations of the Type 1a Anconeus nerve (11) to the different triceps heads. 1: humerus; 2: long head of the triceps; 3: lateral head; 4: medial head; 5: medial epicondyle; 6: lateral epicondyle; 7: ulnar nerve; 8: olecranon; 9: radial nerve; 10: profunda brachii artery; 11: anconeus nerve; 12: nerve branches to the lateral triceps head; 13: tendon of the teres major muscle; 14: nerve branches to the medial triceps head. Anatomical dissection of the distal, intramuscular course of the anconeus nerve (Type 2, yellow star), showing a motor branch to the lateral head of the triceps muscle (green star), before entering the anconeus muscle (yellow cross). Note the topographical passage between the olecranon and the lateral epicondyle.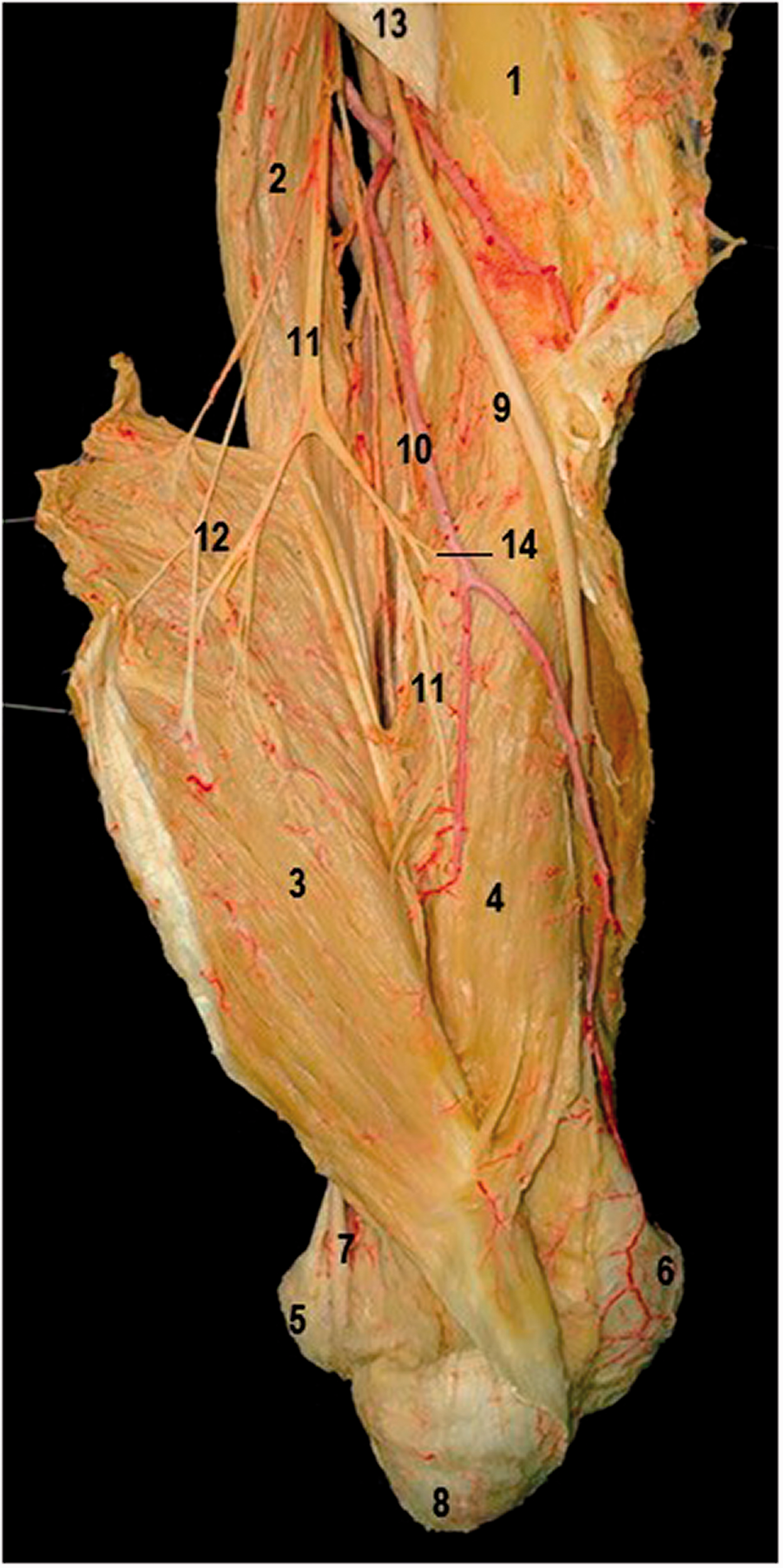
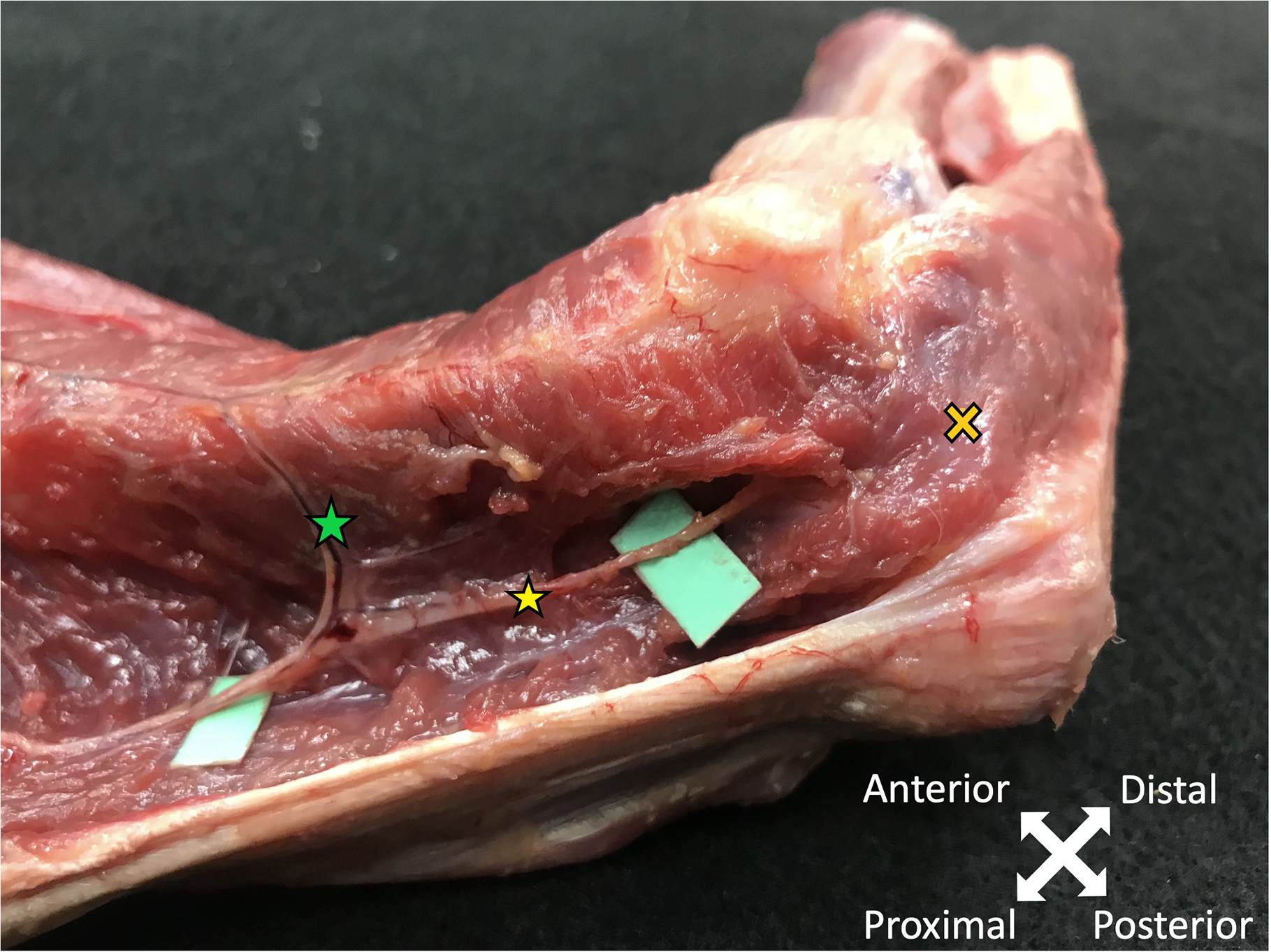
Two different types of anconeus nerves were found: nine nerves also innervated the lateral head of the triceps (Type 1a) (Figure 3), with one of them also providing the origin of the posterior cutaneous nerve of the forearm (Type 1b). The other six nerves only contributed two branches to the innervation of the lateral head (Type 2). In these specimens the lateral head of triceps was mainly innervated by a major, more proximal branch (Figures 5).
Schematic drawing of the three different anconeus nerve (AN) types.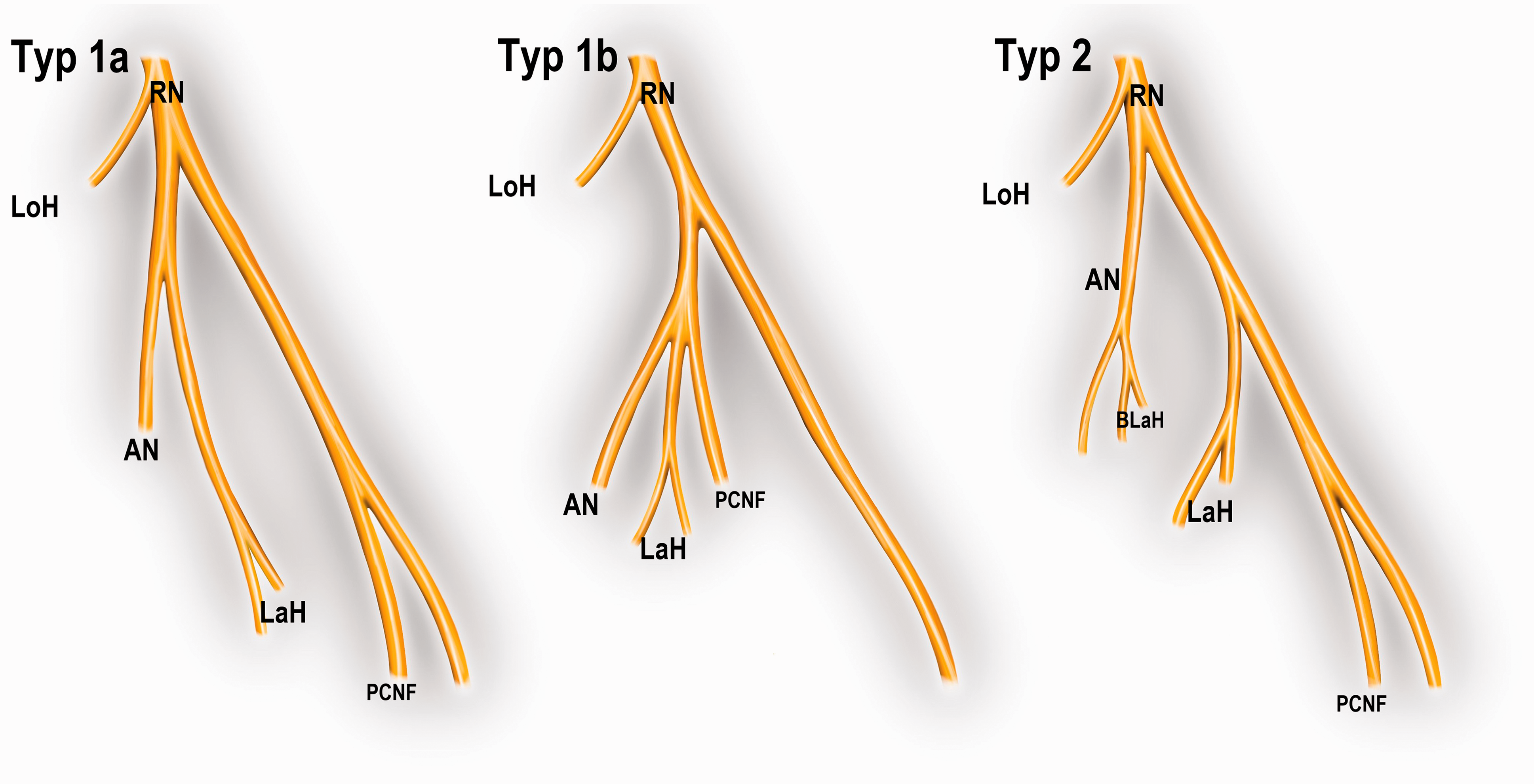
Discussion
We described the topographic course of the anconeus nerve. The origin of the nerve at the trifurcation point of the radial nerve is already a point of controversy: Gerwin et al. (1996) described it at the lateral aspect of humeral diaphysis, in contrast, Özer et al. (2006) found it on the posterior-medial aspect of the humerus. Our findings agreed with those of Özer et al. (2006). Thereafter, the anconeus nerve, which also innervates the medial head of triceps, crosses to the postero-lateral aspect. The nerve travels over the medial head of the triceps, protected by the muscular mass. After exiting the medial head and before piercing into the anconeus muscle lateral to the olecranon, we found the nerve lying on the periosteum and the joint capsule of the elbow, in contrast to Özer et al. (2006). This part was the most difficult to dissect, and probably the most vulnerable part of the nerve. Our finding may be important in avoiding damage to the nerve during exposure of the distal humerus (for instance with a posterior elbow approach).
However, most of posterior elbow approaches do not respect the anconeus anatomy, and denervation is often the consequence (Özer et al., 2005). Such a denervation leads to the impairment of its different functions, including the abduction of the ulna during forearm pronation (Badre et al., 2019; Gleason et al., 1985), prevention of impingement of the lateral portion of the joint capsule during elbow extension and dynamic stabilization of the elbow. Function and posterolateral stability of the elbow may be adversely affected (Badre et al., 2019; O'Driscoll, 2000). In an attempt to avoid denervation, O’Driscoll et al. (2000) introduced the triceps reflecting anconeus pedicle approach as an inter-nervous plane approach. Sun et al. (2015) also described a modification of this approach. Protection of the anconeus nerve is possible in many described approaches; however, surgeons must be aware of the exact position and topography of the nerve.
Knowledge of precise anatomy is also important if the anconeus nerve is used as a donor for neurotization. Although it may seem a contradiction to propose the use of the nerve for transfer, after advocating the importance of its preservation in cases of distal humerus fracture, it is reasonable to do so to treat a more important problem in the upper extremity. This may be the case in high brachial plexus injuries or isolated axillary nerve injuries, as an alternative to the triceps long and/or the upper medial head motor branch (Bertelli and Ghizoni, 2010, 2014; Colbert and Mackinnon, 2006).
The axonal count and diameter of the anconeus nerve, which generally is around 1.5 to 0.5 mm, is similar to that of the triceps long motor branch just proximal to arborization into its muscular insertion (Bertelli et al., 2007; Khair et al., 2016). However, the average length of the anconeus nerve is around 90 mm, whereas that of the triceps long head motor branch is only 30 mm (Bertelli et al., 2007; Wade et al., 2018). In isolated axillary nerve lesions, the damage can be found as far distally as its entrance into the deltoid. This length discrepancy of the donor nerve may be needed to allow for transfer to the axillary nerve terminal divisions. The series by Bertelli and Ghizoni (2014) supports the transfer of the anconeus nerve to the anterior division of the axillary nerve as an effective and reliable method of deltoid reinnervation. Recently, Chepla and Bafus (2018) described the use of the anconeus nerve transferred to the brachialis nerve to restore elbow flexion as an alternative to the Oberlin technique. Transfer to the biceps branch (Figure S1) or to the anteriorly transposed ulnar nerve are further possible options. The mobilization of the anconeus nerve could potentially result in damage to any of the more distal branches, depending on the degree necessary to reach the recipient nerves. We tested the range of the anconeus nerve in the anatomy laboratory and found that it reaches the ulnar and musculocutaneous nerve easily without damaging the more relevant branches of the radial nerve. In an extensive mobilization, the small branches to the medial head of the triceps are mostly at risk (Figure 3). However, we believe that if the use of the entire branch (proximally) to medial head of the triceps as a donor nerve does not cause a clinical impairment in the results presented by Bertelli et al. (2007), release of the more distal branches given by the anconeus nerve may not be clinically relevant either.
In our specimens, we found two types of branching patterns. In the most frequent one (nine specimens), the anconeus (and medial head branch) nerve had a common origin with the lateral triceps head branch. One of these also shared an origin with the posterior antebrachial cutaneous nerve. In the six others, the anconeus nerve gave only two small, more distal branches to the lateral head. In contrast to our results, Bertelli et al. (2007) found that the posterior antebrachial cutaneous nerve had a common origin with the anconeus nerve in half of their specimens.
This study has some limitations. First, anatomical variations in different human populations must be considered. Second, a histological analysis of the number of fibres in the anconeus nerve might refine the anatomical description. Lastly, the number of specimens is small. However, the sample size was sufficient to characterize three different branching patterns.
In conclusion, surgeons should be aware of the course of the anconeus nerve to avoid iatrogenic injuries and for use as a donor for transfer. We also emphasize the critical passage of the nerve on the periosteum and the elbow joint capsule, before it enters the anconeus muscle.
Footnotes
Acknowledgements
We thank the donors and their families.
Declaration of conflicting interests
The authors declare no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
This study was waved by the institutional review board of the Hospital Italiano de Buenos Aires, because it was an anatomical cadaveric study.
Supplemental material
Supplemental material for this article is available online.
