Abstract
It has been suggested that Dupuytren’s disease has an origin in Norse countries. We harnessed data from a genome-wide association study of Dupuytren’s disease and the People of the British Isles study to determine evidence for a Norse origin of Dupuytren’s disease. We computed Wright’s Fixation Index between Orkney (Norse ancestry), Wales (ancient British) and South East England (Anglo-Saxons); compared mean Wright’s Fixation Index in cases versus controls; used permutation to determine any excess of Norse inheritance in disease associated variants; constructed a genetic risk score for Dupuytren’s disease and applied this to the People of the British Isles dataset to look for systematic differences between counties with known high and low levels of Norse ancestry. Finally, chromosome painting was used to see whether Dupuytren’s disease-associated single nucleotide polymorphisms are geographically structured. In all analyses, no evidence was found for an excess of Norse ancestry in Dupuytren’s disease. We conclude that there is no genetic evidence for a ‘Viking origin of Dupuytren’s disease’.
Introduction
Dupuytren’s disease (DD) is a disease of the palmar fascia that causes flexion contractures of the fingers (Townley et al., 2006). It is the most common inherited disease of the connective tissue and its prevalence is increasing (Bebbington and Furniss, 2015). DD is a typical complex disease (Hart and Hooper, 2005). To date, genome-wide association studies (GWAS) have revealed 26 loci predisposing to DD (Dolmans et al., 2011; Ng et al., 2017). These associations have revealed several novel pathophysiological insights and the risk alleles correlate with recurrence of DD after surgery (Riesmeijer et al., 2019).
It has been suggested that DD is a ‘disease of the Vikings’ (Flatt, 2001). The evidence for this assertion comes from two main themes. First, epidemiological studies have shown that DD is more common in European populations and the prevalence seems to be highest in Iceland and Scandinavian countries that were the origin of the Viking Warriors (Hindocha et al., 2009). However, other evidence indicates a substantial prevalence of DD in countries never affected by Viking invasions, such as Japan, Spain, Ethiopia and India (Dasgupta and Harrison, 1996; Egawa et al., 1990; Gebereegziabher et al., 2017; Guitian, 1988) although clearly, with increasing global migration, admixture of populations is occurring with increasing frequency. The second stream of evidence is historical. Whaley and Elliot (1993) provided an account of four ‘miracles’ from Nordic sources, where a condition resembling DD was alleviated by miracle cures. Furthermore, the lack of description of a disease resembling DD in Ancient Greek, Roman, Early Anglo-Saxon or Medieval European writings is further putative evidence of a Nordic origin of the disease (Elliot, 1988).
The People of the British Isles (PoBI) Study sampled DNA from 2039 individuals throughout rural areas of the UK whose four grandparents were born within 80 km of each other (Winney et al., 2012). This strategy allowed investigators to effectively sample the grandparental DNA from rural Great Britain in the latter part of the nineteenth century, before the start of the major population movements of the twentieth century. These data have provided evidence of fine-scale population structure within the UK. Within the UK there are geographical population clusters that can be separated on the basis of their inherited genetic variation (Leslie et al., 2015). There are three major historical sources of this population structure: north-west Germany, France and Spain, with the greatest contribution observed in the current population of rural Wales; northern Germany and Denmark, with the greatest contribution observed in the population of rural south-east England; and the Norse, with the greatest contribution observed in the population of the Orkney Islands.
The most commonly used measure of genetic difference between populations is Wright’s Fixation Index (FST) (Hudson et al., 1992). The higher the index for a particular single nucleotide polymorphism (SNP), the more different the allele frequencies of the SNP are between populations. We used recently reported genetic association results from the British Society for Surgery of the Hand Genetics of Dupuytren’s Disease (BSSH-GODD) (Ng et al., 2017) to provide empirical evidence to investigate whether DD is associated with Norse ancestry.
Methods
Explanation of terms used in this paper.

Cohorts
The recruitment, genotyping, imputation and analysis methods for the BSSH-GODD and PoBI studies have been previously described (Leslie et al., 2015; Ng et al., 2017). Briefly, the BSSH-GODD study included a total of 7912 cases of DD and 12,937 population-based controls. The PoBI dataset consisted of a geographically diverse sample of 2039 individuals from the United Kingdom, which were then compared with 6209 individuals from across Europe to generate signals of historical demographic shifts. We identified the common SNPs between the PoBI dataset and BSSH-GODD dataset and removed multi-allelic SNPs and non-autosomal SNPs, leaving a set of 462,930 SNPs for further analysis.
FST analyses
FST for each SNP was calculated using Hudson’s estimator of FST (Hudson et al., 1992), as implemented in EIGENSOFT (https://www.hsph.harvard.edu/alkes-price/software/). For each analysis, FST was calculated between three comparator groups: Orkney vs Wales, Orkney vs South East, and Orkney vs combined Wales and South East. Because we were interested in those SNPs that differentiated Orkney from the other populations, all negative FST values were set to zero (Garagnani et al., 2009).
First, we compared the summed total FST for Norse ancestry informative markers from PoBI in cases and controls in BSSH-GODD. We hypothesized that if DD were of Norse origin, cases would have a higher average total FST than controls. For the case-control analysis, the total genomic FST was calculated for each individual case or control using the formula:
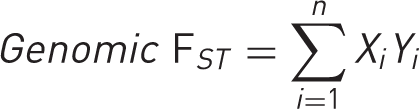
Second, the total FST of the DD-associated SNPs from BSSH-GODD was calculated and compared with the total FST of 10,000 randomly generated permutations of SNP groups of the same size, to generate an empirical p-value for the ‘Viking origin’ of DD-associated SNPs. We hypothesized that if DD were of Norse origin, the total FST our associated SNPs would be in the top 5% of the ranked total FST for the random permutations of SNPs (nominally p < 0.05).
For the permutation analysis, we first determined the SNPs to represent each of the 26 credible sets associated with DD. The PoBI dataset was searched for the statistically most highly associated SNP from each credible set. If the most highly associated SNP from a credible set was not present in the PoBI dataset, we searched sequentially for the next most associated SNP, continuing until a representative variant was found, or all variants in a 99% credible set were exhausted. For five credible sets, there was no representative SNP in the PoBI dataset (lead SNPs: rs117402009, rs2472141, rs1042704, rs61982174, rs34088184), meaning there were 21 DD-associated SNPs remaining for analysis.
Total FST of the remaining 21 DD-associated SNPs was calculated, using each of the three comparator groups. We then permuted 10,000 random sets of 21 SNPs within the PoBI dataset and calculated the total FST of each of these sets in each of the three comparator groups. The permuted sets of random SNPs were ranked by total FST and we examined where in this list the set of DD-associated SNPs was ranked. A nominal threshold of significance of 0.05 was set, with the hypothesis that if DD is associated with Norse ancestry, the total FST of the DD-associated SNP set would be in the top 5% of permuted sets.
Chromosome painting
Next, to assess for localized population structure at the DD-associated loci in the PoBI dataset, we used chromosome painting, comparing the frequency of haplotypes most recently shared within population groups to that between population groups (Lawson et al., 2012). The haplotype structure of PoBI participants at relevant loci was examined. The local painting for the 21 independent Dupuytren’s GWAS hits was extracted and the frequency for which individuals with each population label shared a most recent haplotype with individuals of each other population was calculated, to form a population co-ancestry matrix for each position in the genome. We hypothesized that if DD was caused in individuals by having Norse ancestry at these loci, then we would expect that they have an excess amount of local structure, relative to the genome as a whole. The hypothesis was tested by comparing the within-population co-ancestry to the mean outside-population co-ancestry, by computing the relative frequency of sharing ancestry within a population to that of sharing ancestry with an individual from a different population. To make a fair null hypothesis, we sampled the same number of random genome-wide SNPs that make up the risk score using 500 random samples of 21 SNPs. To ensure that this method has power, we selected a positive control by repeating this analysis for a Lactase-associated SNP – rs4988235 in MCM6. Lactase is under positive selection and therefore has much less population-specific haplotype sharing.
Genetic risk score
Finally, we constructed a genetic risk score (GRS) for DD and applied it to the PoBI dataset, hypothesizing that people from those regions of known high Norse ancestry would have a higher GRS than those from areas with low levels of Norse ancestry. The GRS for DD from the 21 independent Dupuytren’s GWAS hits was calculated for each individual in the PoBI dataset. We then aggregated these scores across the 35 population labels to investigate possible geographical structure associated with known regions of high Norse ancestry, such as the Highlands of Scotland.
Data availability
Data used in this study are available from the European Genome-Phenome Archive (https://ega-archive.org), study numbers EGAS00001000672 and EGAS00001001206
Results
Case-control analysis
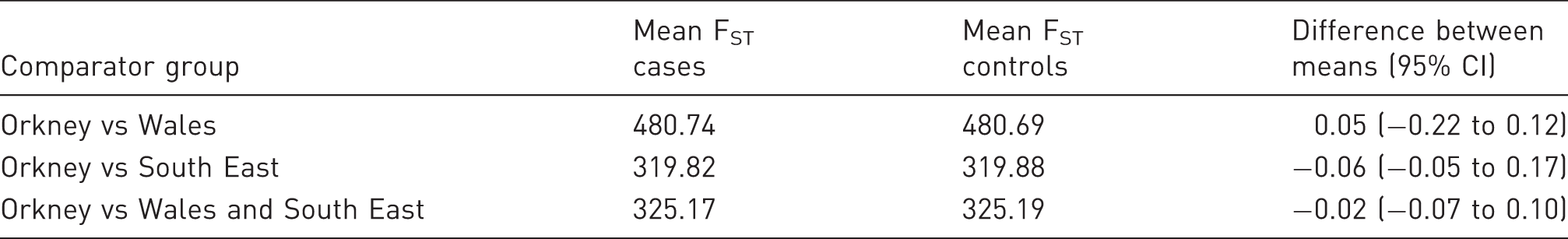
The distribution of genomic FST in both cases and controls closely followed a normal distribution (Figure 1(a)). The mean difference in total FST between cases and controls ranged between −0.06 and 0.05 (Table 2). For all comparator groups, the 95% confidence intervals of the difference between means crossed zero meaning there was no significant difference. If DD were of Norse origin, we would expect that cases would have a higher average total FST than controls.
Wright’s Fixation Index (FST) analysis reveals no evidence of a Norse origin of Dupuytren’s Disease (DD). (a) The mean genomic FST of cases and population controls is not significantly different. The entire cohort was plotted against theoretical quantiles to demonstrate the normality of the data (upper right panel). (b,c,d) Comparison of total FST for DD-associated single nucleotide polymorphisms (SNPs; blue line) compared with randomly permuted sets of SNPs. (b) Orkney vs Wales; (c) Orkney vs South East; (d) Orkney vs Wales and South East. The mean FST for cases and controls in the British Society for Surgery of the Hand – Genetic Origin of Dupuytren’s Disease dataset.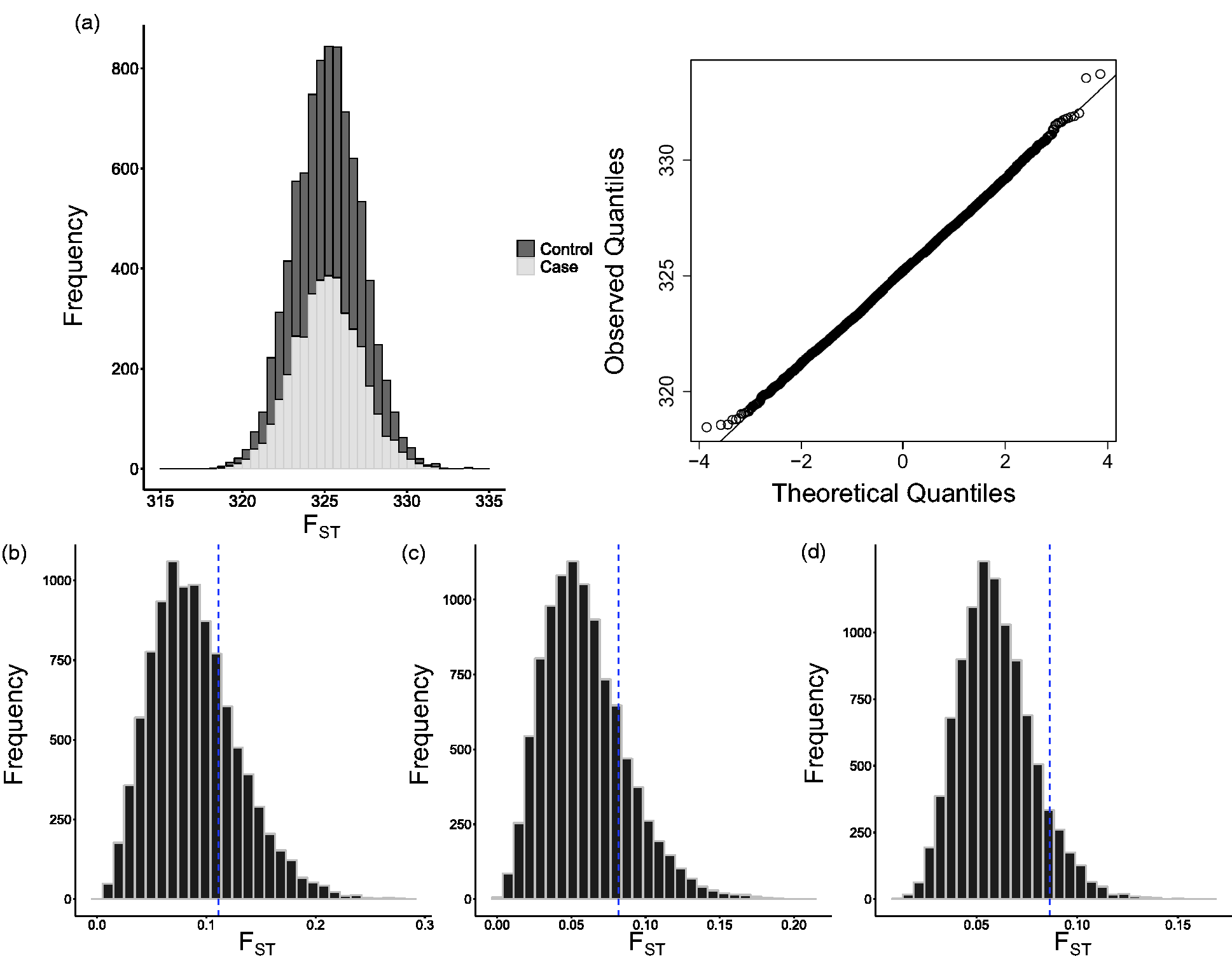
Permutation analysis
For each comparator group, the total FST of the DD-associated SNPs was not within the top 5% of ranked total FST of 10,000 permuted SNP groups (nominally p > 0.05, Figure 1(b,c,d). In Orkney vs Wales, the DD-associated set was on the 74th centile; in Orkney vs South East, it was on the 81st centile; and in Orkney vs Wales and South East, it was on the 92nd centile. If DD were of Norse origin, we would expect that the total FST of our disease-associated SNPs would be higher than randomly generated groups of SNPs.
Chromosome painting
Compared with 500 random samples of 21 SNPs taken from across the genome, the DD-associated SNPs do not show an increase in within-population structure (p = 0.072, Figure 2(a)), which would have been expected had DD been associated with Norse ancestry. This is contrast to the lactase-associated SNP rs4988235, which as expected shows a highly significant decrease in population-specific haplotype sharing (p < 0.001, Figure 2(b)).
Local haplotype structure does not support a Viking introgression of Dupuytren’s disease into the British Isles. (a) Dupuytren’s genetic risk score is not significantly different from random in a haplotype-based test for population structure. (b) Lactase-associated SNP rs4988235 shows a highly significant decrease in local haplotype sharing. Lactase is under positive selection and therefore is expected to show much less population-specific haplotype sharing, and is therefore a positive control in this analysis. (c) Dupuytren’s genetic risk scores are not associated with geography. When a genetic risk score is averaged over the 35 counties of the UK, there is no structure, even in areas with high Viking ancestry such as Orkney (Northernmost population).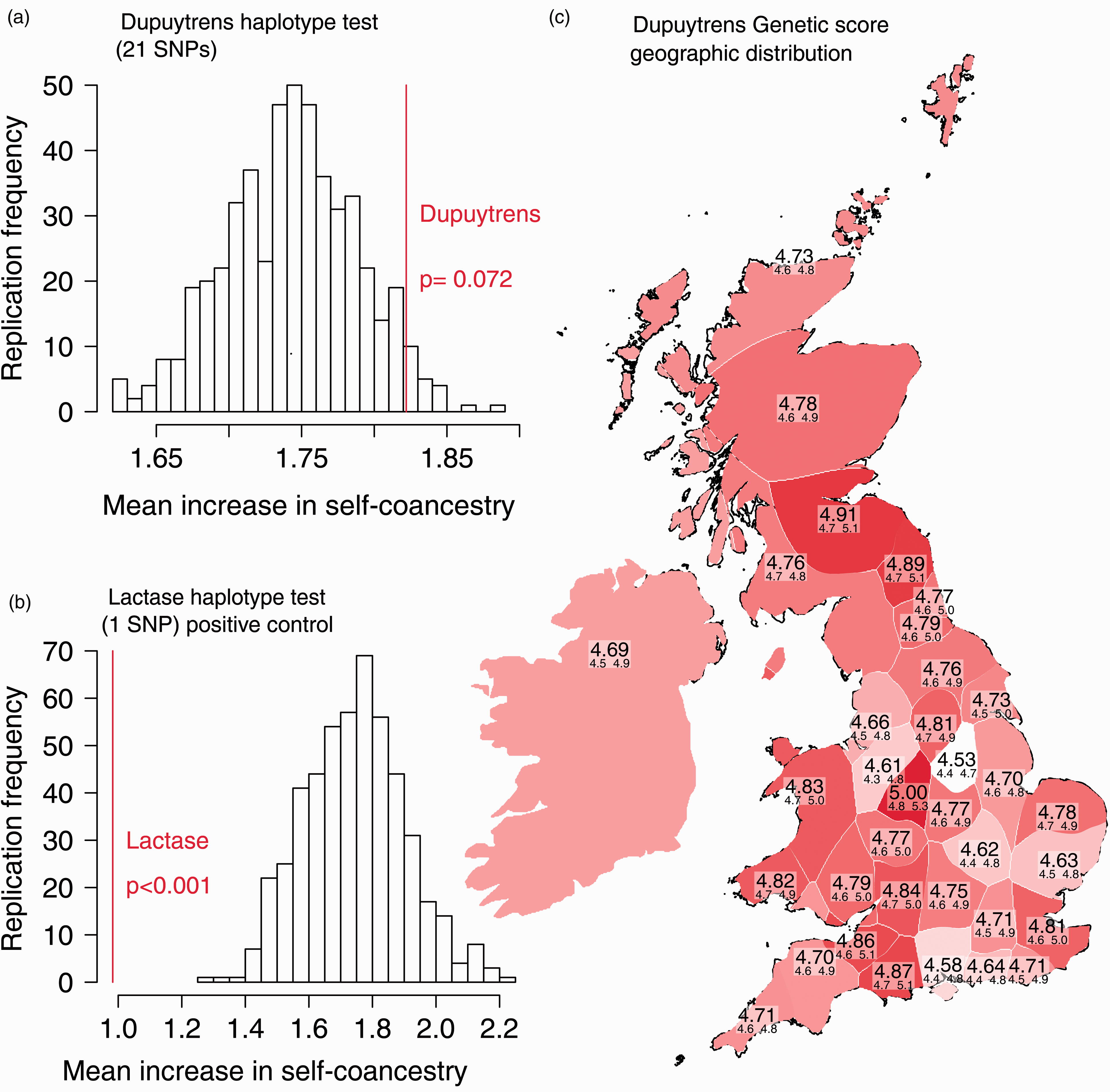
Genetic risk score
The aggregated GRS across the PoBI dataset does not show any geographical clustering in areas of known high Norse ancestry (Figure 2(c)), which would have been expected if the genetic predisposition to DD was derived from Norse ancestry. The 95% confidence intervals for each region overlap, consistent with the notion that DD and Norse ancestry do not cluster together.
Discussion
Our results do not provide support for the hypothesis that DD is a ‘disease of the Vikings’. We found no evidence for an excess of Norse ancestry informative markers in our cases and the total FST of disease-associated SNPs was not in the top 5% of 10,000 randomly permuted SNP groups of the same size.
Furthermore, chromosome painting did not find evidence for localized genetic population structure that would be expected around DD-associated SNPs if there were an excess of Norse ancestry. Finally, a GRS derived from DD-associated SNPs did not show increased geographical clustering around areas of known high Norse ancestry. Other phenotypes, such as educational attainment, show strong associations with geography on this scale (Timpson et al., 2018).
Compared with previous reports (Whaley and Elliot, 1993), our data provide objective evidence of the lack of Norse origin of the disease. Some scepticism as to the Norse origin of the disease has been expressed by Elliot (1988). He commented on the fact that ‘It is surprising that no earlier reference to this condition has been found in the Scandinavian literature’. He also makes reference to both the ‘fragmentary nature of the record which has survived’ and the fact that life expectancy in previous centuries was much lower than the median age of onset of DD; these are both pertinent points that reveal the weakness of the historical case. Furthermore, Elliot (1988) also comments on the lack of records of DD in the medical writings of the missionaries of Iona, the Roman Church in North Germany and North-East England, or the extensive medical writings from mediaeval Europe. If DD had been disseminated around Europe by the Norsemen, mention of it in one of these sources would be expected. Also, Hindocha et al., (2009) stated: ‘Other than the common prevalence of DD in Scandinavia, no objective scientific evidence has been found to date to support and substantiate the “Nordic” origin of the disease’.
In conclusion, our genetic analyses directly contradict the widely held belief, especially popular among patients, that DD is a disease of Norse origin.
Footnotes
Acknowledgements
The authors wish to acknowledge all of the individuals who participated in both studies. In addition, we thank the surgeons and researchers in the BSSH-GODD study who helped with recruitment of affected individuals.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
Ethical approval for this study was obtained from: British Society for Surgery of the Hand Genetics of Dupuytren’s Disease (BSSH-GODD) study – Oxfordshire Research Ethics Committee B/09/H0605/65; People of the British Isles study – NRES Committee, Yorkshire and the Humber – Leeds West, UK 05/Q1205/35.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: DF was supported by an Intermediate Clinical Fellowship from the Wellcome Trust [097152/Z/11/Z]. This work was supported by the NIHR Biomedical Research Centre, Oxford. DJL is supported by the Wellcome Trust and Royal Society [WT104125MA].
Informed consent
Written informed consent was obtained from all subjects before the study.
