Abstract
Ambient exposure to water vapour and oxygen undermines the long-term lubricity of magnetron-sputtered molybdenum disulphide (MoS2) coatings, yet the individual and synergistic contributions of the two species remain poorly quantified. Here we decouple their effects by ageing 1 µm PVD-MoS2 films for 500 h in six precisely controlled gas/humidity atmospheres (dry, 25% RH and 50% RH; N2 or air) followed by micro-tribological testing in dry N2. Complementary “unaged” samples were slid directly in identical environments to probe instantaneous reactivity. Coefficient-of-friction data, Raman spectroscopy, SEM-EDS mapping and cross-sectional TEM reveal three key findings: (i) humidity alone raises steady-state friction by up to 2.5-fold, whereas dry oxygen has little effect; (ii) concurrent H2O and O2 during storage triggers sub-surface penetration of O-species, formation of MoO2/MoO3 within wear scars and inhibition of basal-plane realignment, yielding a persistent friction penalty (µ ≈ 0.12) independent of run-in; (iii) ageing in dry or humid N2 preserves lamellar re-orientation and maintains low friction (µ ≈ 0.05) after only 50 cycles. A mechanistic model is proposed in which adsorbed water lowers the activation barrier for oxygen insertion at defect and edge sites, corroborated by a 2–3 cm−1 blue shift of the E12g Raman band. By isolating the water-assisted oxidation pathway at ambient temperature, this study provides the first quantitative guideline for MoS2 component storage: moisture-free inert gas environments effectively arrest chemical degradation, whereas even moderate humidity in air accelerates tribo-oxidative damage. The insights inform design tools for precision-mechanical assemblies where pre-deployment shelf life is critical.
Introduction
Molybdenum disulphide (MoS2) continues to attract significant attention as a solid lubricant due to its exceptional performance, particularly under vacuum or inert environments. The presence of its lamellar structure, featuring molybdenum atoms sandwiched between sulphur layers and held together by weak van der Waals forces, enables low friction and efficient shear between basal planes. These characteristics make MoS2 coatings indispensable in aerospace applications, including space missions, where reducing wear and friction is critical. 1
Despite its robust performance in controlled conditions, the degradation of MoS2 coatings in environments containing oxygen and water remains a challenge. Recent studies emphasize that the interplay between these environmental factors accelerates tribological deterioration and increases the coefficient of friction (COF). Oxygen and water can disrupt the basal plane shear, leading to increase COF and reduced wear life. 2 Notably, water diffusion into MoS2 structures during exposure leads to oxidation, altering the material's surface chemistry and structure resulting in increased coefficient of friction. 3
Recent research further highlights the role of water in exacerbating these effects by enhancing oxygen diffusion and promoting surface adsorption. For instance, humid environments have been shown to accelerate the transformation of MoS2 to molybdenum oxides, such as MoO2 and MoO3, leading to irreversible changes in structure and lubrication properties. 4 Conversely, environments with controlled humidity, such as dry nitrogen, have been shown to preserve the coating's structural integrity better. 1 Studies employing spectroscopic techniques have demonstrated that surface oxidation predominantly occurs under humid conditions, highlighting water's critical role in promoting oxygen diffusion into the material's near-surface region. 5 Khare and Burris 6 have recently suggested water does not promote oxidation of PVD-deposited films at room temperature hypothesising it directly interferes with lamellar shear through physical bonding, noting oxidation via Energy Dispersive Spectroscopy (EDS) at high temperatures > 100°C. This is consistent with thermal aging, where high-temperature exposure exacerbates oxidation. Çiçek et al. have reported significant changes in structural and tribological properties at temperatures as low as 350 °C. 7
The long-term effects of water and oxygen exposure during storage also raise challenges. Degradation mechanisms such as surface oxidation, adsorption of reactive species, and lamellar misalignment have been observed under prolonged exposure to humid air, highlighting the importance of tailored storage environments. 2 Such changes compromise the reliability of MoS2 coatings in real-world applications, particularly when exposed to alternating environmental conditions prior to deployment. 3 High temperature (∼250°C) aging of MoS2 films has been used to promote oxidation of coatings prior to sliding noting initial high COF (∼0.1–0.3) decreasing to approximately μ ∼ 0.05 after 10 cycles when slid in dry N2 environments, suggesting mechanical removal of oxidised surfaces and preferential formation of basally orientated MoS2 lamella at the interface. 1 Furthermore, basally oriented running films formed during sliding in inert or humid environments have been shown to resist oxidation better, suggesting the importance of pre-aging treatments in inert conditions to minimise environmental degradation. 1 Similar observations have also been noted for aerosol and burnished MoS2 films. Similarly, nitrogen-doped MoS2 coatings have demonstrated superior wear resistance and reduced friction coefficients under vacuum and humid environments, attributed to enhanced mechanical properties and the prevention of oxidation through nitrogen incorporation. 8
While the Relative Humidity (RH) sensitivity of MoS2 friction has been recognised for decades, almost all prior studies have focused on either short-term sliding in controlled atmospheres or high-temperature (> 100 °C) oxidation scenarios. Very few have examined ambient-temperature, multi-week storage in which water vapour and oxygen act simultaneously but at modest partial pressures; conditions that closely mirror real-world shelf life. Moreover, earlier work generally compared “dry” versus “humid” air without isolating the independent roles of H2O and O2 and rarely coupled tribological data with depth-resolved microscopy and spectroscopy. As a result, the mechanistic pathway by which low-temperature water facilitates oxygen ingress and alters lamellar re-orientation remains unresolved, leaving designers without quantitative guidance on how best to store coated components before deployment.
The aim of this study was to interrogate and quantify the independent and combined influences of water vapour and oxygen on the tribological performance, chemistry, and microstructure of magnetron-sputtered MoS2 coatings during prolonged ambient-temperature storage and subsequent sliding, thereby establishing evidence-based guidelines for the safe shelf-life and deployment of MoS2-lubricated components in aerospace and defence applications. Together these contributions close the current evidence gap on low-temperature, long-term environmental degradation of MoS2 and establish a reproducible framework for evaluating solid-lubricant shelf life.
Materials and methods
Materials
Commercially obtained high purity (>99%) molybdenum disulphide (MoS2) coatings were magnetron sputter deposited via Physical Vapour Deposition (PVD) (Miba, UK) to a nominal thickness of 1 μm on 440C stainless steel disc substrates. The substrates had an outer diameter of 10 mm and were metallographically polished utilising a Grinder Polisher (Buehler, DE) to attain a surface roughness (Ra) of 200 ± 50 nm measured by Vertical Scanning Interferometry (Bruker, USA). Firstly, wet grinding of the sample using SiC paper with a coarseness of 120 was undertaken followed by 320, 600, 800 and 1200. The freshly polished sample was then washed with deionised water and dried using compressed air; samples were then packaged and transported for coating deposition. The tribological counter sample used in all tests was an uncoated Ø3 mm 440C stainless steel ball bearing (Simply Bearings, UK).
Environmental control and aging
The environmental parameters were controlled for both ageing and tribological testing using a custom-built gas humidifier system (Figure 1). A dry gas source was fed through a desiccant, then split into two branches, both containing gas flow meters. The first branch remained dry and ran through to an environmental chamber. The second branch was passed through a gas bubbler containing deionised water to humidify the source gas before arriving at the environmental chamber. The humidity of the output gas could be controlled by adjusting the flow of each branch and was measured using a hygrometer, which was integrated in the out-feed flow. (Traceable Hygrometer, Fischer Scientific, UK).
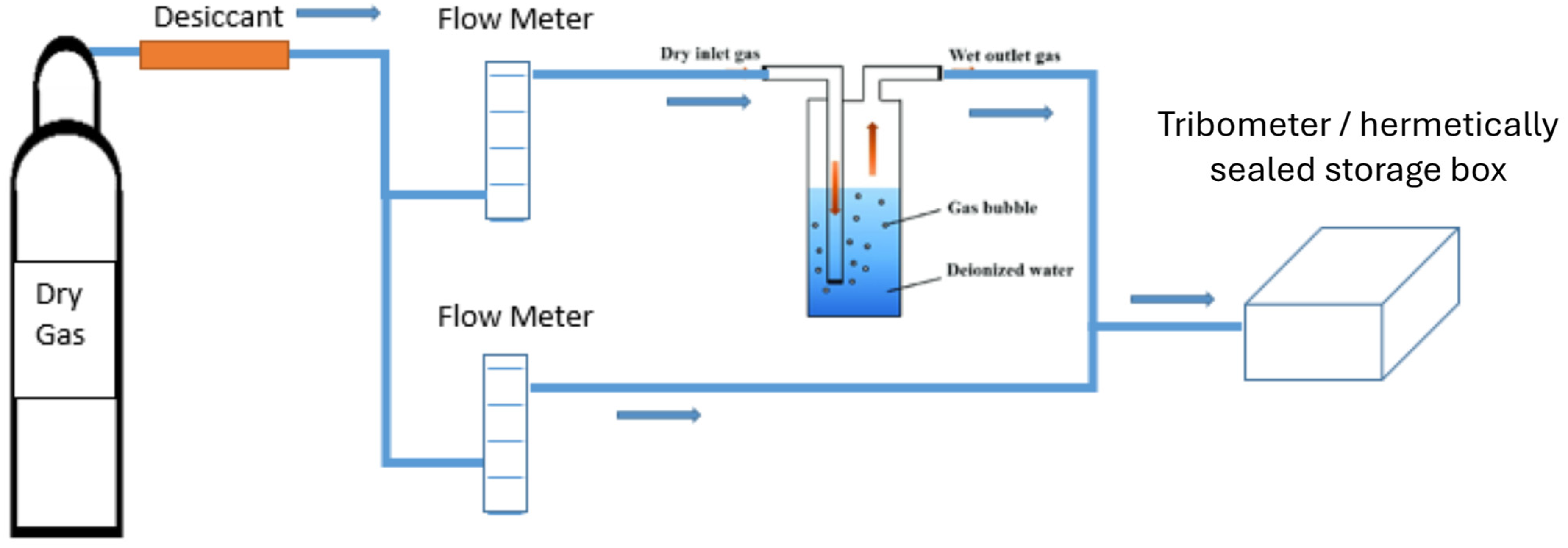
Schematic diagram of custom-built gas humidifier system.
For ageing, the MoS2 coated samples were exposed in the environmental chamber (Sure Seal hermetic chamber, WPI instruments, UK) to six different environments of gas and humidity (N2 or air / dry or 25 or 50%RH). The gas flows were balanced to achieve the desired humidity in the chamber and the chamber was purged for at least 20 min. After the purge period the chamber valves were closed simultaneously, and the samples were stored for a period of 3 weeks (500 h) in the static environment.
Tribological testing
Tribological testing was carried out at room temperature using a micro-tribometer (NTR2, Anton Paar, CH) (Figure 2). Unaged samples were tested in dry, 25% and 50% RH N2 or air. Aged samples were tested in a dry N2 environment. A custom small-volume environmental cell was developed with a hygrometer probe integrated. The chosen testing gas mixture was flowed into the environmental cell for 10 min before the experiment and for the duration of the experiment. The normal force applied was at Fn = 100 mN (Pmax ∼ 450 MPa), with a stroke amplitude of 1 mm and a frequency of 0.5 Hz for a total of 1500 cycles. Each tribology condition was tested at least three times on independent tracks (n = 3) to ensure run-to-run reproducibility and to avoid surface-history effects. The coefficient of friction, μ, was calculated by taking an average of the forward and reverse friction forces within the centre 50% of each cycle. A value for the Coefficient of Friction (COF) at a ‘steady state’ level was calculated by taking the mean ± standard deviation value of cycles 1200–1500.
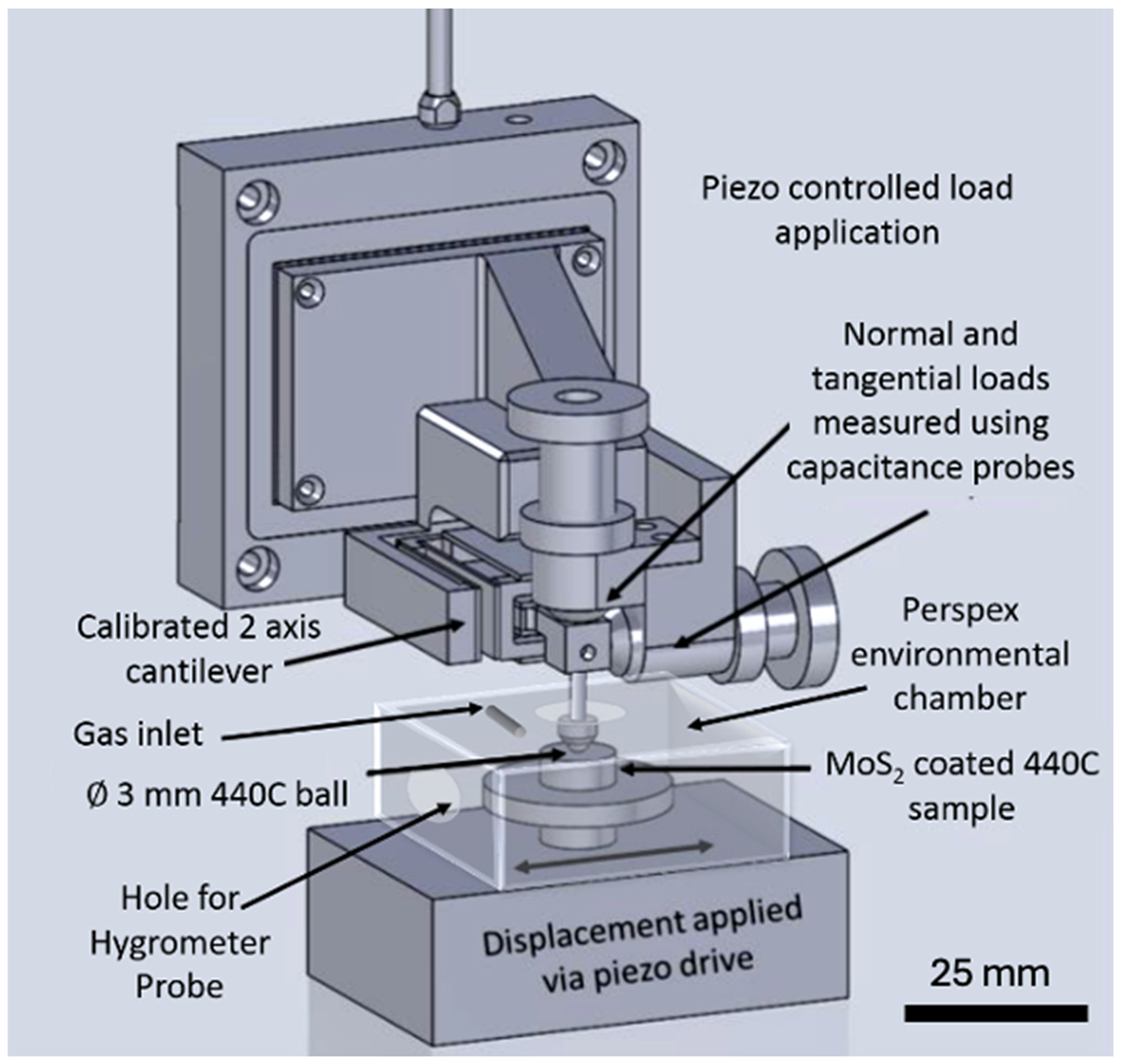
Schematic of Anton Paar NTR2 tribometer with custom local atmospheric chamber.
Scanning electron microscopy and energy-dispersive X-ray spectroscopy
Scanning Electron Microscopy with integrated Energy Dispersive Spectroscopy (SEM-EDS) on a Carl Zeiss EVOMA15: variable pressure SEM with an Oxford Instruments AZtecEnergy EDX System was used to observe the elemental changes on the coatings after ageing and tribology testing. The integrated EDS detector was then used to detect the atomic composition of the coating and wear scar. SEM scans were taken at an accelerating energy of 5 keV to reduce interaction from the steel substrate, as coatings were only 1 μm thick. The elemental composition of the coated surfaces was mapped enabling the spatial distribution of elements to be resolved. Particular attention was drawn to the presence of molybdenum, sulphur, and oxygen.
Raman spectroscopy
Raman spectroscopy was carried out using an InVia spectrometer (Renishaw, UK) in lab air to observe if any changes in the chemical composition had occurred due to aging or application of mechanical shear. A Raman laser with a 488 nm wavelength and a laser power of 1 mW was used to ensure that no laser induced oxidation of the coating took place. Samples were analysed using a short distance 50× objective with a laser spot size of 0.4 mm. Measurements were taken inside and outside of the wear scar on both aged and unaged samples so that effects of aging alone and aging alongside tribology could be seen. Intensity data was normalised between 0 and 1 (at 408–409 cm−1). Raman measurements were carried out with short collection times to minimise the risk of sample heating, and these measurements were repeated over a defined period. The spectra were then integrated to improve the signal-to-noise ratio. The spectrometer routinely operated with access to the first-order Si reference at 520.7 cm−1. The system has a spectral resolution of ∼1 cm−1 and shifts larger than ∼1–1.5 cm−1 were taken to be physically meaningful.
Transmission electron microscopy
A FEI Helios G4 CX Dual Beam was used to prepare Focussed Ion Beam (FIB) wafer thin samples to be analysed on a Transmission Electron Microscope (TEM). Samples were initially Ir coated to prevent charging before wafer sections were collected across the wear scar. A primary protective Pt layer (200–300 nm) was deposited over the area of sample collection using an electron beam and this was followed by further Pt using a Ga+ ion beam. Material was milled to an approximate depth of 10μm and further thinned before being partially cut free from the substrate. The milled section was removed using a Kleindiek micromanipulator and attached to a Cu TEM grid by way of Pt deposition. The sample lift-out was further thinned using Ga+ ions to produce electron transparent sections. Samples were examined using an FEI Titan Themis Cubed 300 TEM at 185,000× with an electron voltage of 300 kV.
Results
Tribology results
Effect of the sliding environment (unaged samples)
The effect of the sliding environment on unaged samples is shown in Figure 3. For both the N2 (Figure 3(a)) and the air (Figure 3(b)) environment COF increases with the amount of humidity present: approximately a 2.5× increase for 50% RH as compared to dry gas. The COF is also increased in air as compared to N2 for each of the humidity conditions.
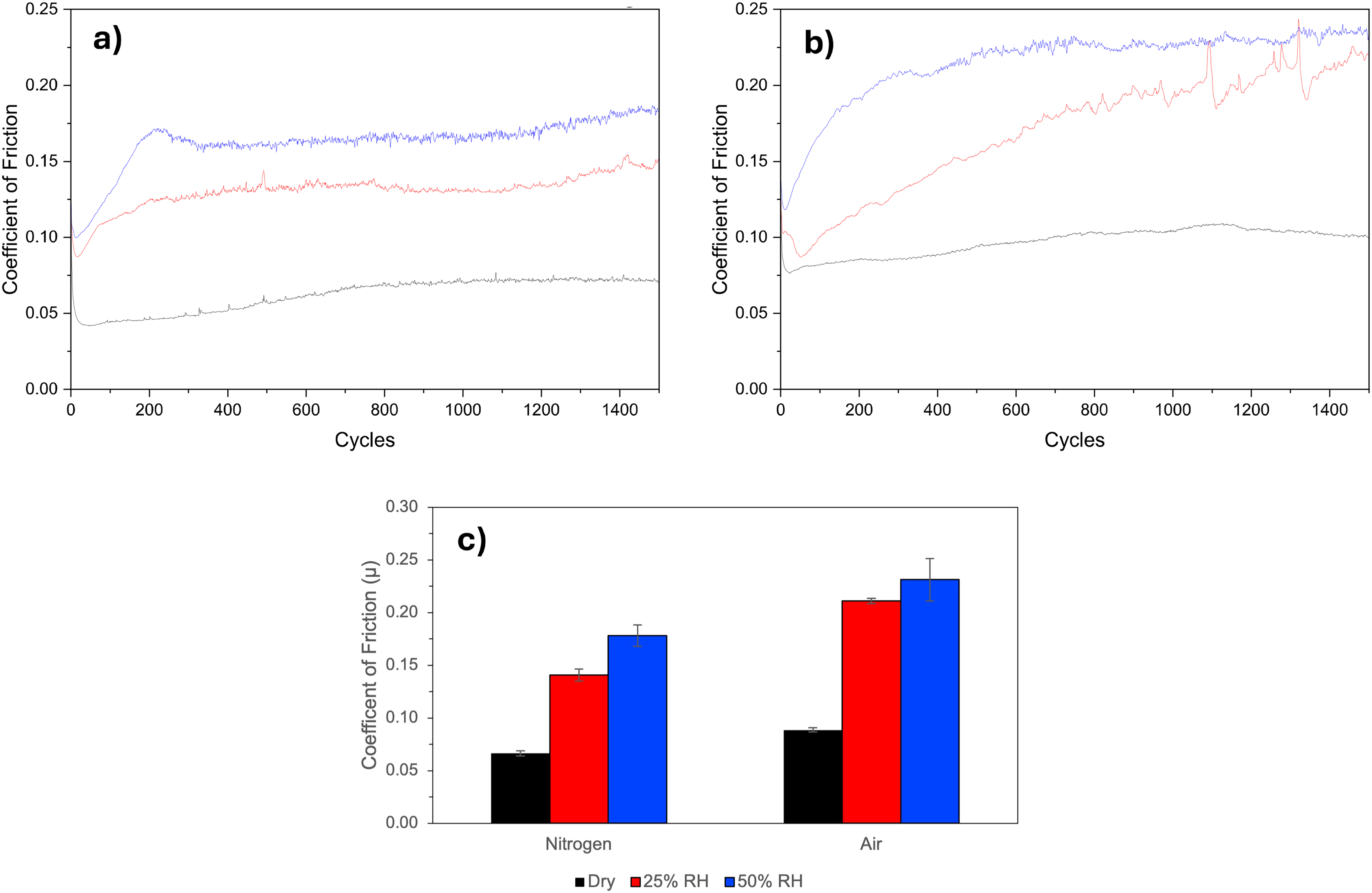
Coefficient of friction data for the ‘unaged’ samples showing the effect of the sliding environment when unaged samples were tested in a range of environments. a) tested in dry (black), 25% (red) and 50% relative humidity N2 (blue)
In all cases the COF drops rapidly over the first 50 cycles to a minimum. For those environments with 25% or 50% RH the COF then increases over a number of cycles reaching a plateau steady state. The COF in these cases typically reaches the plateau by 200–500 cycles with the exception of 25% RH air which is arguably reaching the plateau at the end of the experiment (1500 cycles). For those environments with dry gas (air or N2) the rise in COF is less pronounced over some 800 cycles. The average COF for the six tests is shown in Figure 3(c).
Effect of the ageing environment (aged samples)
The effect of the ageing environment on samples is shown in Figure 4. The COF in the N2 aged samples is largely independent of humidity present in the ageing environment (Figure 4(a)): the dry N2 and 25% RH data sets are very similar, and the 50% RH data is arguably only somewhat greater COF (20–30%) than the dry N2. It is noteworthy that this behaviour is similar to that exhibited by unaged samples tested in dry N2 (see the black data set in Figure 3(a)). The COF for the air aged samples (Figure 4(b)) increases with the amount of humidity present analogous to the effect of humidity on the sliding environment (see 3.1.1), although the effect is less pronounced.
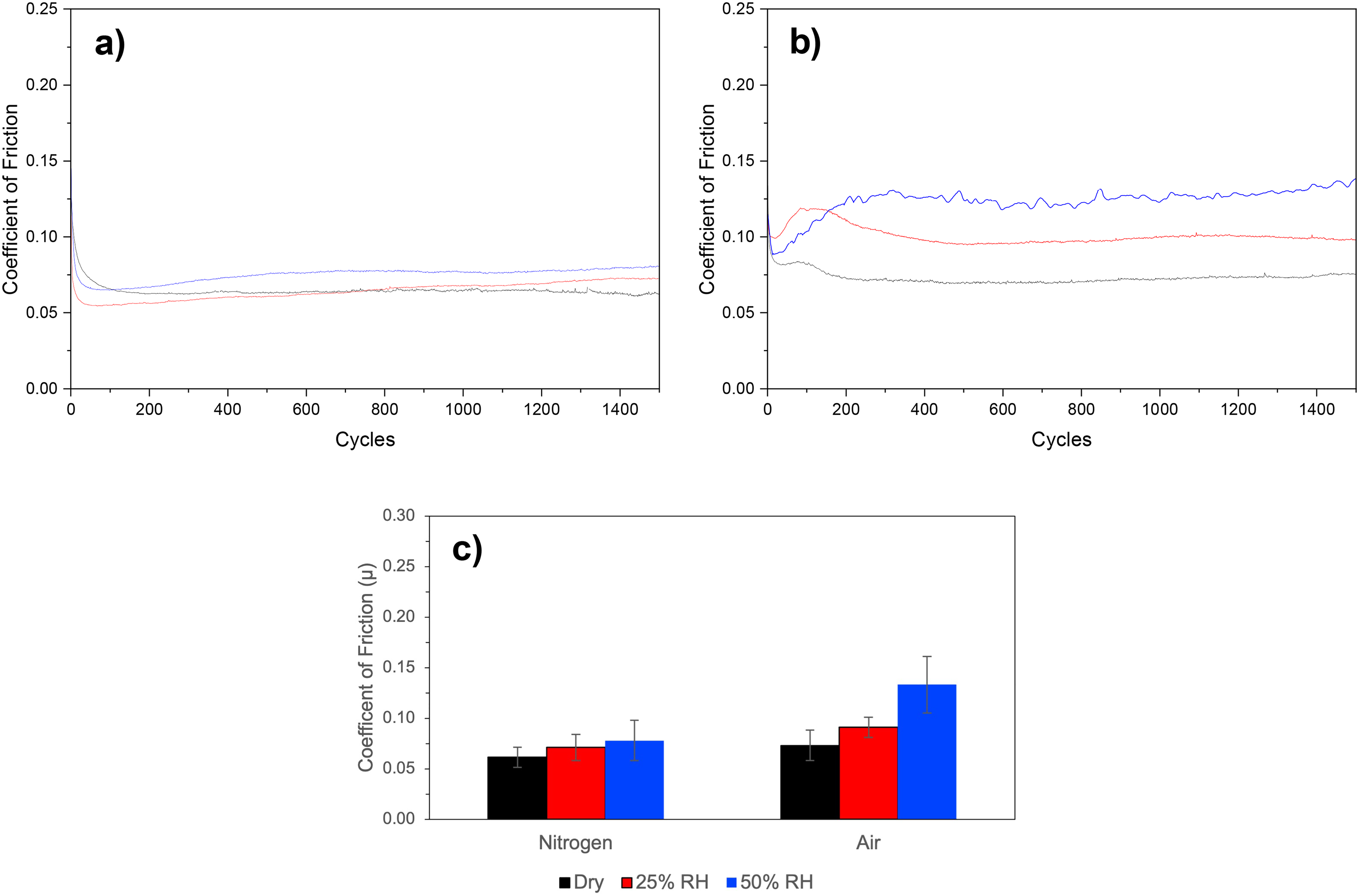
Coefficient of friction data for the ‘aged’ samples showing the effect of the ageing environment when subsequently tested in a dry N2 environment.
Samples stored in N2 (dry, 25% RH and 50% RH) exhibited similar development over the tests, that is an initial rapid drop in COF over the first 50 cycles followed by a steady state COF of between 0.06 and 0.075 for the remainder of the experiment; this is similar behaviour to the unaged sample tested in dry N2 (black data set in Figure 3(a)).
The aged sample that had been stored in air at 50% RH showed a similar development to those unaged samples tested in 25% and 50% RH air and N2, that is of an initial minimum over 20 cycles followed by an increase to a plateau COF by 200 cycles. The remaining air-aged samples (dry air and 25% RH air) exhibited an initial minimum over the first 40 cycles, but this was followed by a further local maximum at 75–125 cycles which then tended to a lower steady state level. In each case the COF then remained largely unchanged through to the end of the experiment.
Surface analysis
Energy-Dispersive X-ray spectroscopy
Figure 5 shows the Oκα1 EDS signal in the wear scar of the MoS2 coated stainless steel disc for the ‘unaged’ sample tested in dry N2 and the various ‘aged’ samples tested in dry N2. The intensity of oxygen signal is more concentrated in the wear scar for all samples and this is generally at localised points. The intensity of the O signal both within the wear scar and outside of it increases with humidity. In each case the air aged samples exhibited greater intensity of oxygen within the wear track when compared to the N2 aged samples (at any given RH). In addition, it is noteworthy that the ‘unaged’ sample tested in dry N2 still exhibited some above-background O signal within the wear track despite no extended exposure to water or air; this sample, unsurprisingly, most closely resembled the sample aged in dry N2 for 500 h. Finally, the ‘aged’ sample exposed to 50% RH air exhibited the most consistent oxygen signal increase across the entire wear track.

Oκα EDS maps of wear scars of the post-test MoS2 coated stainless steel disc for the ‘unaged’ sample tested in dry N2 and the various ‘aged’ samples tested in dry N2. In each case the samples were exposed to the ageing environment for 500 h.
Raman
Raman spectra of the post-test MoS2 coated stainless steel disc for the ‘unaged’ sample tested in dry N2 and the various ‘aged’ samples tested in dry N2 are shown in Figure 6. In each case a comparison is made between undisrupted surface away from the wear scar (on the left) and disrupted surface at the wear scar (on the right). In almost all cases bands for MoS2 at 287, 383 (
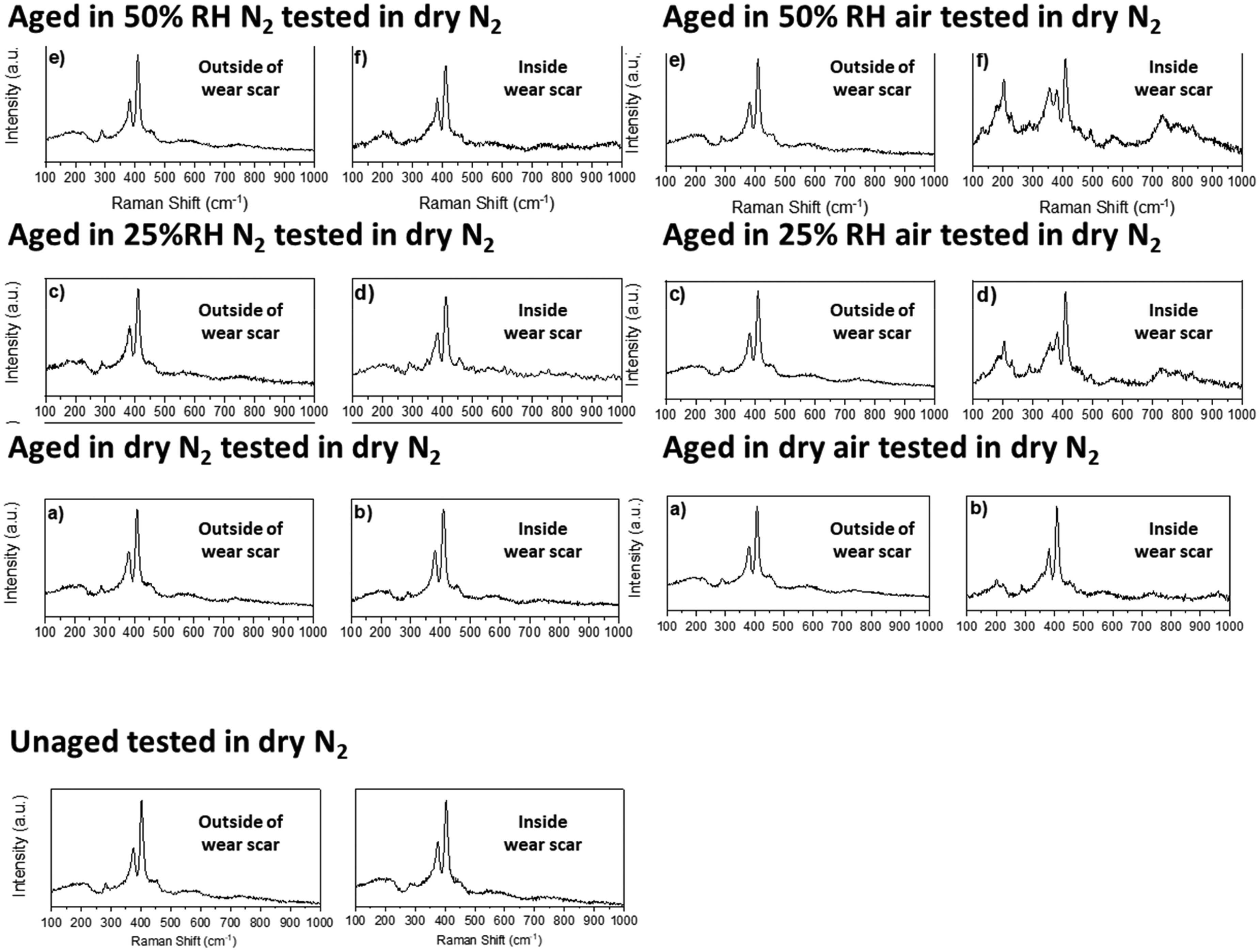
Raman spectra of post-test MoS2 coated stainless steel disc for the ‘unaged’ sample tested in dry N2 and the various ‘aged’ samples tested in dry N2. In each case the samples were exposed to the ageing environment for 500 h.
The only two exceptions to this are the two samples ‘aged’ in air with H2O present (25% and 50% RH). In these cases (far upper right of Figure 6) the undisrupted surface is still characteristic of MoS2 only. In contrast, the Raman spectra of the wear scars indicate formation of MoO2 (202, 227, 363, 489, 565 and 728 cm−1) and, to a lesser extent, MoO3 (197, 819 cm-1). The proportion of signal of these two oxides is greater at 50% RH in air as compared to 25% RH in air.
The spectra for the two samples ‘aged’ in 25% and 50% RH air also exhibited bands at ∼140, ∼180 and ∼780 cm−1 that could not be assigned. The
Transmission electron microscopy
Transmission Electron Microscopy observations on the wear scar showed MoS2 frictionally transformed to a depth of 25–75 nm (Figure 7). Of the four samples examined only the two samples aged in N2 (dry N2 and 50% RH N2) showed significant crystallographic reorientation with basal orientation of lamellar parallel to the sliding direction. The remaining two samples aged in air (dry air and 50% RH air) did not show significant crystallographic reorientation associated with enhanced lubricity. The degree of transformed depth (δ) further correlated with aging environment and the presence of H20; δ50% RH N2 = 42.15 ± 4.03, δ50% RH Air = 65.86 ± 13.49, δdry N2 = 37.53 ± 9.31 and δdry N2 = 57.31 ± 1.94. No film failure was seen during testing.

Transmission electron micrographs of coating material on the steel substrate. Micrographs are cross-sections of the coating material with sliding direction left to right on the images. Key: [A] protective Pt, [B] Ir conducting layer, [C] C layer from previous SEM analysis, [D] MoS2 frictionally transformed, [E] MoS2 bulk.
Discussion
The aim of this study was to explore the impact of both sliding and ageing environments on the tribological performance of MoS2 PVD-coatings, using a combination of tribological, spectroscopic, and microscopic techniques. For the sliding environment, the focus was on understanding how humidity and oxygen interact with the MoS2 film during tribological testing, leading to changes in the coefficient of friction. In contrast, the ‘ageing’ protocols examine how different storage conditions—varying in humidity and oxygen content—affect the chemical and structural integrity of the MoS2 film and its subsequent tribological behaviour over longer term non-harsh exposures. The findings highlight the importance of environmental factors, both during storage and testing, in determining the performance of MoS2 coatings and provide new insights into the mechanisms driving their degradation.
Effect of the sliding environment
Where the effect of the sliding environment was investigated samples were ‘unaged’ in the sense that they were controlled as much as possible between formation and use, and that the samples were nominally identical. With the exception of the ‘unaged’ sample tested in dry N2, all samples saw an initial reduction in COF followed by a steady increase. The initial decrease (over first 50 cycles) in COF is attributable to the ‘run-in’ period where the cycles cause a removal of any initial oxygen and water contaminated interface.1,9 The results show that this is independent of the environment of the test, albeit the minima COF achieved was environmentally dependant (i.e., environment dependant).
The subsequent increase in COF over ∼100–500 cycles (or 1500 cycles in the case of 25% RH/air) indicates that tribological shear allows the MoS2 coating to be further ‘activated’ by the humidity and/or air that is present in the test environment at the contacting interface. The degree to which the eventual COF is increased over the ‘run-in’ COF is proportional to the presence (and extent) of humidity as well the presence of air, as would be predicted by an oxidation mechanism. The effect is seen in the presence of humidity/N2 but is enhanced when humidity and O2 is present from the air. Khare and Burris 6 showed that the COF is strongly dependent on humidity (but independent of O2) below 100°C, and above 100°C they showed that the COF is less strongly dependent to O2 (but independent of H2O). Serpini 10 also noted that there was little difference in friction response when alternating between dry N2 and air, which appears to agree with Khare and Burris. In our ambient temperature dry N2 and air data, however, we have observed O2 playing a synergistic role in the evolution of the coefficient of friction values. These observations are consistent with other studies and can be explained through the continued exposure of reactive edge sites and defects within the coating, which have a high affinity for water binding to S vacancies.11,12 Furthermore, the intermittent friction spikes observed at 25% RH after ∼800 cycles (Figure 3(b)) may be attributed to transient disruption/re-formation of the MoS2 running film/transfer layer at the MoS2–440C interface. At this intermediate humidity, patchy water adsorption and associated changes in interfacial adhesion/third-body behaviour may produce a metastable sliding state, giving brief excursions to higher shear strength before the basally oriented low-shear tribofilm is re-established. However further work is required to confirm this and interrogate the interplay between interfacial reaction kinetics, thermodynamics and COF.
The ‘unaged’ sample tested in dry N2 exhibited some EDS O signal both away from the wear scar and inside the wear scar (Figure 5). The small O signal away from the wear scar likely arises from the deposition process 13 and adventitious water vapour adsorption on MoS2 coatings during brief air exposure between formation, testing, and analysis, providing a baseline for comparison with later EDS results. The fact that the product of this reactivity is not visible to Raman spectroscopy (Figure 6) would suggest that it is probably adsorbed water related rather than from oxidation.
Effect of the ageing environment
For samples ‘aged’ in N2 for 500 h and then slid in dry N2, the tribology tests indicate that the ageing environment (i.e., the presence and extent of humidity) is not a factor in determining the COF when compared to ‘unaged’ testing in a dry N2 environment. All tribological tests show an initial decrease in COF over the first 50 cycles which is, again, attributable to a ‘run-in’ period. The EDS results indeed show that there is an increase in O signal intensity away from the wear scar and that this may be related to the humidity of the storage environment or initial O within the deposited coating. Raman spectroscopy results did not indicate formation of oxides. This is consistent with previous studies hypothesising adsorbed water and/or hydroxyl species on the MoS2 surface generated during the storage period. 12 Within the wear scars of these samples, a “patchiness” is observed in the distribution of the EDS O signal. It is not clear if the patchiness is related to localised oxidation due to coating defects or damage, as site specific Raman spectroscopy, or to changes in the coefficient of friction (COF). It is possible that this variation in the EDS O signal within the wear scars results from adsorbed water due to post-tribology exposure; this aspect requires further interrogation.
For the sample ‘aged’ in dry air for 500 h and then slid in dry N2, the behaviour is largely indistinguishable from those ‘aged’ in the N2 environments. The one notable exception to this comparison is the more complex start of the test with perhaps a first minimum after 50 cycles and a second minimum after 200 cycles. This may indicate that some surface oxidation (from storage in dry air) influences the early COF but is quickly removed and does not influence the later parts of the test. An explanation for this more complex running in behaviour maybe related to enhanced Mo-O-Mo interactions 14 through longer N2 + O2 exposure. 15
For samples “aged” in air at 25% and 50% relative humidity (RH) and then slid in a dry N2 environment, COF increases throughout the experiment. This suggests that the effect is linked to H2O (or its derivatives) permeating the MoS2 coating to at least the depth assessed during tribological tests. Furthermore, this effect is directly related to humidity levels and occurs only in the presence of O2. These results support the idea that ‘ageing’ in the presence of humid air results, at least to some extent, in reaction at lamella edge sites and that this reaction results in an interruption of the formation of larger, aligned MoS2 during the ‘run-in’ phase. 9 Additionally, our data suggests that this effect is constant throughout these tests implying that the mechanism responsible is not restricted only to the near surface layer and any ‘poisoned’ region is not removed through tribological processes. Restricted basally orientation MoS2 lamella is further confirmed by TEM analysis shown in Figure 7.
The EDS data for the samples ‘aged’ in N2 (dry, 25% RH and 50% RH) again show an increase in the O signal away from the wear scar and without any Raman MoO2 or MoO3 signature. Once again indicating some degree of water adsorption on the surface post-tribology test or localisation of oxidation not observed by the Raman: similar to samples ‘aged’ in humid N2. Similar observations were made for samples ‘aged’ in air, albeit with increased EDS O signal detected outside and within the wear track. This also increases with humidity. In the case of samples aged in 25% and 50% RH air, some of this O signal intensity is related to oxidation products since both MoO2 and MoO3 are detected in the wear scar by Raman spectroscopy; the proportion of the Raman signal of these two oxides (relative to MoS2) increases with the humidity of the ageing storage environment. Comparison of Raman spectroscopy inside and outside the wear track suggest these were formed because of mechano-chemical interactions, which in turn affect both run-in and final COF. From this systematic investigation, ‘O’ species penetrate the coating to a significant depth and is implicated in altered run-in dynamics and an increased COF. Interestingly, in the presence of H2O, COF is further increased and increases with RH.
Overall, the mechanism suggests that the “O species” detected by EDS in all environments arise partly from water adsorption, without a Raman signature when oxidation is minimal, and partly from the formation of Mo oxides, which occurs when oxygen is available, such as in air or at higher relative humidity (RH). Ab initio calculations indicate that water alone is not thermodynamically favourable for efficiently oxidizing MoS2. 16 However, in oxygen-containing environments, water can lower the barrier for oxidation reactions, particularly at defect or edge sites. 16 Water vapor can accelerate the oxidation of MoS2 by facilitating oxygen activation and interacting with defect sites. Under humid conditions, water molecules may adsorb onto the MoS2 basal plane or edges, participating in reactions that replace sulphur with oxygen, leading to the formation of MoOx species. 17 Similar observations of accelerated oxidation due to the combined presence of O2 and H2O have been reported in the semiconductor literature. 18 The interplay of water's physical adsorption, its role in enhancing localised oxidation under mechanical stress/input of kinetic energy further lowering thermodynamic activation barriers, and the subsequent formation of non-lubricious oxide phases explains both the spatial variation (inside versus outside the wear track) and the environmental dependence of friction behaviour.
It is noteworthy that the MoS2
The TEM observations suggest that the samples aged in N2 showed significant basal orientation compared to the air aged samples. This supports the idea that the films only experience very surface level changes with humidity in N2 (we suggest adsorbed water and/or hydroxyl development) that do not affect how the samples are subsequently ‘run-in’. In contrast, the sample aged in 50% RH air showed significantly less uniformity after tribological testing which is in agreement with changes in the chemical characteristics of the film affecting the re-orientation and therefore the COF. The dry air aged sample exhibited some reorientation of film lamellar but not to the degree of the N2 stored samples. Again, this manifests as a slightly increased steady-state coefficient of friction when compared to ‘unaged’ or ‘aged’ MoS2 coatings tested in N2 only environments.
As mentioned earlier, Khare and Burris reported that water alone did not promote MoS2 oxidation at room temperature and that friction remained essentially independent of oxygen below 100 °C. This is not inconsistent with our findings. In our study, neither dry/purged storage nor exposure to oxygen in the absence of moisture led to measurable changes in COF (Figure 4). The sputtered PVD coatings examined here may have a more open, defect-rich, columnar morphology compared to aerosol deposited MoS2 layers, which increases the density of edge and grain-boundary sites available for adsorbate-assisted reactions. Under prolonged ambient storage, simultaneous H2O and O2 adsorption appears to activate these sites, producing the small Raman shifts and an increase in COF we report. Our results therefore extend the earlier observations to a more oxidation-susceptible coating architecture and to longer, static exposures at room temperature. Figure 8 shows the proposed room-temperature, water-assisted oxygen degradation pathway for magnetron-sputtered MoS2 coatings based on these studies observations.
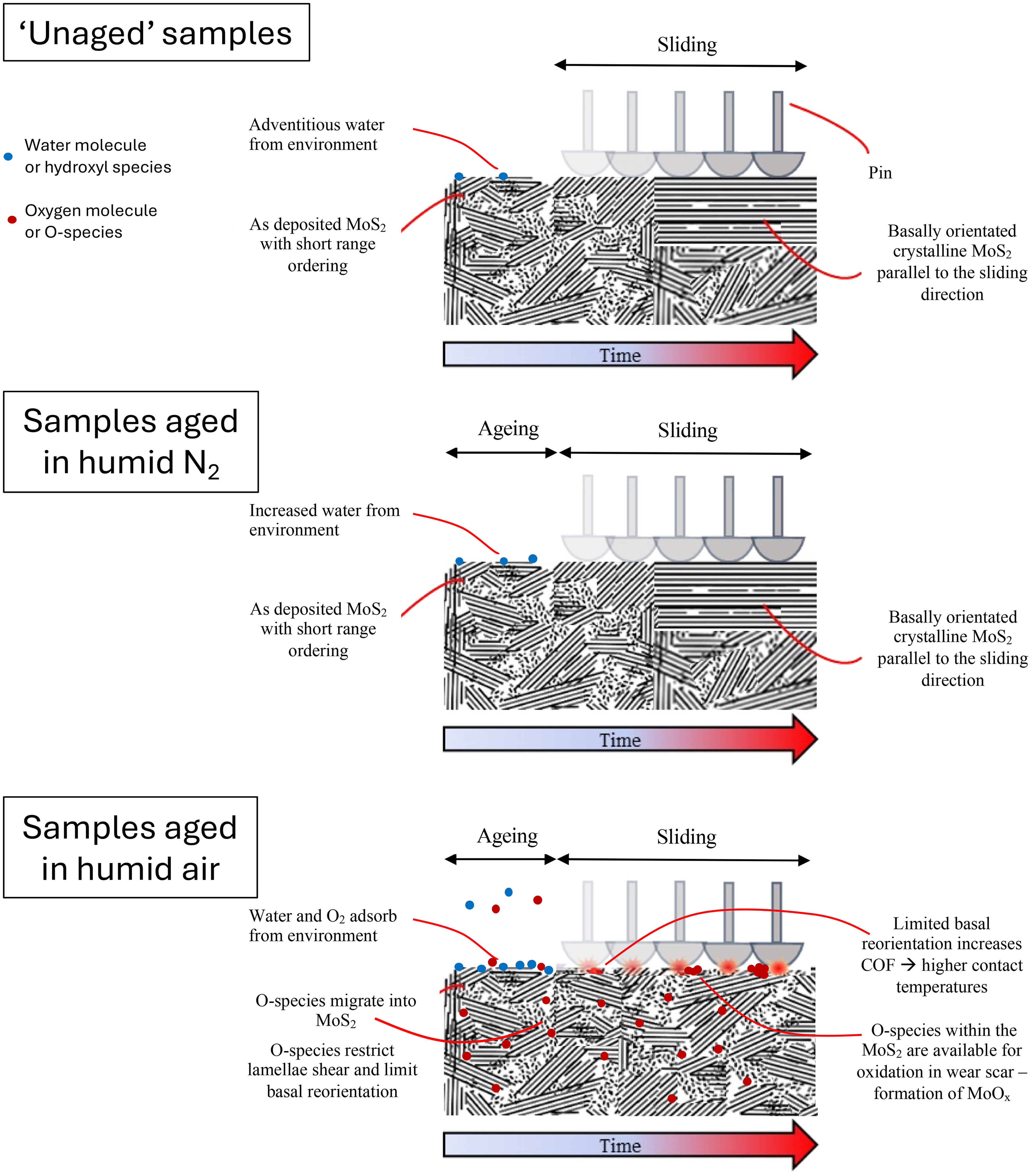
Proposed room-temperature, water-assisted oxygen degradation pathway for magnetron-sputtered MoS2 coatings. (1) Pristine PVD film on steel. (2) Humid N2 storage leads only to reversible H2O adsorption. (3) Concurrent H2O and O2 in air (i.e., humid air) lower the activation barrier for oxygen insertion at defect/edge sites. (4) During sliding, films aged in inert gas rapidly realign their basal planes, delivering low friction (µ ≈ 0.05). Films aged in humid air retain mis-oriented lamellar as a result of oxygen incorporation in the MoS2, causing a persistent friction penalty (µ ≈ 0.12) and an increased flash temperature. Oxygen incorporated within the MoS2 film is available to chemically react at the contact point and forms MoOx in the wear scar.
The insights gained from this study have direct and wide-ranging industrial value. By showing that even moderate humidity in air (≥ 25% RH) doubles the steady-state coefficient of friction whereas both dry and humid nitrogen cause no measurable degradation, We define a practical storage threshold of <25% relative humidity, ideally approaching 0% RH, for PVD-MoS2-coated components. Adopting a simple dry-nitrogen purge storage systems, with a humidity indicator for verification, may extend storage shelf-life of tribological components and mitigate unforeseen failure due to coating degradation. The storage protocol may be transferable to MEMS gyroscopes, vacuum-pump seals and semiconductor-handling arms, providing new insights that contribute to enhance safety and efficacy of other engineering tribological systems.
Conclusions
This study set out to isolate and quantify the individual and combined effects of water vapour and oxygen on the tribological, chemical and microstructural evolution of 1 µm PVD-MoS2 coatings stored at room temperature. Ageing was performed for 500 h in six tightly controlled gas/humidity environments, followed by micro-tribological testing and correlative SEM-EDS, Raman and TEM analysis. The principal conclusions are:
To our knowledge, this is one of the first systematic ambient-temperature studies on sputtered PVD MoS2 that directly links combined H2O–O2 exposure during storage to both measurable chemical modification and degraded tribological performance, complementing recent reports on environment-assisted changes in MoS2 films.1,4 Multiple ageing conditions, replicate tests, and correlative characterisation underpin the robustness of the findings. The resulting storage protocol and mechanistic insight equip aerospace, defence and precision-instrument engineers with evidence-based shelf-life limits, reducing first-motion failures and informing emerging international standards. Future work will extend the approach to doped MoS2 variants, cyclic humidity exposure and in-situ spectroscopy to refine the kinetic model of low-temperature tribo-oxidation.
Footnotes
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Novel Surface Engineering Strategies for Dry-Sliding Contacts, (grant number EP/N509681/1).
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
