Abstract
The research evaluates the impact of Sargassum algae methyl ester (SAME20) with 50 and 100 ppm silicon dioxide (SiO2) nanoparticles on the performance and emissions of a single-cylinder, four-stroke diesel engine. SiO2 nanoparticles were characterized using FTIR, SEM, and TEM. Diesel, SAME20, SAME20 + 50 ppm of SiO2, and SAME20 + 100 ppm of SiO2 blends were tested at 1500 rpm under loads from 25% to 100%. The SAME20 + 100 ppm of SiO2 blend outperformed others, improving brake thermal efficiency by 1.43% and reducing brake-specific fuel consumption by 5.35% compared to diesel. It also decreased HC, CO, and smoke emissions by 11.12%, 18.1%, and 16.2%, respectively, with a slight NOx increase. Tribological analysis using a four-ball tribometer showed the SAME20 + 100 ppm of SiO2 blend reduced the coefficient of friction by 76.89% and wear scar diameter by 59.19%. SEM analysis confirmed reduced surface wear and damage, highlighting improved engine and tribological performance.
Introduction
Biodiesel is gaining attention as an alternative fuel due to price volatility, industrial growth, and climate change concerns. However, challenges such as feedstock shortages, high production costs, and competition with food crops remain.
1
Researchers are increasingly focusing on eco-friendlier and more cost-effective algae-based biofuels than crop-derived biofuels.
2
Algae offers a high growth rate (30 to 100 times faster than conventional crops), high lipid content, and superior efficiency compared to terrestrial seed crops.3,4 Various researchers have explored algae biodiesel to assess engine performance and emission characteristics. In a single-cylinder diesel engine, biodiesel's performance and emission characteristics derived from microalgae
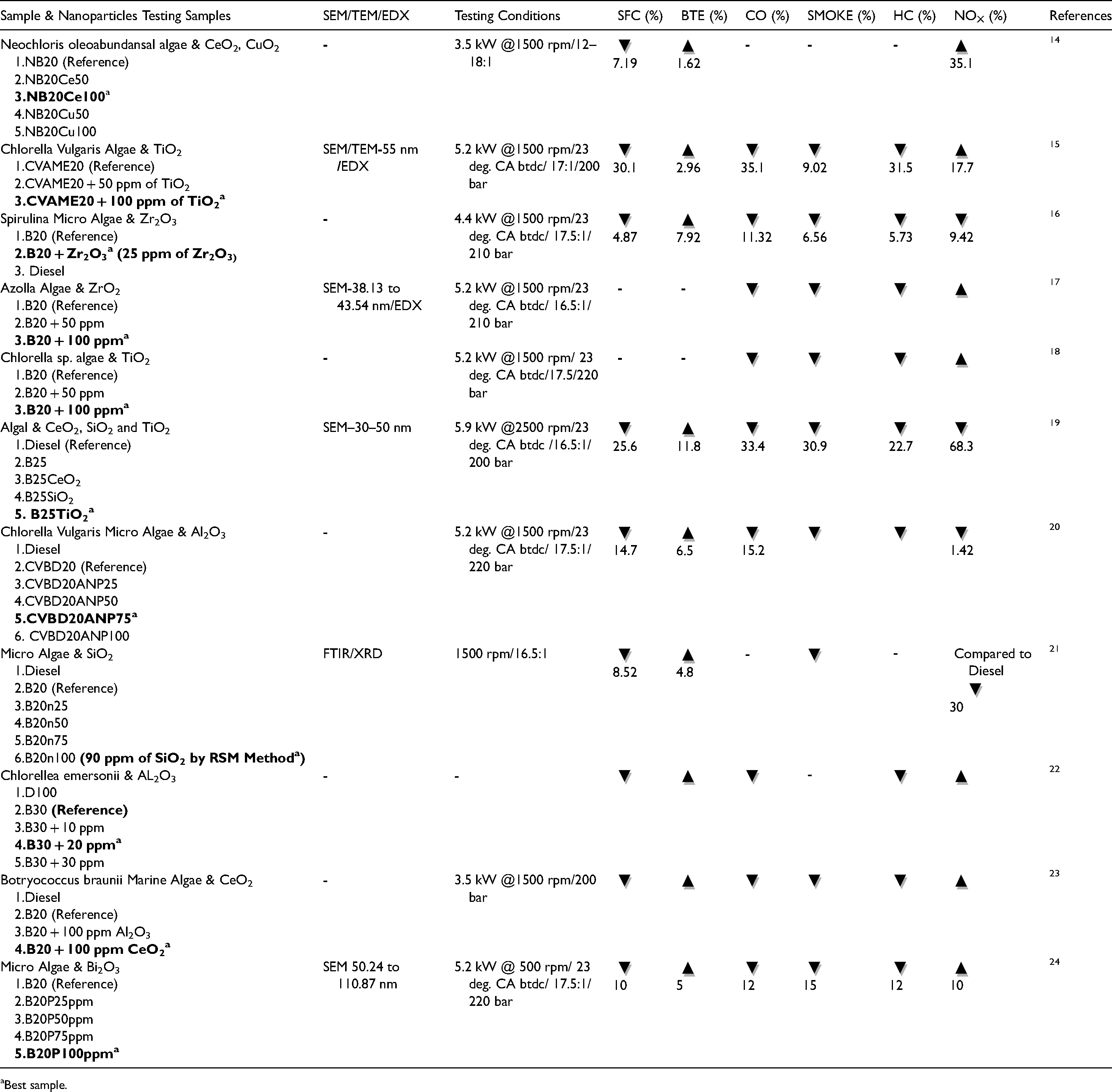
Overview of previous studies on engine performance and emissions using various algae biodiesel blends with metal oxide nanoparticles.
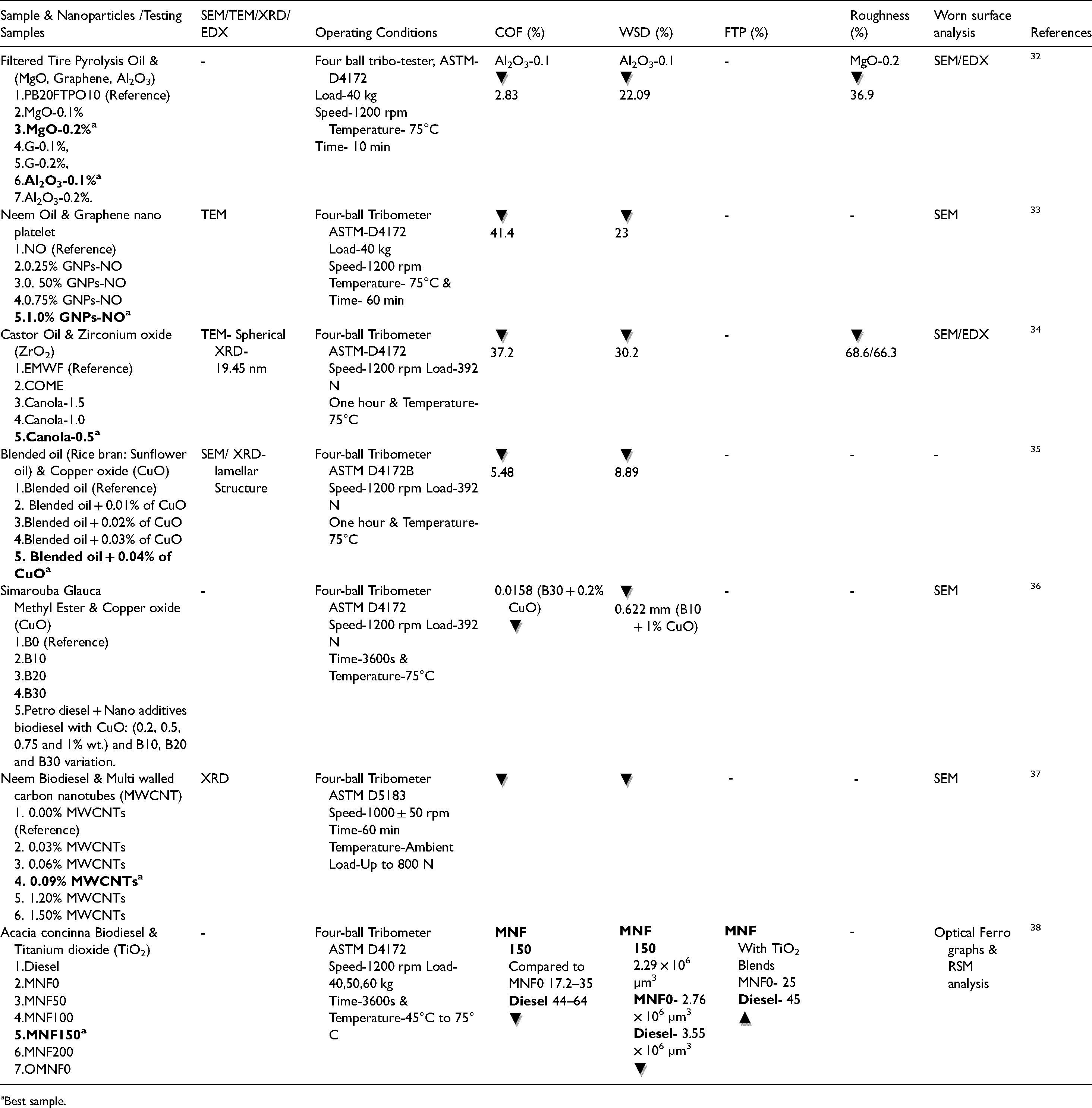
Summary of existing studies on tribological analysis of biodiesel blends enhanced with various metal oxide nanoparticles.
Experimental setup and procedure
Sargassum algae oil
Among algae feedstocks,
Extraction of algae oil and transesterification process
Silicon dioxide
Silicon dioxide nanoparticles have garnered significant attention as nano additives in internal combustion engine fuel blends due to their unique properties, including high thermal stability, excellent dispersibility, and enhanced catalytic activity. Table 3 demonstrates the chemical and physical characteristics of attributes of silicon dioxide.
Illustrates the chemical and physical features of silicon dioxide.

Testing fuel preparation
A magnetic stirrer formulated the fuel blend SAME20, composed of 80% pure diesel and 20% S
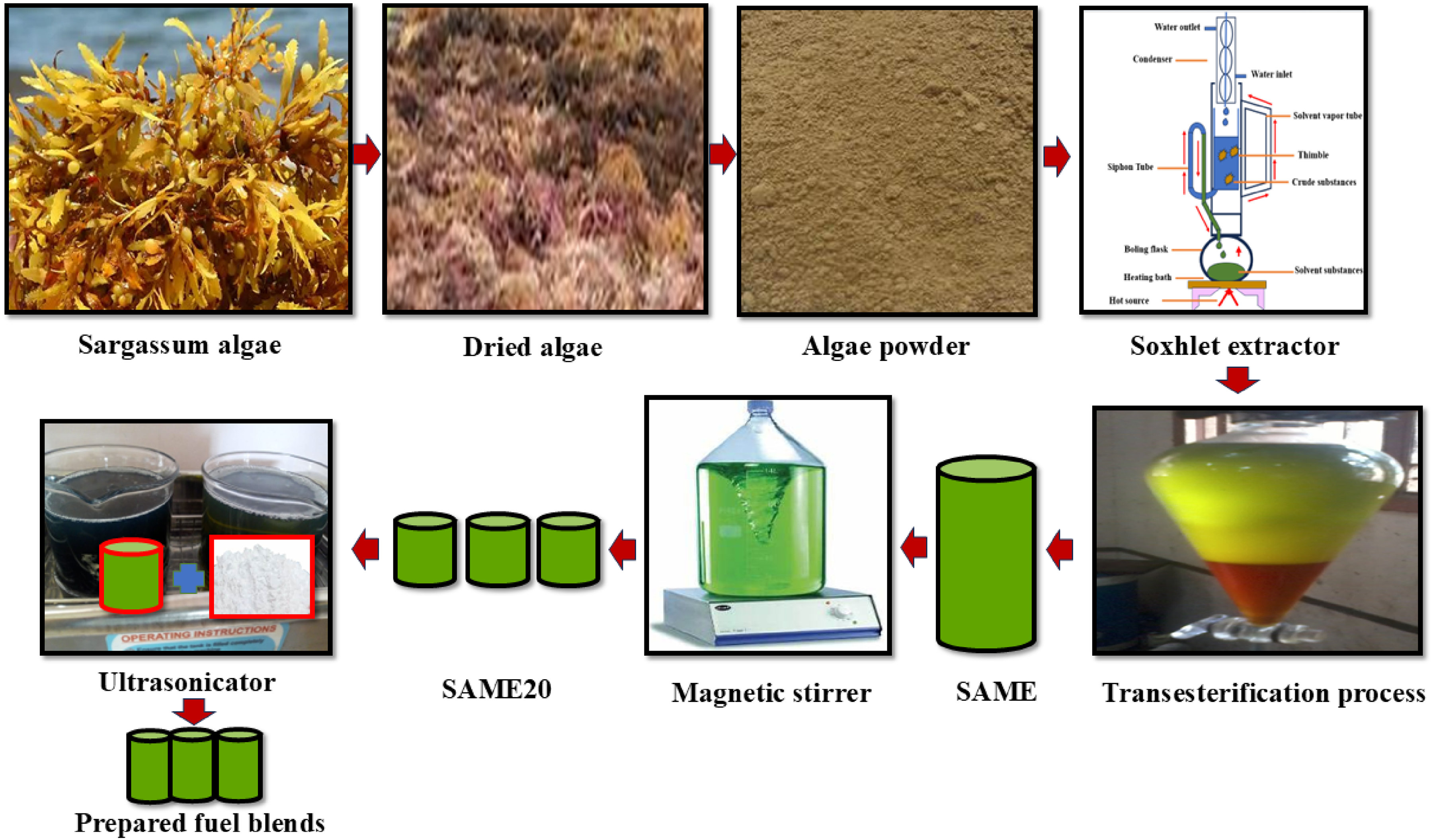
Schematic representation of the overall sample preparation process.
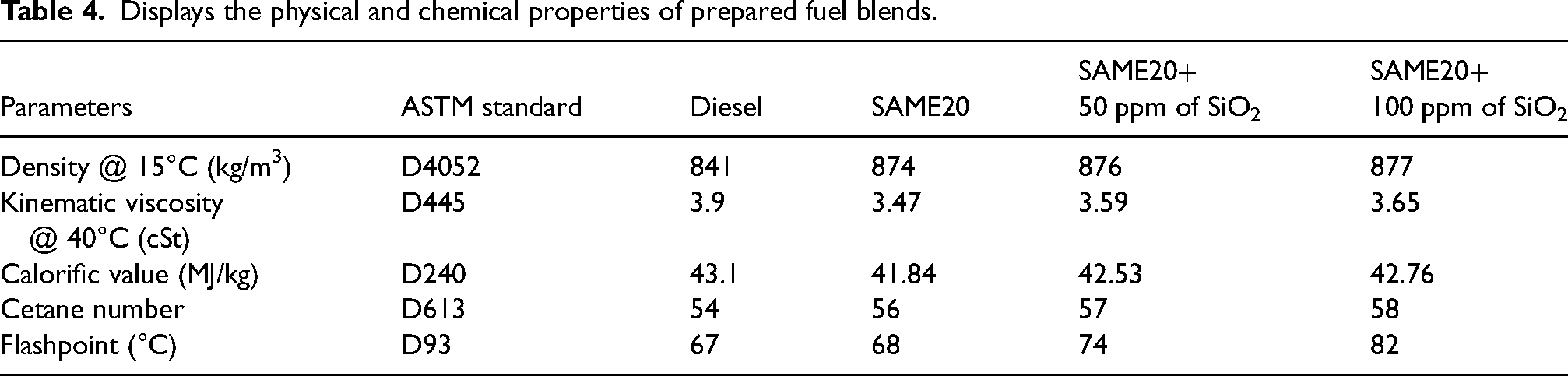
Displays the physical and chemical properties of prepared fuel blends.
Engine experimental setup
The experiments utilized a Kirloskar single-cylinder diesel engine with direct injection and natural aspiration. The engine was set with an injection timing of 23° bTDC and a fuel injection pressure of 210 bar. It featured a 6.5 L fuel tank, a connecting rod assembly with a diameter of 203 mm, a cylinder bore of 87.5 mm, and a stroke of 110 mm. The engine generated 5.2 kW of power while maintaining a coolant temperature of 80°C. A data acquisition (DAQ) system connected to the setup measured parameters such as fuel flow, air flow, fuel consumption, and cylinder pressure. The DAQ system was connected to a computer running Enginesoft software, enabling real-time data acquisition of combustion parameters (net heat release rate), performance metrics (BTE and BSFC), and cylinder pressure. The AVL 437C smoke meter was used to monitor smoke emissions. The AVL 444 Di-gas analyzer was used to measure exhaust emissions, such as HC, NOx, and CO. Before data collection, the engine was driven for 15 min using diesel fuel to reach steady-state conditions, during which the water supply temperatures at critical points were monitored. Afterward, the system was purged of diesel fuel, and biodiesel, both with and without nanoparticle blends, was introduced, ensuring the fuel supply system was free from air blockages. To ensure consistency, a stable air temperature was maintained throughout the experiments, enabling precise evaluation of the engine's performance and emission characteristics for each test fuel. Figure 2 shows a schematic illustration of the test engine.
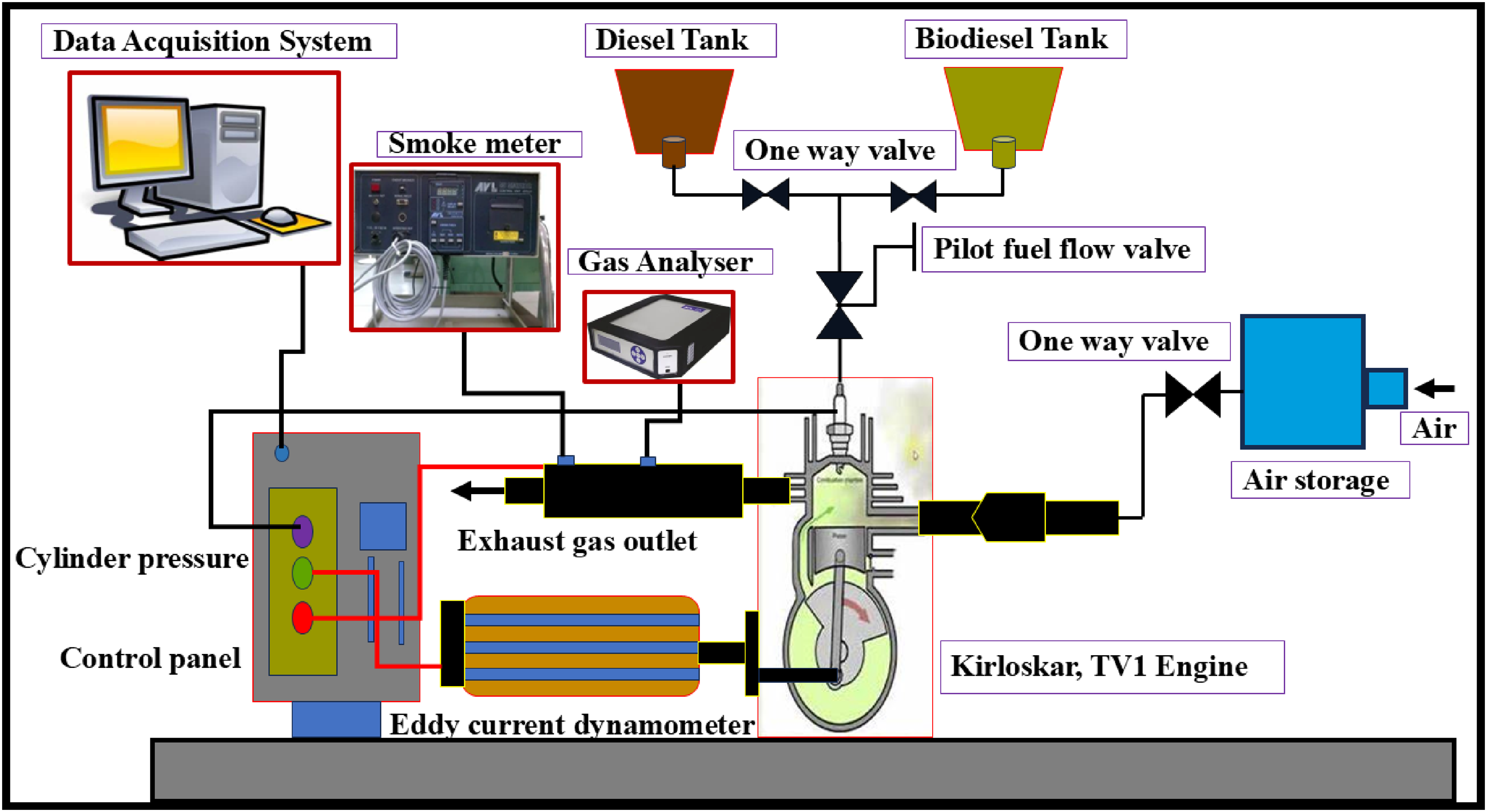
Schematic illustration of the engine experimental arrangement.
Four-ball tribometer experimental setup
A four-ball tribometer (DUCOM TR30L-LAS) was used to conduct tribological tests according to ASTM D4172, as illustrated in Figure 3.The apparatus has three steel balls at the bottom and one at the top, which are attached to a collar at the end of a vertical spindle, while the remaining three balls are in a pot of lubricant. The operation involves the rotation of the lower balls against the stationary upper ball, with approximately 10 ml of the test lubricant added to the steel cup for each trial. The four-ball tribometer is equipped with a computer for monitoring friction torque and calculating the coefficient of friction. This experiment was conducted under carefully controlled conditions: 1200 rpm speed, 15 kg load, 3600 s duration, and 75°C temperature. The wear scar diameters on the three lower balls were measured and averaged at the end of the experiment to calculate the average wear scar diameter. The balls were prepared for subsequent surface analysis by carefully cleaning them with a polymer brush and running water to remove debris without causing significant abrasion.

Four-ball tribometer equipment.
Result and discussions
This section provides experimental findings about the performance and emissions of a diesel engine powered by
FTIR spectrum of SiO2
The bonds and functional groups in SiO2 were identified using Fourier transform infrared. Figure 4 shows the FTIR spectrum of SiO2. The spectrum of FITR exhibits the graphical information in the form of wavelength (cm−1) and the transmittance (%) for SiO2 nanoparticles. The provided FTIR spectrum shows characteristic peaks that confirm the presence of silica. The strong absorption at 1082.90 cm−1 indicates Si-O-Si stretching vibrations, while the peak at 796.03 cm−1 corresponds to Si-O-Si bending vibrations. These features suggest a silica structure. Additionally, the peak at 3221.11 cm−1 indicates O-H stretching vibrations, which could imply the presence of surface hydroxyl groups or adsorbed water. This is further supported by the H-O-H bending vibration observed at 1637.47 cm−1. Overall, the spectrum confirms that the sample is a silica-based material with some degree of water adsorption.
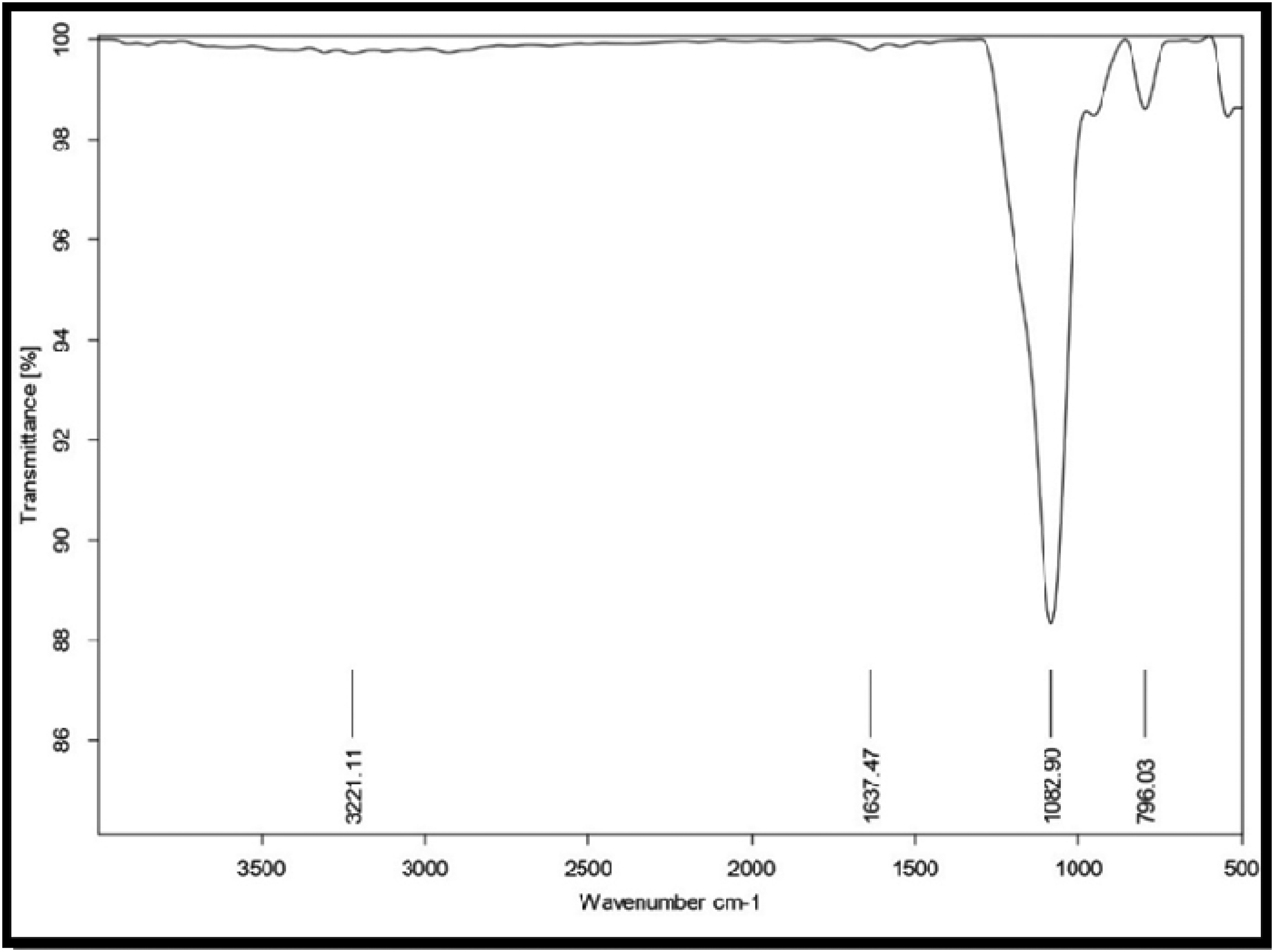
FTIR spectrum of SiO2.
Structural and morphological analysis of SiO2 nanoparticles
Scanning electron microscopy offers high-resolution imaging, allowing precise analysis of material surface structures. The SiO₂ nanoparticles were characterized using a scanning electron microscope operated at 20 kV. Figure 5(a) illustrates an SEM image of the SiO₂ nanoparticles, where the brighter areas indicate their crystalline structure. The image also reveals loosely formed agglomerates at a resolution of 20 μm, with the nanoparticles appearing nearly spherical. Figure 5(b) provides a transmission electron microscopy image that shows detailed information on the nanoscale shape, structure, and crystallinity of SiO2 nanoparticles. TEM analysis confirms that the nanoparticles are within a 30–60 nm size range.

(a) SEM (b) EDX image of SiO2.
Brake-specific fuel consumption
BSFC, also known as SFC, measures how much fuel is required to produce one unit of power (one kilowatt) over an hour of engine operation. 43 As shown in Figure 6(a), the BSFC values varied across different fuel blends. At peak load, the BSFC ranged from 0.28 kg/kWh for diesel in the SAME20 to 0.284 kg/kWh for the same in the SAME20 + 50 ppm of SiO2, 0.274 kg/kWh for the SAME20 + 100 ppm of SiO2, and 0.265 kg/kWh for the SAME20. These findings indicate that the SAME20 fuel had a higher brake-specific fuel consumption than other blends due to its lower heating value. In contrast to traditional diesel, the BSFC decreased by 2.14% for the SAME20 + 50 ppm of SiO2 and 5.35% for the SAME20 + 100 ppm of SiO2. However, SiO2 nanoparticles enhanced combustion efficiency by promoting better fuel atomization, improving the air-fuel mixture, and ensuring complete combustion. This reduced brake-specific fuel consumption as the engine could extract more energy from the same fuel. Overall, incorporating nanoparticles into fuel blends improved fuel quality and efficiency, as evidenced by lower BSFC values. Other researchers have reported similar results. Adding TiO2, Cr2O3, and SiO2 to Calotropis gigantea seed oil significantly reduces the BSFC. At higher engine loads, the BSFC values decrease for the tested fuels: diesel is approximately 0.3 g/kWh, CGSB20 is 0.46 g/kWh, CGSB20 + Cr2O3 is 0.33 g/kWh, CGSB20 + SiO2 is 0.37 g/kWh, and CGSB20 + TiO2 is 0.41 g/kWh. This indicates that increasing engine load reduces the BSFC. Furthermore, dispersing Cr2O3 nanoparticles in CGSB20 results in less BSFC than other tested samples. 44
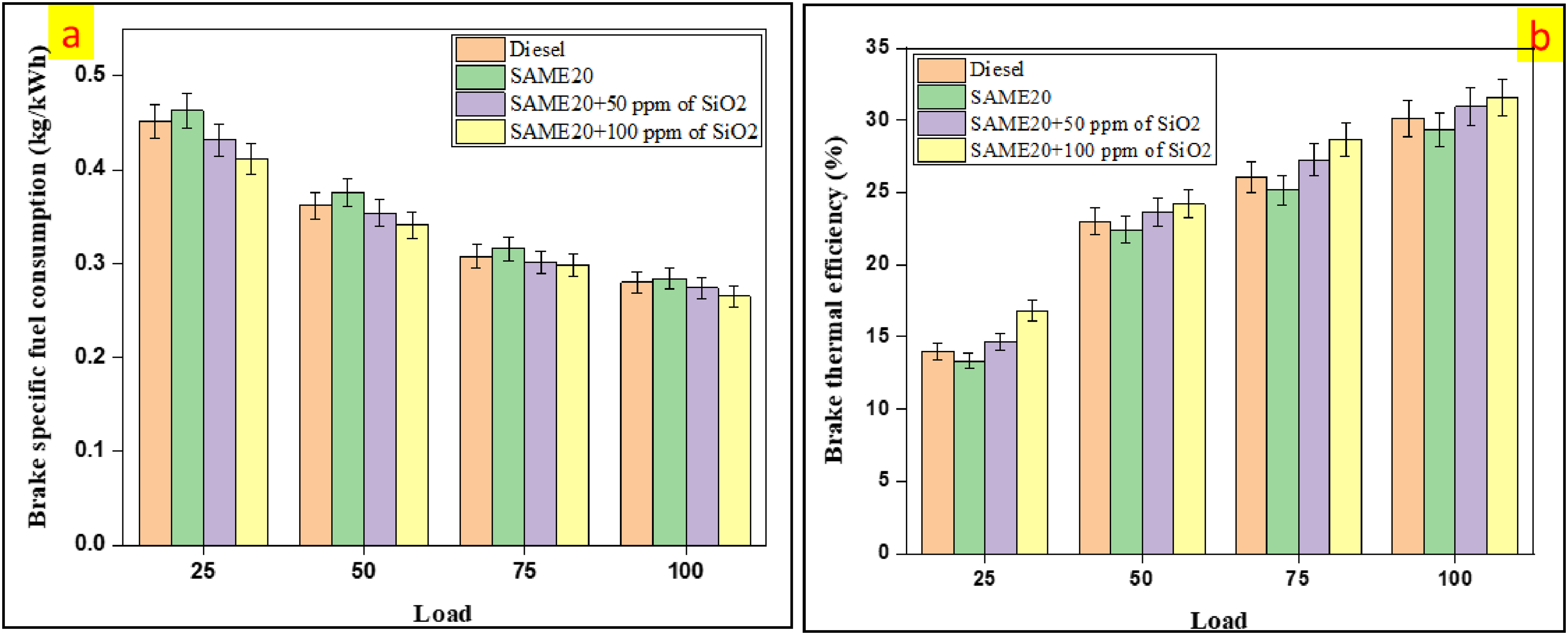
(a) The variation of BTE and (b) BSFC with different load conditions.
Brake thermal efficiency
Brake thermal efficiency is the ratio of an engine's brake power output to the heat energy input from its fuel. Figure 6(b) illustrates how brake thermal efficiency changes with engine load while utilizing various fuel blends. The BTE values at 100% engine load were 30.13% for diesel, 29.34% for SAME20, 30.95% for SAME20 + 50 ppm of SiO2, and 31.56% for SAME20 + 100 ppm of SiO2. The physical properties of the SAME20 fuel, including its lesser heating value, higher flash point, density, and viscosity, contributed to the relative decrease in BTE compared to the other fuel blends. Specifically, the lower combustion chamber temperatures associated with the higher flash point of SAME20 resulted in incomplete fuel combustion. In contrast, biodiesel blends containing nanoparticles enhanced BTE. The SAME20 + 100 ppm of SiO2 blend demonstrated the maximum BTE value across all load conditions. BTE increased by 0.82% at a SiO2 concentration of 50 ppm and by 1.43% at 100 ppm of SiO2 compared to diesel. Conversely, SAME20 exhibited a BTE that was 0.79% lower than diesel. SiO2 nanoparticles, due to their high thermal conductivity, improved heat transfer within the combustion chamber, leading to more efficient combustion and enhanced thermal efficiency. The even distribution of SiO2 in the fuel facilitated better atomization and combustion, reducing energy losses and increasing power output for the same amount of fuel. A higher concentration of nanoparticles increased the combustion rate, leading to improved energy conversion and higher BTE. Consistent with prior research, adding CeO2 nanoparticles to biodiesel at 20–80 ppm concentrations increased BTE by up to 1.5%, with the most significant improvement observed at 80 ppm. This improvement is attributed to the faster heat release during combustion as the CeO2 nanoparticle concentration increases. 45
Carbon monoxide emission
Carbon monoxide emissions arise from the incomplete combustion of fuels in engines. Biodiesel usually contains 10 to 12% higher oxygen content than conventional diesel, which enhances complete combustion and reduces CO emissions. Figure 7(a) depicts the CO emissions associated with various fuel blends in relation to load. The carbon monoxide emission is inversely proportional to engine load. Diesel fuel exhibits significantly greater CO emissions compared to biodiesel blends. At maximum load conditions, the CO emissions were recorded at 0.33% for the diesel, 0.31% for the SAME20, 0.28% for the SAME20 + 50 ppm of SiO2, and 0.27% for the SAME20 + 100 ppm of SiO2. The results indicate that CO emissions are reduced when SiO2 nanoparticles are added to SAME20. Compared to diesel, the SAME20 + 50 ppm of SiO2 blend demonstrated a 15.5% reduction in carbon monoxide emissions. In contrast, the SAME20 blend exhibited higher CO emissions than other tested samples due to its inability to achieve complete combustion as effectively as the nanoparticle-enhanced blends. Moreover, the physicochemical properties of SAME20 may lead to lower combustion temperatures and incomplete carbon oxidation, resulting in increased carbon monoxide emissions. SiO2 nanoparticle fuel blends enhance combustion efficiency by facilitating more complete carbon oxidation, which reduces carbon monoxide formation. An increased concentration of SiO2 nanoparticles further accelerates the oxidation of carbon monoxide, effectively lowering its emissions. The maximum reduction of CO emissions was recorded with SAME20 + 100 ppm of SiO2 under peak load conditions, reaching 18.1%. Other researchers have also observed similar findings. Under maximum load conditions, the DSOME-B20 + CuO 75 ppm blend resulted in a 14.28% reduction in carbon monoxide emissions compared to the DSOME-B20 alone. 46
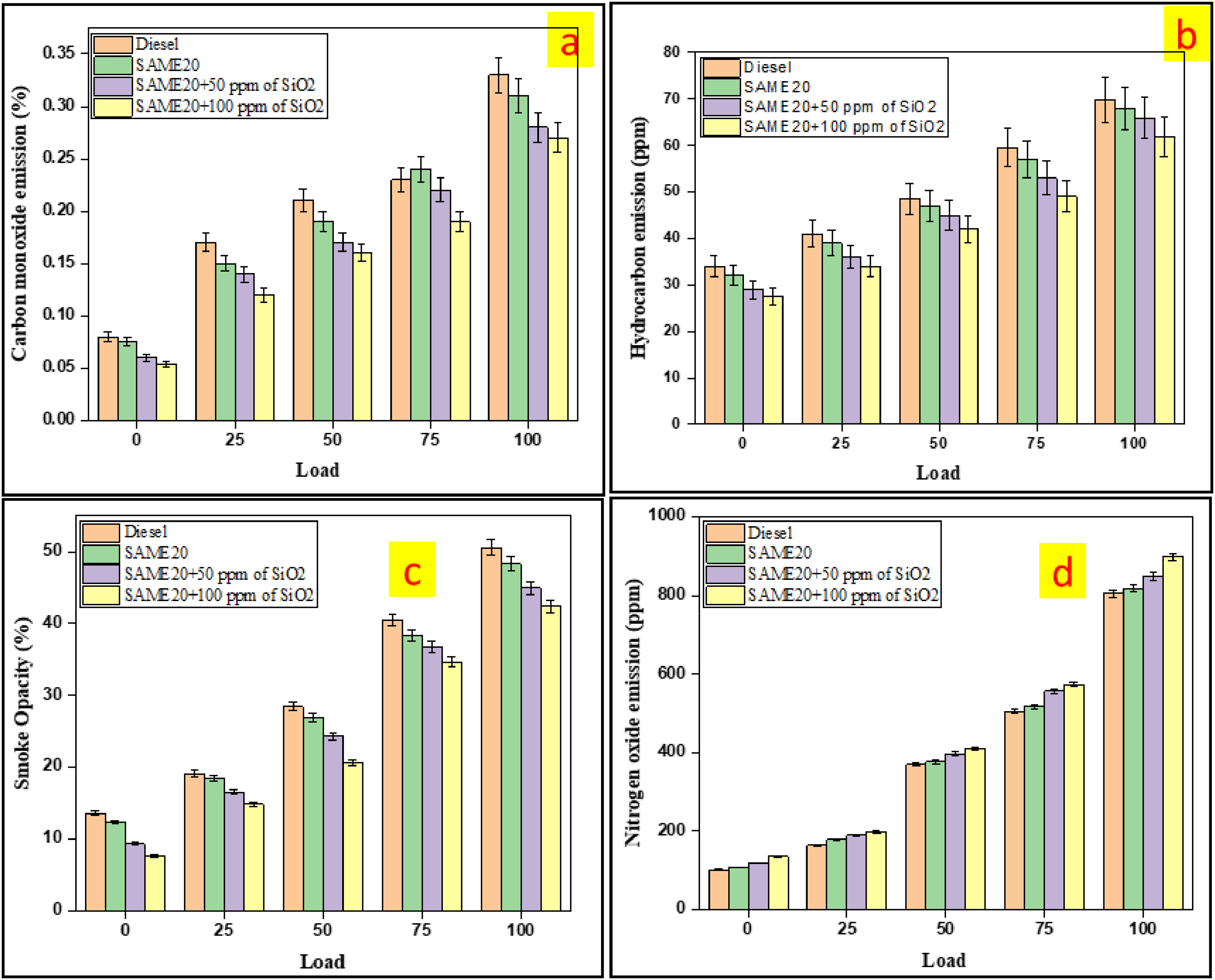
(a) The variation of CO (b) HC (c) Smoke and (d) NOx emission with load different load conditions.
Hydrocarbon emission
The primary source of HC emissions is incomplete fuel combustion, which generates hydrocarbons as a byproduct. Various factors influence these emissions, including the cylinder, the cylinder wall, wall temperatures, the oxygenation characteristics of biodiesel, and the cetane number. These characteristics lead to various patterns in hydrocarbon emissions from diesel engines. Figure 7(b) illustrates the variation in unburned hydrocarbon emissions across various engine loads and fuel mixtures. It has been observed that the amount of hydrocarbon emissions increases in proportion to the engine load. At maximum load, the HC emissions in the exhaust were recorded as 69.8 ppm for diesel, 68 ppm for SAME20, 66 ppm for SAME20 + 50 ppm of SiO2, and 62 ppm for SAME20 + 100 ppm of SiO2. The maximum hydrocarbon emissions observed for diesel fuel blends compared to other tested fuel blends. The catalytic activity and surface area of SiO2 nanoparticle particles improve fuel oxidation and promote complete combustion. These nanoparticles can be mixed with biodiesel to enhance combustion and significantly reduce hydrocarbon emissions. As the concentration of SiO2 increases, combustion further improves, resulting in substantial decreases in HC emissions. Compared to diesel, SAME20 resulted in a 2.57% lower HC emission, SAME20 + 50 ppm of SiO2 showed a 5.57% decrease, and SAME20 + 100 ppm of SiO2 exhibited an 11.12% reduction in HC emissions. These results align with findings from other researchers. They found that incorporating TiO2 nanoparticles into jatropha biodiesel significantly reduced hydrocarbon emissions by approximately 22% compared to base fuel. 47
Smoke emission
The variation in engine load significantly influences smoke emissions during the combustion process. As the engine loads increase, more fuel is supplied relative to the available oxygen, resulting in a fuel-rich mixture that exceeds the prescribed air-fuel ratio. This imbalance leads to incomplete combustion, causing increased smoke emissions from the exhaust. Factors contributing to diesel engine smoke pollution include high combustion rates, fuel atomization, fuel injection dynamics, and rich air-fuel zones. The variation in smoke emissions across different fuel blends and loads is illustrated in Figure 7(c). At maximum load, the recorded smoke emissions were as follows: 50.6% for diesel, 48.3% for the SAME20, 44.9% for the SAME20 + 50 ppm of SiO2, and 42.4% for the SAME20 + 100 ppm of SiO2. Silicon oxide nanoparticles, acting as combustion catalysts, enhance the oxidation of carbon-based particulates, thereby reducing smoke formation. This is attributed to the higher concentration of SiO2 nanoparticles, which promotes more complete combustion and lower smoke emission levels. Similar to previous studies, research examining the effect of CeO2 and Al2O3 in tamarind seed oil methyl ester (TSOME) found that the reduction in smoke emissions for TSOME, TSOME + CeO2, and TSOME + Al2O3 were 15%, 17%, and 16.8%, respectively, all of which were lower than the smoke emissions of diesel fuel at maximum load. 48
Nitrogen oxide emission
Nitrogen oxides are generated through the high-temperature reaction of oxygen and nitrogen in exhaust gases. Several variables affect the formation of nitrogen oxides, including the spray pattern and the physical characteristics of the fuel, like fuel atomization and the presence of nanoparticles within the fuel.49,50 Figure 7(d) plots NOx emissions for each test fuel against engine load. Increasing engine load leads to higher NOx formation, potentially due to elevated exhaust gas and cylinder temperatures. At maximum load, nitrogen oxide emissions were recorded for the 804 ppm diesel, 817 ppm for the SAME20, 849 ppm for the SAME20 + 50 ppm of SiO2, and 898 ppm for the SAME20 + 100 ppm of SiO2. Including nanoparticles in biodiesel blends significantly enhances fuel combustion efficiency, leading to elevated in-cylinder temperatures and increased NOx emissions. The findings from the study reveal that SiO2 nanoparticles included fuel blends that exhibit a noticeable increase in NOx emissions. The NOx emissions compared to pure diesel demonstrated a 1.62% increase for SAME20, a 5.60% increase for SAME20 + 50 ppm of SiO2, and an 11.69% increase for SAME20 + 100 ppm of SiO2. This phenomenon is attributed to the SiO2 nanoparticles accelerating the combustion process, resulting in an approximate 8% increase in cylinder pressure. This rise in pressure reduces the nitrogen oxidation temperature threshold in the air. Furthermore, as the engine load increases, the amplified pressure intensifies the combustion rate within the cylinder, promoting NOx formation in line with Zeldovich's thermal NOx formation mechanisms. Other researchers have observed similar outcomes. Incorporating graphene oxide into waste cooking oil led to an increased NOx emission. Experimental results indicated that the B20GO60 fuel blend produced 19.4% higher nitrogen oxide emissions at peak load than the B20GO20. 51
Tribology
This study utilizes
Coefficient of friction
The coefficient of friction describes the frictional resistance during relative motion and is calculated in a four-ball tribometer as the friction force divided by the average load. A lower COF signifies improved lubrication and reduced wear, whereas a higher COF indicates increased frictional resistance and wear. As illustrated in Figure 8(a), the coefficient of friction exhibits variations across different fuel blends. The coefficient of friction was 0.1242 for the diesel, 0.0529 for the SAME20, 0.0447 for the SAME20 + 50 ppm of SiO2, and 0.0287 for the SAME20 + 100 ppm of SiO2. Because SiO2 nanoparticles are challenging and chemically inert, they serve as excellent anti-friction agents. Dispersed in fuel or lubricant, they form a protective layer between moving components, reducing metal-to-metal contact and lowering friction coefficients. Increasing SiO2 concentrations increases lubrication effectiveness, reducing COF significantly. This helps extend the life of engine components and reduce energy loss due to friction. The coefficient of friction was reduced by 57.4% for SAME20, 64% for SAME20 + 50 ppm of SiO2, and 76.89% for SAME20 + 100 ppm of SiO2, compared to diesel. Previous studies have reported similar findings. Notably, the 0.2 wt.% CuO nano-additive blend exhibited the lowest average coefficient of friction, achieving an 80% reduction than pure diesel. This significant enhancement is attributed to the rolling action of the 0.2 wt% CuO nanoparticles at the contact points. 36
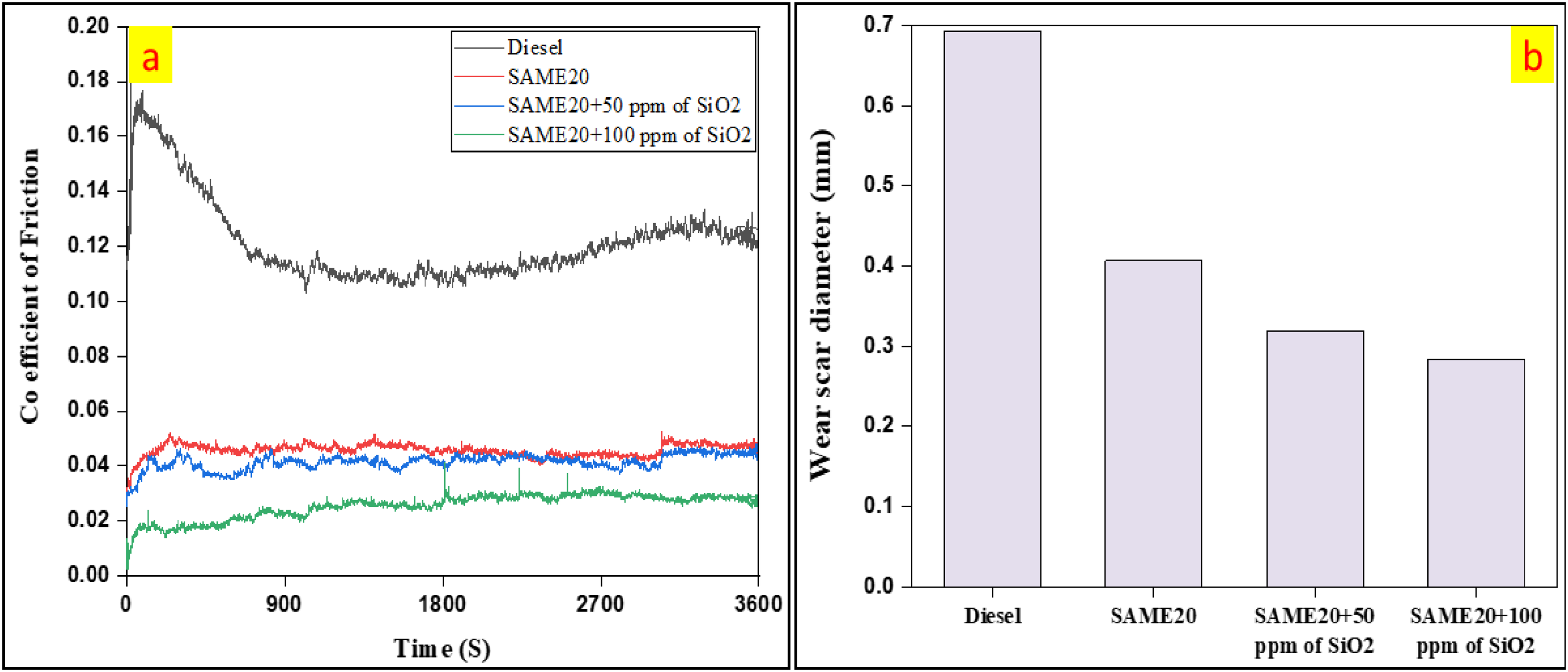
(a) The variation of COF and (b) WSD with different fuel blends.
Wear scar diameter
The wear scar diameter refers to the size of the wear mark that forms on the surface of a test ball after a specific duration of contact and load in a four-ball tribometer test. It indicates the wear resistance of lubricants and materials under frictional conditions. Generally, a larger wear scar diameter signifies more significant wear and material loss, while a smaller diameter suggests better wear resistance. The wear scar diameter differed among the various fuel blends, as illustrated in Figure 8(b). The wear scar diameter was 0.6924 mm for the diesel, 0.4066 mm for the SAME20, 0.3192 mm for the SAME20 + 50 ppm of SiO2, and 0.2828 mm for the SAME20 + 100 ppm of SiO2. SiO2 nanoparticles form a protective lubricating layer that minimizes wear between moving surfaces. Their hardness and spherical morphology help reduce abrasion and adhesive wear, resulting in a smaller wear scar diameter in tribological tests such as the four-ball tribometer. Higher concentrations of SiO2 further enhance surface protection, leading to reduced wear and improved tribological performance. Compared to diesel, the wear scar diameter was reduced by 41.27% for SAME20, 53.89% for SAME20 + 50 ppm of SiO2, and 59.15% for SAME20 + 100 ppm of SiO2. Other researchers have reported similar findings, and they observed a 37% reduction in wear volume loss with the incorporation of CuO nanoparticles and a 33% reduction with SiO₂ nanoparticles in coconut oil. 52
SEM analysis for worn surface balls
The scanning electron microscope analysis provides significant insights into the anti-wear characteristics of the ball surfaces. Figure 9(a) and (d) revealed that worn surfaces exhibited clear signs of metal-to-metal contact, characterized by numerous scratches and black spots. Also, deep grooves and polishing marks indicate severe abrasive wear, while the observation of cracks and pits suggests the involvement of fatigue wear mechanisms. Additionally, extensive material removal confirms the presence of severe adhesive wear. In contrast, Chemical interactions between nanoparticles and the mating surfaces formed thin lubricating films, enhancing lubrication significantly. These nanoparticles acted as rolling elements and improved lubrication efficiency. By filling voids, influenced by their shape and size, the nanoparticles contributed to a smoother surface finish, slowing wear progression and resulting in a polished surface. As the concentration of SiO2 nanoparticles increased, the depth and prominence of furrows in the wear scars diminished. SiO2 nanoparticles functioned as third-body particles, forming stable deposits on the contact surfaces, which reduced shear stress during sliding. These deposits were more effective than debris generated by the sliding materials, leading to a smoother wear path. This mechanism minimized wear caused by abrasive and adhesive interactions. SAME20 blends containing SiO2 nanoparticles exhibited smoother worn surfaces between asperities than other blends, with fewer visible grooves and a significant reduction in wear scar diameter, as shown in Figure 9(b) and (c). Overall results indicated that SAME20 + 100 ppm of SiO2 blend showed better anti-wear properties.

SEM image of worn surface balls (a) SAME20, (b) SAME + 50 ppm of SiO2 (c) SAME + 100 ppm of SiO2, (d) diesel.
Conclusion
This experimental study explores SEM analysis revealed crystalline structures and agglomeration, improving the surface morphology. TEM confirmed that the SiO2 nanoparticles had a nanoscale size ranging from 30 to 60 nm and were effectively dispersed. Incorporating SiO2 nanoparticles into biodiesel blends made from The SAME20 + 100 ppm of SiO2 nanofuel blend demonstrated significant emission reductions compared to conventional diesel, including 18.1% for CO, 11.12% for HC, and 16.2% for smoke. The findings indicated that the SAME20 + 100 ppm of SiO2 blend exhibited improved tribology performance, with a 76.89% decreased coefficient of friction and a 59.19% reduction in wear scar diameter compared to diesel. Scanning electron microscopy revealed that the SAME20 + 100 ppm of SiO2 blend revealed reduced surface wear and damage compared to diesel.
The findings of this study indicate that nanoparticles significantly improve engine performance and emission characteristics. However, further investigation is needed to assess the long-term effects of nanoparticles and conventional fuels on the engine and injection system. Future studies will focus on examining the behavior of diesel engines under varying compression ratios, injection pressures, injection timings, and exhaust gas recirculation conditions. Additionally, the authors suggest conducting long-term experiments to evaluate the impact of nanoparticles on engine wear.
Footnotes
Credit author statement
J. Arunprasad: Conceptualization, Methodology, Investigation, Writing – original draft. Michael D. Atkins: Review and editing.
Data availability
Data will be made available on request.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
