Abstract
This study evaluates the tribological performance of Fe-based composite coatings for aerospace and automotive applications, focusing on wear resistance at elevated temperatures. The coatings, comprising 15% WC-Co and Cr3C2 in an SS316L matrix, were deposited onto maraging steel via HVOF spraying. Microstructural analysis and adhesive strength testing confirmed strong particle bonding, with the coatings achieving a hardness of 711 ± 14 HV. Dry sliding wear behaviour was assessed using a ball-on-disc tribometer with alumina as the counterface under 10 N and 30 N loads at 25 °C, 300 °C, and 600 °C. Results indicated a decrease in the coefficient of friction and specific wear rate with increasing load and temperature. At 600 °C, wear rates were reduced by ∼97.56% (10 N) and ∼97.12% (30 N) than uncoated steel. Abrasive wear dominated at room temperature, while adhesive and oxidative wear mechanisms emerged at 600 °C, attributed to protective glaze layer formation.
Introduction
The increasing demand for superior substances with tailored properties has driven innovation in alloy design within various industries, including the aerospace, automotive, and energy production sectors.1,2 Iron (Fe) alloys have remained the subject of extensive research related to their remarkable properties, such as high strength, good toughness, cost-effectiveness, and abundant availability on Earth, making them crucial materials in aerospace, mining, metallurgy, manufacturing, agriculture, and construction. 3 The limited wear resistance and low hardness have confined their applicability in high-temperature sliding situations. 4 There is a need to explore the combined effect of WC and Cr3C2 reinforcement in SS316L coatings, which has not been extensively studied in the existing literature, and the synergistic benefits of both carbides, particularly at elevated temperatures, remain underexplored and to improve the capabilities of iron-based alloys.5–7 It is vital in ensuring optimal performance, reliability, and safety of various components and systems. 8 Components working in harsh environments such as high temperatures are bound to get degraded and worn, resulting in material loss and eventually leading to failure. 9 Applying protective surface coatings onto a metal substrate is a highly effective strategy for significantly enhancing tribological performance and extending service life, even in harsh conditions.10,11
Accordingly, thermal spray coating has been applied further to improve the mechanical characteristics of these iron-based alloys. These processes have many benefits, and it is an exceptionally versatile and adaptable method for applying feedstock powders onto various surfaces12,13 and protecting the substrate material. Numerous research studies have explored various thermal spray techniques to deposit composite materials. These methods include laser cladding and surface remelting,14,15 high-velocity oxyfuel (HVOF), 16 atmospheric plasma spray technique,17,18 high-velocity air fuel method, 19 arc spray, 20 detonation gun, 21 kinetic spray, 22 to create composite coatings with enhanced deposition efficiency and higher cooling rates. Among the numerous thermal spraying techniques, the HVOF coating technique is widely used23,24 to combat wear-related issues.25,26
Xiao et al. studied the laser cladding of iron (Fe) coatings reinforced with tungsten carbide (WC) particles, examining various mass fractions of WC. Their findings demonstrated that incorporating WC significantly enhanced the microhardness and wear resistance of the coatings. 27 In their study, Huebner et al. investigated the development of coatings with 20%WC and 30%WC. They extensively analyzed the phase composition and microstructure of these coatings. The study showed that as the volume percentage of tungsten carbide within the coatings increased, there was a corresponding improvement in wear resistance, hardness, and compressive strength. On the other hand, the results indicated that higher tungsten carbide content negatively impacted the composite's yield strength and toughness.28,29 Tungsten carbide (WC) is considered an exceptional reinforcement phase due to its high melting point and excellent wettability with Fe-based alloys, making it highly effective in enhancing the material's properties. 30 The 316L stainless steel, is a highly durable alloy of iron, nickel, chromium, and additional elements like molybdenum. Its exceptional resistance to corrosion makes it a popular choice for coating applications in marine engineering.31,32 Recent studies have shown that applying HVOF-sprayed 316L stainless steel coatings on magnesium alloys improves wear and corrosion resistance. Additionally, the adhesion between the coatings and the alloys is significantly strengthened.33,34
Carbide-based composite coatings offer improved durability against wear and possess higher hardness and toughness. 35 Regarding mechanical characteristics and production procedures, the combined effect of tungsten carbide (WC) and cobalt (Co) is highly valued. This is because they offer a great combination of wettability, hardness, and toughness. 36 Cobalt is often combined with tungsten carbide (WC) owing to its exceptional bonding characteristics and advantageous microstructural properties. 37 Carbide-based thermal spray coatings, particularly those created using the HVOF process, offer exceptional resistance to degradation in extreme conditions due to their remarkable hardness and chemical stability. The hardness of the carbide particles plays a crucial role in determining the wear resistance of the carbide coating.19,38–42 Based on the available literature, there is a clear gap in research regarding the effects of integrating chromium carbide (Cr3C2) and tungsten carbide (WC) into SS316L stainless steel coatings. The existing studies have primarily focused on different material systems or reinforcement materials, and there is a lack of comprehensive investigation into the impact of these carbides on the microstructural properties, wear resistance, and overall performance of SS316L coatings. Furthermore, previous studies on assessing the tribological behaviour of these carbide-reinforced SS316L coatings at high temperatures are limited. Therefore, there is a critical need for research to comprehensively investigate how carbide additions influence the high-temperature wear properties and durability of SS316L coatings, particularly under demanding operational conditions.
The increasing demand for superior materials with tailored properties has driven significant advancements in the development of alloy systems for various industries, including aerospace, automotive, and energy production. Iron-based alloys have remained a focus of research due to their high strength, toughness, cost-effectiveness, and abundant availability. Nevertheless, their limited wear resistance and mechanical stability in high-temperature environments have restricted their application in demanding conditions. To overcome these limitations, various reinforcement strategies, including the incorporation of carbides such as tungsten carbide (WC) and chromium carbide (Cr3C2), have been explored to enhance wear resistance and hardness.
Recent studies have primarily investigated coatings with either WC or Cr3C2 reinforcement individually. For example, WC-based coatings offer high hardness and wear resistance but tend to be brittle and susceptible to oxidation at elevated temperatures. On the other hand, Cr3C2 reinforced coatings exhibit better oxidation resistance and adhesive properties but may not achieve the same hardness as WC based coatings. Despite these advancements, a critical research gap remains regarding the synergistic effect of combining both WC and Cr3C2 within Fe-based coatings, particularly under elevated temperatures. Exploring this synergy offers the potential to develop coatings that balance hardness, wear resistance, and oxidative stability.
The present study aims to address this gap by investigating the microstructural and tribological performance of SS316L coatings reinforced with a combination of WC and Cr3C2. Through this research, we seek to demonstrate how the properties of these carbides enhance the coatings performance, especially in high-temperature environments, making them suitable for aerospace and automotive applications.
This paper examines the influence of carbide reinforcement on SS316L precursor powder, focusing on its impact on tribological properties. The study hypothesises that the developing oxide phases from the carbides could serve as a lubricating layer, thereby reducing the friction coefficient and wear, particularly at high temperatures. To validate this hypothesis, wear assessments using a tribometer (ball-on-disk), a standard method for evaluating friction and wear. The wear behaviour of the coated samples was analysed across a range of temperatures, specifically 25 °C, 300 °C, and 600 °C, and under varying loads of 10 N and 30 N. Thus, the as-sprayed samples underwent a series of comprehensive tests to assess their properties. The analysis encompassed several techniques, including X-ray diffraction (XRD) for phase identification, energy dispersive spectroscopy (EDS) for elemental mapping, Vickers microhardness testing for hardness measurement, and 3D profilometer analysis to assess wear depth profiles and rates.
Materials and methods
Coating details
The feedstock powders used for coating applications included SS316L, WC-Co, and Cr3C2, acquired from suppliers Oerlikon Metco in India and Indo Mim Pvt. Ltd in Doddaballapur, India. Image J software determined these powders’ particle size distribution and average values were calculated for precision. The detailed chemical composition of each powder is provided in Table 1. To create the coating mixture, SS316L powders were blended with WC-Co and Cr3C2 powders in specific ratios using a mechanical mixer for 4 h to ensure uniform distribution. The chosen substrate for this study was Maraging Steel 250, sourced from Chamunda Industries in Mumbai, India. This surface preparation was cleaned with acetone and dried using high-pressure air to enhance the adhesion strength between the coated layer and the substrate material. Before coating, the substrate surface was prepared through alumina grit blasting to enhance adhesion.
Elemental composition of the substrate and feedstock in weight %.
Elemental composition of the substrate and feedstock in weight %.

Aum Surface Technologies in Bengaluru, India, employed the HVOF thermal spray method to deposit a top layer of precursor powders onto the maraging steel. The specific spray variables employed in the HVOF process included hydrogen fuel gas flow rates maintained at 550 Standard Liters Per Minute (SLPM) to ensure a robust and consistent energy source. Additionally, the nitrogen carrier gas, serving as a dual role of carrier gas and coolant during the HVOF spraying technique, was set to flow at 30 SLPM, while airflow was managed at 200 SLPM. The powders fed into the process at a rate of 20 grams per minute with the gases. The spray distance was positioned at a distance of 140–200 mm from the target surface. Furthermore, the oxygen flow rate of 200 SLPM was maintained to fuel the combustion process. The feedstock powders were introduced into a spray gun (specifically, the DJH 2600 model from Sulzer Metco, Inc., Westbury, NY), where they melted owing to combustion. The temperature control was thoroughly maintained throughout the process. These molten particles were then forced toward the maraging steel surface by a high-velocity gas stream, ensuring effective deposition.
The top surface analysis and microstructure of the deposited coatings are examined in the cross-section. The specimen is cut through a wire EDM machine, and the sectioned part is mirror polished using 400 to 2500-grit emery paper along with diamond paste and velvet cloth. The coating thickness laterally the cross-section is measured using a backscattered electron image. Top surface morphologies and cross-sectional views are analyzed using a Fe-SEM (7610 FPLUS, Jeol, Japan) and an optical microscope (Zeiss Axiovert 200 MAT, Germany). The coating layer comprises of a fully melted and partially melted region, and the compositions present were determined by a point, line, and area analysis using EDS.
After the wear test the surface roughness and wear profile track of coated samples were evaluated by means of a 3D non-contacting optical profilometer instrument (ST400, NANOVEA, USA). Further, the study of furrows refers to the grooves or scratches formed on surfaces due to wear, friction, or material displacement during contact and motion. These furrows, often caused by abrasive particles or surface asperities, are critical indicators of surface roughness and degradation. 43 The mean density of furrows helps quantify the extent of wear and is used to assess the frictional properties of a material. Higher furrow densities typically lead to increased friction and wear. 44 These furrows are essential for the longevity and functionality of mechanical components in motion. Before the measurements, the samples were cleaned with isopropyl alcohol, free from foreign objects, then dipped in an ultrasonic bath, and then the readings were taken.
Phase analysis
The feedstock powders and coating specimen phases have been identified using an X-ray diffractometer to analyze the different phases in the coating layer, equipped (Empyrean 3rd Gen, Malvern PANalytical, Netherlands) through; anode material is Cu Kα radiation and diffractometer system applied is EMPYREAN. The data points are taken at 2°/min scanning speed and step size of 0.026° in the 10 to 90° range. Xpert high score software was used to analyse, and ICDD data was extracted.
Microhardness measurements
The hardness of the as-sprayed coatings was evaluated using a Vickers microhardness tester (Model: MVH-S-AUTO, produced by Omni Tech, Bengaluru, Karnataka, India). This assessment was conducted in accordance with the ASTM E384 standard. 45 Readings were obtained from mirror-finished cross-section samples, starting with the substrate and progressing to the interface, then with coatings. A force of 300 grams were applied to create an indentation and held for 10 s. The indentations were spaced 50 micrometers apart. The coating density was determined by employing a pycnometer and the water immersion technique in accord with the ASTM C-135-96 standards. A mechanical dissection method was used to remove the coating from the substrate, resulting in a peeled coating weighing 1–2 grams.
Scratch test
Scratch tests were carried out on cross-sections of coatings to assess the adhesion/cohesion of thermal sprayed coatings in accordance with ISO 27307 standards. The equipment utilised here in the study is a TR-101 scratch tester from DUCOM, India. Before the test, the samples surface was polished to a mirror finish using grit emery paper and diamond paste, and the average surface roughness measured was Ra = 0.02 microns. The test employed a Rockwell C-type conical diamond indenter with a rounded tip of 200 µ radii. It made four unidirectional scratch passes with a constant normal load of 10 N, 25 N, 50 N, and 75 N. The scratch length is 4 mm, and the scratch velocity is 0.1 mm/sec. After the test, an optical microscope was used to inspect and analyze the scratch grooves to determine the type of failure in the test, three scratches under constant load were performed at each load to ensure consistent and reliable results. The projected cone surface area (Acn) was calculated using optical microscopy to determine its length (a) and width (b), and it is given by Acn = 1/2(a × b).3,46
Tribological test
A ball-on-disc tribometer (THT 1000 and TRB3, Anton Paar, Austria) was utilised to conduct tribological testing on samples measuring 25 mm × 25 mm × 8 mm, following ASTM standard G99-05. The testing equipment consisted of a ball holder assembly and an upper arm supporting a high-quality alumina ball (6072 HRC hardness, 6 mm diameter) that made contact 47 with the as-sprayed sample fixed inside the circular housing chamber with a wedge. For high-temperature wear testing, the chamber was preheated to the required temperature for approximately 30 min before commencing the test. Temperature was continuously monitored using a sensor. Once the compartment reached the desired temperature, the testing process began, and the temperature was consistently maintained throughout the tests. The experiments were carried out at different temperatures (ambient 25 °C, 300 °C, and 600 °C) and loads (10 N and 30 N); the speed was kept constant at 200 rpm and a sliding distance 1000 m. Three tests were conducted on each as-sprayed sample, and average values were obtained to assess the results’ consistency and precision.
Raman spectrometer
A Raman spectrometer (Renishaw Centrus 2R4P63, with Wire 5.4 software, UK) was employed in a continuous scan mode to examine the oxide layer composition on both worn and unworn surfaces. Raman spectra were consistently recorded with a 10-s exposure time and two accumulations, covering a Raman shift range from 100 to 1600 cm−1.
Results and discussion
Morphology of the precursor powder
The images in Figure 1 illustrate the surface characteristics of the feedstock powders. In Figure 1(a), the surface of SS316L appears nearly spherical, while the WC-Co elements, as shown in Figure 1(b), display mostly elliptical shapes with some spherical particles. On the other hand, Cr3C2 exhibits irregular and spherical shapes, as depicted in Figure 1(c). Furthermore, the EDS spectrum in Figure 2 reveals the primary elements Fe, Cr, Ni, WC, Mo, and C and small traces of cobalt present in the feedstock powders. Both WC-Co and Cr3C2 powders exhibit a rough surface topography.

SEM micrograph showing powder morphology (a) SS316L, (b) WC-Co, (C) Cr3C2.
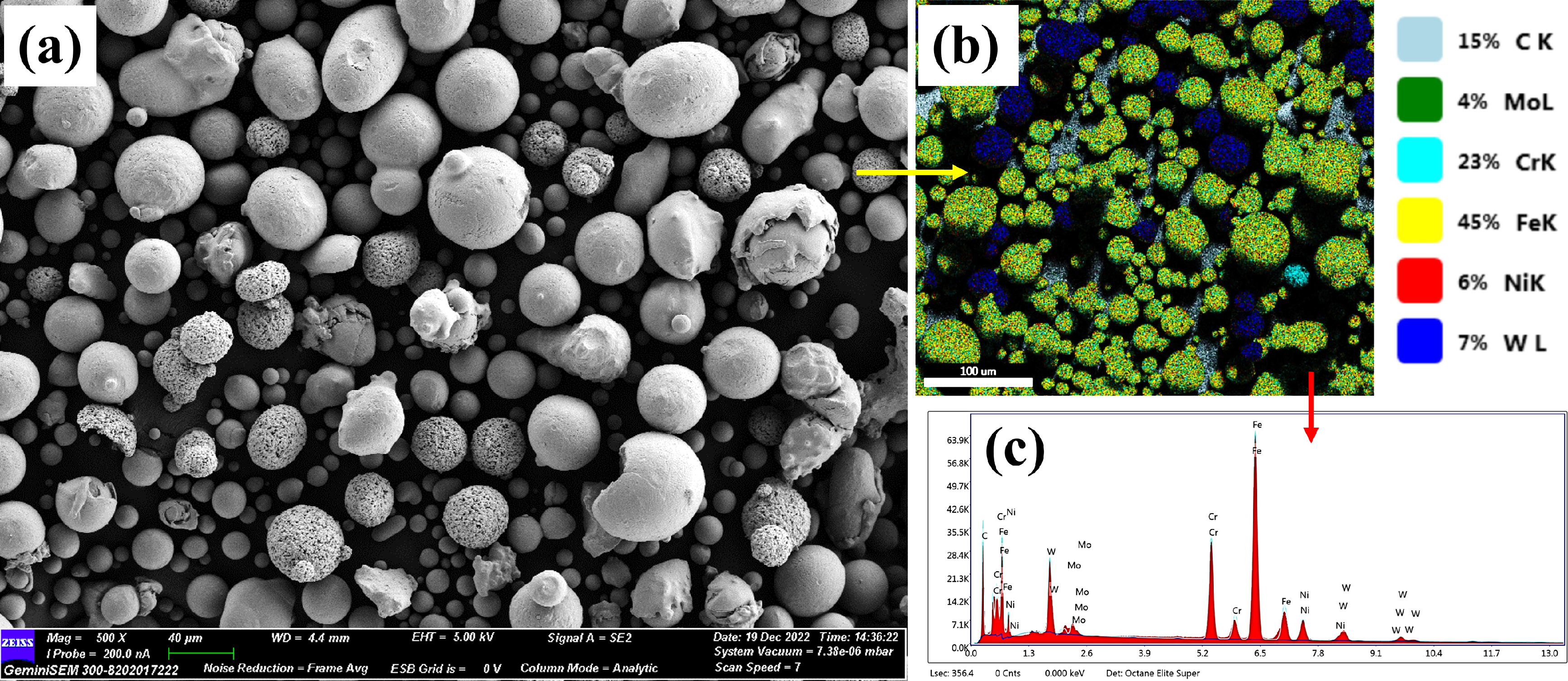
Elemental mapping of the feedstock powder.
The mean particle diameters of SS316L, WC-Co, and Cr3C2 powders are determined to be 32.34 µm, 23.35 µm, and 22.51 µm, respectively. The particle size and morphology of the feedstock powder significantly affect deposition characteristics during the heating process, influencing the cooling behaviour of each splat. Particle size distribution plays a critical role in coating quality. Smaller particles promote denser coatings with fewer pores, while larger particles may lead to increased porosity and weaker adhesion. The particle size in coating materials significantly affects the surface roughness of the coating. Larger particles in the coating can result in a rougher texture, while smaller particles help achieve a smoother finish. Our current study used well-defined particle sizes to ensure uniform deposition, but we recognise that variations in size distribution can affect hardness, wear resistance, and adhesion strength.48,49 The feedstock particle size, morphology, and process parameters substantially influence the performance and overall quality of the thermally sprayed coatings.2,50
The coatings produced from Fe-based powder and carbides using the HVOF as-sprayed technique displayed in Figure 3(a) have a compact microstructure, with Fe as the primary component and additional elements with WC-Co and Cr3C2. The surface features of the as-sprayed coatings are illustrated in Figure 3(c). The dark grey area labelled as point 1 contained Fe, signifying enhanced metallurgical bonding among the coating and the substrate. 51 The light grey area indicated in point 2 contains WC reinforcement, which significantly improves its resistance to high temperatures and prevents noticeable deterioration. 52 The majority of WC-Co powder particles experienced substantial fracturing and deformation as they collided during flight, causing them to depart significantly from their original spherical shape. Consequently, the resulting coating displayed an uneven and irregular form. 50 The area indicated at point 3 in light grey is characterised by high concentrations of chromium and carbon. The dark spot corresponds to the presence of voids. These voids are a result of splat shrinkage during the solidification process, and they can be found in multiple locations across the coating. 53 The compact arrangement is formed owing to the partial melting of carbides, leading to enhanced hardness and dense structure.

Micrograph image of coated surface (a) coated surface, (b) surface morphology, (c) cross-section of the coatings.
Results depicted by EDS at a particular location in Figure 3 clearly reveal the existence of iron (Fe), chromium (Cr), nickel (Ni), tungsten carbide (WC), cobalt (Co), and carbon (C). The mean coating thickness accessed across the cross-section was 211 ± 15 µm, depicted in Figure 3(b). A uniform distribution is expected to improve the adhesion strength of the coatings by facilitating the integration of WC and Cr into the maraging steel substrate and iron into the coating. Adding WC and Cr to the coating enhances its wear resistance and hardness, resulting in a durable protective layer over the maraging steel substrate. This coating is applied using the HVOF spraying process, which ensures a strong bond among the coating and the substrate while preserving the mechanical properties of the base materials. The combination of these components in the upper layer demonstrates the uniform distribution of the coating, leading to improved surface properties and overall performance of the alloy.
In Figure 4, the XRD pattern indexing for both the feedstock powders and coatings of the Fe-based composition is depicted. The SS316L-15%WC-Co-15% Cr3C2 powder shows prominent peaks of FeNi and Cr-Fe-Ni-C, with minor peaks of WC and Co2Mo3. After the coating deposition, the significant peaks consist of FeNi and Cr-Ni-Fe-C, while WC and Co2Mo3 are observed as minor peaks.
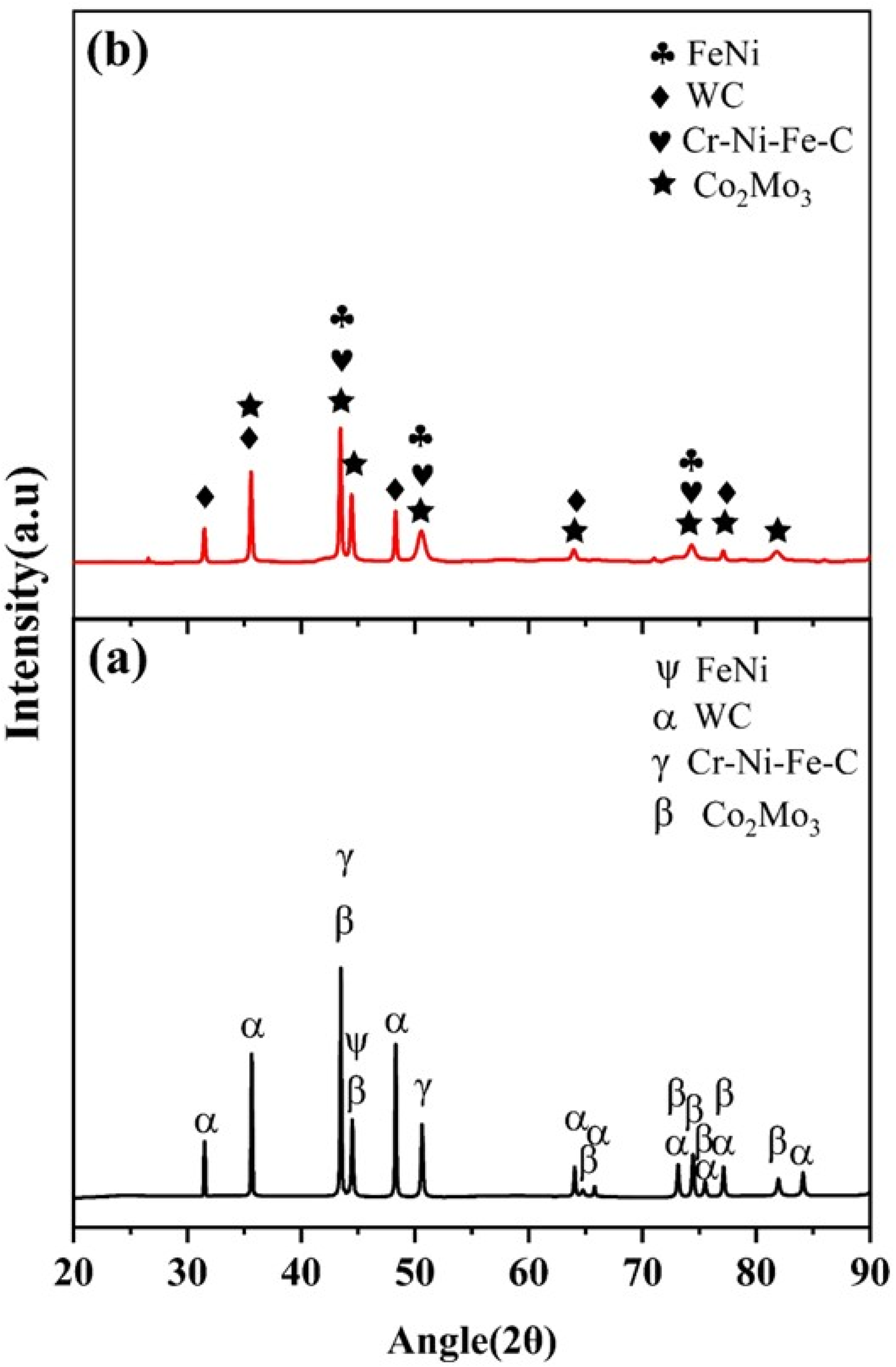
XRD graphs of (a) feedstock powders and (b) coating at 25 °C.
The XRD data of the SS316L-15%WC-Co-15%Cr3C2 coated sample reveal specific peaks and their corresponding planes along with the ICDD data. The FeNi peaks are indexed to (111), (200), (220), (311), and (222), with ICDD card number 00-023-0297. Cr-Ni-Fe-C peaks are indexed to (111), (200), (220), (311), and (222), with ICDD card number 00-031-0619. WC peaks are indexed to (001), (100), (101), (110), (111), (200), (102), and (201) planes, with ICDD card number 01-073-0471. Co2Mo3 peaks are indexed to (311), (002), (410), (330), (202), (212), (411), (331), (532), and (413) with ICDD card number 03-065-5102. Furthermore, austenite phase peaks were revealed in Stainless steel powder and the deposited coatings.54,55
Mechanical hardness
The hardness test revealed that the substrate possesses a micro-hardness of 349 ± 10 HV0.3, while the SS316L-15%WC-Co-15%Cr3C2 coating exhibits a significantly higher hardness of 711 ± 14 HV0.3, as depicted in Figure 5. The as-deposited coatings demonstrate characteristics such as rapid cooling rates, the formation of partial oxides during deposition, and the presence of metallic phases, all of which contribute to the observed elevated hardness at room temperature. WC and Cr3C2 powders possess significantly higher hardness than SS316L. The incorporation of both Cr3C2 and WC improves hardness by introducing hard carbide phases into the SS316L matrix. Cr3C2 also enhances wear resistance by forming stable, high-temperature oxides, which protect the coating from wear. Additionally, Cr3C2 mitigates the oxidation of WC, preserving the coatings mechanical properties under extreme conditions. 56 The literature suggests that combining WC and Cr3C2 into stainless steel enhances both hardness and wear resistance. WC-based coatings, in particular, stand out from cemented carbides by providing a strong combination of hard WC particles within a metallic matrix. These metal matrix composites are highly valued for their superior performance in challenging environments, demonstrating outstanding resistance to elevated temperatures and varying loading conditions while maintaining excellent mechanical and tribological properties.57–59
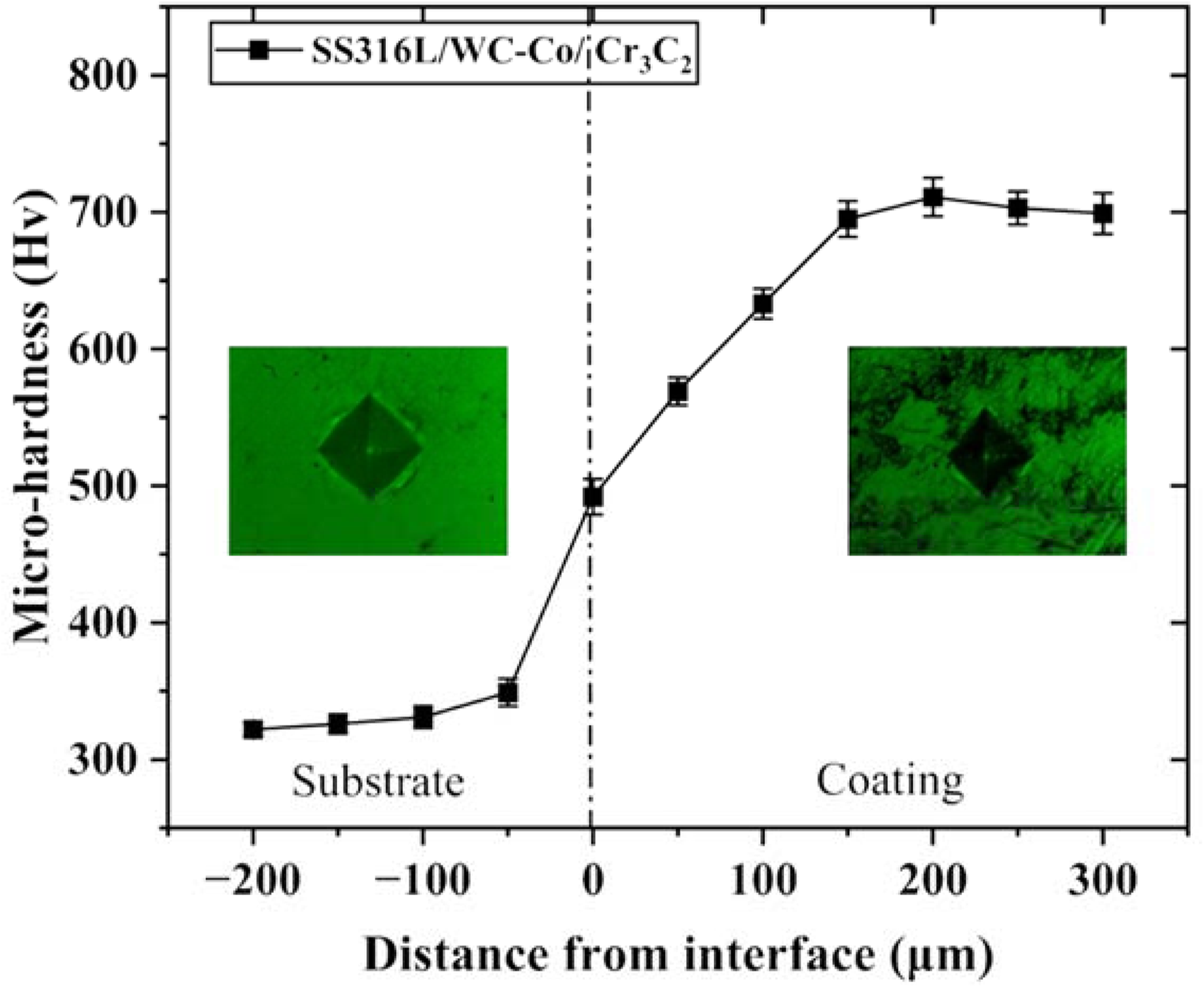
Hardness graph from the substrate to coating.
The SS316L-15%WC-Co-15%Cr3C2 coatings exhibit a porosity of 1.95 ± 0.15, a surface roughness (Ra) of 7.661 µm, and a density of 8.71 g/cc.
The scratch behaviour depicted in Figure 6 is observed in the cross-section of the coatings under various constant normal loads. The coating SS316L-15%WC-Co-15%Cr3C2 sample showed no signs of failure or cracks at loads of 10 N, 25 N, and 50 N (Figure 6(a) to (c)). At a load of 75 N, as shown in Figure 6(d), cracks began to form within the coating, oriented perpendicular to the direction of the scratch. This suggests cohesive failure within the material, where the internal bonds of the coating layer itself start to break down. Additionally, signs of adhesive failure were also observed, indicating a loss of bonding between the coating and the underlying surface depicted in Figure 6(e), (f) and (g). These combined failures highlight the materials susceptibility to both internal and interfacial weaknesses under increased loading conditions. The enhanced adhesion is due to the presence of solid-state WC particles, which are securely embedded and bonded by the liquid matrix phase of Co particles during the spraying process. The Co matrix acts as a binder, ensuring stronger cohesion between the WC particles and providing improved bonding with the substrate, resulting in superior adhesive performance. 60
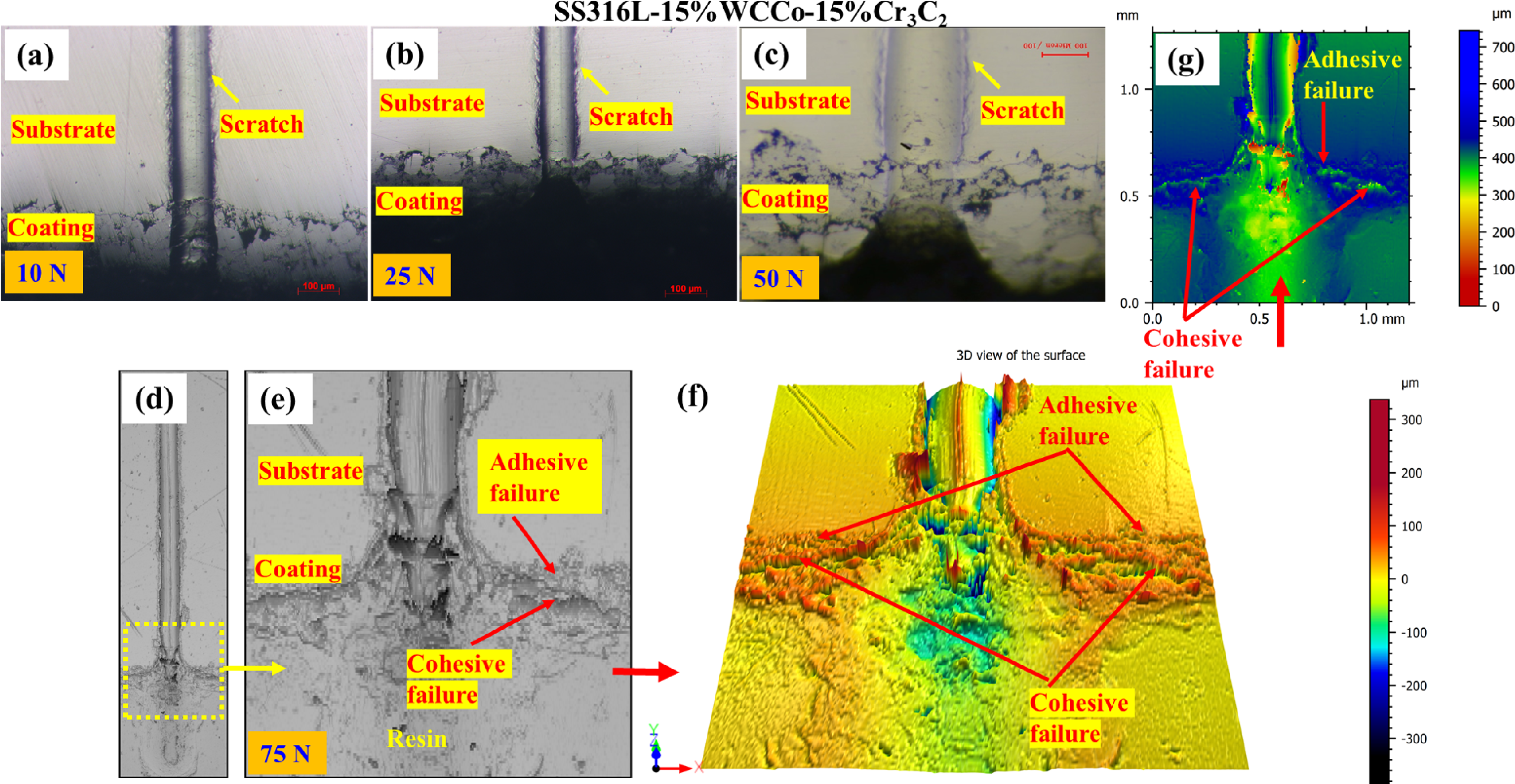
Cross-section image of scratch test viewed through optical microscopy and 3D optical profilometer of SS316L-15%WC-Co-15%Cr3C2 at varying loads (a) 10 N, (b) 25 N, (c) 50 N, and (d) and (e) 75 N, along with (f) and (g) respectively.
Wear and frictional behaviour of the deposited samples
A dry sliding wear test was conducted via a ball-on-disk tribometer to examine the substrate and coated samples friction and wear characteristics. The test was performed under varying loads of 10 N and 30 N across different temperatures of 25 °C, 300 °C, and 600 °C, with a rotational speed of 200 rpm and a sliding distance of 1000 metres. Figure 7(a) and (b) show the wear rate of SS316L-15%WC-Co-15%Cr3C2 coatings under these test conditions, demonstrating the effect of temperature and applied load on wear performance.
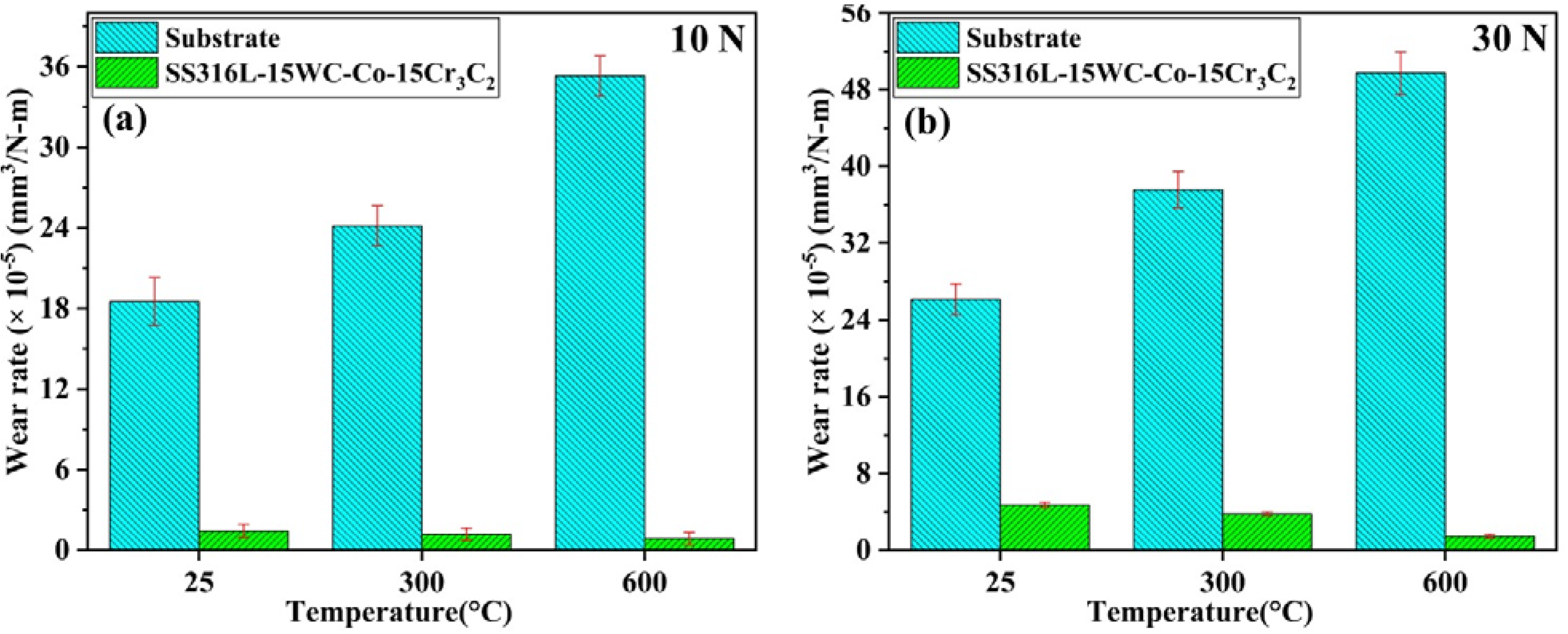
Wear rate v/s temperature graph at different temperatures & loads.
The wear rate of SS316L-15%WC-Co-15%Cr3C2 coatings shows a noticeable decrease as the temperature increases at room temperature (25 °C) to 600 °C across both applied loads. Specifically, the wear rates at 25 °C, 300 °C, and 600 °C are recorded as 1.44 × 10−5 mm3/N-m, 1.19 × 10−5 mm3/N-m, and 0.86 × 10−5 mm3/N-m for a 10 N load, and 4.71 × 10−5 mm3/N-m, 3.79 × 10−5 mm3/N-m, and 1.43 × 10−5 mm3/N-m for a 30 N load, respectively. This substantial decrease in wear rates is primarily ascribed to developing a dense oxide layer on the top surface of the coatings, which acts as a solid lubricant, thereby minimising direct contact among the coated surfaces and the counter body.
Adding chromium carbide (Cr3C2) to Fe-based composite coatings enhanced microstructural properties and tribological performance compared to only tungsten carbide (WC). Cr3C2 enhances phase stability at elevated temperatures compared to WC. This stability can lead to better performance in high-temperature applications. Cr3C2 can help reduce porosity in the coatings owing to its denser structure, resulting in improved density. Lower porosity leads to enhanced wear resistance and corrosion resistance. The interaction between Cr3C2 and WC can create a composite structure that combines the desirable properties of both carbides. The inclusion of Cr3C2 can refine the microstructure of the coating, leading to a finer grain size. A finer microstructure generally contributes to improved mechanical properties, including hardness and toughness. Regarding tribological performance, Cr3C2 facilitates the formation of Cr2O3 oxides at high temperatures, which act as solid lubricants, reducing friction and minimising wear during sliding. Coatings with Cr3C2 improved performance in abrasive wear environments, as they are less prone to delamination than pure WC coatings.2,61
The wear rate of the SS316L-15%WC-Co-15%Cr3C2 coating is significantly less than that of the substrate at various temperatures and loads. At room temperature, the wear rate is reduced by 92.22%, which decreases further to 95.07% at 300 °C and 97.56% at 600 °C under a 10 N load. When the load is increased to 30 N, the wear rate remains significantly lower across all tested temperatures, with reductions of 81.98% at room temperature, 89.91% at 300 °C, and 97.12% at 600 °C. The superior wear resistance of the SS316L-15%WC-Co-15%Cr3C2 coating is mainly due to developing a protective glaze layer on the coating surface, enhancing its durability at elevated temperatures. A recent study by Islas Encalada et al. investigated Cr3C2-reinforced cobalt-based coatings applied via HVOF, finding that the incorporation of Cr3C2 facilitated the formation of protective oxide layers at high temperatures, further enhancing wear resistance. 62 Earlier work by Matikainen et al. on Cr3C2-based coatings showed that Cr3C2-25NiCr performed best at 700 °C due to the development of a protective tribo-layer, while Cr3C2-37WC-18NiCoCr experienced reduced wear resistance due to WC particle oxidation. 63 Similarly, Bolelli et al. compared Cr3C2–NiCr coatings applied by HVOF and HVAF, noting that HVOF coatings exhibited more carbide dissolution, impacting wear performance and thermal expansion compatibility with steel substrates. 64 Current research also demonstrates improved wear resistance at both room and elevated temperatures, with the enhancement at higher temperatures attributed to the incorporation of hard particle reinforcements and the formation of protective oxide layers, which together enhance the coatings durability.
At 25 °C to 300 °C, the worn surface morphology of the coating revealed crack propagation, pull-out, delamination, and fractures, indicating abrasive surface wear as the primary mechanism. At 25 °C to 300 °C, no prominent oxide layer was formed. As a result, there is direct contact between surfaces and counter material. When the temperature rises from 300 °C to 600 °C, a smooth surface with minimal pull-out and no crack formation is observed. This indicates that wear rate control is achieved by developing an oxide film on the worn surface. The oxide layer can reduce the contact between the severities of tribo-pairs, consequently reducing the wear rate and friction coefficient of coatings. This implies that the primary wear mechanisms of coatings at 600 °C are oxidative and adhesive wear.
Figure 8(a) and (b) shows the difference in friction coefficients between SS316L-15%WC-Co-15%Cr3C2 coatings and substrates at different temperatures and normal loads. As the temperature rises from 25 °C to 600 °C, the average friction coefficient of the SS316L-15%WC-Co-15%Cr3C2 coating decreases.
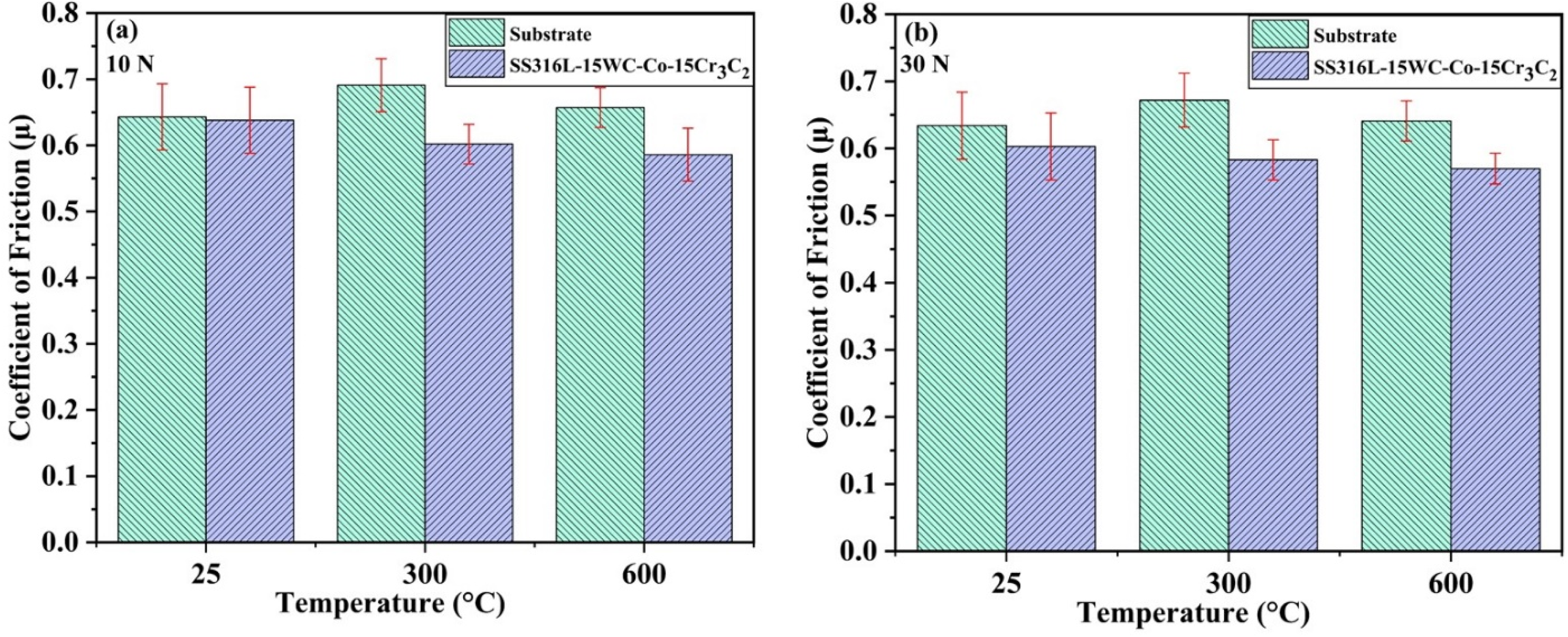
COF v/s temperature graph at different temperatures & loads.
Under a 10 N load, the average friction coefficient for the SS316L-15%WC-Co-15%Cr3C2 coating is 0.63 at 25 °C, 0.60 at 300 °C, and 0.58 at 600 °C. When the load is increased to 30 N, the average friction coefficient at these temperatures drops slightly to 0.60, 0.58, and 0.57, respectively. This trend indicates that frictional force does not scale linearly with applied load, with the friction coefficient reducing as the load increases.
Compared to the base material, the SS316L-15%WC-Co-15%Cr3C2 coating showed a reduction in the coefficient of friction of 0.7% and 12.87% at 25 °C and 300 °C, respectively, under a 10 N load. Under a 30 N load, the reduction was 4.88% at 25 °C and 13.24% at 300 °C. At 600 °C, the coating exhibited a 10.87% lower coefficient of friction at 10 N and an 11.07% lower coefficient of friction at 30 N associated with the substrate, highlighting the superior frictional properties of the coating at high temperatures.
The decrease in the coefficient of friction (CoF) observed in HVOF sprayed iron-based composite coatings at elevated temperatures can be attributed to various interrelated mechanisms. One of the most significant contributors is the formation of protective oxide layers, notably Fe2O3, WO3, NiWO4, and NiCr2O4, which developed on the surface of the coatings. These oxide layers function as effective solid lubricants, significantly reducing the extent of direct contact between surfaces and thereby minimising friction. In addition to forming these oxides, the high temperatures promote the development of glaze or tribo-layers. These specialised surfaces enhance lubrication properties and protect against material removal during operation. As the temperature rises, the predominant wear mechanism undergoes a transition. In contrast, three-body abrasive wear is the primary concern at room temperature. During elevated temperatures, there is a shift in the wear dynamics towards oxidative wear and adhesive wear. This change contributes to the smoothening of sliding surfaces and plays a crucial role in further lowering the frictional forces experienced during sliding contact.65,66
The average friction coefficient decreases noticeably with increasing applied load. This decrease in friction implies that the coatings tribological performance improves with increased loads. 26 Aristizabal et al. found that the average coefficient of friction for WC-Co and WC-Ni-Co-Cr coatings decreased as oxide layers developed at high temperatures. These oxide layers were found to have a lubricating effect. 67 The rise in temperature resulting from the development of the protective layer led to a substantial decrease in the friction coefficient. 68
Subsequently the coated samples surpassed the maraging steel substrate in terms of wear resistance under both loading conditions. Across a range of loads and temperatures, the coatings average COF was consistently lower than the substrate. This suggests that the coatings not only provided superior wear resistance, but also contributed to more stable and frictionless behaviour under different operating conditions.
The wear test involves subjecting the coatings to high temperatures (600 °C) and analysing the worn surface using XRD phase analysis, as shown in Figure 9. The change in phases from ambient temperature to high temperature could have an important impact on the wear resistance of the coatings. The protective oxide layer formed in the SS316L-15%WC-Co-15%Cr3C2 coating consists of Fe2O3, Ni W O4, WO3, and Ni Cr2O4, which were identified with ICDD numbers 01-084-0308, 01-072-0480, 01-087-2389, and 01-076-2184 respectively. These oxide phases lead to the formation of glaze layers, which act as a protective barrier, resulting in improved wear resistance. These findings provide compelling evidence of the coatings performance under high temperatures and their ability to reduce friction. Through the coating deposition, unheated tungsten carbide (WC) and chromium carbide (Cr3C2) particles adhere to the metal matrix, playing a vital role in enhancing the coatings wear resistance. 69
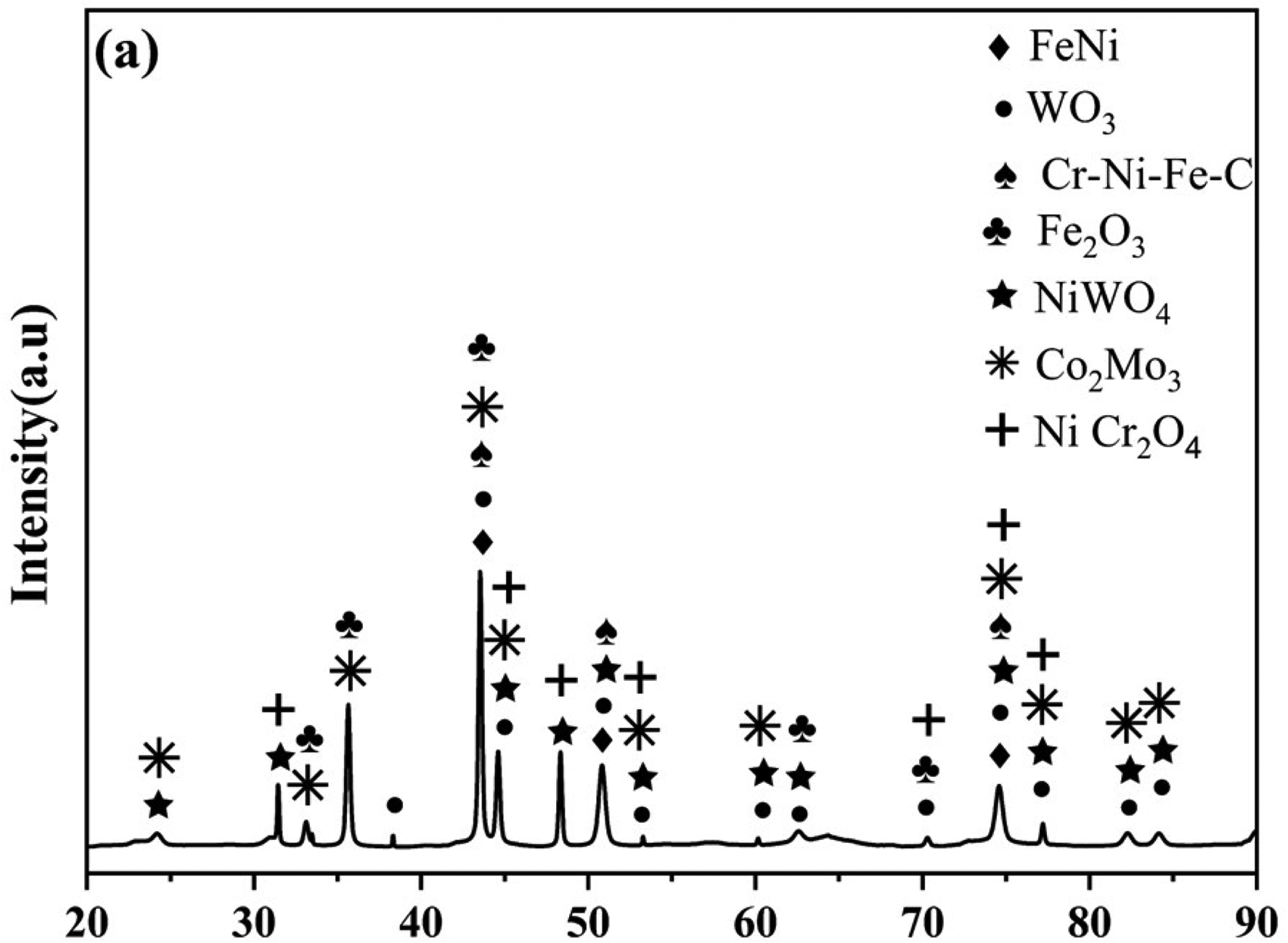
XRD graph at elevated temperature (600 °C).
At room temperature, the surfaces of materials engage in direct interaction, leading to abrasive wear characterised by the creation of furrows and the generation of debris. At 25 °C, the worn track surface exhibits cracks, flaking pits, and delamination. This indicates the predominance of abrasive wear mechanisms.
As the temperature rises, these processes undergo a notable transformation; oxide layers such as iron oxide (Fe2O3), tungsten trioxide (WO3), nickel tungsten oxide (NiWO4), and nickel chromium oxide (NiCr2O4) begin to develop on the surface of the material. These oxide layers serve a crucial role, evolving into a smooth, lubricating glaze that markedly reduces material removal. This shift enables the onset of adhesive wear mechanisms, where localised bonding occurs between the opposing surfaces, allowing for enhanced performance under varying conditions. Notably, at a temperature of 600 °C, these protective oxide layers become increasingly stable and form continuous shields over the surface. This stability minimises friction and significantly boosts wear resistance, demonstrating the critical interplay between temperature and wear mechanisms. The transition from abrasive wear to adhesive and oxidative wear is influenced significantly by the temperature-induced formation of oxide layers.
At a temperature of 600 °C, the surface material and wear debris undergo oxidation, leading to the formation of various oxides. These oxides are responsible for creating a lubricating oxide layer on worn surfaces, which acts as a lubricant. The oxides accumulate more rapidly than they are removed, forming a dense and continuous oxide lubrication layer on the worn tracks due to the softening at high temperatures and external loads. This intact oxide lubricating layer effectively separates the surfaces in contact during sliding, thereby declining the wear rate and the friction coefficient. The prime wear mechanisms of the coatings at 600 °C are attributed to oxidation wear. 70
Figure 10 represents the surface morphology of the SS316L-15%WC-Co-15%Cr3C2 coatings. When subjected to a 10 N load at ambient temperature, as shown in Figure 10(a) and (b), the primary wear mechanisms are abrasion (grooving), surface fatigue (resulting in furrows, pits, and delamination), and debris formation.

Fe-SEM images of worn scars at ambient (25 °C) and elevated temperature (600 °C).
Upon increasing the temperature from 25 °C to 600 °C and the loads from 10 N to 30 N, depicted in Figure 10 (c) and (d). The wear test revealed a noticeable reduction of wear rates with respect to temperature owing to the sliding action, which appeared to be produced by the pulling out the maximum splats, indicating severe wear. With the rise in temperature, protective oxide layers formed on the worn-coated surfaces, effectively preventing further coating loss at higher loads and leading to a reduction in wear over time.
The wear mechanism of the coating involves the gradual buildup of glaze layers, a process known as oxidative wear, followed by the subsequent formation of cracks in the oxidised surfaces. With increasing temperature, excessive heat leads to the development of an oxide layer. For the SS316L-15%WC-Co-15%Cr3C2 coating, the resultant oxide layers include Fe2O3, Ni W O4, WO3, and Ni Cr2O4. The line analysis using Energy Dispersive X-ray Spectroscopy (EDAX) on wear scar of the SS316L-15%WC-Co-15%Cr3C2 coatings at room temperature revealed presence of iron (Fe), nickel (Ni), tungsten (W), chromium (Cr) and carbon (C), as well as traces of molybdenum (Mo), cobalt (Co), and manganese (Mn), are depicted in Figure 11.
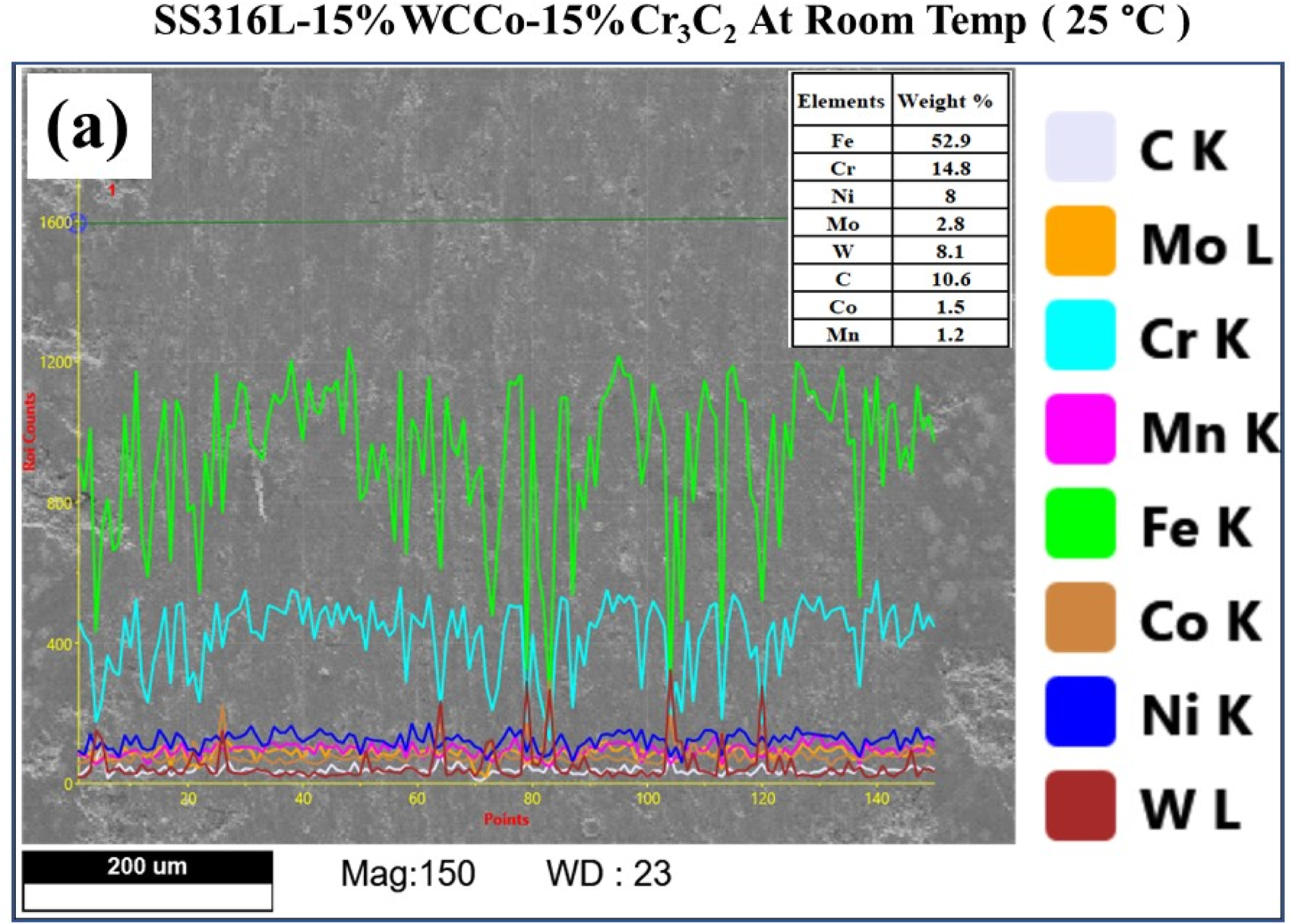
Line scanning analysis of worn scar image at ambient temperature (25 °C).
At 30 N load the worn scar at elevated temperature, as revealed in Figure 12, the outcomes of the EDAX line study revealed the presence of oxygen (O), iron (Fe), tungsten (W), and chromium (Cr) at high temperatures. The oxygen content was found to be 26.5 wt.%, confirming the presence of protective oxides or glaze layers. Notably, when the temperature reached 600 °C, smooth tribo layers formed on the worn surfaces, displaying visible adhesive grooves indicating adhesive wear. Moreover, a thin oxide layer developed between the interacting surfaces, serving as a protective layer that significantly improved the overall tribological performance.
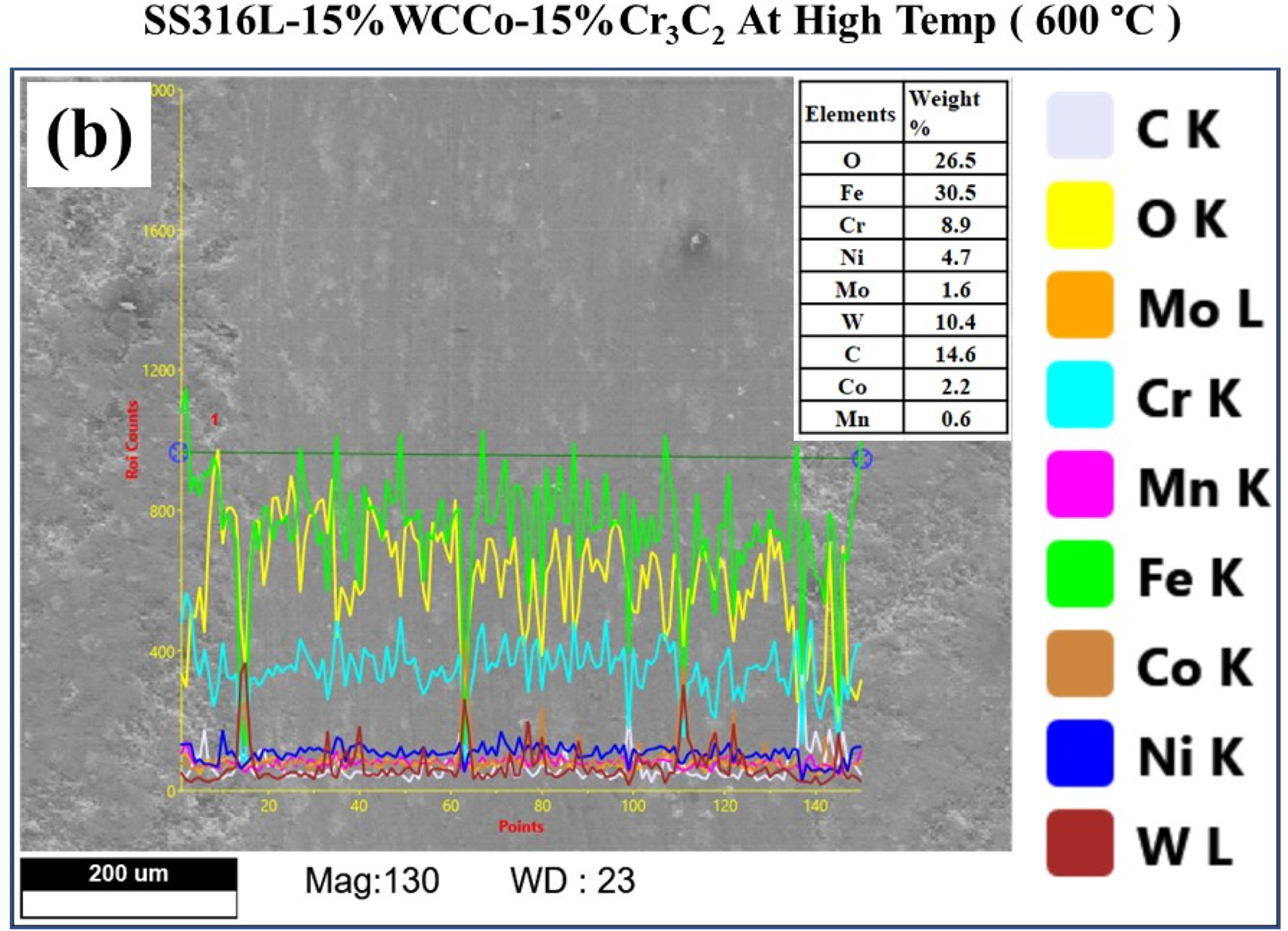
Line scanning analysis of worn scar image at elevated temperature (600 °C).
Figure 13 illustrates the wear worn contour of SS316L-15%WC-Co-15%Cr3C2 coatings under varying conditions, including temperatures (25 °C, 300 °C, and 600 °C) and loads (10 N and 30 N). The analysis indicates that with an upsurge in temperature from 25 °C to 600 °C, the wear track depth and width of the coatings decreases. At room temperature (25 °C), the worn scar of the coatings shows parallel plough lines, suggesting the occurrence of abrasive particles produced through the dry sliding wear test. However, at 600 °C, these parallel furrows vanish from the wear track profile. The depth of the SS316L-15%WC-Co-15%Cr3C2 coatings was approximately 42.6 μm at 25 °C for 10 N, 34.1 μm at 300 °C, and 34.6 μm at 600 °C. For a load of 30 N, the wear depths at 25 °C were 102.1 μm, 89.2 μm at 300 °C, and 46.7 μm at 600 °C. It was observed that with increasing temperature, material removal decreased for both loads. The SS316L-15%WC-Co-15%Cr3C2 coatings demonstrated improved wear resistance at elevated test temperatures.
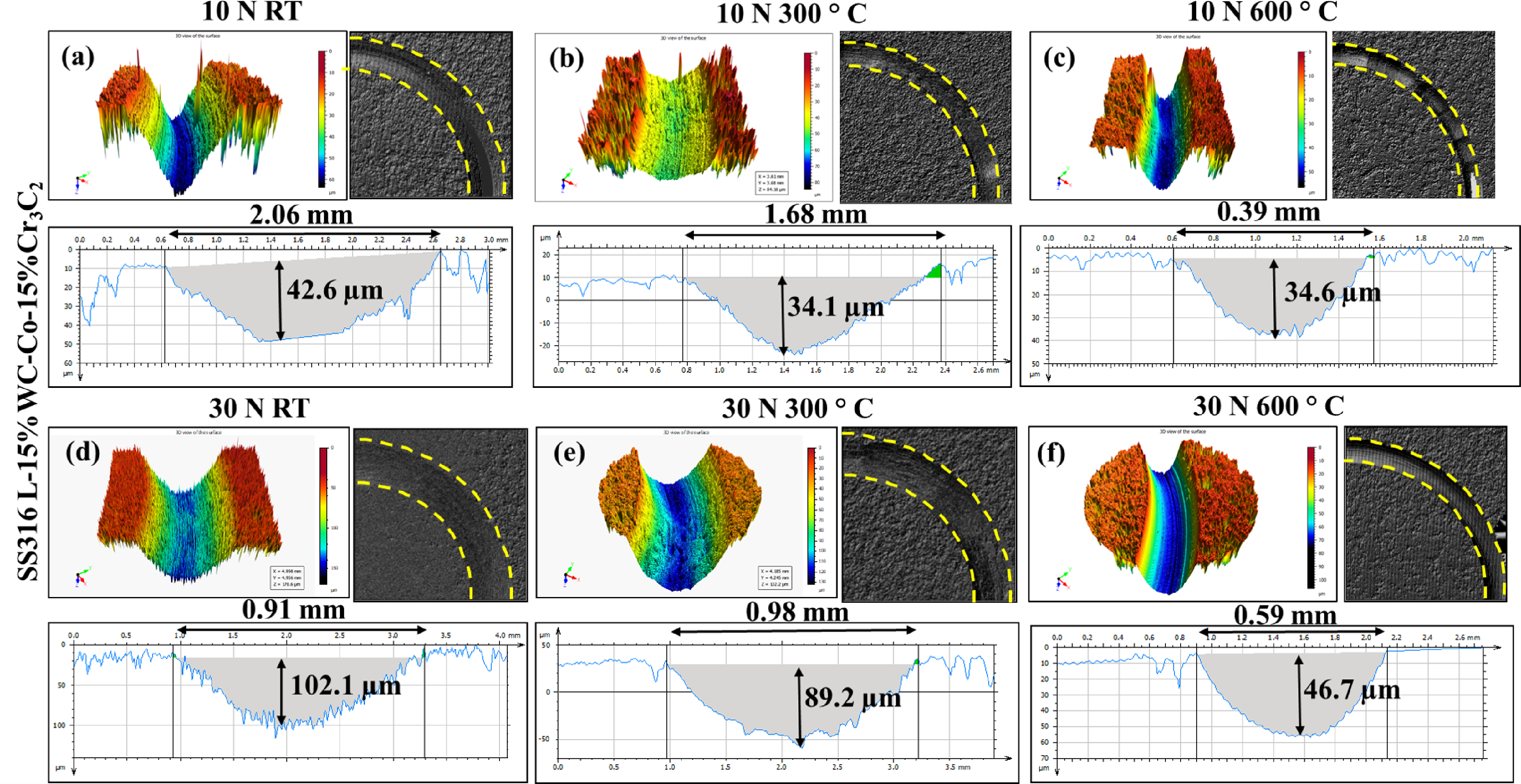
Worn scar analysis image taken from 3 D profilometer at different temperatures (25 °C, 300 °C, and 600 °C) and loads (10 N and 30 N).
Furrows are grooves or scratches that form on surfaces due to wear and friction during contact and motion. These features, often caused by surface asperities, serve as critical indicators of surface degradation. 43 The mean density of furrows helps quantify wear and is crucial for assessing the frictional properties of a material. Generally, a higher furrow density leads to increased friction and wear. 44 In this study, the mean density of furrows for the coatings was measured at different temperatures and loads shown in Figure 14. At 25 °C and under a 10 N load, the mean furrow density was 84.62 cm/cm², increasing to 90.32 cm/cm² at 300 °C. Then reduced at 600 °C, decreasing to 74.98 cm/cm². For a load of 30 N, the mean density of furrows at 25 °C was 75.44 cm/cm², dropping to 63.74 cm/cm² at 300 °C and further to 58.01 cm/cm² at 600 °C. These results indicate that as the temperature increases, material removal decreases for both loads.
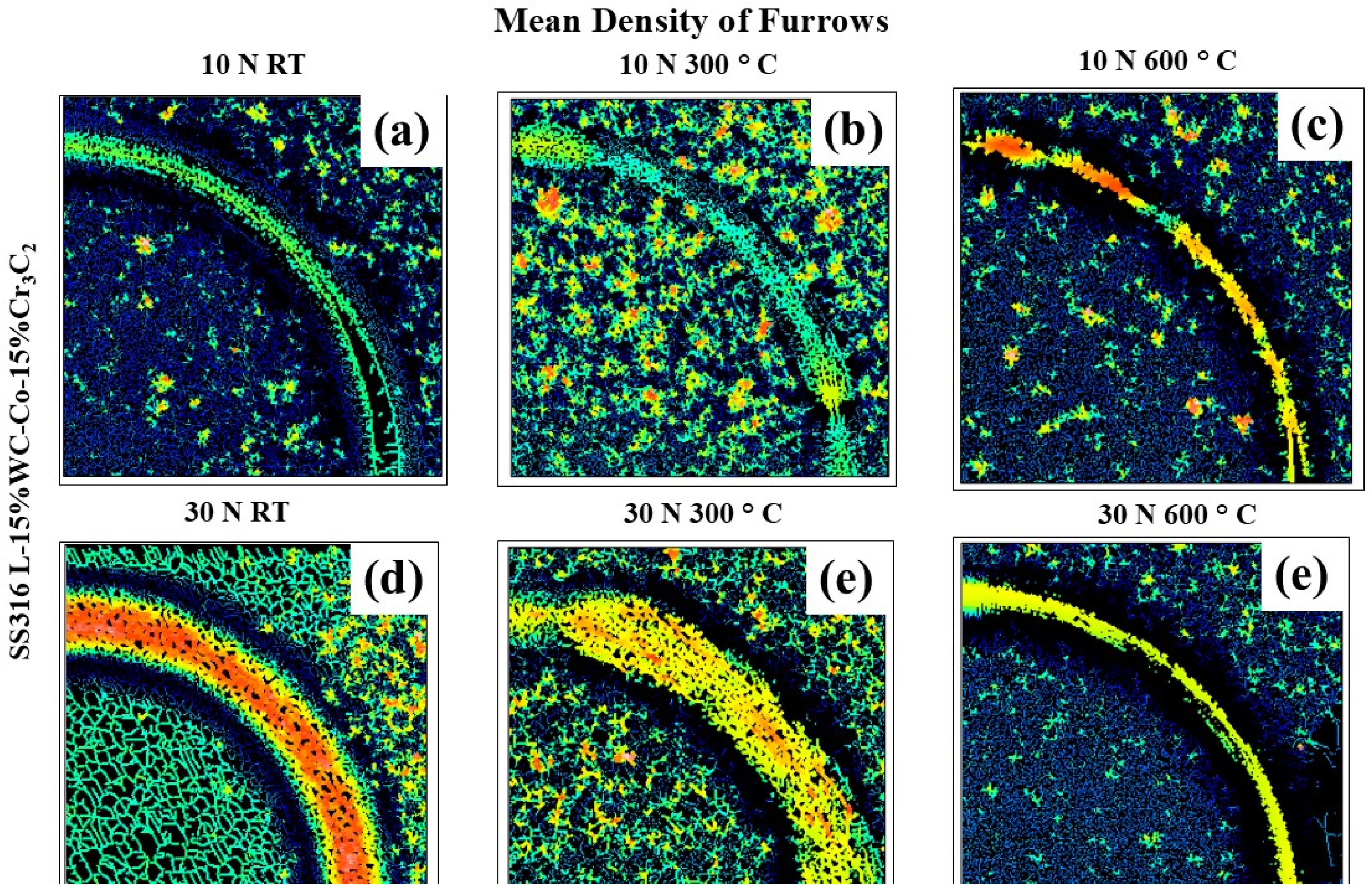
Furrows analysis image taken from 3 D profilometer at different temperatures (25 °C, 300 °C, and 600 °C) and loads (10 N and 30 N).
The mean density of furrows at a 30 N load was reduced more when compared to a 10 N load. The reduction in furrow density was measured to be 10.84% at 25 °C, 29.42% at 300 °C, and 22.63% at 600 °C. This indicates that as the load increased, there was a noticeable decrease in furrow density, suggesting improved resistance to wear under higher loads, particularly at elevated temperatures. Controlled furrowing at elevated temperatures may enhance lubricant retention and improve surface performance, contributing to better wear resistance. 71 This was attributed to the development of protective oxide or glaze layers. Significantly, at a temperature of 600 °C, smooth protective layers developed on the worn surfaces.
At 25 °C, abrasive wear is the predominant mechanism observed, characterised by the formation of distinct furrows and the removal of material resulting from direct contact between surfaces. As the temperature increases to 300 °C, the situation changes slightly: a layer of oxides begins to form on the surfaces. These oxide layers help to reduce friction but still permit a moderate level of material removal, indicating a transition in wear characteristics. When temperatures reach 600 °C, a shift occurs towards adhesive and oxidative wear, where the formation of protective tribo-layers plays a crucial role. These tribo-layers not only provide lubrication but also significantly diminish the rate of wear experienced by the components. 48 These observations suggest that the coatings exhibit optimal performance at elevated temperatures, positioning them as ideal candidates for applications in the aerospace and automotive sectors. In these industries, components frequently face high-temperature environments and demand superior wear resistance.
Raman spectroscopy proved highly effective in characterising worn surfaces and identifying oxide phases formed during wear. It offers valuable insights into the composition of surface layers, enabling the detection of specific oxide compounds formed as a result of sliding wear under different temperature conditions.72,73 The Raman spectroscopy was carried out on the wear track samples of room temperature and elevated temperature shown in Figure 15(b), (c), (e) and (f). During room temperature sliding wear, no metal oxide peaks appeared in the Raman spectra for the worn sample surfaces shown in Figure 15(a). However, at 600 °C, Raman spectroscopy detected oxide layers of Fe2O3 (224 cm−1, 293 cm−1, 409 cm−1), WO3 (703 cm−1), NiWO4 (703 cm−1, 1322 cm−1), Co2Mo3 (409 cm−1, 1588 cm−1), and NiCr2O4 (224 cm−1, 293 cm−1)74–76 displayed in Figure 15(d). These Raman spectroscopy results closely aligned with the XRD phase analysis presented in Section 3.5.2. The presence of various oxide phases on the worn surfaces at 600 °C, contributed to a reduction in both frictional coefficients and specific wear rates.
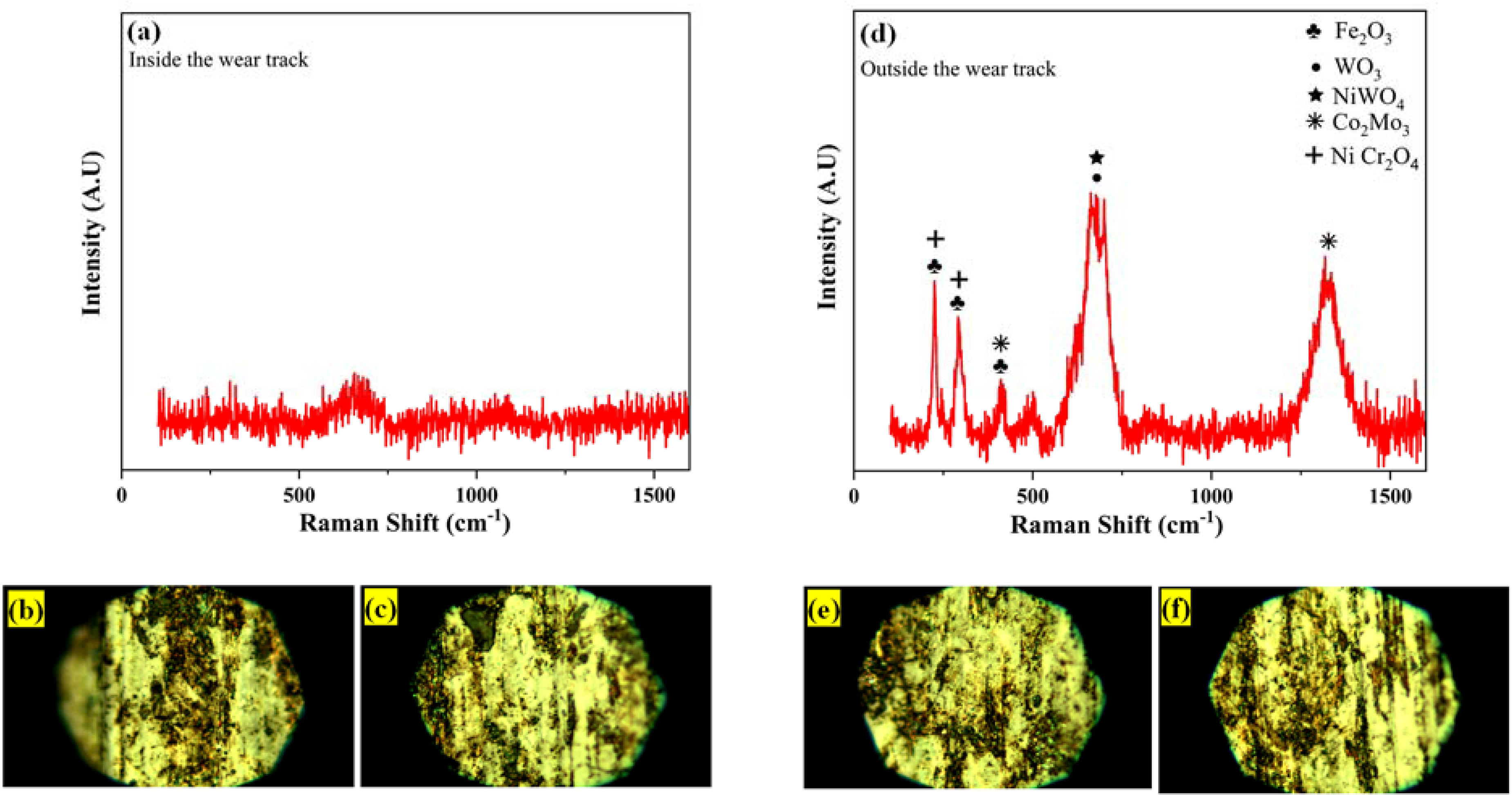
Raman graph after wear testing at (a) 25 °C and (d) 600 °C.
The research involved the use of SS316L-15%WC-Co-15%Cr3C2 coating powder, which was applied onto the surface of maraging steel 250 substrates using the HVOF spraying process. The coating microstructure was analyzed, and its mechanical properties were assessed. The wear performance of this coating was investigated through a ball-on-disc tribometer at room and elevated temperatures under normal loads (10 N and 30 N). The experiment yielded the following outcomes.
The SS316L-15%WC-Co-15%Cr3C2 composite coatings have a thickness of 211 ± 15 µm and a hardness of 711 ± 14 HV0.3. The porosity, surface roughness, and density of the SS316L-15%WC-Co-15%Cr3C2 coatings are recorded as 1.95 ± 0.15, 7.661 µm (Ra), and 8.71 g/cc, respectively. The wear rate of the maraging steel substrate was significantly high under normal functional loads of 10 N and 30 N, as well as at different temperatures. However, upon the application of the SS316L-15%WC-Co-15%Cr3C2 coating onto the maraging steel substrate, a notable decrease in the wear rate was observed. This improvement is ascribed to the development of a protective oxide layer within the SS316L-15%WC-Co-15%Cr3C2 coating, which includes Fe2O3, Ni W O4, WO3, and Ni Cr2O4. The SS316L-15%WC-Co-15%Cr3C2 exhibits consistently lower wear rates than the substrate across various temperatures and loads. At room temperature, the wear rate is 92.22% lower, decreasing to 95.07% at 300 °C, and 97.56% at 600 °C under a 10 N load. When the load is increased to 30 N, the wear rate remains significantly lower at all temperatures, showing a reduction of 81.98% at room temperature, 89.91% at 300 °C, and 97.12% at 600 °C. The results from the wear scar analysis conducted using a 3D non-contacting optical profilometer clearly indicate that there is less material removal as the temperature increases. Further, the mean density of furrows at a 30 N load was reduced more when compared to a 10 N load. The reduction in furrow density was measured to be 10.84% at 25 °C, 29.42% at 300 °C, and 22.63% at 600 °C. The SS316L-15%WC-Co-15% Cr3C2 coating exhibits wear resistance at 25 °C owing to its microstructure, which primarily experiences abrasion characterized by furrows and delamination. However, at 600 °C, the coating wear resistance improves as it forms a tribo-layer with self-lubricating properties. As temperature rises, the friction coefficient gradually reduces, and the wear mechanism changes from abrasive to adhesion wear and oxidation wear. Raman's study at room temperature sliding wear found no metal oxide peaks appeared for the worn sample surfaces. However, at 600 °C, Raman spectroscopy detected oxide layers of Fe2O3, WO3, NiWO4, Co2Mo3, and NiCr2O4. The presence of various oxide phases on the worn surfaces at elevated temperatures contributed to a reduction in both frictional coefficients and specific wear rates.
The findings of this study present an innovative composite coating to improve the wear resistance of the metals exposed to elevated temperatures in industrial utilisation.
Footnotes
Acknowledgments
The authors sincerely acknowledge the Department of Mechanical Engineering and Central Research Facility (CRF), National Institute of Technology Karnataka, for providing a research facility. The authors are also grateful to the Sri Jayachamarajendra Govt. Polytechnic Bengaluru for higher education support under QIP.
Author contributions
Chandramouli T V was involved in experiments, analysis, and original draft writing. Dr Sharnappa Joladarashi1 was involved in conceptualisation, discussion, and review—editing. Dr Ramesh M R was involved in conceptualisation, discussion, and review—editing.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
