Abstract
Skidding, which causes strip-surface defects between rolls and strips, is more likely to occur during the cold rolling of high-strength steel (HSS) than during that of ordinary steel. This occurs when the friction coefficient between them is low. However, the cause of the low friction coefficient during the cold rolling of HSS necessitates further investigation. This study conducted rolling tests using a synthetic hydrocarbon with oleic acid and methyl oleate as friction modifiers to compare the boundary friction coefficients of HSS and ordinary steel. Consequently, the friction coefficient of the HSS was lower than that of ordinary steel, regardless of the type of friction modifier applied. Furthermore, neutron reflectometry revealed that thicker adsorption films were formed on the HSS than on ordinary steel. Moreover, the thicker friction modifier film formation on HSS than on ordinary steel probably resulted in a lower friction coefficient during the cold rolling of HSS.
This is a visual representation of the abstract.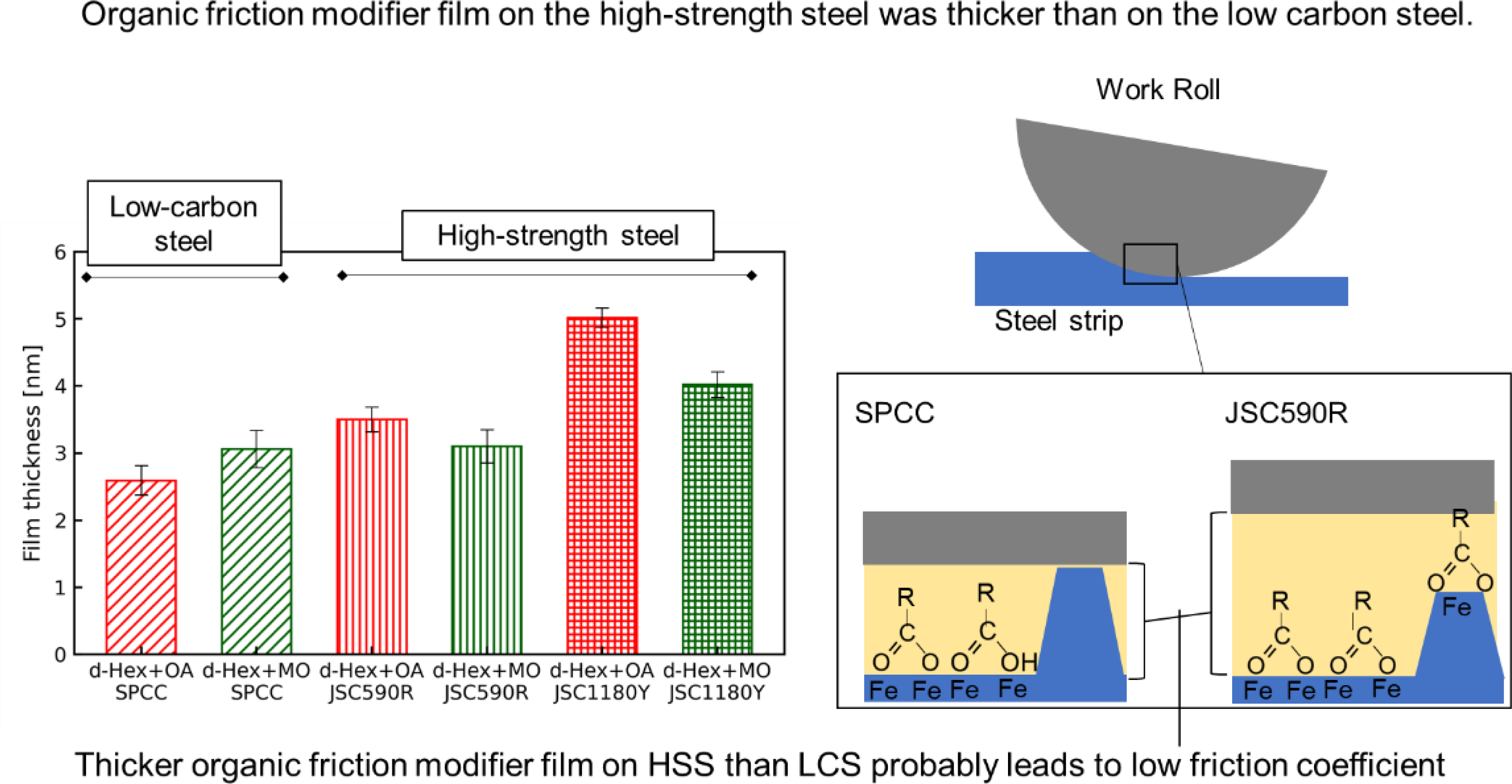
Keywords
Introduction
The increasing demand for lower fuel consumption and improved crash safety in automobiles has resulted in increased demands for lightweight and robust vehicle bodies. Therefore, materials such as aluminium, magnesium and carbon fibre-reinforced polymers have been used in automobile parts. They are aid in realising lightweight bodies and are beneficial in terms of reducing global CO2 emissions. Although multiple materials are used in automobile bodies, high-strength steel (HSS) is still considered effective in the production of car parts. 1 HSS must be thinner to further increase the strength and weight of automotive components and broader to improve the yield of automobile parts. Furthermore, the rolling of HSS with tensile strengths of 1–1.2 GPa has been in operation. 2
Each rolling mill has a limit on the rolling load, which is the reaction force that the work rolls of the rolling mill receive from the rolled material, to prevent surface fatigue defects on the rolls owing to contact stress between the backup and work rolls and cracking owing to stress concentration in the housing. In the production of such high-strength materials, the rolling load is extremely high, and this limit can be easily reached. The rolling force significantly depends on the friction coefficient between the work rolls and strips, and lubrication systems can effectively reduce the friction coefficient and rolling load.3–5
However, when the friction coefficient becomes significantly low, skidding occurs between the work rolls and rolled strips, particularly HSS strips.6,7 The skidding occurs when the circumferential speed of the work roll
When skidding occurs during rolling, the relative sliding velocity of the roll bite increases, resulting in heat scratches. In addition, the strip speeds at the entry and exit of the rolling mill and the tension on the steel strip become unstable. This results in strip-thickness fluctuations in the product owing to ineffective automatic-gauge-control. Results from rolling tests revealed that the friction coefficient was lower for HSS than that for ordinary steel; thus, it was concluded that skidding easily arises from the lower friction coefficient in HSS rolling. 9 Tight friction control is required to maximise and stabilise production throughput. Elucidating the mechanism underlying the low-friction coefficient in HSS rolling is beneficial for achieving this control.
The inside of a roll bite exists in a mixed-lubrication state in which the boundary and hydrodynamic lubrication areas are mixed. The friction coefficient under hydrodynamic lubrication was significantly lower than that under boundary lubrication. Therefore, in cold rolling, the friction coefficient depends significantly on the friction coefficient of the boundary lubrication and contact ratio, which is a proportion of the boundary lubrication regime to total contact area. 10 The authors suggested that the difference in the contact ratio between HSS and ordinary steel during rolling was one reason for the differences in the friction coefficients between them. 9 Although Boemer et al. focused on hydrodynamic lubrication in the roll-bite and the tried to simulate the flow of rolling oil inside roll bites during high-tensile steel rolling, the difference in friction coefficient between HSS rolling and ordinary steel rolling has not been clarified. 11 However, the differences in the boundary friction coefficients between HSS and ordinary steel are yet to be clarified. This could be one of the causes of the differences in the friction coefficients when these materials are rolled.
In boundary lubrication, friction modifiers, such as fatty acids and fatty acid esters, adsorb and form films on the friction surface, thereby reducing friction. However, these films often break at certain places, causing microscale metal-metal adhesion, which is defined as the real contact area. Therefore, the friction force is considered to be the shear strength of the film or the metal adhesion strength. 12 In other words, the boundary friction coefficient is influenced by two factors: the friction coefficient in the real contact area and the lubricity provided by the friction modifier film.
The friction coefficient in the real contact area on the friction surface is the ratio of the shear stress on the friction surface to the hardness of the material with lower flow stress on the two surfaces. Since the hardness of steel
Further, the lubricating properties of friction modifier films have been investigated using friction tests such as ball-on-disk tests. Simic et al. 15 proposed fatty acid adsorption mechanisms for steels. They suggested that rubbing induced chemisorption through the carboxylic acid group and the formation of iron carboxylate structures. In addition, the friction coefficient under boundary lubrication is dependent on the molecular structure of the friction modifiers, such as the carbon chain length, number of double bonds and functional groups.16,17
In addition, the friction coefficient varies depending on the composition of the workpiece material, even when the same friction modifier is used. Murakami et al. conducted a ball-on-disk test using ethanol as a lubricant with iron (Fe) and iron silicide (FeSi2) discs and silicon carbide (SiC) tool balls as workpiece materials. 18 The friction coefficient was lower when the working material was FeSi2 than when Fe was used. Moreover, they suggested that the lower friction was probably owing to the silicon alkoxide formed on the surface through the reaction between ethanol and FeSi2.
These studies indicate that material hardness may affect the friction coefficient. Further, they suggest that the friction coefficient is dependent on the reactivity between the rolled material and friction modifiers, as well as the structure of the friction modifier. The hardness and composition of HSS differ from those of ordinary steel because elements such as Si and Mn are added to HSS to improve its tensile strength and to stabilise the residual austenite at room temperature to ensure workability. 19 As reported by Murakami et al., 18 the composition difference between HSS and ordinary steel may affect the adsorption film formation of the friction modifier, which results in the differences in the boundary friction coefficient.
However, no previous studies have focused on the boundary lubricity of HSS during cold rolling or evaluated the effects of the hardness of the rolled material and friction modifier on the rolling lubricity compared with ordinary steel. In cold rolling process, the huge amount of oil is used and the cost of friction modifiers is expensive. Therefore, using friction modifiers that are bio-based, environmentally friendly, and provide effective lubrication has environmental and economic benefits.
This study investigated and compared the effects of friction modifiers on the rolling lubricity of HSS with those of ordinary steel during rolling. First, to elucidate the effect of material hardness on the boundary friction coefficient, ball-on-plate friction tests, which facilitate the evaluation of the boundary friction coefficient regime, were performed on SPCC steel. The flow stress and hardness were changed via the application of strain through rolling. Second, to evaluate the rolling lubricity indices, such as the forward slip and friction coefficient, rolling tests were conducted on ordinary SPCC steel (tensile strength: 270 MPa), JSC590R steel (tensile strength: 590 MPa) and HSS specimens. Synthetic hydrocarbons (poly-alpha-olefin, PAO) with oleic acid or methyl oleate added as friction modifiers were used as test oils. They are biodegradable and they could be good candidates to meet the environmental concerns. In addition, fatty acids and esters are usually added to industrial rolling oil as friction modifiers. Furthermore, to consider the influence of the test oil and test material on rolling lubricity, we analysed the adsorption film of the rolling oil formed on the surface of the steel sheet after rolling using X-ray photoelectron spectroscopy (XPS) and high-sensitivity infrared reflection–absorption spectroscopy (IRRAS). Third, ball-on-plate friction tests were performed on HSS and ordinary steel workpieces to clarify whether the relationship between the friction coefficient obtained in the rolling test and the test oil or test material was owing to the difference in boundary lubrication. Finally, a quantitative evaluation of the adsorption film thickness of the friction modifier on the surfaces of the HSS and ordinary steel was performed using neutron reflectometry, and the effect of the combination of the specimen and friction modifier on the rolling lubricity was discussed.
Friction test using Bowden–Leben machine to elucidate material hardness effect on friction
Test method
In this test, the boundary friction coefficient was compared for the SPCC, whose surface hardness was varied via the addition of strain via rolling. Friction measurements were performed using a Bowden–Leben machine. The SPCC coil (hereafter referred to as SPCC) was cut and used as a specimen. The SPCC coils rolled at reductions of 10% (hereafter referred to as SPCC r = 10%) and 50% (hereafter referred to as SPCC r = 50%) using a rolling mill, as described in Section ‘Comparison of rolling lubricity between HSS and ordinary steels and the chemical analysis of steel sheet surfaces after rolling’, were cut and used as specimens. These specimens were 25 mm wide and 60 mm long. They were then polished to a surface roughness of 0.03–0.04 µmRa. Steel balls were fabricated from SUJ-2 with diameters of 6 mm, and the load on the specimens was 10 N. The maximum Hertzian contact stress under these conditions was about 1.1 GPa. The hardness values of the specimens were measured using a Vickers hardness tester HM-102 (Mitutoyo Corporation). The measurements were performed at three different positions on each specimen and the average values are shown in Figure 1. The Vickers hardness values for SPCC, SPCC r = 10% and SPCC r = 50% were 120, 154 and 218 HV, respectively, and the hardness value for JSC590R was 179 HV. Further, PAO (INEOS Oligomers, US) and PAO with 5 wt% oleic acid (Tokyo Chemical Industry, Japan) or methyl oleate (Tokyo Chemical Industry, Japan) were used as test oils. The kinematic viscosity of PAO at 30 °C is 3 × 10−5 m2 /s, similar to actual rolling oil. The sliding conditions using PAO are referred to as PAO and those using PAO with oleic acid or methyl oleate are referred to as PAO, PAO + OA and PAO + MO, respectively. The specimens were heated to 100 °C, the surfaces were supplied with the test oil to maintain them in a state of oil bath lubrication. Further, 30 reciprocating runs of 10 mm in each direction were performed at a sliding speed of 10 mm/s. Thirty reciprocating strokes were performed three times for each oil condition at different positions, and the average values of the friction coefficients obtained were compared.
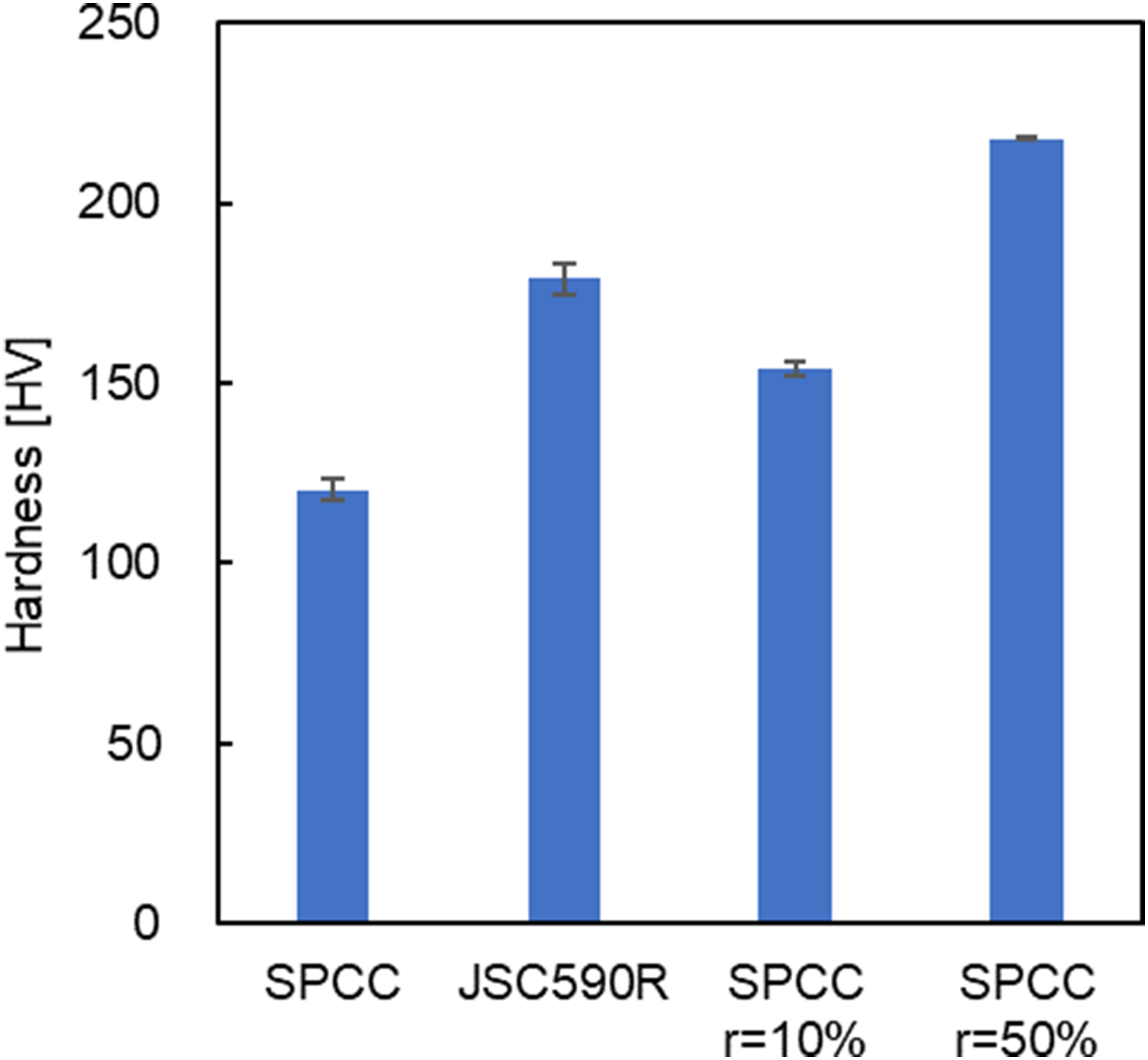
Comparison of the Vickers hardness for SPCC, JSC590R, SPCC rolled at a reduction of 10% (SPCC r = 10%) and SPCC rolled at a reduction of 50% (SPCC r =50%).
Results
The friction coefficient of SPCC as a function of the number of sliding cycles is shown in Figure 2(a). In PAO, the friction coefficient increased from 0.1 immediately after the start of sliding, and it exceeded 0.3 after seven sliding cycles. At that point, the stick–slip phenomenon occurred because of scratches between the steel ball and specimen, and the sliding was terminated. The friction coefficient of PAO + OA was lower than that of PAO (approximately 0.15). This value did not change significantly, even when the number of sliding cycles increased. PAO + MO had a lower friction coefficient than PAO + OA (approximately 0.1). Similarly, this value did not change significantly, even when the number of sliding cycles increased. The friction test results for SPCC r = 10% and SPCC r = 50% are shown in Figure 2(b) and (c), respectively. The friction coefficients of PAO + MO were lower than those of PAO + OA for both SPCC r = 10% and SPCC r = 50%. The friction coefficients for each condition were similar to those of the SPCC, as shown in Figure 2(a). Regardless of the strain applied to the specimen during rolling, the friction coefficients for each condition ranged as 0.1–0.35 for PAO, 0.1–0.2 for PAO + OA and 0.1–0.15 for PAO + MO. These friction test results show that the influence of the steel hardness on the friction coefficient during sliding was negligible under the conditions of the present friction tests.
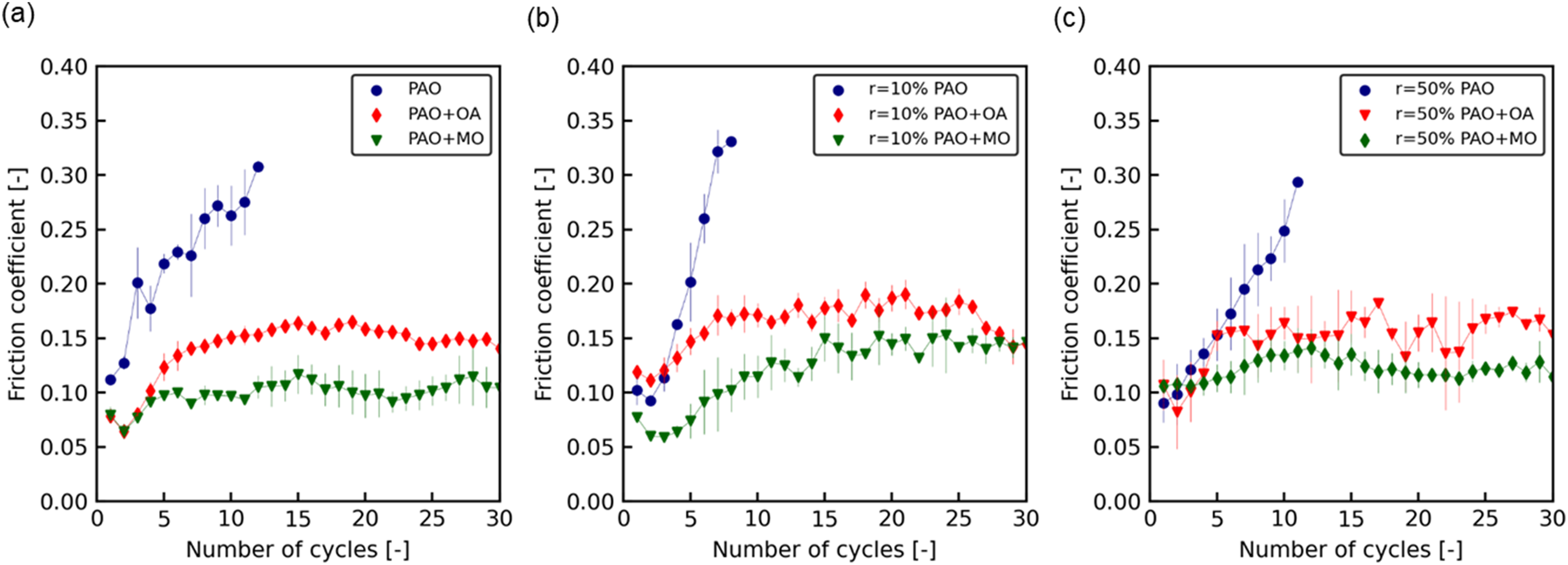
Friction coefficient as a function of the number of sliding cycles measured in the sliding test for: (a) SPCC (not rolled), (b) SPCC rolled at a reduction of 10% (SPCC r = 10%) and (c) SPCC rolled at a reduction of 50% (SPCC r = 50%).
Comparison of rolling lubricity between HSS and ordinary steels and the chemical analysis of steel sheet surfaces after rolling
Experimental methods
Rolling tests
Rolling lubricity indices such as the forward slip and friction coefficient were evaluated through rolling tests using SPCC and JSC590R steels as ordinary and HSS specimens, respectively. The measurements were performed in a single-stand pilot mill with a four-height configuration. The rolling test conditions are listed in Table 1. This pilot mill differs from industrial mills in roll diameters and rolling speed. The industrial mills have roll diameters of 500–600 mm, while the lab mill has a diameter of 110 mm. When the roll diameter is small, the amount of oil drawn into the roll bite reduces. 20 The rolling speed of industrial mills is about 1000 m/min, while the rolling speed of the pilot mill is 50 m/min. This slow rolling speed also reduces the amount of oil drawn into the roll bite. Since the pilot mill's small roll diameter and slow rolling speed lead to a decrease in the hydrodynamic lubrication regime, we could evaluate the friction coefficient at where the boundary lubrication regime was dominant.
Experimental conditions of the rolling experiment.

The rolling load was measured using strain gauges attached to the reduction cylinders by varying the reduction from 10 to 30% while maintaining the back tension at 98 MPa and front tension at 118 MPa which were applied to the steel strip. The rolling speed was at 50 m/min. The surfaces of the work rolls were polished with emery paper to a surface roughness of 0.3 μm Ra, which is equivalent to the surface roughness of a tandem rolling mill's work rolls. The surface roughness of SPCC and JSC590R before rolling was 0.9 µmRa and 0.8 µmRa, respectively,
Under identical rolling conditions, such as reduction and front-rear tension, the position of the neutral point depends on the friction coefficient. Thus, if the friction coefficient is high, the position of the neutral point is closer to the roll bite entry. Whereas, if it is low, it is closer to the roll bite exit. Therefore, the forward slip also varies with the lubrication condition of the roll bites and it is lower when friction is low.
The SPCC and JSC590R specimens were coils with width and thickness of 50 and 1.0 mm, respectively. PAO and PAO with 5 wt% oleic acid or methyl oleate were used as test oils. These were the same as those described in Section ‘Friction test using Bowden–Leben machine to elucidate material hardness effect on friction’. The rolling conditions using PAO are referred to as PAO, and those using PAO with oleic acid or methyl oleate are referred to as PAO, PAO + OA and PAO + MO, respectively. The test oil was supplied via air atomisation. The test oil was heated to 50 °C in a vessel and fed at a flow rate of
High-sensitivity infrared reflection–absorption spectroscopy
Fourier-transform infrared (FTIR) spectroscopy was performed using an FT/IR-6100 spectrometer (JASCO Corporation). The measurement range was 100 µm × 100 µm, and the incidence angle was 83°. After the steel strips were rolled at a reduction of 30%, they were cut into 50 mm squares and used as specimens for analysis without washing.
X-ray photoelectron absorption spectroscopy
The X-ray photoelectron absorption spectrometer used was Phi-Quantes (ULVAC Phi). The X-ray source for the irradiated X-rays was a single-crystal spectroscopic Al Kα (
Friction test using a Bowden–Leben machine
SPCC and JSC590R coils were used in the rolling test, and the plates were cut from the coils in the pre-rolled state and used as specimens for the friction test. Hereafter, these are referred to as SPCC and JSC590R, respectively. The lubricants used in the rolling tests were PAO, PAO + OA, and PAO + MO. The other sliding conditions were similar to those described in Section ‘Friction test using Bowden–Leben machine to elucidate material hardness effect on friction’.
Results
The rolling load of the SPCC as a function of reduction is shown in Figure 3(a). The rolling load increased with reduction in all cases. The rolling load for PAO was the highest at all reductions, whereas those of PAO + OA and PAO + MO were approximately 10% lower than that of PAO. The forward slip as a function of the reduction is shown in Figure 3(b). The forward slip decreased with increasing reduction in all cases. PAO exhibited a higher forward slip than PAO + OA and PAO + MO at all reductions, whereas that of PAO + MO was lower than that of PAO + OA.
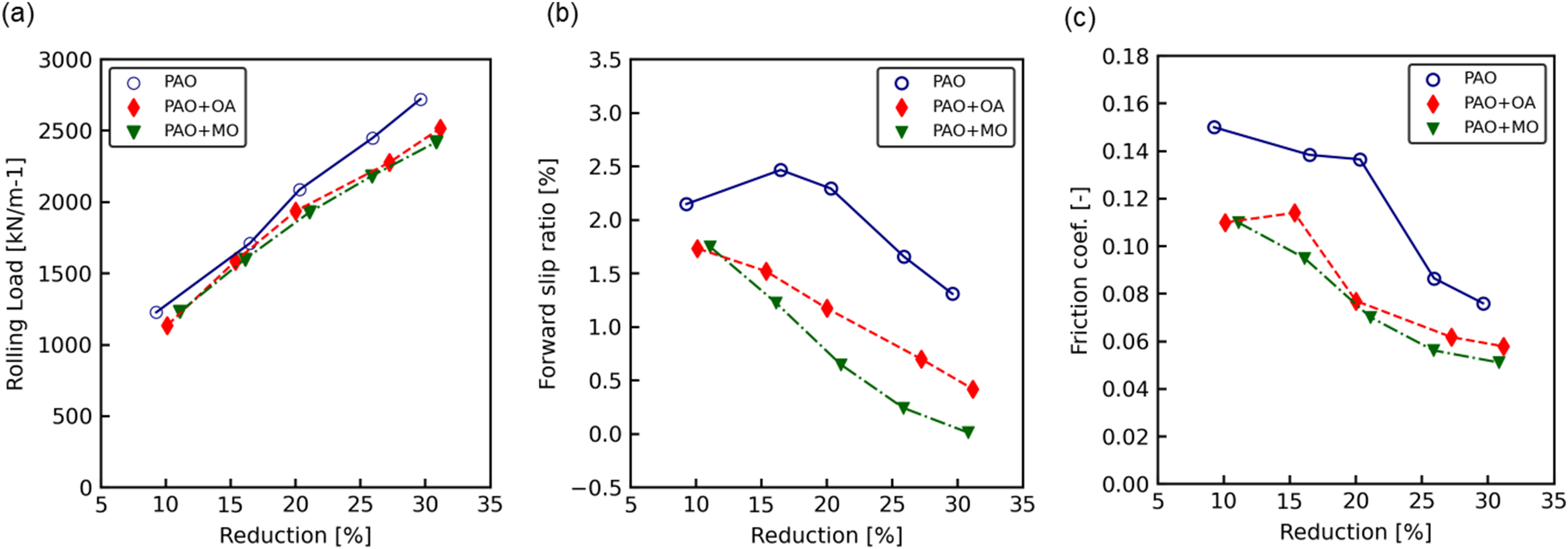
Rolling test result for SPCC for: (a) specific rolling force, (b) forward slip and (c) friction coefficient.
Using the rolling analysis model, 21 which considers noncircular roll flattening, we calculated the friction coefficient from the rolling load shown in Figure 3(a) and the forward slip shown in Figure 3(b). This model was based on Orowan's model 22 and Jortner's roll-flattening calculation model. 23 The calculated friction coefficient as a function of the reduction is shown in Figure 3(c). The friction coefficient was the highest for PAO and the lowest for PAO + MO. For all oil conditions, the temperature increased with increasing reduction, and was approximately 80 °C at a reduction of 30%. After rolled at a reduction of 30%, the surface roughness of ordinary steel was about 0.3 μmRa.
The rolling load as a function of reduction in JSC590R is shown in Figure 4(a). The rolling load increased with an increase in the reduction under all oil conditions. The forward slip as a function of the reduction is shown in Figure 4(b). The forward slip decreased with an increase in the reduction. The friction coefficient of JSC590R as a function of the reduction is shown in Figure 4(c). The friction coefficient was highest for PAO and lowest for PAO + OA. The strip temperature after rolling as a function of the reduction in JSC590R increased with an increase in the reduction and was approximately 120 °C at a reduction of 30%. After rolled at a reduction of 30%, the surface roughness of ordinary steel and JSC590R were 0.3 ± 0.03 μmRa. In addition, since the contact between the work rolls and the steel strip is instantaneous, a wear amount of the steel strip is negligible. In addition, the work rolls, which are in constant contact with the steel strip, showed little wear under this rolling test conditions.
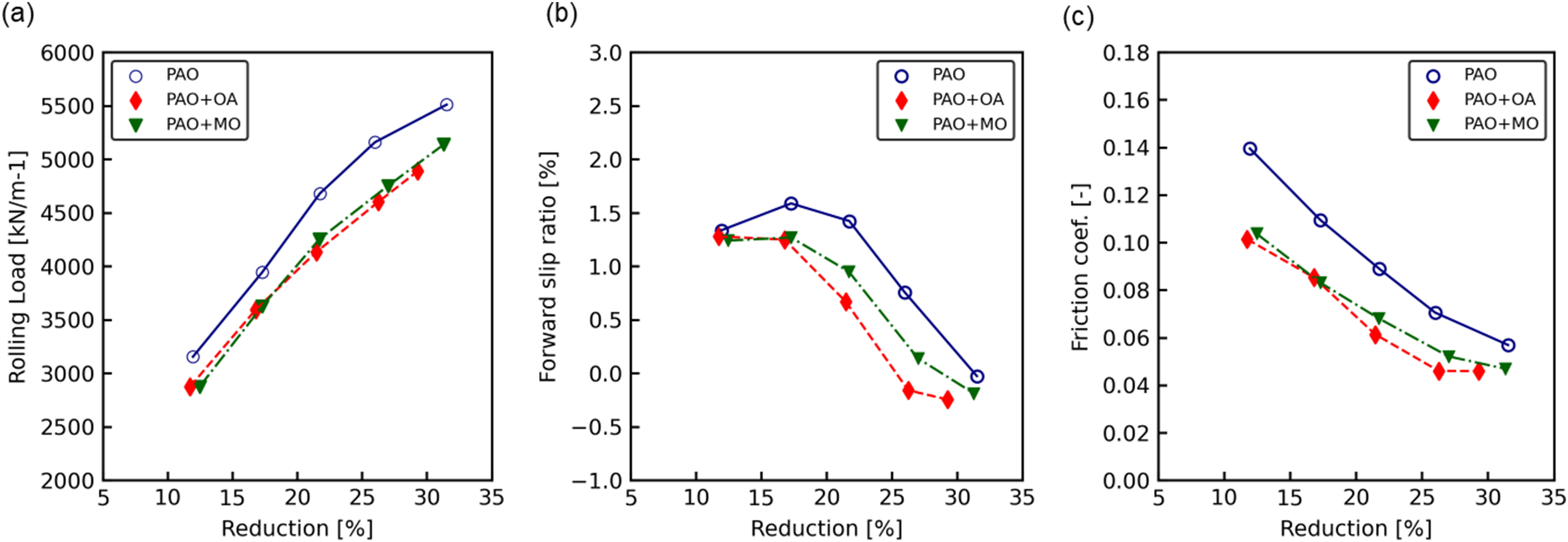
Rolling test result for JSC590R for: (a) specific rolling force, (b) forward slip and (c) friction coefficient.
Friction modifiers dissociate from the surface and lose their friction-reducing function when the temperature of the adsorbed surface exceeds the transition temperature, which is approximately 200 °C. 24 Therefore, under the conditions of this experiment, where the maximum strip temperature was 120 °C, the friction modifier was functional. Thus, the difference in lubricity observed between the test oils can be attributed to the friction modifier.
The results of IRRAS analyses of the SPCC and JSC590R surfaces after rolling are shown in Figure 5(a) and (b), respectively. Figure 5(a) shows that the C–C/C–H bond was present at 1464 cm−1 and the C=O bond at 1700–1800 cm−1 was not observed in PAO. Absorption owing to iron oleate was also observed at approximately 1500–1600 cm−1 in PAO + OA. In contrast, PAO + MO exhibited the absorption of the C=O bond of the ester at 1745 cm−1; however, in contrast to the case of PAO + OA, no absorption peak for iron fatty acids was observed. As shown in Figure 5(b), the absorption peak of the C–C/C–H bonds on the JSC590R surface was observed at 1464 cm−1 for PAO, but not at 1700–1800 cm−1, where C=O bonding was present. For PAO + OA, an absorption peak of iron oleate was observed at 1500–1600 cm−1, as on the SPCC surface; however, the absorption intensity of COOH at 1710 cm−1 was smaller than that on the SPCC surface. Although an absorption peak of iron oleate was observed at 1500–1600 cm−1, that of COOH at 1710 cm−1 was smaller than that of the SPCC surface. The ratio of the absorption intensity of iron oleate at 1596 cm−1 to the absorption intensity of oleic acid at 1710 cm−1 was 1.6 for SPCC and 5.6 for JSC590R. This indicates that the proportion of iron oleate present on the surface of JSC590R was greater than that of SPCC. For PAO + MO, the absorption peak of the C=O bond of the ester was observed at 1745 cm−1. However, in contrast to the case of PAO + OA, no iron oleate absorption peak was observed for PAO + MO. These IRRAS results show that when oleic acid was used as a friction modifier, oleic acid and iron oleate were in the oil film on the surface of the both the SPCC and JSC590R after rolling. Iron oleate was formed by the chemisorption of oleic acid on the surface of a steel sheet during rolling. Moreover, the proportion of iron oleate in the oil film was significant for JSC590R. When methyl oleate was used as the friction modifier, the oil film on the surface of the steel sheet after rolling contained methyl oleate in both the SPCC and JSC590R samples. The absence of iron oleate suggests that most of the methyl oleate was physically adsorbed onto the steel sheet surface.
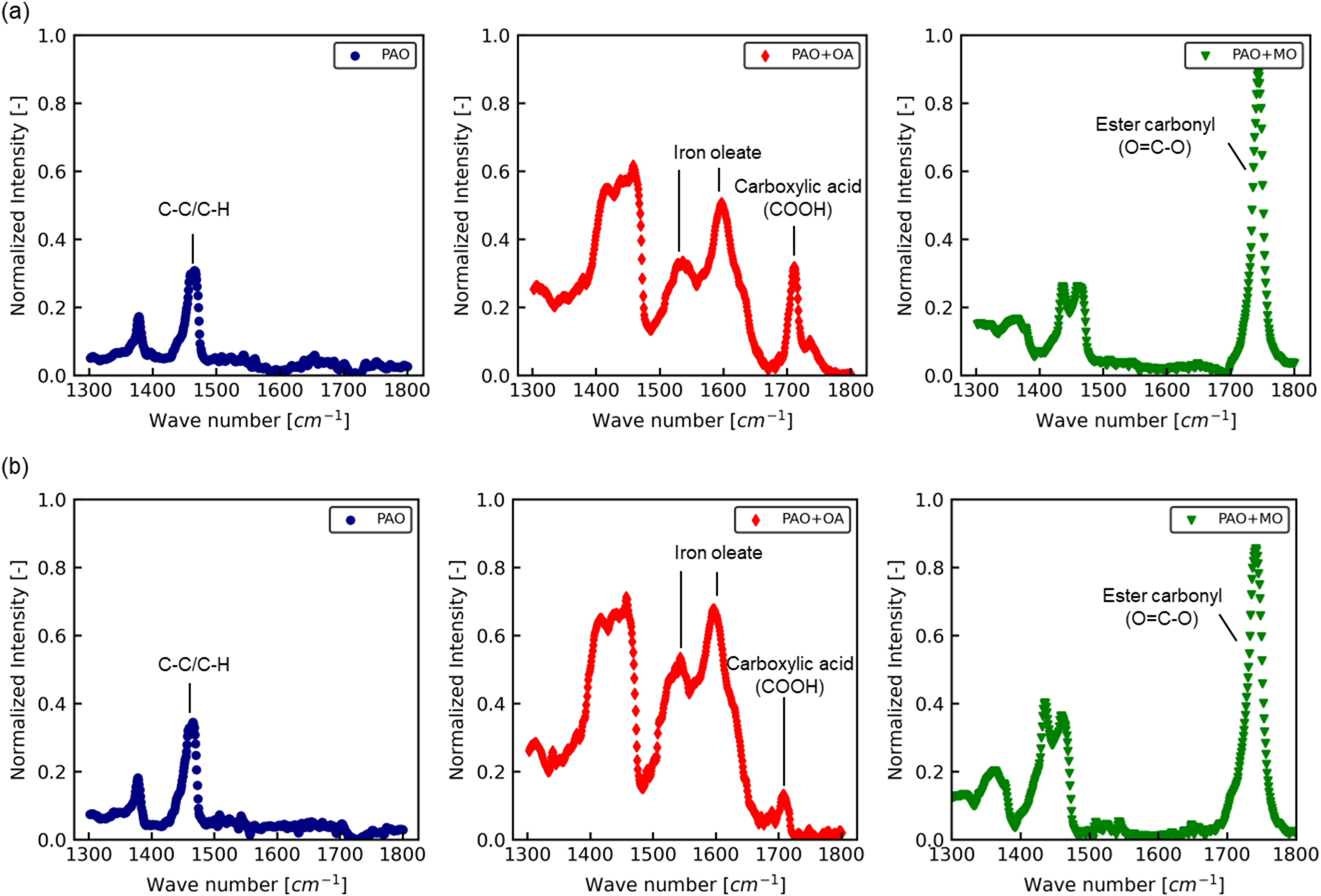
Ir spectra after rolling with PAO, PAO + OA and PAO + MO for: (a) SPCC surface and (b) JSC590R surface.
The C1s spectra obtained from the XPS measurements of the SPCC and JSC590R surfaces after rolling are shown in Figure 6(a) and (b), respectively. The binding energies of each chemical species, identified by fitting the C1s spectra, are listed in Table 3. In the C1s spectra of the SPCC surface shown in Figure 6(a), peaks corresponding to carbide, C–O and O=C–O bonds were observed for PAO. In contrast, peaks corresponding to C–O and O=C–O bonds were observed for PAO + OA and PAO + MO. In the C1s spectra of the JSC590R surface shown in Figure 6(b), as well as for the SPCC surface, carbide, C–O and O=C–O bond peaks were observed for PAO, whereas C–O and O=C–O bond peaks were observed for PAO + OA and PAO + MO. The peak area ratios of each chemical species determined by fitting are shown in Figure 7. In both the SPCC and JSC590R specimens, the area ratios of the C–O and O=C–O bond peaks for PAO + OA and PAO + MO were higher than those for PAO. The XPS results suggested that oleic acid and methyl oleate with carbonyl groups in the PAO + OA and PAO + MO test oils were chemisorbed onto the steel sheet surface to form a film.
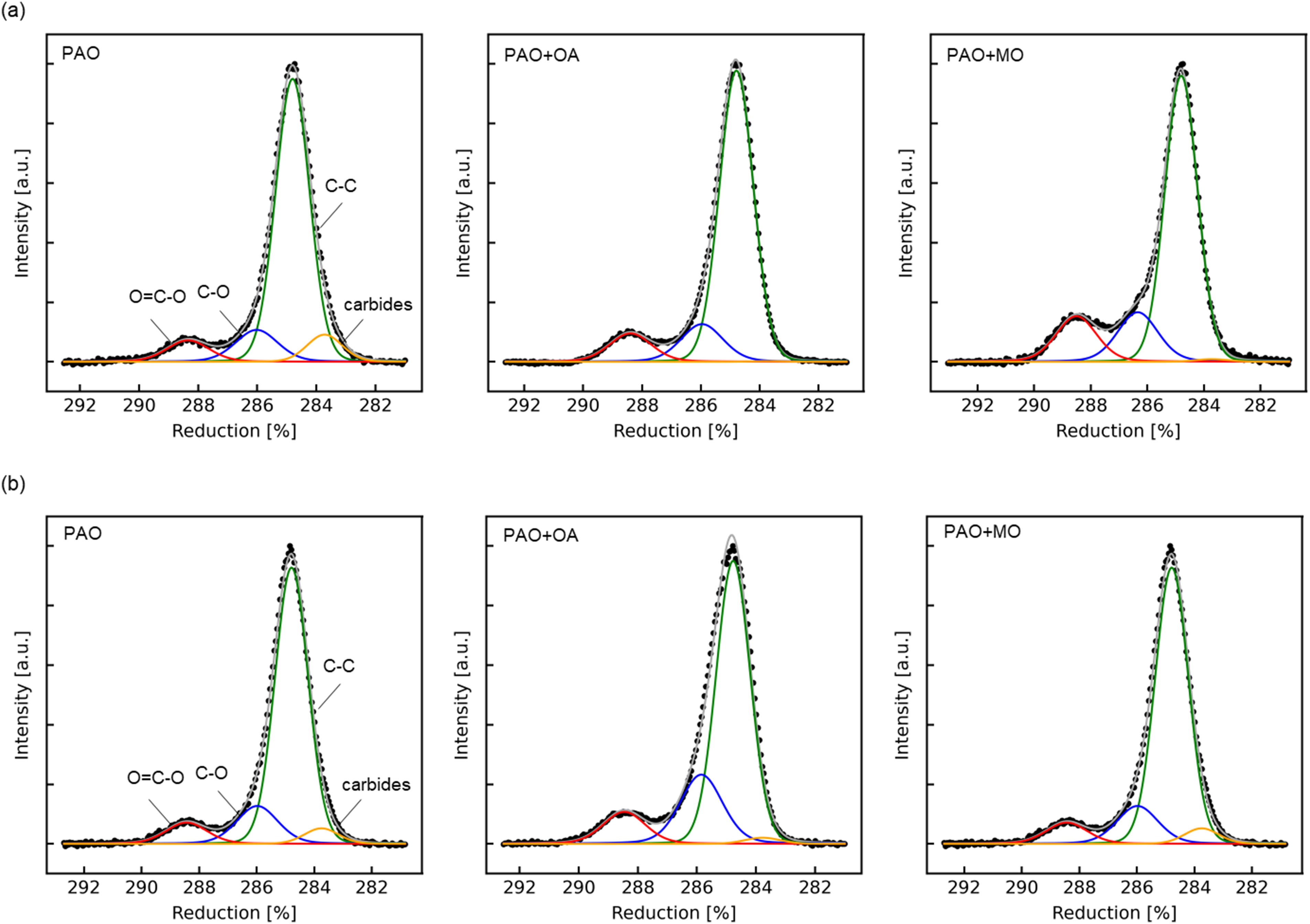
XPS spectra after rolling with PAO, PAO + OA and PAO + MO for: (a) SPCC surface and (b) JSC590R surface.
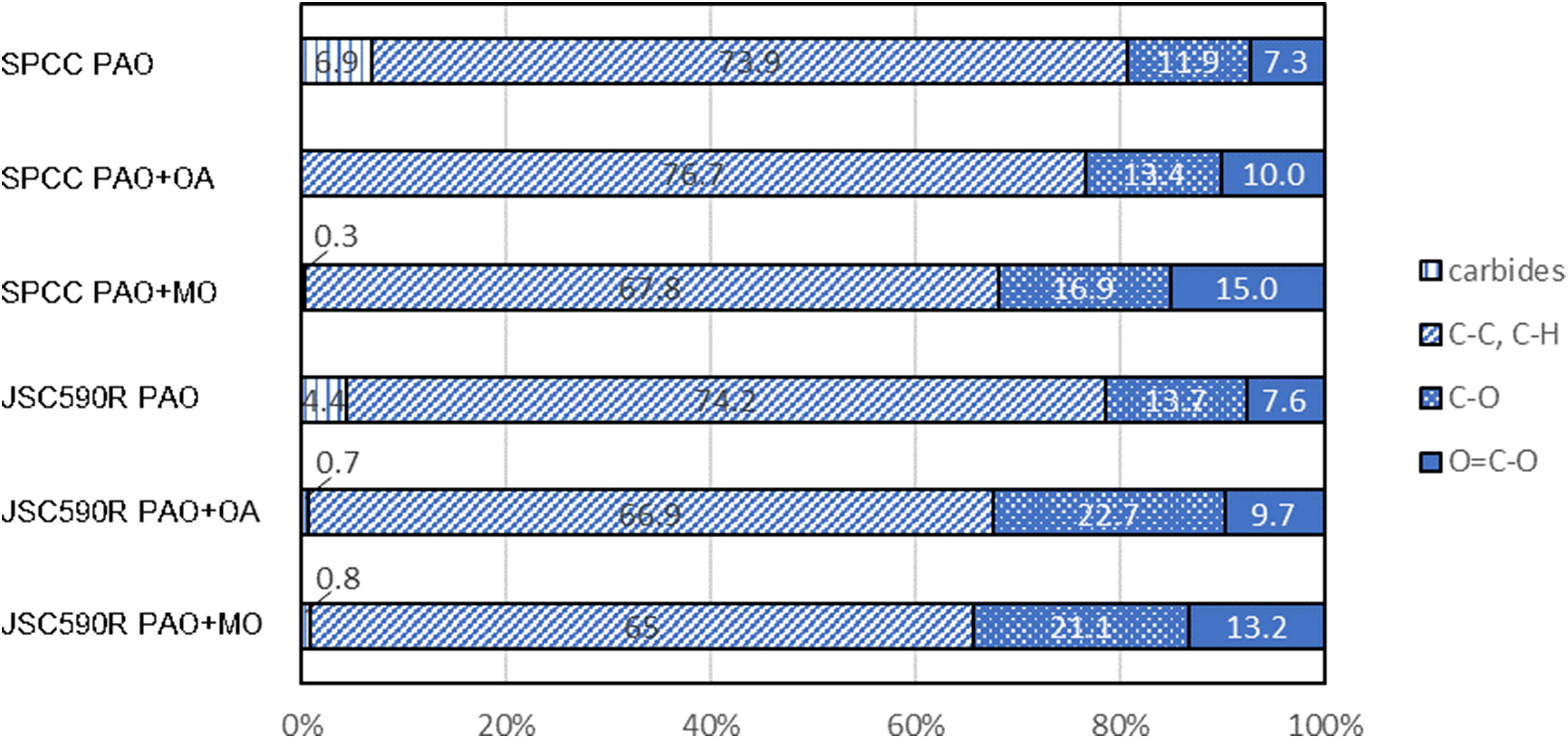
Comparison of XPS peak areas for each sample.
The friction coefficient of SPCC as a function of the number of sliding cycles is shown in Figure 8(a). In PAO, the friction coefficient increased from 0.1 immediately after the start of sliding, and it exceeded 0.3 after seven sliding cycles. At that point, the stick–slip phenomenon occurred, which was attributed to scratches between the steel ball and the specimen, and the sliding was terminated. The friction coefficient of PAO + OA was lower than that of PAO (µ = 0.15), and this value did not change significantly even if the number of sliding cycles increased. Further, PAO + MO had a lower friction coefficient than PAO + OA (µ = 0.1), and this value did not change significantly even when the number of sliding cycles increased.
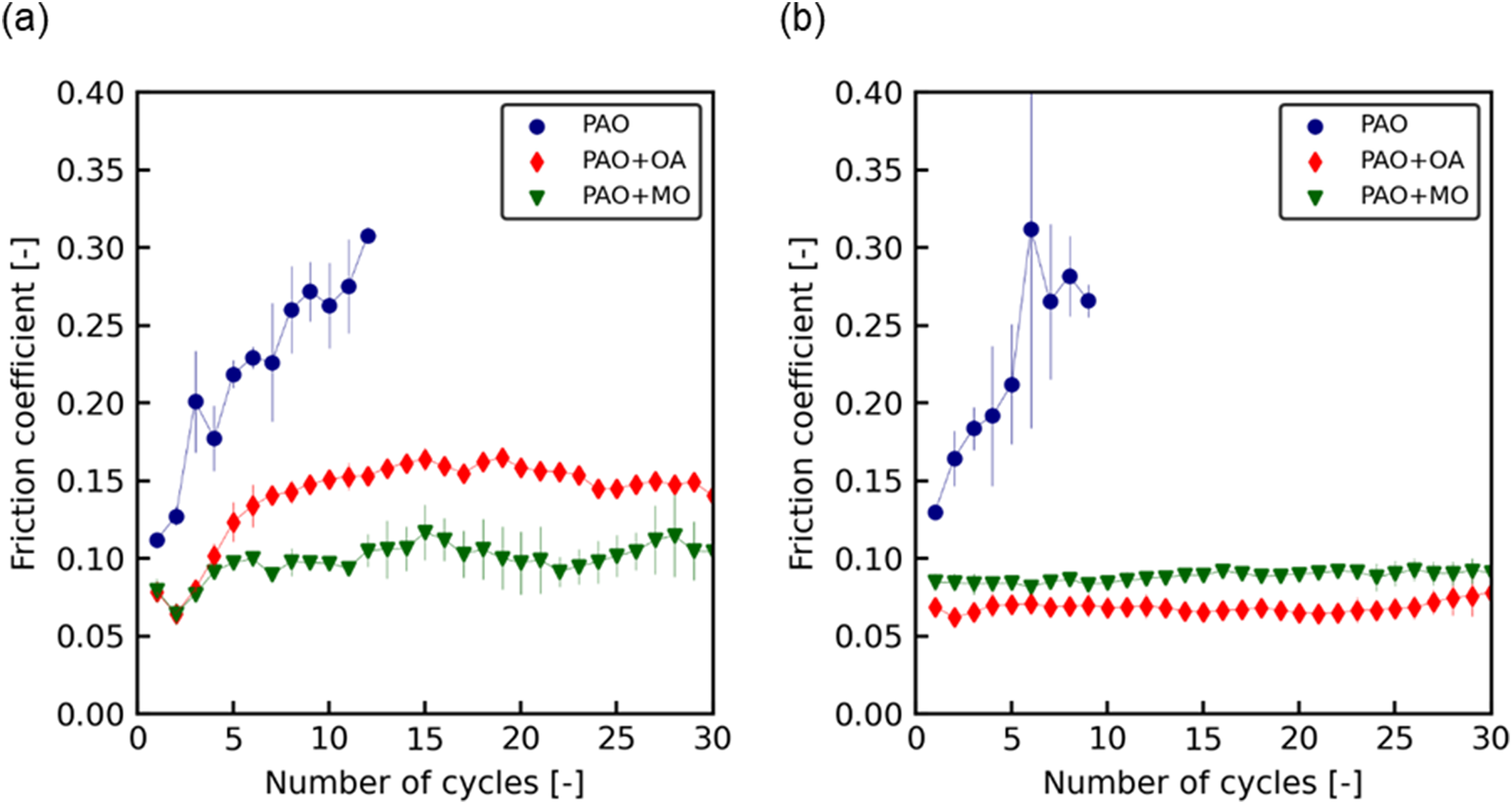
Friction coefficient as a function of the number of sliding cycles measured by the sliding test for: (a) SPCC and (b) JSC590R.
The friction coefficient as a function of the number of sliding cycles for JSC590R is shown in Figure 8(b). Similar to the case of SPCC, the friction coefficient of PAO increased from 0.1 to 0.3 with an increase in the number of sliding cycles. After nine sliding cycles, stick–slip occurred because of scratches between the steel ball and specimen, and sliding was halted.
The friction coefficient of PAO + OA was lower than that of PAO (µ = 0.06). Moreover, this value did not change significantly as the number of sliding cycles increased. According to these results, the friction coefficient was lower with methyl oleate when SPCC was used as the specimen and lower with oleic acid when using JSC590R. This trend corresponded to the forward slip and friction coefficient results of the rolling tests.
Neutron reflectometry
Experimental methods
Neutron reflectometry is useful for measuring organic compounds that are difficult to distinguish using X-rays. Consequently, neutron reflectometry has been used to assess the thickness and density of friction modifier films adsorbed onto metal surfaces. The correlation between the friction modifier adsorption film thickness or density measured by neutron reflectometry and lubricity has been assessed using friction tests.25,26
Measurements were performed using a time-of-flight neutron reflectometer, SOFIA, at MLF BL 16 of the Japan Proton Accelerator Research Complex (J-PARC). The reflectivity measurement enables us to investigate structural analysis in the sub-nanometer depth direction, which requires substrates with sub-nanometer or less roughness. To investigate the interface between liquid and solid in the sub-nanometer depth direction, neutron beam incidence from the substrate side, and a mirror-polished silicon block (50 × 50 × 10 mm) with high transparency is prepared for neutron beam. A metallic plate is used as the sputtering target and sputtered onto the surface of a mirror-polished silicon block (50 × 50 × 10 mm) to form a metallic film on the surface. 27 To evaluate the effect of the concentration of the alloying elements such as Si and Mn in the steel on the friction modifier film thickness, in addition to the SPCC and JSC590R steels used in the rolling and friction tests, JSC1180Y steel with a tensile strength of 1.2 GPa class was used as a sputtering target. As shown in Table 2, JSC1180Y contained the most alloying elements among the three steel grades listed in Table 2. The silicon block was placed in a holder, and the inside of the holder was filled with test oil such that the test oil was in contact with the metallic film on the silicon block surface during the neutron reflectometry measurements. All the measurements were performed at room temperature. Deuterated hexadecane (d-hexadecane) and d-hexadecane with 5 wt% oleic acid and methyl oleate were used as the friction modifiers. The conditions for each test oil were defined as d-hex, d-hex + OA and d-hex + MO. The scattering length density (SLD) of d-hexadecane is 6.8 × 10−6 Å−2. A neutron beam can be injected from the substrate and the interface between the thin metallic film and test oil can be directly observed. 28 Furthermore, the difference in the scattering length enables discrimination between deuterium and protium; therefore, layers formed by friction modifiers with only protium and layers of deuterated d-hexadecane solvents can be distinguished. To evaluate the effect of the concentration of the alloying elements such as Si and Mn in the steel on the friction modifier film thickness, in addition to the SPCC and JSC590R used in the rolling and friction tests, JSC1180Y with a tensile strength of 1.2 GPa class was used as a sputtering target. As shown in Table 2, JSC1180Y contained the most alloying elements among the three steel grades listed in Table 2.
Specifications of samples.
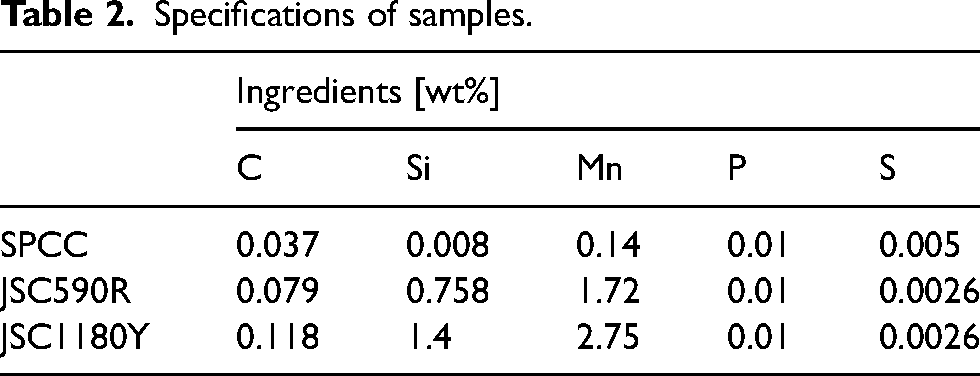
Summary of C1s spectra main results.
In the neutron reflectometry measurements, neutron beams were incident from the side of the silicon block at incident angles of 0.3°, 0.6° and 1.2° to the interface between the thin metal film and the test oil. Neutron beams reflected from the interface and transmitted through the silicon block were identified using a detector, and the reflectivity profile was obtained by normalising the intensity of the neutron beam to that of the incident neutron beam. The reflectivity profile was measured in air to confirm the structure of the metallic film. Subsequently, only d-hexadecane, the base oil, was poured into the sample holder to investigate the structure of the interface between the base oil and metal. Thereafter, the sample oil wherein d-hexadecane was mixed with a friction modifier was poured into the sample holder. Consequently, the structure of the base oil and metal interface was examined. The thickness and density of the friction modifier adsorption layer were then evaluated by comparing the reflectivity profile, which indicated the change in reflectivity, with the scattering vector Q, which indicated the change in the wavenumber vector of the neutron beam caused by its reflection of the neutron beam on the surface of the substrate. Q is expressed by Equation (3), where λ is the wavelength and θ is the angle of incidence.
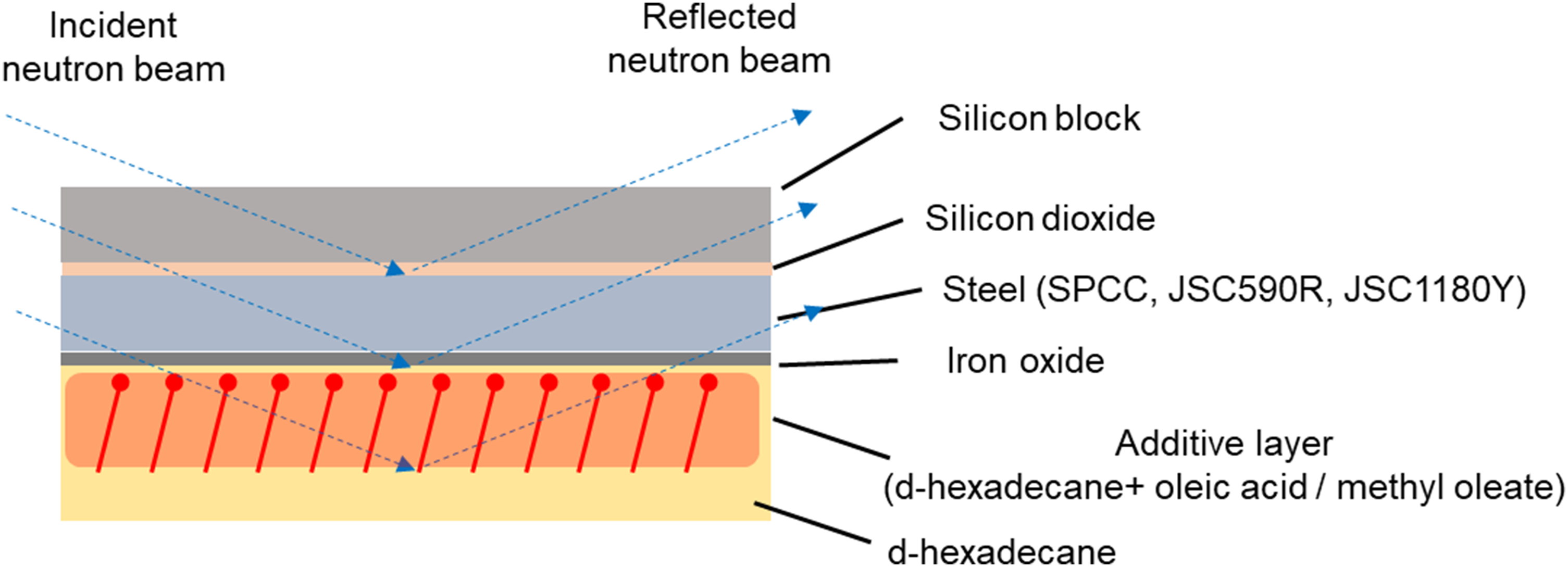
Schematic of neutron reflectometry measurements to investigate the film thickness and density of the adsorbed additive layer.
Results
The reflectivity profiles of Si blocks sputtered with SPCC, JSC590R and JSC1180Y steel grades are shown in Figure 10(a), (b) and (c), respectively. The solid lines indicate the fitting results based on Parratt theory. In all the analyses, the error between the measured values and the fitting results, that is,
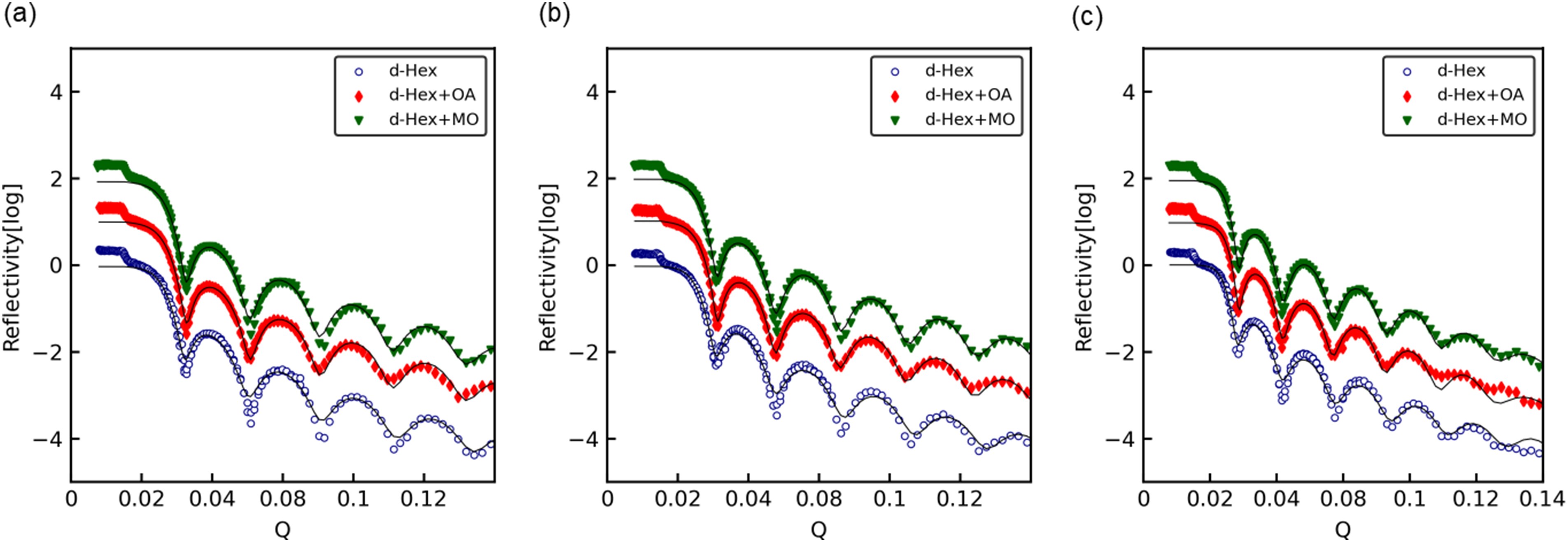
Neutron reflectivity versus scattering vector profiles with optimum fitting lines for: (a) SPCC, (b) JSC590R and (c) JSC1180Y.
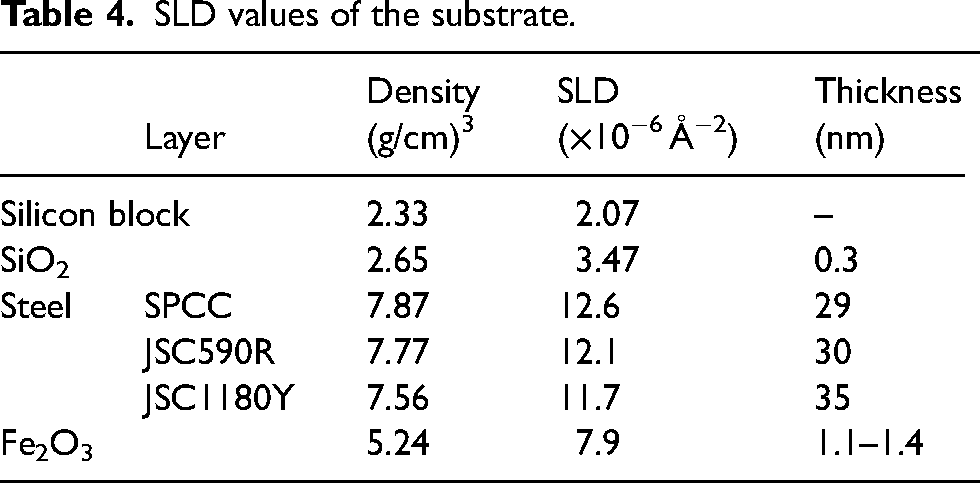
To assess the thickness and density of the adsorbed layer on the substrate surface owing to the addition of a friction modifier to d-Hex, we determined the SLD by fitting the reflectivity profiles, as shown in Table 4. Oleic acid and methyl oleate were not deuterated in this measurement; therefore, they were not detected using neutron reflectometry. Therefore, SLD was calculated from the thickness and density of the d-hexadecane molecular film in the friction modifier adsorption layer on the substrate. The density (D%) of the friction modifier adsorption layer was determined using the ratio of the SLD of the adsorption layer (SLDad) to that of the deuterated d-hexadecane (SLDdHex), as shown in Equation (4). The SLD values of the adsorption layers under each condition are listed in Table 5.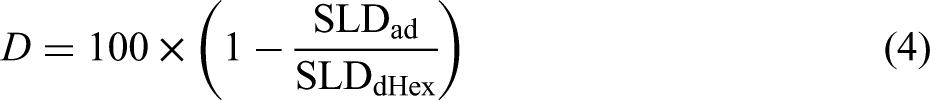
SLD values of the substrate.
Optimal fitting SLD data (×10−6 Å−2) for the friction modifier layer.

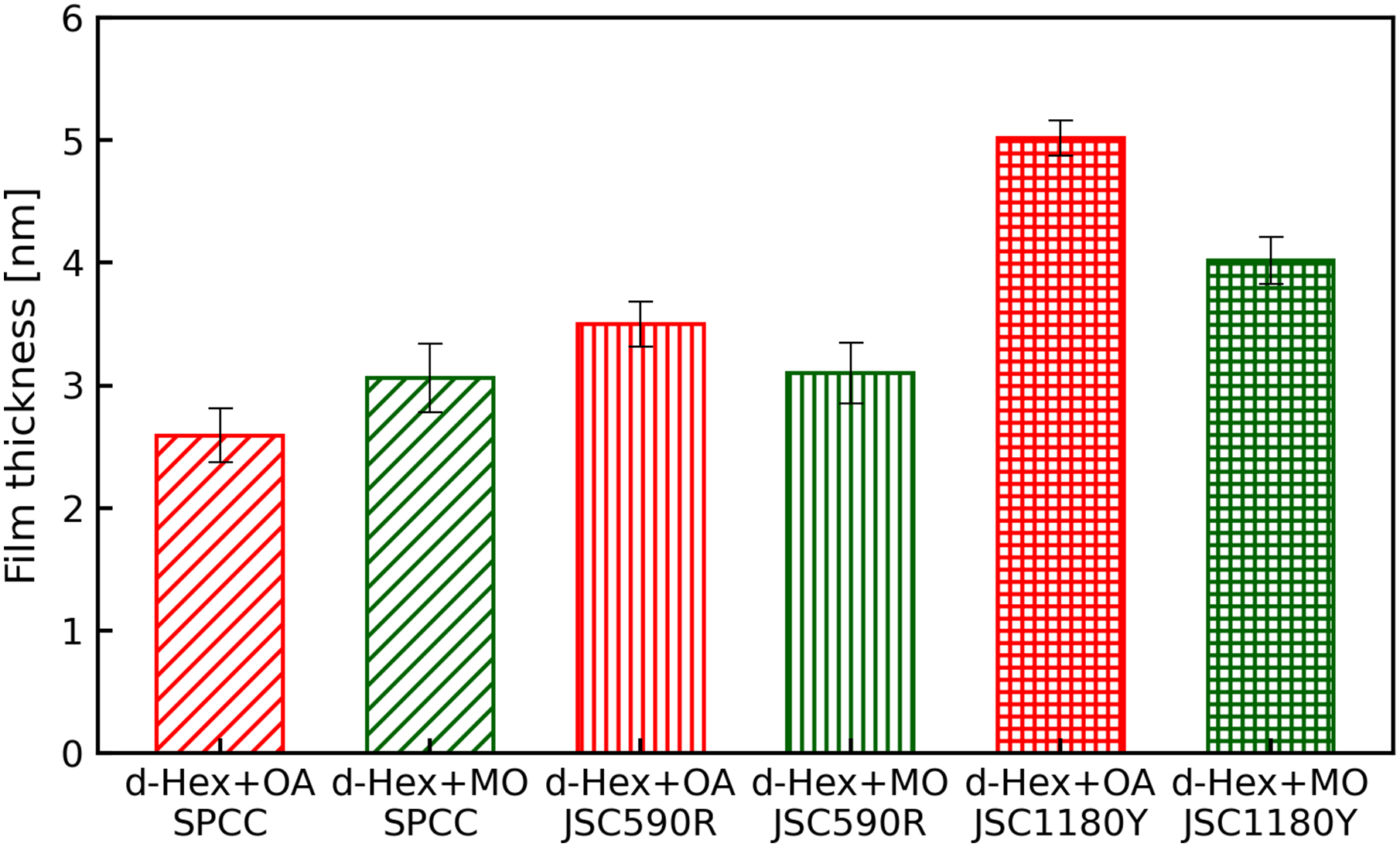
The estimated thickness of the adsorption layer obtained from the fitting analysis is shown in Figure 11. If the number of neutron counts is insufficient when acquiring NR profiles, the error in the estimated film thickness become significant. In this study, we acquired a sufficient number of neutron counts to minimise the error in the estimated film thickness as much as possible.
Film thickness of the organic frictional modifiers calculated from neutron reflectivity.
The adsorption thicknesses of oleic acid and methyl oleate estimated from d-Hex + OA and d-Hex + MO were 2.6 and 3.1 nm on SPCC, 3.5 and 3.1 nm on JSC590R and 5.0 and 4.0 nm on JSC1180Y, respectively. Thus, the films were thicker for JSC590R than for SPCC, in case of oleic acid, and even thicker for JSC1180Y than for JSC590R.
The densities of the adsorption layers, estimated from the fitting analysis, are shown in Figure 12. The densities of oleic acid and methyl oleate were estimated using d-Hex + OA and d-Hex + MO, respectively; they were 48% and 31% for SPCC, 53% and 40% for JSC590R and 55% and 44% for JSC1180Y, respectively. The density of oleic acid was higher than that of methyl oleate in all samples. When comparing the densities of the adsorption films on different substrates, the densities of both oleic acid and methyl oleate were higher for JSC590R than for SPCC, and even higher for JSC1180Y than for JSC590R.
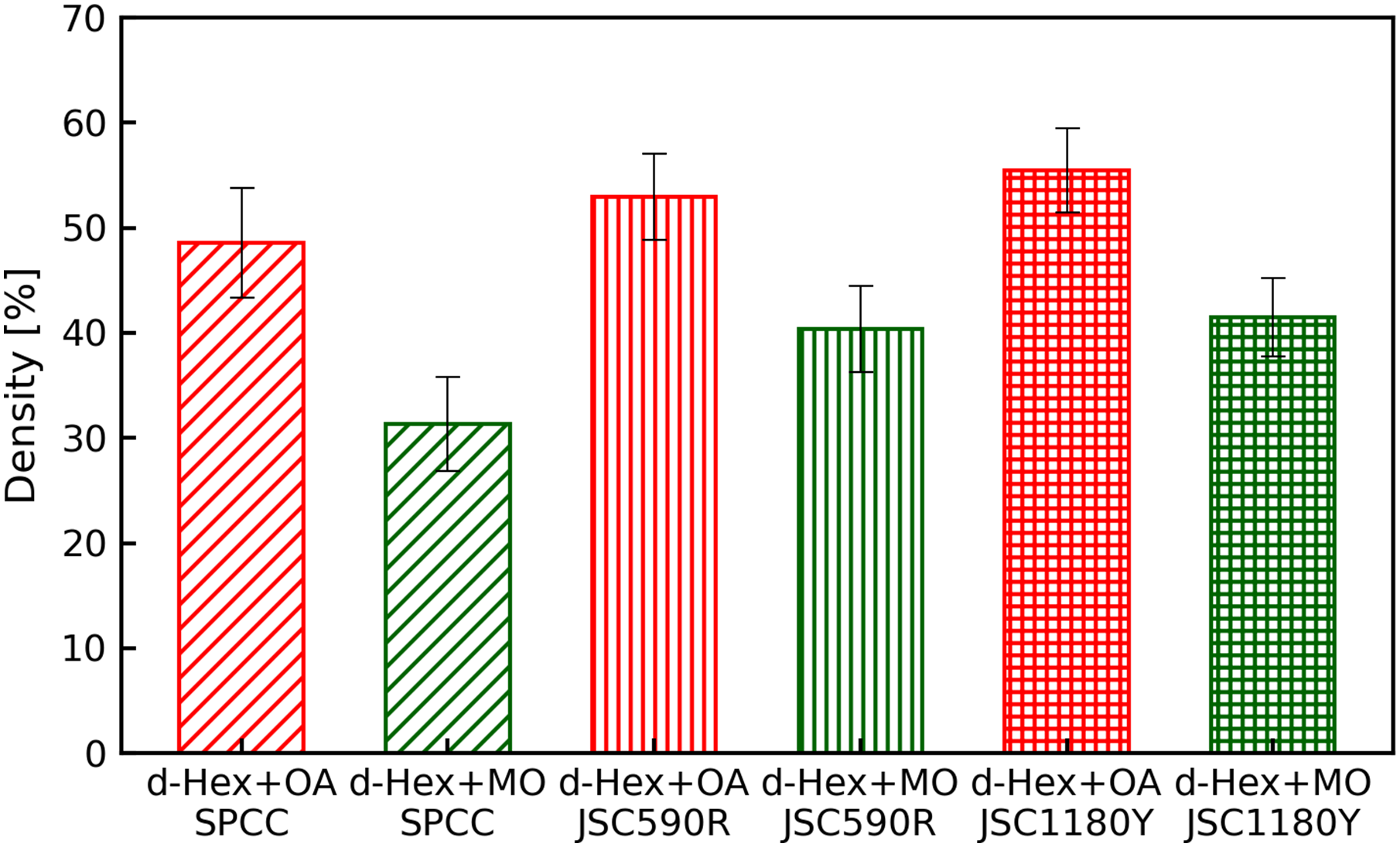
Film density of the organic frictional modifiers calculated from neutron reflectivity.
Discussion
Difference in friction coefficient between JSC590R and SPCC rolling
A comparison of the friction coefficients of JSC590R and SPCC, which are the indicators of rolling lubricity, is shown in Figure 13. The friction coefficient of JSC590R was lower than that of the SPCC at a reduction of 30%, regardless of whether oleic acid or methyl oleate was used as the friction modifier, as shown in Figure 13. Certain XPS results revealed that when oleic acid and methyl oleate were measured, O=C–O bond peaks were observed at 289.2–289.3 eV.31,32 In this study, the XPS results indicated the chemisorption of friction modifiers because the measurements were performed on SPCC and JSC590R plates washed with hexane. As shown in Figure 6, the peak of the O=C–O bond observed in PAO + OA and PAO + MO was 288.4 eV, which is 0.8–0.9 eV lower than that of the single bond. The shift in the bond energy to the lower energy side indicates the occurrence of electron exchanges between the carboxyl group and the iron atoms of the substrate. Thus, we conclude that oleic acid and methyl oleate reacted chemically with the steel sheet surface. As shown in Table 3, the peak area fraction of the O=C–O bond was not significantly different for SPCC and JSC590R. However, the peak area fraction of the C–O bond was approximately 10% larger in JSC590R than that in SPCC for both PAO + OA and PAO + MO. Although the identification of substances with C–O bond structures was impossible in this study, these results suggest that chemisorbed films of oleic acid and methyl oleate are more likely to form on the JSC590R surface than on the SPCC surface.
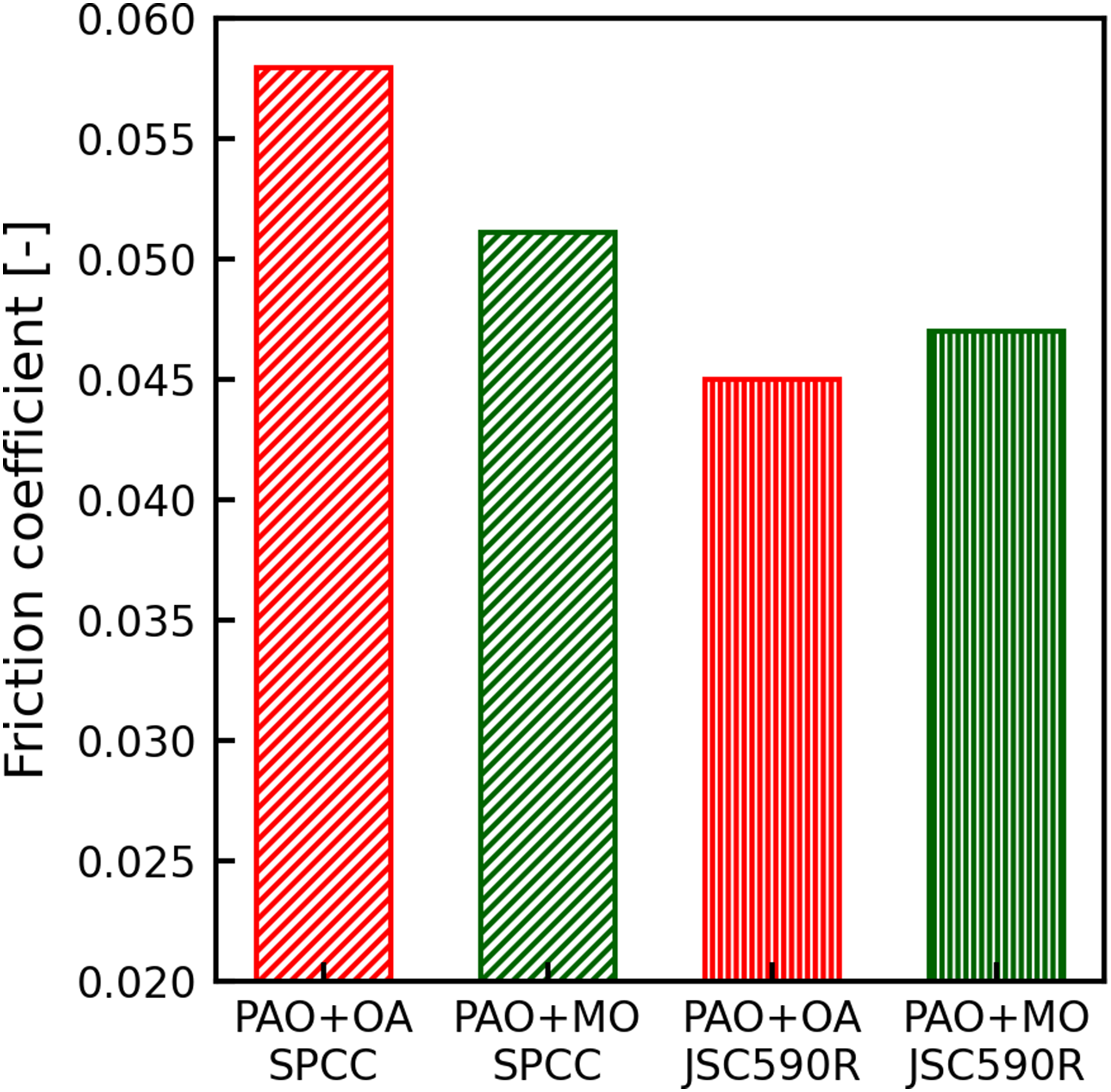
Comparison of the friction coefficient obtained from the rolling experiment at a reduction of 30%.
Based on the results of the adsorption thicknesses of the friction modifiers on various substrate surfaces obtained using neutron reflectometry, as shown in Figures 12 and 13, the film thickness and density of the friction modifier were more prominent on the JSC590R surface than on the SPCC surface. Furthermore, these values were the most significant for JSC1180Y. It appears that alloying elements, such as Si and Mn, in steel affected the friction modifier film thickness. However, it is necessary to elucidate why high concentrations of these elements increase the thickness.
As the neutron reflectometry used in this test was limited to very smooth surfaces, the results shown in Figure 11 refer to the adsorption film thickness of the friction modifier on a solid surface in the absence of rolling or rubbing. However, neutron reflectometry can be used to quantitatively evaluate the adsorption film thickness of a friction modifier on a substrate surface. Therefore, an adsorption film of equivalent thickness to that measured by neutron reflectometry probably formed inside the roll bite during SPCC and JSC590R rolling. Fry et al. suggested the minimum adsorption film thickness required for friction reduction was 0.6 nm, and the friction coefficient was lower under the larger adsorption film thickness. 33 This minimum film thickness was equivalent to the monolayer height of the friction modifiers. In other words, friction can be reduced if friction modifiers form monolayers. The adsorbed film thickness of the friction modifier on each substrate surface, as shown in Figure 11, was approximately four times the minimum required thickness of 0.6 nm for friction reduction. Therefore, the difference in rolling lubricity between JSC590R and SPCC was caused by the difference in the thickness of the adsorbed film of the friction modifiers.
Thus, the XPS and neutron reflectometry results indicate that the chemisorption of oleic acid and methyl oleate on the roll bites was more advanced during JSC590R rolling than during SPCC rolling and that a thicker friction modifier adsorption film was formed on the surface through physical and chemical adsorption. The results also suggested that these phenomena were responsible for the lower friction coefficient during JSC590R rolling than during SPCC rolling.
Difference between JSC590R and SPCC in the type of friction modifier with the best lubricity
In both rolling and friction tests, the friction modifiers that exhibited the best lubricity differed between SPCC and JSC590R, with the lowest friction coefficients obtained when methyl oleate and oleic acid were used for SPCC and JSC590R, respectively. The IRRAS results shown in Figure 5(a) and (b) indicate the substances in the adsorption film formed by physisorption and chemisorption of the friction modifiers on the steel sheet inside the roll bites. In the IRRAS measurements, greater amount of iron oleate than oleic acid was detected when oleic acid was used as the friction modifier on the surface of the steel sheets after rolling using both SPCC and JSC590R. However, when methyl oleate was used as the friction modifier, only methyl oleate was detected in the absence of iron oleate. This result suggests that oleic acid is primarily chemisorbed on the steel strip and methyl oleate is primarily physisorbed on the steel strip. A comparison between SPCC and JSC590R showed that the abundance ratio of iron oleate on the surface of JSC590R was higher than that on SPCC. This result indicates that JSC590R is more likely to form a chemisorption film during rolling when oleic acid is used as the friction modifier. Furthermore, Figure 11 shows that the adsorption film thicknesses of oleic acid and methyl oleate on the SPCC surface were 2.6 and 3.2 nm, respectively, indicating that the film thickness of methyl oleate is greater than that of oleic acid.
In contrast, the adsorption film thicknesses of oleic acid and methyl oleate on the JSC590R surface were 3.5 and 3.1 nm, respectively, indicating that oleic acid formed a thicker film on the substrate than methyl oleate. These results suggest that the friction coefficient on the SPCC surface was lowest when methyl oleate was used because the adsorption film was thicker for methyl oleate than for oleic acid. However, the JSC590R surface had the lowest friction coefficient when oleic acid was used because oleic acid was more chemisorbed than the SPCC surface, and the adsorption film of oleic acid was thicker than that of methyl oleate.
There are some steel grades such as silicon steel and manganese steel that contain more of each element than HSS. In this study, we found oleic acid is an effective friction modifier for HSS and it possibly an effective friction modifier also for silicon steel or manganese steel.
Based on the findings of this study, we will consider developing a cost-effective and efficient method for applying friction modifiers to new lubrication system. For example, Moreover, we will investigate the possibility of using environmentally friendly and sustainable friction modifiers that can provide effective lubrication during cold rolling while minimising environmental impact.
Conclusion
This study elucidated the cause of the low friction coefficient during the cold rolling of HSS. We compared the effects of friction modifiers on lubricity during the rolling of HSS and ordinary steel focusing on the boundary lubricity of them. In these tests, PAO with oleic acid and PAO with methyl oleate were used as the test oils. In addition, the SPCC, JSC590R and JSC1180Y films were sputtered onto a silicon block surface, and the adsorption film thicknesses of various friction modifiers on the surface were evaluated using neutron reflectometry. The findings of this study are summarised as follows.
Friction tests indicated that the effect of the work material hardness on the friction coefficient was negligible. Moreover, methyl oleate in SPCC and oleic acid in JSC590R acted as reducing the friction coefficient, which correlated with rolling test results. This correspondence between the rolling and friction test results suggests that the lubricating properties of friction modifiers in boundary lubrication significantly influenced the rolling friction coefficient under this study's conditions. Under rolling, JSC590R exhibited a lower friction coefficient than SPCC, and an analysis of the steel sheet surface after rolling using XPS showed that oleic acid and methyl oleate were more progressively chemisorbed on the roll bites during the rolling of JSC590R compared to that of SPCC. Evaluation of the film thickness via neutron reflectometry revealed that a thicker friction modifier film was formed on the JSC590R surface than on the SPCC surface. The progression of chemisorption of these friction modifiers and the thicker adsorption film on JSC590R than on SPCC likely caused the lower friction coefficient in JSC590R rolling than in SPCC rolling. The friction modifiers exhibiting superior lubricity differed between SPCC and JSC590R, with methyl oleate and oleic acid reducing the friction coefficient in SPCC and JSC590R, respectively. Chemical analysis of the steel sheet surface after rolling also confirmed that the ratio of iron oleate on the surface of JSC590R was higher than that of SPCC and that chemisorption with the friction modifier was more advanced. Furthermore, neutron reflectometry showed that the adsorption film thickness of methyl oleate was greater than that of oleic acid on the SPCC surface. In contrast, the oleic acid was thicker than the methyl oleate on the JSC590R surface. This suggested that oleic acid reacted more strongly with the JSC590R surface to form a film that exhibited better lubricity than the SPCC surface. Neutron reflectometry showed the adsorption film thickness and density of both oleic acid and methyl oleate friction modifiers tended to increase for steels with higher concentrations of added alloys. Alloying elements, such as Si and Mn, in steel may affect the adsorption of friction modifiers on the steel surface.
These results clearly show that the friction coefficient in cold rolling depends significantly on the boundary friction coefficient. In particular, this study reveals the combination of steel grade and friction modifier influences rolling lubricity. These are important results for improving tight friction control during the cold rolling of HSS and avoiding problems such as skidding between the work rolls and strips.
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The authors received no financial support for the research, authorship and/or publication of this article.


