Abstract
Background:
Acute kidney injury (AKI) within the intensive care unit (ICU) is common but evidence is limited on longer-term renal outcomes. We aimed to model the trend of kidney function in ICU survivors using estimated glomerular filtration rate (eGFR), comparing those with and without AKI, and investigate potential risk factors associated with eGFR decline.
Methods:
This observational cohort study included all patients aged 16 or older admitted to two general adult ICUs in Scotland between 1st July 2015 and 30th June 2018 who survived to 30 days following hospital discharge. Baseline serum creatinine and subsequent values were used to identify patients with AKI and calculate eGFR following hospital discharge. Mixed effects modelling was used to control for repeated measures and to allow inclusion of several exploratory variables.
Results:
3649 patients were included, with 1252 (34%) experiencing in-ICU AKI. Patients were followed up for up to 2000 days with a median 21 eGFR measurements. eGFR declined at a rate of −1.9 ml/min/1.73m2/year (p-value < 0.001) in the overall ICU survivor cohort. Patients with AKI experienced an accelerated rate of post-ICU eGFR decline of −2.0 ml/min/1.73m2/year compared to a rate of −1.83 ml/min/1.73m2/year in patients who did not experience AKI (p-value 0.007). Pre-existing diabetes or liver disease and in-ICU vasopressor support were associated with accelerated eGFR decline regardless of AKI experience.
Conclusions:
ICU survivors experienced a decline in kidney function beyond that which would be expected regardless of in-ICU AKI. Long-term follow-up is warranted in ICU survivors to monitor kidney function and reduce morbidity and mortality.
Keywords
Introduction
Acute kidney injury (AKI) incidence is high in patients with critical illness, at approximately 40%–60%, compared to 10%–15% of the general hospital population.1 –4 Survival following critical illness is improving but many patients develop long-term morbidity requiring increased healthcare resources following discharge. 5
Following AKI, patients have been demonstrated to be at increased risk of chronic kidney disease (CKD) and worsening of pre-existing CKD, and this risk increases with AKI severity.6 –10
Conventionally, acquisition of long-term renal outcomes for analysis requires studying patients until they develop kidney failure or require dialysis or a kidney transplant. This requires the study of a large cohort as kidney failure events are relatively infrequent, even in high-risk populations. 11 However, eGFR slopes have been verified as a viable surrogate for clinical endpoints in randomised control trials and are a validated tool in assessment of long-term kidney function.12 –14 Emerging evidence supports the use of repeated data measurements, particularly using eGFR as a marker of kidney function, to inform long-term monitoring and follow-up in ICU survivors. 15
Renal injury incurred during ICU may lead to longer-term renal dysfunction, however, evidence is limited. A study by Haines et al. found a rapid initial decline in post-discharge eGFR in ICU survivors followed by a slower decline up to 7 years follow-up. 15 Consequently, the authors advocated for a post-ICU follow-up clinic to assess long-term kidney function and provide intervention to reduce morbidity and mortality. 15 Although the National Institute for Health and Care Excellence (NICE) recommended in their 2023 update that patients who experience in-hospital AKI should have a clinical review within 3 months or sooner if they are deemed to be high risk, 16 historically, actual follow-up rates have varied and were generally low.17 –21 As ICU survivors may present a group at higher risk of poor kidney outcomes in both the short and longer term, it may be more appropriate to identify and prioritise patients at the highest risk of accelerated longitudinal decline in kidney function. Furthermore, precise predictors of outcomes following ICU-associated AKI, as well as post-ICU discharge care, have been identified as areas for further study in this emerging field. 22
This study sought to model the trend of post-ICU discharge kidney function in ICU survivors; to compare post-discharge kidney function in patients who were diagnosed with in-ICU AKI to those who were not; and to investigate potential risk factors associated with kidney function decline.
Methods
This observational cohort study used retrospective analysis of an existing dataset of routinely gathered prospectively acquired data from patients’ ICU admission and subsequent follow-up. 4 Ethics approval was granted by the NHS Health Research Authority, London-Surrey Research Ethics Committee (REC reference: 18/LO/2060).
Patient population
All patients aged 16 or older admitted to two large Scottish general adult ICUs (Glasgow Royal Infirmary and the Queen Elizabeth University Hospital, serving approximately 1.1 million people) between 1st July 2015 and 30th June 2018 were identified. The study population consisted of ICU survivors, determined as patients alive at 30 days following hospital discharge to avoid patients discharged for palliative care from influencing results. Exclusion criteria included long-term kidney replacement therapy (KRT) or prior kidney transplantation. Sample size was determined by number of patients identified over the pre-determined study period.
Data collection
Patients were identified using the Scottish Intensive Care Society Audit Group (SICSAG) Wardwatcher™ database which has capture of all patients admitted to ICU. Patient identifiers were then input into the Strathclyde Electronic Renal Patient Record (SERPR) database (VitalPulse, UK). SERPR has a record of all patients receiving long-term KRT across the West of Scotland as well as active interfaces that automatically retrieve data from laboratory, radiology and death records. This includes historical laboratory data from 01/01/2009 onwards, ensuring important outcomes were available for this study. Full details are described in previously published work on the same patient cohort. 4 Patients without available eGFR data following discharge were considered lost to follow-up and excluded from all analyses.
Follow-up data were retrieved from routinely gathered eGFR measurements at subsequent hospital or general practice appointments. eGFR measurements from the 30 days following hospital discharge were not included to mitigate any acute effects of illness and ICU on kidney function. However, it should be noted that the reason for collection of the eGFR measurements is unknown, so although measurements within 30 days of discharge were not included, subsequent measurements could have been taken at a point of acute illness and eGFR therefore may not have stabilised.
Baseline demographic variables including age, sex, admitting specialty, APACHE II score, admission diagnosis and type of organ support were retrieved from the SICSAG database. Admitting specialty was categorised as medical or surgical. Admission diagnoses were organised into groups according to coding in the SICSAG database and diagnoses such as sepsis were grouped regardless of causative pathogen. Organ support was categorised based on receipt of invasive mechanical ventilation, vasopressor support (infusion of vasoactive medication including vasopressors or inotropes) and KRT. Pre-existing comorbidities were identified using data in ICU electronic patient records and grouped according to cardiovascular disease (including chronic hypertension and ischaemic heart disease), respiratory disease (including obstructive airway disease and interstitial lung disease), liver disease (including alcoholic and non-alcoholic fatty liver disease, hepatitis or cirrhosis) diabetes mellitus (of all types) and cancer (all types and stages).
AKI determination
Pre-admission serum creatinine results were used to calculate a baseline value for each patient. Either the median value from 8 to 365 days before admission, or, if these data were unavailable, the lowest value in the week prior to admission was used. Patients without data available for classification of baseline kidney function were excluded. This serum creatinine value was used to calculate baseline eGFR using the CKD-EPI equation. 23 This reference value was then used to diagnose AKI during ICU admission using Kidney Disease Improving Global Outcomes (KDIGO) 2012 classification. 21 The initial injury was identified as the point at which AKI criteria were met for the first time and severity of AKI was classified using the highest creatinine value from the duration of injury. Patients who received KRT whilst in ICU were automatically classified as having AKI stage 3. AKI was classified using only serum creatinine values due to a lack of urine output data. Patients who were readmitted to ICU during the study period were not re-evaluated for AKI on their second or any subsequent admissions.
Statistical analysis
Statistical analysis was conducted using the statistical software R (The R Foundation; R version 4.2.2).
For baseline demographics, continuous variables were summarised using median values and interquartile range (IQR) and compared using Wilcoxon rank sum test. Categorical variables were summarised using proportions with 95% confidence intervals (95%CI) and compared using Pearson’s Chi-squared test.
Mixed effects modelling was used throughout determination of eGFR trend to control for repeated measures and missing data, and to allow inclusion of several exploratory variables. The dataset was capped at a maximum follow-up of 2000 days due to increased variability in eGFR measurements after this due to significantly fewer patients having follow-up data available after this time point.
When investigating potential risk factors associated with kidney function decline, the complete dataset was divided into two cohorts: patients who experienced in-ICU AKI and those who did not. Initial unadjusted models were built for each variable in each cohort and univariable p-values < 0.1 were included in the multivariable adjusted models. Two adjusted models for each cohort were created: one investigating the effect of pre-ICU variables and one investigating the effect of in-ICU variables on longitudinal kidney function. Multivariable p-values < 0.05 were regarded as statistically significant.
Results
3649 patients met the inclusion criteria (Figure 1) with 134,038 eGFR measurements (median 21 per patient). The minimum and maximum follow up times were 6 days and 2000 days respectively (median 1229 days). Main causes for ICU admission were sepsis (20.0%), malignancy (13.0%), gastrointestinal disease (excluding perforation; 8.7%) and trauma (8.1%). Of the 3649 patients, 1252 (34.3%) experienced AKI whilst in ICU. Patients who experienced AKI were older (median age of 60 compared to 56); had a higher APACHE II score (19 compared to 12); a lower baseline eGFR (83 ml/min/1.73m2 compared to 95 ml/min/1.73m2); a lower calculated eGFR on discharge (78 ml/min/1.73m2 compared to 102 ml/min/1.73m2); and there was a higher proportion with a past medical history (PMH) of cardiovascular disease, liver disease and diabetes (Table 1).
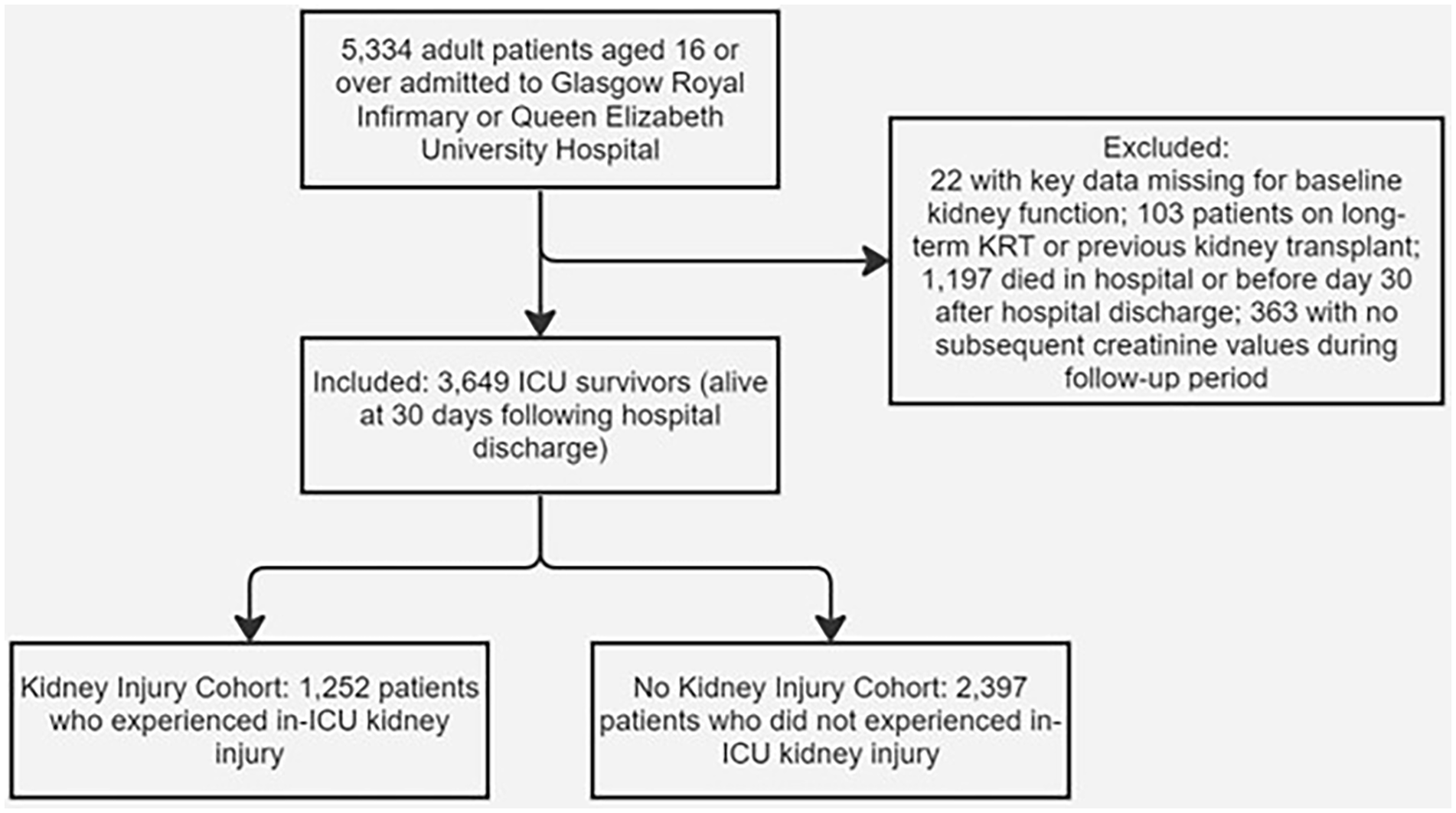
Generation of the final cohort included in analysis.
Baseline patient demographics grouped according to kidney injury in ICU.
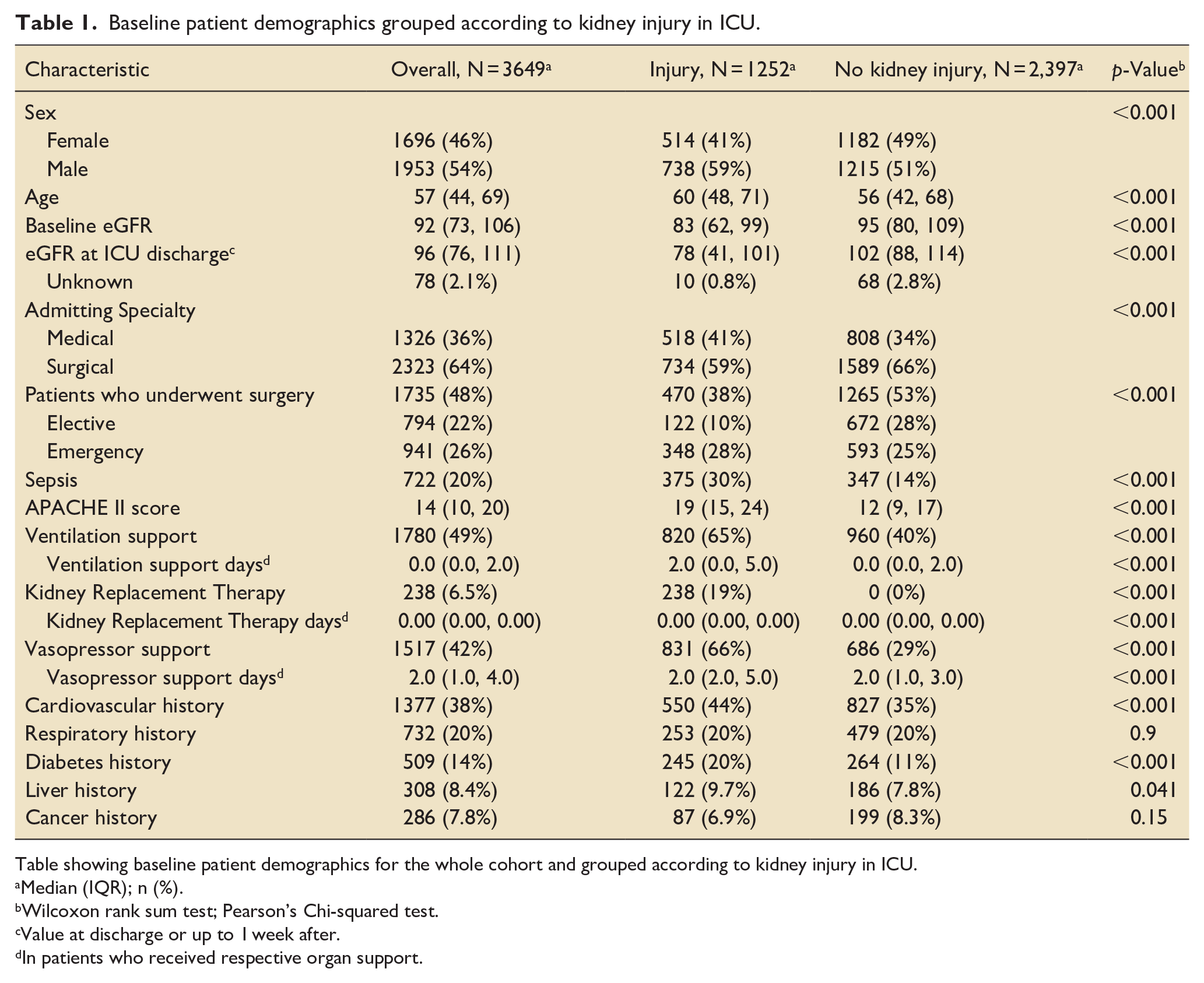
Table showing baseline patient demographics for the whole cohort and grouped according to kidney injury in ICU.
Median (IQR); n (%).
Wilcoxon rank sum test; Pearson’s Chi-squared test.
Value at discharge or up to 1 week after.
In patients who received respective organ support.
In the cohort of ICU survivors with AKI, 37%, 20% and 43% of patients experienced AKI stages 1, 2 and 3 respectively.
Renal function in ICU survivors
Initial linear mixed effects modelling of all ICU survivors identified a decline in eGFR over 2000 days at a rate of −1.9 ml/min/1.73m2/year (p-value < 0.001). After adjustment for age, a decline in eGFR was observed at a rate of −1.28 ml/min/1.73m2/year (p-value < 0.001; Table 2).
Summary of models built to explore eGFR rate of decline and level in the whole patient cohort and with interaction for kidney injury.
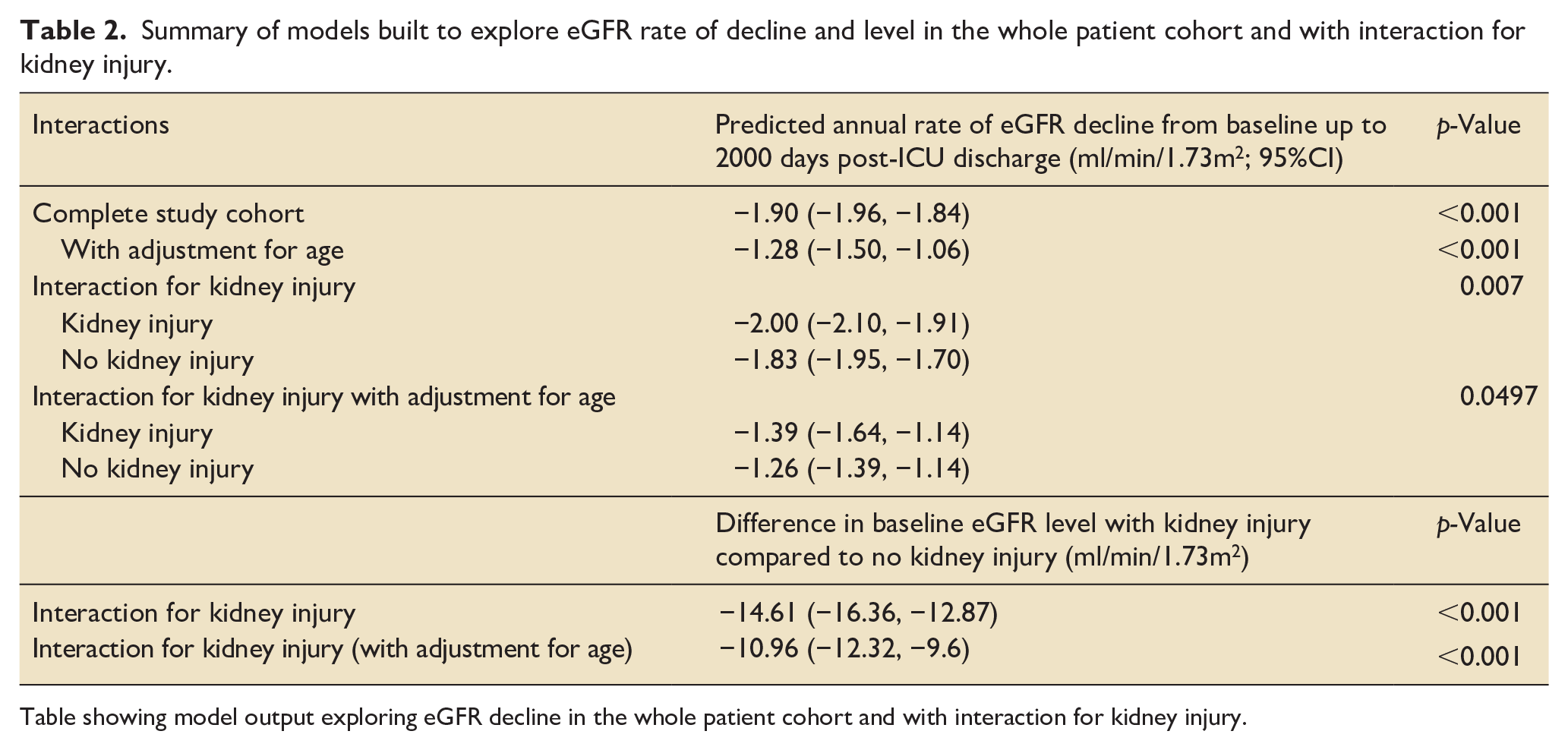
Table showing model output exploring eGFR decline in the whole patient cohort and with interaction for kidney injury.
eGFR declined at a rate of −2.00 ml/min/1.73m2/year in patients who experienced in-ICU AKI, compared to −1.83 ml/min/1.73m2/year in patients who did not (p-value = 0.007 for interaction; Table 2). Comparison of the two eGFR slopes can be viewed in Figure 2.
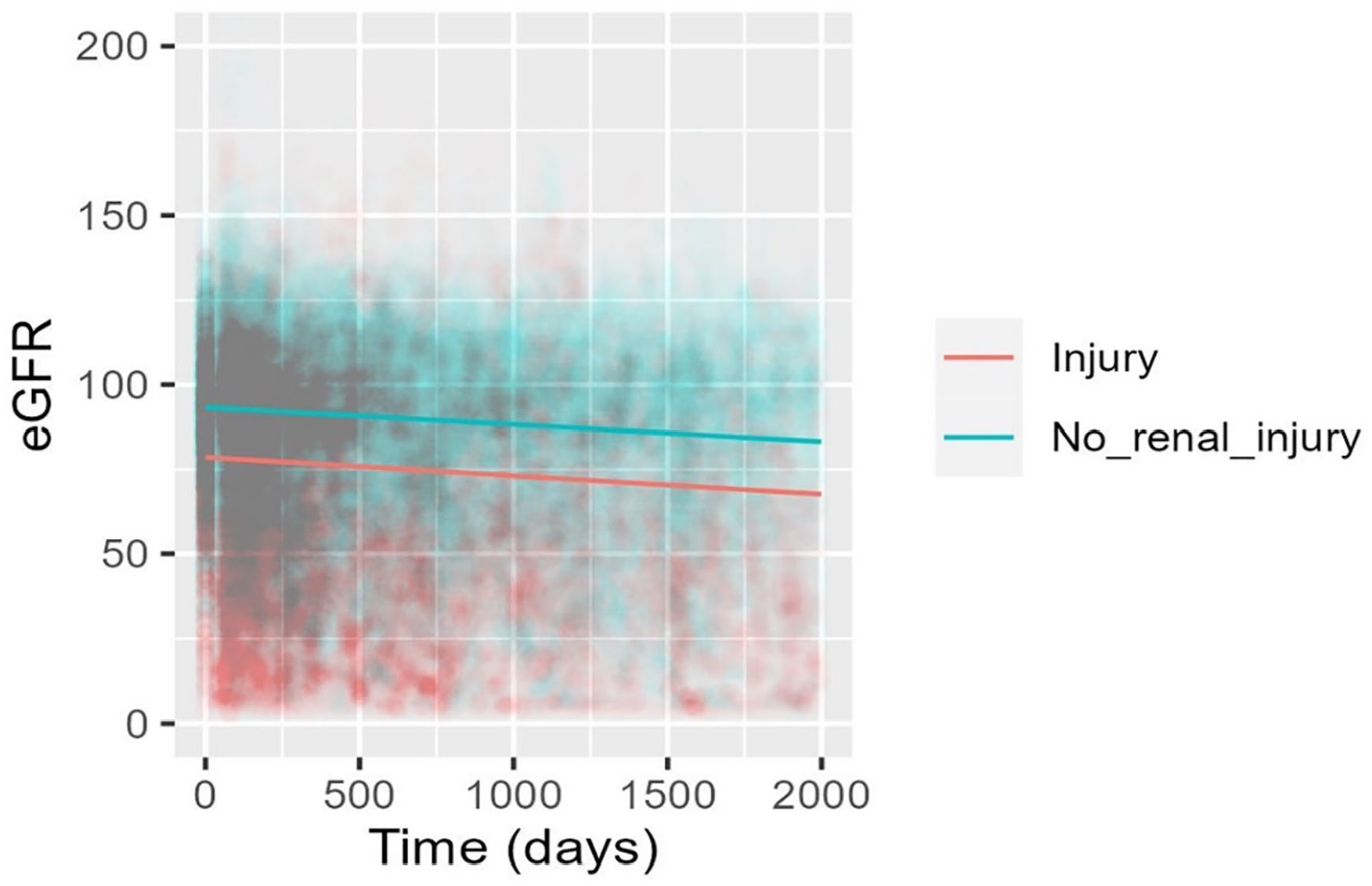
Predicted eGFR by in-ICU kidney injury, adjusted for age (years).
After adjustment for age, the eGFR slope declined at a rate of −1.39 ml/min/1.73m2/year in patients who experienced AKI compared to −1.26 ml/min/1.73m2/year in patients who did not (p-value = 0.0497).
Identifying risk factors in patients without AKI
Factors associated with accelerated eGFR decline in patients without AKI in ICU are summarised in Table 3 and Supplemental Table 1.
Results of univariable and multivariable modelling for association between in-ICU variables and post-discharge eGFR results in patients who did not experience kidney injury in ICU.
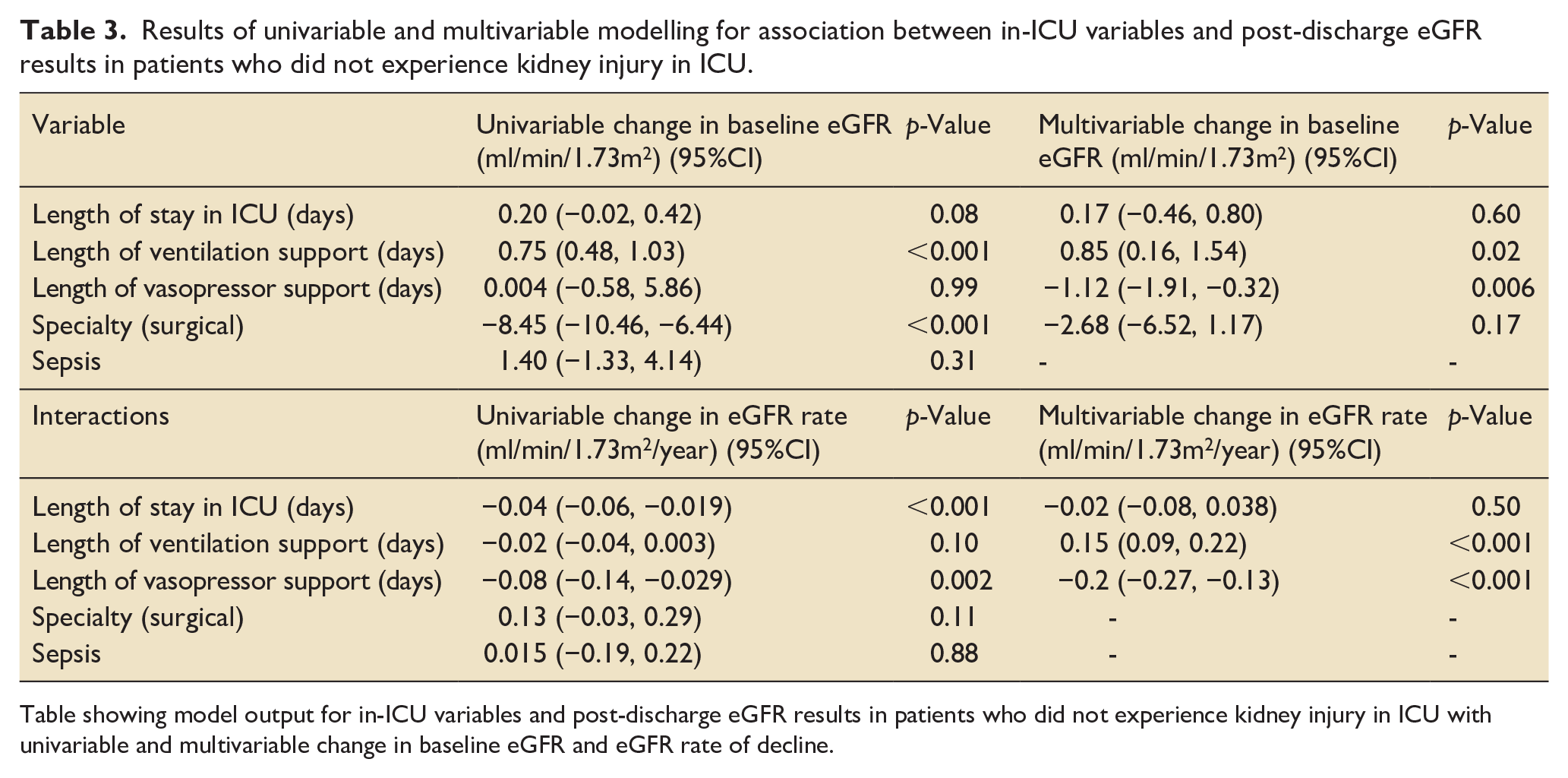
Table showing model output for in-ICU variables and post-discharge eGFR results in patients who did not experience kidney injury in ICU with univariable and multivariable change in baseline eGFR and eGFR rate of decline.
Pre-ICU variables
Pre-ICU variables associated with an increased rate of eGFR decline following ICU discharge included pre-existing diabetes (−0.53 ml/min/1.73m2/year, p-value < 0.001) and pre-existing liver disease (−2.30 ml/min/1.73m2/year, p-value < 0.001). A history of respiratory disease was associated with a reduced decline in eGFR following ICU discharge of 0.27 ml/min/1.73m2/year (p-value = 0.004).
In-ICU variables
Length of vasopressor support was associated with an accelerated annual rate of eGFR decline of −0.20 ml/min/1.73m2/year per day of vasopressor support (p-value < 0.001). Length of ventilation support was associated with a reduced annual rate of decline in eGFR by 0.15 ml/min/1.73m2/year per day of support (p-value < 0.001).
Identifying risk factors in patients with AKI
Results of the univariable and multivariable models for patients with AKI are summarised in Supplemental Tables 2 and 3.
Pre-ICU variables
Pre-existing conditions associated with an increased annual rate of eGFR decline were: cardiovascular disease (−0.64 ml/min/1.73m2/year, p-value < 0.001); liver disease (−0.68 ml/min/1.73m2/year, p-value < 0.001); diabetes (−1.75 ml/min/1.73m2/year, p-value < 0.001); and cancer (−0.75 ml/min/1.73m2/year, p-value = 0.001).
A history of respiratory disease was associated with a reduced annual rate of eGFR decline of 0.39 ml/min/1.73m2/year (p-value = 0.004).
In-ICU variables
AKI stage was associated with an increased annual rate of eGFR decline of −1.21 ml/min/1.73m2/year for stage 2 and −1.85 ml/min/1.73m2/year for stage 3 compared to stage 1 (p-value < 0.001 for both).
With regards to organ support, length of ventilation support was associated with a reduced rate of eGFR decline by 0.09 ml/min/1.73m2/year per day of support. Length of receipt of KRT was also associated with a reduced rate of eGFR decline by 0.11 ml/min/1.73m2/year per day of support (p-value < 0.001 for both). There was also a significant reduction in baseline eGFR of −0.50 ml/min/1.73m2 per day of KRT (p-value = 0.04).
Length of vasopressor support was associated with an accelerated annual rate of decline in eGFR by −0.06ml/min/1.73m2/year (p-value < 0.001) per day of support.
Finally, sepsis was associated with a reduced annual rate of eGFR decline of 1.54 ml/min/1.73m2 (p-value < 0.001).
Discussion
This study of ICU survivors demonstrated an annual decline in eGFR beyond what would be expected due to age alone.24 –28 While previous studies have suggested the annual decline in eGFR would be expected to be between 0.6–0.9 ml/min/1.73m2 due to a year’s increase in age,24 –28 ICU survivors experienced a decline of −1.9 ml/min/1.73m2/year. Anticipating that some of this decline may be age-related, age adjusted analysis demonstrated eGFR decline in ICU survivors of −1.28 ml/min/1.73m2, a difference of 0.62 ml/min/1.73m2, consistent with expected age-related decline. These results are consistent with the findings of Haines et al. that ICU survivors experience continued decline in kidney function in the years following ICU discharge. 15
AKI incidence in our study was found to be 34.3% which is lower than the 40%–60% in-ICU AKI incidence stated in literature.1 –4 Although critical care populations are heterogenous, the lower incidence found in our study is likely due to exclusion of patients who did not survive up to 30 days following ICU discharge from the study population. As AKI is associated with high mortality, it is likely that in-ICU AKI incidence in our cohort was higher.
In contrast to the findings of Haines et al. who noted renal decline to be similar in patients who experienced in-ICU AKI to those who did not, this study observed an accelerated longitudinal rate of eGFR decline in ICU survivors who experienced in-ICU AKI compared to those who did not. 15 However, although the difference in eGFR decline between the two cohorts was statistically significant, an increased annual rate of decline of −0.13 ml/min/1.73m2 (accounting for age) in patients who experienced AKI compared to those who did not is not clinically significant and falls below the rate of eGFR slope decline which is considered significant in CKD trials. 12 More relevant perhaps, is the finding that ICU survivors experienced an accelerated rate of eGFR decline in the years following discharge, above that which is attributable to ageing alone, regardless of in-ICU AKI experience. This suggests that ICU survivors are at risk of accelerated decline in kidney function warranting dedicated follow-up regardless of AKI experience in ICU.
This study also explored factors associated with accelerated kidney function decline. Existing factors associated with accelerated eGFR decline following ICU-discharge in all patients included a past medical history of diabetes or liver disease. Diabetes mellitus is a known risk factor for CKD and patients with diabetes are more likely to develop end stage kidney disease (ESKD). 29 The association between liver and kidney disease has also long been known, with hepatorenal syndrome a clinical example of this, 30 and a recent meta-analysis found that non-alcoholic fatty liver disease (NAFLD) was associated with a moderately increased risk of CKD stage⩾ 3, according to KDIGO classification. 31 Therefore, it is unsurprising that we found pre-existing diabetes or liver disease to be associated with accelerated decline in post-ICU kidney function.
A history of malignancy was associated with an accelerated rate of eGFR decline in the patient cohort who experienced in-ICU AKI. This could be a result of nephrotoxic chemotherapeutic agents used in some cancer treatment making the kidneys susceptible to greater damage following in-ICU injury.32,33 However, not all patients with cancer will have received chemotherapy or nephrotoxic treatment so further research is required before drawing more definitive conclusions.
Cardiovascular disease was not associated with any changes in eGFR slope in the no AKI cohort, however, was associated with an increased rate of decline in the AKI cohort. Cardiovascular and kidney disease are known to be linked, with cardiovascular factors including hypertension increasing CKD risk.34,35 As such, the fact that there was no association in the no AKI cohort is surprising. Liver and kidney disease share many cardiovascular and metabolic risk factors meaning these comorbidities may account for some of the expected association seen in cardiovascular disease.35,36
The fact that pre-existing respiratory disease was associated with a decreased rate of eGFR decline in both cohorts is surprising and there is no current physiological explanation for this. The statistically significant difference in eGFR is very small however, and likely not clinically relevant. A possible explanation for this finding is that the statistical significance could be a random observation.
This study also explored factors related to ICU stay on subsequent kidney function. Increasing length of vasopressor support was associated with an increased rate of eGFR decline. This association could be as great as an accelerated decline of −1.4 ml/min/1.73m2/year in a patient who did not experience AKI and received a week of vasopressor support. This is highly clinically significant and identifies a group of ICU survivors at potentially greater risk of long-term accelerated kidney function decline.
Vasopressors are routinely used in ICU in the management of conditions such as vasodilatory shock, following cardiovascular surgery and after myocardial infarction. 37 Adverse effects of vasopressors include excessive vasoconstriction which can result in organ under-perfusion, with the kidneys being particularly vulnerable due to their dense microvasculature. 37 Increasing duration of vasopressor support could therefore increase the risk of kidney under-perfusion and subsequent damage, resulting in accelerated eGFR decline.
In patients who experienced AKI, length of receipt of KRT was associated with a reduced rate of eGFR decline. This is contrary to findings by Soum et al. who found increasing length of receipt of KRT to be a risk factor for developing severe, long-term CKD in critically ill patients requiring KRT for AKI treatment. 38 One explanation for our findings is that there was a significant difference in baseline eGFR of −0.50 ml/min/1.73m2 per day of KRT suggesting that patients who require KRT in ICU have a lower pre-ICU baseline eGFR. Therefore, this could result in a relative reduction in eGFR decline compared to patients with a higher baseline, explaining the observed protective effect.
Length of ventilatory support was associated with a reduced rate of eGFR decline in both cohorts. One possible explanation for this could be that muscle wasting as a result of increased duration of ventilatory support could have resulted in lower serum creatinine measurements and therefore overestimation of eGFR resulting in a reduced rate of eGFR decline.
An in-ICU diagnosis of sepsis was associated with a reduced rate of eGFR decline in patients who experienced AKI, although the difference was negligible. Again, this is surprising as sepsis is a common cause of AKI and is associated with renal vasoconstriction and reduced oxygenation which can impair kidney function.39,40 Sepsis is associated with significant morbidity and mortality, 40 therefore, this observed association could be explained by survivor bias with surviving patients more likely to have been in better health prior to ICU admission and therefore suffering from less consequent kidney damage.
Implications of our findings
Our finding of accelerated eGFR decline in ICU-survivors concurs with the conclusion made by Haines et al. that long term follow-up of renal function, potentially through community eGFR monitoring, is warranted in ICU-survivors to reduce the morbidity and mortality associated with CKD. 15 However, follow-up of all ICU-survivors may not be feasible.17,18 Therefore ICU-survivors should be risk stratified according to pre-existing comorbidities and length of vasopressor support to prioritise those at greatest risk.
Strengths and limitations
A strength of this study was that it utilised a large dataset likely sufficiently powered for the statistical analyses undertaken. Patients included were from two centres with a mixed general ICU population and can likely be considered representative of the UK critical care population. 15 However, AKI is a syndrome with high heterogeneity with regards to causes and not all phenotypes will follow the same disease pattern. Furthermore, the use of mixed effects modelling accounted for repeated measures, missing values and several exploratory variables.
Limitations of this study include those associated with retrospective study design and these results should be viewed as exploratory rather than explanatory. Furthermore, the eGFR equation is not validated in patients under the age of 18, but due to patients aged 16–17 being occasionally admitted to the general adult ICUs investigated in this study, 19 patients in this age group were included. However, these patients are likely to exhibit adult physiology and as only 19 patients were included this is unlikely to have influenced results. Patients who were dialysis dependent on hospital discharge were also included. eGFR calculation is not representative of kidney function in those who are dialysis dependent, however, due to the relatively small number of patients in this group (n = 17), these eGFR measurements are also unlikely to have influenced results.
A lack of urine output data to determine in-ICU AKI likely resulted in underestimation of AKI incidence. Furthermore, inclusion of patients without serum creatinine readings prior to the week before admission and using lowest serum creatinine in the week before admission might have resulted in inaccuracies as this value could have already be influenced by illness and therefore not represent a true baseline.
The CKD-EPI equation used to calculate eGFR solely used serum creatinine as a measure of kidney function. Serum creatinine is influenced by factors including muscle mass which can fall during ICU admission resulting in overestimation of kidney function. 41 This could have particularly affected the AKI patient group due to AKI being associated with increased morbidity and therefore likely increased muscle wasting, as well as any patients who underwent limb amputation. Therefore, patients in these groups could have experienced greater longitudinal decline in kidney function than our results show. Although, muscle mass is usually regained to some extent following ICU discharge so therefore effects could be smaller in those with longer follow up. Future studies could utilise Cystatin C measurement as a biomarker for kidney function as it is not associated with muscle mass, to greater reflect kidney function in these patient groups. 41 Alternatively, proteinuria could be used as a marker of kidney damage and predictor of CKD progression.
eGFR follow-up data were gathered from routinely collected measurements. As follow-up of kidney function tends to occur more often in patients with worse renal function, 17 this introduces potential bias. Moreover, due to difficulties with model convergence, two final multivariable models were built for each patient cohort. This means that any interaction between pre-ICU and in-ICU variables hasn’t been accounted for in the two separate models. Finally, as many of the variables investigated are inherently linked, potential confounding makes interpretation of results more uncertain.
Conclusion
This study found that ICU survivors experienced accelerated decline in kidney function represented by eGFR slopes. Whilst patients with in-ICU AKI suffered an accelerated rate of eGFR decline compared to those without in-ICU AKI, all patients suffered accelerated rate of eGFR decline compared to that expected by age alone. Pre-existing comorbidities, AKI stage and increasing length of vasopressor support were associated with accelerated eGFR decline. Long-term follow-up is warranted in high-risk ICU survivors to monitor kidney function and reduce morbidity and mortality associated with CKD. Further work is required to help risk stratify ICU survivors at highest risk of accelerated decline in kidney function and determine feasibility of more widespread follow-up of kidney function.
Supplemental Material
sj-docx-1-inc-10.1177_17511437241308673 – Supplemental material for Longitudinal trend in post-discharge estimated glomerular filtration rate in intensive care survivors
Supplemental material, sj-docx-1-inc-10.1177_17511437241308673 for Longitudinal trend in post-discharge estimated glomerular filtration rate in intensive care survivors by Rebecca M Glendell, Kathryn A Puxty, Martin Shaw, Malcolm AB Sim, Jamie P Traynor, Patrick B Mark and Mark Andonovic in Journal of the Intensive Care Society
Footnotes
Acknowledgements
The authors would like to acknowledge the NHS Health Research Authority, London-Surrey Research Ethics Committee who gave ethics approval for this research.
Authors’ contributions
MA, MS, MABS, PM, JT and KP contributed to the conception and design of the work. RG performed statistical analysis and initial drafting of the manuscript with subsequent input from MA, MS, MABS, PM, JT and KP. All authors were responsible for interpretation of the data, decision to submit for publication and have approved the final version of this manuscript.
Availability of data and materials
The data that support the findings of this study are available on request from the corresponding author, [RG]. The data are not publicly available due to their containing information that could compromise the privacy of research participants.
Ethics approval and consent to participate
Ethics approval was granted by the NHS Health Research Authority, London-Surrey Research Ethics Committee (REC reference: 18/LO/2060). The study utilised data routinely gathered during patients’ ICU admission and follow-up period therefore individualised participant consent was not sought.
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: PM reports lecture honoraria from Astrazeneca, Pharmacomsos, Astellas, GSK and Boehringer Ingelheim outside the submitted work. The authors declare no other potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
