Abstract
Background:
The formation of anastomoses between the pulmonary arteries and pulmonary veins, or the pulmonary and the bronchial circulation, is part of normal foetal lung development. They persist in approximately 30% of adults at rest, and open in almost all adults during exertion. Blood flowing through these anastomoses bypasses the alveolar surface and increases in such shunting can thus cause hypoxaemia. This is now known to contribute to the pathogenesis of hypoxaemia in COVID-19 disease. We here provide evidence to support a similar role in influenza A infection.
Illustrative case presentation:
We describe a case of influenza A infection associated with severe hypoxaemia, poorly responsive to supplemental oxygen and which worsened following the application of continuous positive airway pressure (CPAP), despite the presence of a normal physical examination, chest radiograph and echocardiogram. This combination suggests a significant intrapulmonary (extra-alveolar) shunt as a cause of the severe hypoxaemia. The shunt fraction was estimated to be approximately 57%.
Discussion and conclusion:
Intrapulmonary vascular shunts can contribute substantially to hypoxaemia in viral infection. Seeking to understand the pathogenesis of observed hypoxaemia can help guide respiratory therapy. Mechanistic research may suggest novel therapeutic targets which could assist in avoiding intubation and mechanical ventilatory support.
Case presentation
A 46-year-old female presented to the Emergency Department (ED) with a 1-day history of a non-productive cough, fever and profound lethargy following recent contact with her parents, who were suffering ‘flu-like’ symptoms but who had tested (lateral flow) negative for Covid-19.
She had primary progressive multiple sclerosis and used a wheelchair to mobilise. She was taking rivaroxaban as treatment for a pulmonary embolism, diagnosed 7 months earlier.
On presentation to ED, she was normotensive (129/70 mmHg), but tachycardic (109 beats/min), febrile (38.5°C) and hypoxaemic (oxygen saturation on pulse oximeter (SpO2) only maintained at 99% with 15 L oxygen delivered via non-rebreathe face mask): a paramedic ambulance crew had found her to be tachypnoeic and cyanosed, and the oxygen they administered was being continued. On examination, the patient had a respiratory rate of 32 breaths/min with high respiratory excursion, consistent with a high tidal volume and therefore high minute volume. Chest expansion, percussion and auscultation were all normal. There were no clinical features of deep venous thrombosis, the patient confirmed that she had missed no doses of her oral anticoagulant, she had not suffered pleuritic chest pain and there were no pleural rubs. Twelve-lead electrocardiography revealed only sinus tachycardia and chest radiography was completely normal. White cell count was 16.8 × 109/L (neutrophils 16.1 × 109/L), CRP 25 mg/L. The remaining biochemistry and blood counts were within normal range.
She tested negative for SARS-CoV-2 using a point-of-care isothermal amplification, then Reverse Transcription Polymerase Chain Reaction amplification (RT-PCR).
A possible occult bacterial chest infection was treated with antibiotics (Piperacillin with Tazobactam 4500 mg TDS) and steroids (hydrocortisone 100 mg IV stat, then two 40 mg doses of oral prednisolone for 2 days). Oral oseltamivir (75 mg BD) was administered, and oral anticoagulation was continued.
Six hours following admission to the ward her clinical condition worsened. SpO2 was 89% despite administration of 15 L/min O2 via a non-rebreathe mask, with a partial pressure of oxygen in arterial blood (PaO2) of 7.7 kPa (57.75 mmHg; normal 11.0–14.4 kPa (82.5–108.0 mmHg)). SpO2 was independent of posture (i.e. sitting vs lying). Bicarbonate and lactate concentrations, base excess and carboxyhaemoglobin and methaemoglobin proportions were all normal. PaO2 continued to fall to 7.4 kPa (55.7 mmHg) despite initiation of high flow nasal oxygen (HFNO; 60 L/min, FiO2 0.8). A trial of continuous positive airway pressure (CPAP) to improve oxygenation was quickly discontinued as, despite good patient compliance and the mask being well-fitted, SpO2 decreased to 80% (rebounding back to 90% upon CPAP cessation). The patient’s blood pressure remained unchanged during the trial of CPAP.
Worsening oxygenation with the therapeutic trial of CPAP and the limited improvement following supplemental oxygen suggested the presence of an intra-cardiac or intra-pulmonary vascular shunt. Using the values above, (PaO2/FiO2 ratio: 69.6), and with reference to Reske et al., 1 shunt fraction on HFNO was estimated to be circa 56.7% (75% prediction interval: 41.2–71.1).
Transthoracic echocardiography revealed normal right ventricular dimensions and function, with normal pulmonary artery pressures. Contrast echocardiography, performed with agitated saline and a brief (due to dyspnoea) Valsalva manoeuvre, revealed no evidence of right-left intra-cardiac shunting.
In terms of the cause of hypoxia, pulmonary embolus was unlikely, given the history, sustained use of anticoagulants, lack of evidence of lower limb deep vein thrombosis, absence of rubs and absence of clinical/electrocardiographic/echocardiographic right heart strain; pulmonary oedema was likewise excluded clinically and through imaging, as was extensive alveolar consolidation; a significant right-to-left cardiac shunt was excluded by echocardiography. Platypnea-orthodeoxia (low oxygen saturation and dyspnoea in the upright posture, improved by recumbent positioning) can be caused by cardiac shunt, but was absent. It was concluded that the hypoxia was substantially driven by an intrapulmonary vascular shunt and on that basis, and given the detrimental response to CPAP, intubation and mechanical ventilation were not instigated.
The following morning, respiratory viral swabs were reported to be negative for respiratory syncytial virus (RSV) and parainfluenza, but positive for influenza A. By this time, her oxygen requirement had reduced to 0.45 via HFNO set at 60 L/min, resulting in a PaO2 13.0 kPa (97.5 mmHg). The following day, her oxygenation improved further, and she was supported only with 1 L/min O2 via nasal cannula with SpO2 of 96%. Five days later (a week after hospital admission), she had made a complete recovery and was discharged home.
Discussion
We describe a case of influenza A infection associated with severe hypoxaemia – worsening following the application of CPAP – despite the presence of a normal physical examination, chest radiograph and echocardiogram. This combination suggests a significant intrapulmonary (extra-alveolar) shunt as a cause of the severe hypoxaemia.
Diagnostic approach
Pulmonary infection can cause hypoxaemia due to the presence of inflammatory lung oedema and loss of aeration (e.g. atelectasis and consolidation). The oxygen content of the mixed venous blood perfusing poorly-ventilated or consolidated alveolar units remains largely unchanged flowing through the lung, and this low ventilation: perfusion (V:Q) ratio causes hypoxaemia, given that the normally ventilated and perfused areas of the lung cannot compensate for the alveolar units at very low V:Q. The estimated shunt fraction (the proportion of cardiac output or pulmonary blood flow perfusing non-ventilated lung units) can also be greater than expected based on the extent of alveolar aeration loss, when inflammation blunts the normal mechanisms of hypoxic vasoconstriction (HPV), 2 or even when it causes paradoxical pulmonary arteriolar vasodilatation. 3 These vascular changes lead to a further increase in perfusion of hypo/non-ventilated alveolar units and a further increase in the shunt fraction and consequent hypoxaemia, which becomes disproportionate to the anatomical shunt of diseased lung. While computed tomography (CT) was not performed, a completely normal physical lung examination and chest radiography was apparently incompatible with the lack of response to supplemental oxygen. Furthermore, a normal haemodynamic state makes severe hypoxaemia due to low central venous oxygen saturation (ScvO2) unlikely. In addition, echocardiography and electrocardiography were reported as normal, and there were no clinical features of pulmonary oedema or intracardiac shunt. A new acute pulmonary embolism was considered unlikely, as there was no evidence of lower limb oedema, pulmonary hypertension or RV pressure overload. Additionally, the patient was fully concordant with oral anticoagulation.
The diagnostic approach for detecting intracardiac and intrapulmonary shunts typically involves contrast echocardiography, where agitated saline or contrast medium is injected to visualise bubble passage. Provocation tests, such as the Valsalva manoeuvre or cough, are used to increase right atrial pressure above left atrial pressure, enhancing shunt detection. The ‘three to six rule’ is a helpful rule which states that bubbles appearing in the left atrium within three cardiac cycles after appearing in the right atrium suggest an intracardiac shunt (like a patent foramen ovale), while bubbles appearing after three to six cardiac cycles suggest an intrapulmonary shunt. These echocardiographic signs together with the gas exchange and examination can aid differential diagnosis of hypoxaemia unrelated to an alveolar pathology. 4 Unfortunately, as we were only able to perform a bedside echocardiogram, with a brief Valsalva manoeuvre in the context of marked tachypnoea, it was not possible to confirm the presence or timing of pulmonary venous bubbles.
Given that more common causes of hypoxaemia had been reasonably excluded, the most plausible mechanism was a form of extra-cardiac, intra-pulmonary, but extra-alveolar, right-to-left vascular shunting. This is the only mechanism able to cause a significant uncoupling between the observed shunt fraction (e.g. hypoxaemia) and the lack of parenchymal lung or heart disease. In other words, there should be an anatomical mechanism – triggered by an inflammatory stimulus – which led to desaturated venous blood joining the systemic circulation in the bronchopulmonary anatomy without perfusing the alveoli, which are otherwise normally aerated and ventilated.
Background
These are anastomoses either between the pulmonary arteries and pulmonary veins or the pulmonary and the bronchial circulation. In both cases lung precapillary anastomoses open and directly connect the pulmonary arterial and the venous circulation, or the pulmonary and systemic circulation, so that pulmonary blood bypasses the alveoli and therefore does not contribute to gas exchange, resulting in shunt and hypoxaemia. Such shunts typically respond poorly to supplemental oxygen, given that deoxygenated venous blood is being ‘bypassed’ around alveolar units. 5
Intrapulmonary anastomotic shunts between pulmonary arteries and veins are part of the normal development of the foetal lung and contribute to the normal alveolar growth. 6 There is also increasing evidence that these anastomoses persist in ~30% of adults at rest 7 and in almost all adults during exertion, 8 where they contribute to increased shunting and widening alveolar-arterial gradient. Such a dramatic contribution of intrapulmonary shunting, as seen in this case, has not been previously reported in a patient with influenza A.
Intrapulmonary anastomotic shunting in Covid-19
These anastomoses may become more widely prominent in pathological conditions when cardiac output increases (e.g. fever, inflammation, sepsis) or when increased respiratory effort or positive airway pressure (e.g. CPAP as in our case) cause an increase pulmonary arterial pressure and lead to a higher proportion of pulmonary blood flow passing through these anastomoses, which may function as a ‘pop-off mechanism’ to limit further increases in pulmonary arterial pressure. Microvascular pulmonary thromboses are identified in 57% of patients who died from COVID-19 and 24% who died from H1N1 Influenza A, 9 and these might ‘open up’ (normal anatomical and physiological) intrapulmonary arteriovenous anastomoses (IPAVAs) by raising flow and pressure in other vascular units, 10 in a similar manner to that which occurs during exercise.8,11 Vasoconstriction in other small pulmonary vessels might have a similar impact on other less-affected lung units. 12
Such a mechanism of hypoxaemia was initially hypothesised in patients with COVID-19, where a lack of relationship between PaO2/FiO2 and the quantity of non-aerated lung tissue was demonstrated. 13 This mechanism was subsequently confirmed to play an important role in the pathogenesis of hypoxaemia in COVID-19 disease. 14 Vascular perfusion abnormalities and increased vascularity and congestion of lung parenchyma and their relationship with hypoxaemia and outcomes have since been confirmed by many authors. 15 A more direct anatomical evidence of intrapulmonary bronchopulmonary anastomoses (IBAs) as a potential source of right-to-left shunt with profound hypoxaemia was described by Galambos et al. 16 Functionally, these anastomoses have been demonstrated by the delayed detection of microbubbles in the left ventricle and arterial circulation following venous injection of agitated saline,17,18 which supports the presence of large anatomical vascular shunts, 10 as such saline bubbles are too large to cross the pulmonary vasculature. 19 Like IPAVAs, IBAs are not normally perfused but can be recruited in response to an increase in pressure or blood flow20,21 and contribute to hypoxaemia in Covid-19 and in other inflammatory lung pathologies.16,22,23 Histopathological examination of the lungs of three patients with Covid-19 demonstrated prominent IBAs in all. 16 It has also been shown that the effect on hypoxaemia can be amplified by neovascularisation induced by inflammation. 24
The total amount of oxygen delivered to the pulmonary arteries and flowing through the lung is the product of the Oxygen content of the mixed venous blood (CvO2) and the pulmonary blood flow (Qt) which is equivalent to cardiac output. The arterial oxygen delivery is product of the Oxygen content of the arterial blood (CaO2) and the cardiac output. The CaO2 is the weighted average of the blood flowing to non-ventilated alveolar units (shunted blood) which is equivalent to the Oxygen content of the mixed venous blood (CvO2) multiplied by the proportion of the blood flowing to non-ventilated alveolar units (i.e. shunt fraction, Qs/Qt) (1 in the Figure 1) + the Oxygen content of the mixed venous blood that, after perfusing ventilated alveolar units becomes fully oxygenated (end-capillary blood, CcO2) multiplied by the non-shunted proportion of blood flow (1 − Qs/Qt) (2 in the Figure 1). The higher the number of the non-ventilated units, the higher the shunt fraction, the lower the arterial oxygenation.
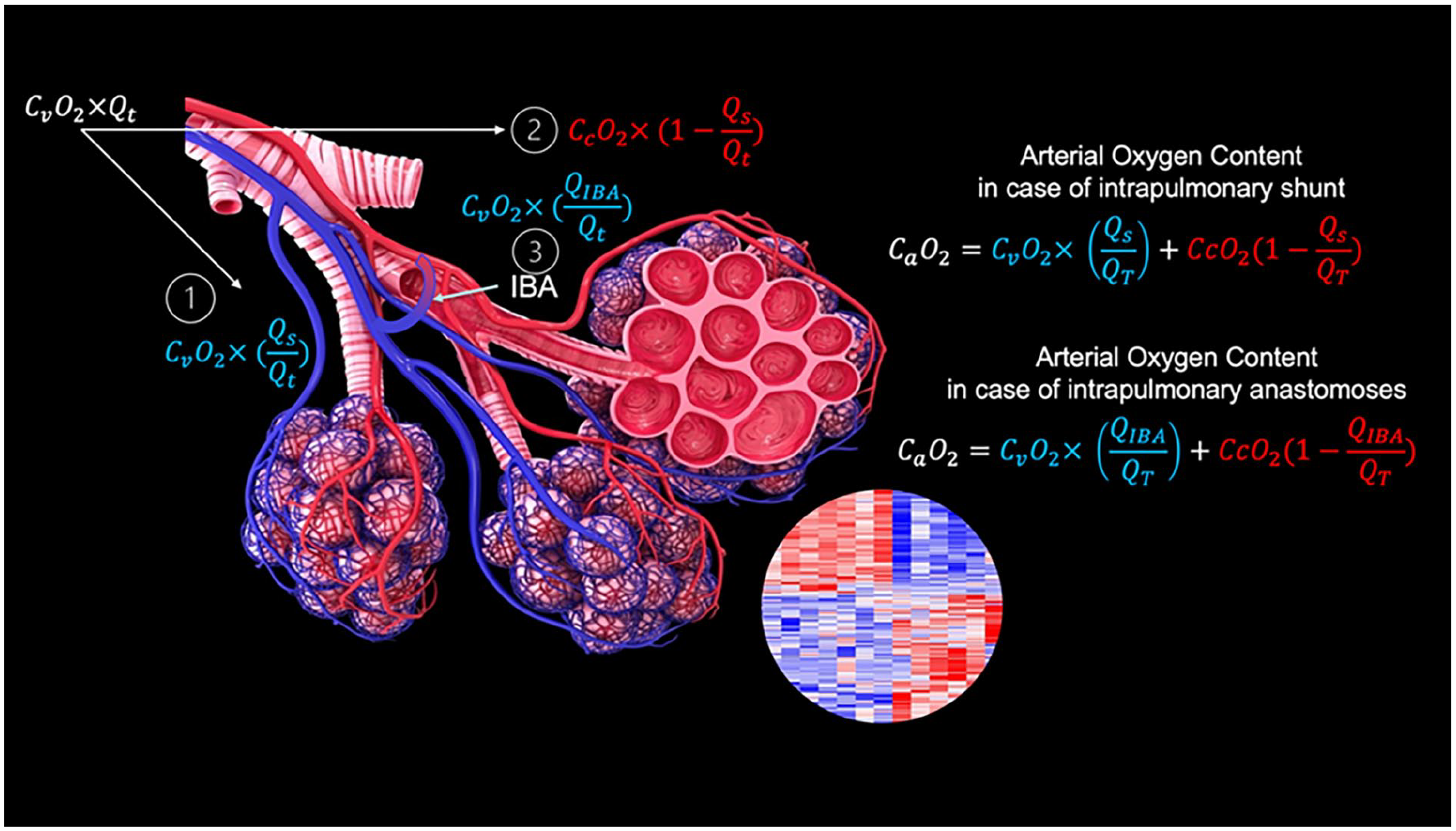
Schematic representation of hypoxaemia in intrapulmonary shunting and the effect of intrapulmonary bronchopulmonary anastomoses.
However, in the presence of intrapulmonary bronchopulmonary anastomoses (IBA), mixed venous blood flows directly from the pulmonary artery to pulmonary vein therefore bypassing the alveoli (3 in the Figure 1). This, in essence, is equivalent to shunted blood which is not affected by the inspired oxygen as the blood will not perfuse alveoli. This blood has the same oxygen content as the mixed venous blood and decreases considerably CaO2 in proportion to its quantity (QIBA/Qt).
Exemplar case and rapid recovery
The presence of extensive intravascular shunting in our patient would explain her clinical presentation and features, as well as her poor response to supplemental inhaled oxygen. It would also account for her SpO2 falling with the application of CPAP (which can cause microvascular compression, an increase in pulmonary arterial pressure and a possible increase in shunt flow). Right ventricular dysfunction seemed unlikely given the patient’s previous good health, and the absence of suggestive electrocardiographic and echocardiographic findings. Nor was there clinical evidence of significant dehydration. Nonetheless, it is possible that some degree of occult right ventricular systolic impairment may have been present in the context of modest underfilling, and that these may have worsened the shunt when CPAP was applied. A sustained, long and intense Valsalva manoeuvre was not possible, given the patient’s dyspnoea, and it is thus possible that a small right-to-left cardiac shunt was missed.
The patient’s very rapid recovery suggests an associated rapid resolution in shunt fraction, to which viral clearance may have contributed. While Oseltamivir is often given to treat influenza A, research suggests that there is limited benefit to its use.25,26 Steroids are commonly given in the treatment of community acquired pneumonia and of Covid-19 (and were initiated by the attending medical team on this basis, given the gravity of the hypoxaemia observed). The use of both oseltamivir and steroids in the treatment of influenza and community acquired pneumonia is currently being investigated as part of the REMAP-CAP trial. 27
Inhalation of 100% oxygen can reduce IPAVAs flow, 21 although the mechanism underlying this remains unclear. 28 A reduction in sepsis, and normalisation of cardiac output may also have decreased shunted flow and contributed to the rapid recovery. Among patients with Covid-19 pneumonitis, intrapulmonary shunting resolved fully in most, albeit over a very much longer timeframe than that observed in our patient. 29
Conclusion
Intrapulmonary vascular shunts can contribute substantially to hypoxaemia in viral infection. Seeking to understand the pathogenesis of observed hypoxaemia can help guide respiratory therapy. Mechanistic research may suggest novel therapeutic targets which could assist in avoiding intubation and mechanical ventilatory support.
Footnotes
Acknowledgements
H.M. is supported by the National Institute for Health Research’s Comprehensive Biomedical Research Centre at University College Hospitals, London.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship and/or publication of this article.
Informed consent
Informed consent was obtained for patient information to be published.
