Abstract
Background:
Despite high rates of cardiovascular disease in Scotland, the prevalence and outcomes of patients with cardiogenic shock are unknown.
Methods:
We undertook a prospective observational cohort study of consecutive patients with cardiogenic shock admitted to the intensive care unit (ICU) or coronary care unit at 13 hospitals in Scotland for a 6-month period. Denominator data from the Scottish Intensive Care Society Audit Group were used to estimate ICU prevalence; data for coronary care units were unavailable. We undertook multivariable logistic regression to identify factors associated with in-hospital mortality.
Results:
In total, 247 patients with cardiogenic shock were included. After exclusion of coronary care unit admissions, this comprised 3.0% of all ICU admissions during the study period (95% confidence interval [CI] 2.6%–3.5%). Aetiology was acute myocardial infarction (AMI) in 48%. The commonest vasoactive treatment was noradrenaline (56%) followed by adrenaline (46%) and dobutamine (40%). Mechanical circulatory support was used in 30%. Overall in-hospital mortality was 55%. After multivariable logistic regression, age (odds ratio [OR] 1.04, 95% CI 1.02–1.06), admission lactate (OR 1.10, 95% CI 1.05–1.19), Society for Cardiovascular Angiographic Intervention stage D or E at presentation (OR 2.16, 95% CI 1.10–4.29) and use of adrenaline (OR 2.73, 95% CI 1.40–5.40) were associated with mortality.
Conclusions:
In Scotland the prevalence of cardiogenic shock was 3% of all ICU admissions; more than half died prior to discharge. There was significant variation in treatment approaches, particularly with respect to vasoactive support strategy.
Keywords
Introduction
Cardiogenic shock represents a severe presentation of cardiovascular disease, where decreased cardiac output results in reduced end-organ perfusion and ultimately multi-organ failure. 1 Mortality is high, with cohort studies from the United States, Australia and Europe reporting in-hospital death rates of 35%–50%.2 –6 In the United States studies suggest the prevalence of CS has increased over the last two decades, particularly in the group with aetiologies other than acute myocardial infarction as compared to acute myocardial infarction-related shock. 7 In the United Kingdom, data from registries suggest cardiogenic shock complicates up to 13% of ST-segment elevation myocardial infarctions. 8
As in other acute conditions with high mortality, a relationship between larger centre case volume and better outcomes has been suggested in cardiogenic shock.9,10 Some have advocated for hub-and-spoke networks to coordinate care and provide specialist input, including the delivery of mechanical circulatory support.11,12 The joint British Cardiovascular Society – Intensive Care Society guideline statement in 2022 called for the development of such networks of care to improve outcomes of patients with cardiogenic shock. 13 In Scotland, despite cardiovascular disease being the leading cause of premature death, 14 no formal shock networks exist. The lack of epidemiological data on prevalence of cardiogenic shock and outcomes is an obstacle to potential improvements in pathways of care.
We aimed to describe the prevalence, management and outcomes of patients presenting to critical care with cardiogenic shock in Scotland, and to identify factors associated with in-hospital mortality.
Methods
Study design, setting and participants
We conducted a prospective observational cohort study of all patients with cardiogenic shock admitted to an acute care hospital (the EPOCHS (
All patients admitted to either an intensive care (ICU) or coronary care unit (CCU) were screened for inclusion. Inclusion criteria were all of: (1) adult patients (age ⩾16) receiving critical care at Level 2 (high dependency) or above as defined by the UK Intensive Care Society, 16 regardless of physical location in the hospital; (2) a clinical diagnosis of cardiogenic shock; (3) hypotension, defined as systolic blood pressure (SBP) < 90 mmHg for ⩾30 min or need for pharmacological or mechanical support to maintain SBP ⩾90 mmHg; and (4) clinical or biochemical evidence of hypoperfusion, defined as at least one of: (a) serum lactate > 2 mmol L−1; (b) rise in serum creatinine ⩾2 times baseline or urine output ⩽0.5 mL kg−1 h−1; (c) new serum alanine transaminase (ALT) > 160 IU L−1; (d) cold or mottled extremities; (e) new altered mental status without alternative cause.
Patients were excluded if shock arose following cardiac surgery, if there was an alternative cause for shock (e.g. sepsis, haemorrhage), or if the ceiling of treatment was ward-level and therefore invasive therapies were deemed inappropriate. Patients admitted following out-of-hospital cardiac arrest (OHCA) were included only if they met the above criteria, the cause of cardiac arrest was a cardiac condition (e.g. myocardial infarction, overdose of cardiotoxic drugs, ventricular arrhythmia) and there was significant evidence that the cause of shock was primarily cardiogenic (e.g. echocardiographic evidence of reduced cardiac output).
Data extraction
In accordance with the NHS Health Research Authority/UK Medical Research Council research ethics framework, ethical approval was not required for this service evaluation that use deidentified data available from the patient record (IRAS Project ID #317909). Specific approval was obtained for the collection of data at each hospital from the local Caldicott Guardian.
Data were extracted from paper or electronic record systems dependent on the infrastructure of each participating site and uploaded by investigators to a secure data storage system (REDCap, Vanderbilt University, Nashville, TN) hosted by the Surgical Informatics Group, Usher Institute, University of Edinburgh. Where patients were transferred between multiple hospitals participating in the study, physiological data from the index presentation was used along with outcome data from the final hospital site in their admission.
Data on patient demographics, comorbidities (coronary artery disease, hypertension, diabetes, heart failure, chronic kidney disease, severe chronic lung disease (defined as >1 hospital admission/year or long-term oxygen therapy), adult congenital heart disease and pregnancy), the Rockwood clinical frailty score (as adjudged by the admitting clinician), aetiology of cardiogenic shock, admission location and prior cardiac arrest (defined as cardiopulmonary resuscitation (CPR) or defibrillation prior to unit admission) were recorded. Serum lactate and pH were taken from the sample performed closest to unit admission. Society for Cardiovascular Angiographic Intervention (SCAI) grade for cardiogenic shock was assessed by the reporting clinician at the point of presentation to critical care. 17
In order to estimate the prevalence of cardiogenic shock as a proportion of all critical care admissions, the denominator was derived from data from the Scottish Intensive Care Society Audit Group (SICSAG). 18 Admissions to coronary care units were excluded from this analysis, as these units are not included in the SICSAG database.
Outcomes collected included in-hospital mortality, length of stay in the ICU and the use of mechanical cardiac support.
Statistical analysis
Continuous data are presented as median (interquartile range, [IQR]) and compared with Mann-Whitney or Kruskal-Wallis test as appropriate; categorical data are presented as n (%) and compared with chi-squared test. The annual prevalence of cardiogenic shock in ICUs was estimated by doubling the number of admissions during the 6-month study period and dividing by the number of admissions in each ICU over the previous calendar year.
Multivariable logistic regression was used to identify associations with survival. Factors known to be associated with the primary outcome were selected a priori for inclusion in the base model. These included age, sex, out-of-hospital cardiac arrest and serum lactate at presentation. In addition, backward stepwise regression was performed to identify other factors associated with the primary outcome to create two further models. The first model included non-modifiable factors already present at presentation. The second model included additional modifiable factors, such as treatment in the ICU.
All other variables were entered into the multivariable regression model if they were associated with outcome on the univariable analysis (p < 0.10). Variables were assessed for co-linearity using visual inspection of correlation plots and the Pearson’s correlation coefficient, and removed from the model if significant co-linearity existed.
All statistical analysis was conducted using R version 4.2.2 (R Foundation for Statistical Computing, Vienna, Austria), and code is available on request to the corresponding author.
Results
Patient characteristics
After exclusion of five patients who were entered in the study database twice as they were treated in two different hospitals during the same presentation, a total of 247 patients from 13 sites were included. This comprised 40 patients from the pilot phase (July 2022) and 207 patients from the main study period (November 2022 to April 2023 inclusive).
The median age of patients with cardiogenic shock was 65 [IQR, 55–72] years, 60% were male, and the median BMI was 26.1 [24.0–30.0] (Table 1). The majority of patients (60%) had a Rockwood clinical frailty score of 1–3 (very fit to managing well); a quarter (23%) had a score of 4–5 (vulnerable to mildly frail) and 16% had a score 6–9 (moderately frail or worse). The commonest comorbidity was hypertension (39%) followed by coronary artery disease (26%), diabetes mellitus (25%) and heart failure (21%).
Baseline characteristics associated with mortality in 247 patients with cardiogenic shock.
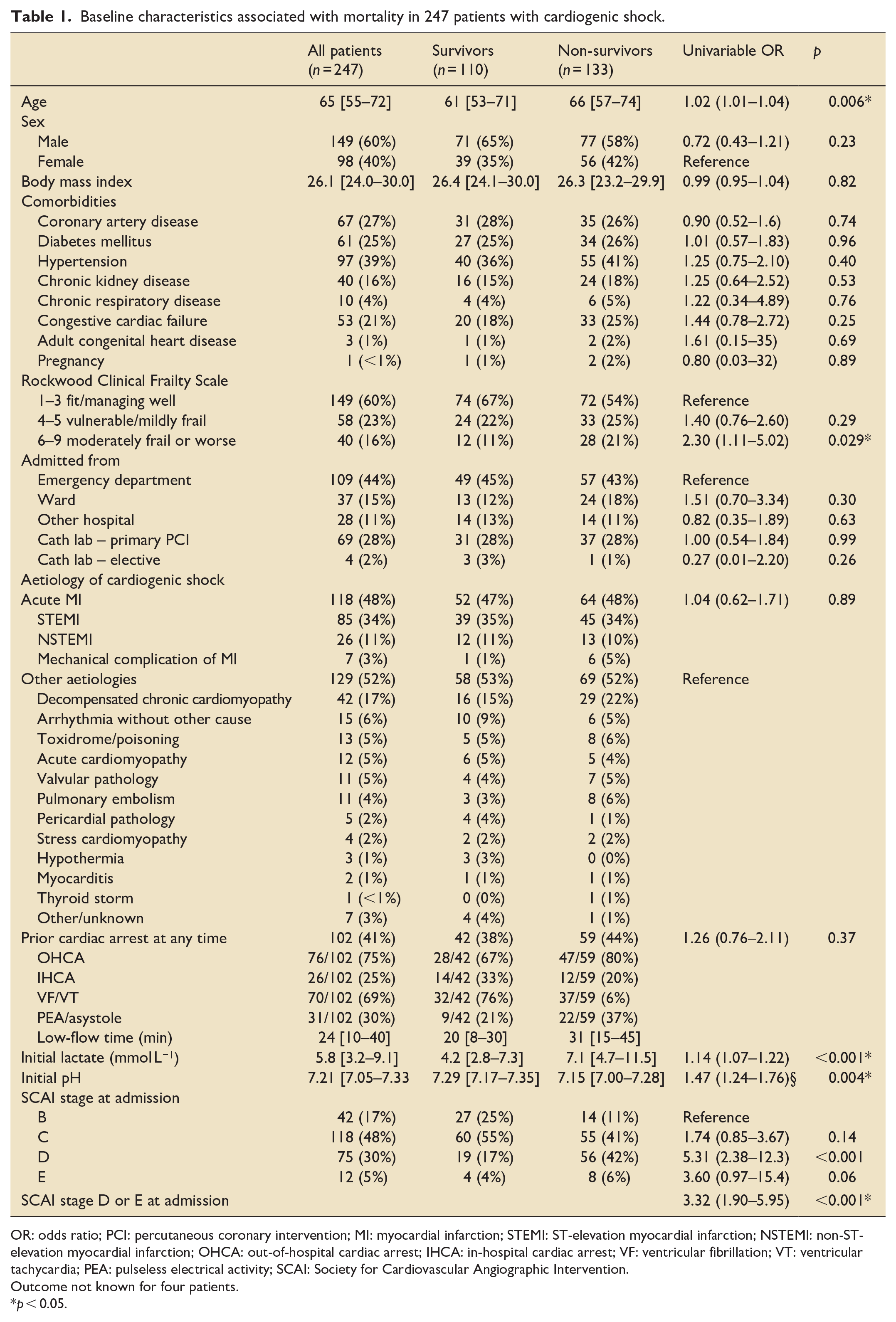
OR: odds ratio; PCI: percutaneous coronary intervention; MI: myocardial infarction; STEMI: ST-elevation myocardial infarction; NSTEMI: non-ST-elevation myocardial infarction; OHCA: out-of-hospital cardiac arrest; IHCA: in-hospital cardiac arrest; VF: ventricular fibrillation; VT: ventricular tachycardia; PEA: pulseless electrical activity; SCAI: Society for Cardiovascular Angiographic Intervention.
Outcome not known for four patients.
p < 0.05.
The aetiology of cardiogenic shock was acute myocardial infarction in 48% of patients, acute heart failure due to decompensation in those with known cardiomyopathy (17%), primary arrhythmia (6%), poisoning, acute heart failure due to a de novo presentation of cardiomyopathy, and valvular pathology (all 5%) and pulmonary embolism (4%). Less common causes included stress cardiomyopathy, hypothermia, myocarditis and thyroid storm (Table 1).
At unit admission, median initial lactate was 5.8 mmol L−1 and median pH 7.21; 42 patients (17%) were Society of Cardiovascular Intervention (SCAI) class B; 48% class C; 30% class D and 5% class E. Four in ten patients (41%) had a cardiac arrest prior to unit admission, of whom 75% had an out-of-hospital cardiac arrest and 25% an in-hospital cardiac arrest. Median duration of cardiac arrest was 24 [10–40] min.
Investigations and treatment
Echocardiography was performed within 48 h of unit admission in 84% of patients (Table 2). In those who presented with cardiogenic shock secondary to acute myocardial infarction, coronary angiography was performed in 80% (92% of survivors and 69% of non-survivors). Approximately three-quarters of patients (76%) received an arterial line (94% of those in intensive care and 19% of those in coronary care), 11% a pulmonary artery catheter and 6% had another cardiac output monitor.
Management characteristics associated with mortality in 243 patients with CS.
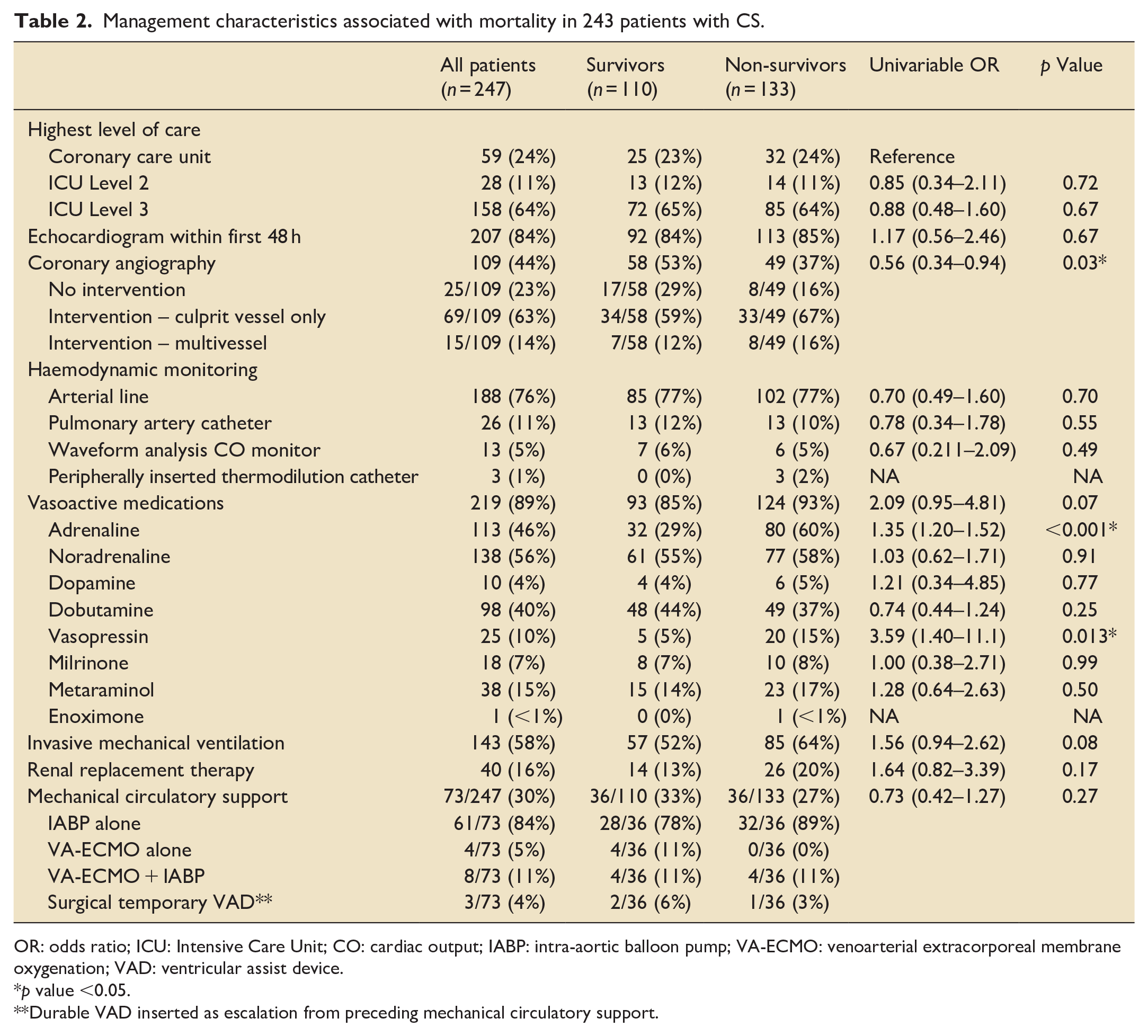
OR: odds ratio; ICU: Intensive Care Unit; CO: cardiac output; IABP: intra-aortic balloon pump; VA-ECMO: venoarterial extracorporeal membrane oxygenation; VAD: ventricular assist device.
p value <0.05.
Durable VAD inserted as escalation from preceding mechanical circulatory support.
In total, 89% of patients received at least one vasoactive medication. The most frequently utilised was noradrenaline (56%), followed by adrenaline (46%) and dobutamine (40%). Regarding the initial vasoactive medication (excluding drugs given during CPR), adrenaline (29%) was most frequent, followed by noradrenaline (26%), dobutamine (24%) and metaraminol (14%). Invasive mechanical ventilation was used in 58% and renal replacement therapy in 16%.
Mechanical circulatory support was used in 30% of patients. Of these, most received an intra-aortic balloon pump (IABP) alone (84%), with just 5% receiving veno-arterial extracorporeal membrane oxygenation (ECMO) alone and 11% veno-arterial ECMO plus an IABP. Whilst no patient received a percutaneous ventricular assist device, three underwent escalation to a surgically-implanted temporary ventricular assist device. In the total cohort, five patients (2%) received cardiac transplantation.
Mortality and associated factors
Follow-up for in-hospital mortality was available in 98% (243 of 247 patients), of whom 133 (55%) patients died (Table 3). In those who survived in whom the discharge destination was recorded, 79/83 (95%) were discharged to their usual place of residence, with 4/83 (5%) discharged to a nursing home or hospice. One patient who was still alive awaiting discharge from the unit at >30 days at close of data collection was counted as alive for the purpose of mortality analyses.
Outcomes for 243 patients with CS.

Non-modifiable risk factors for in-hospital mortality in the univariable analysis included age (odds ratio [OR] 1.02 per year, 95% confidence interval [CI] 1.01–1.04, p = 0.006), clinical frailty score of 6 or greater (OR 2.30, 95% CI 1.11–5.02, p = 0.029), initial lactate (OR per 1 mmol L−1 increase 1.14, 95% CI 1.07–1.22, p < 0.001), initial pH (OR per 0.1 reduction 1.47, 95% CI 1.24–1.76, p = 0.004) and SCAI stage D or E on presentation (OR 3.32, 95% CI 1.90–5.95, p < 0.001) (Table 3). Aetiology was not significantly associated with outcome (acute myocardial infarction vs other, OR 1.04 (95% CI 0.62–1.71, p = 0.89); nor was pre-admission cardiac arrest (OR 1.26 (95% CI 0.76– 2.11), p = 0.37).
Modifiable risk factors for in-hospital mortality in the univariable analysis included use of adrenaline (OR 1.35, 95% CI 1.20–1.52, p < 0.001), and vasopressin (OR 3.59, 95% CI 1.40–1.1, p = 0.013) (Table 3). Coronary angiography was associated with a lower risk of death at 30 days (OR 0.56, 95% CI 0.34–0.94, p = 0.03) although only approximately half of (48%) the patients had cardiogenic shock as a consequence of acute myocardial infarction. Neither use of a pulmonary artery catheter (OR 0.78, 95% CI 0.34–1.78, p = 0.55) nor mechanical circulatory support (OR 0.73, 95% CI 0.42–1.27, p = 0.27) were associated with death at 30 days.
Multivariable regression models are displayed in Table 4. In a model accounting only for non-modifiable risk factors, with a-priori inclusion of age, pre-admission cardiac arrest and lactate, significant associations with mortality were observed with age (OR 1.04 per year (95% CI 1.02–1.06, p < 0.001), lactate (OR 1.10 per mmol L−1 (95% CI 1.03–1.19, p = 0.010)) and SCAI stage D or E at presentation (OR 2.16 (95% CI 1.10–4.29, p = 0.009)). When modifiable risk factors were added to this model, use of adrenaline (OR 2.73 (95% CI 1.40–5.40, p = 0.001) and vasopressin (OR 4.21 (95% CI 1.39–15.1, p = 0.008) retained their association with mortality.
Multivariable logistic regression models.
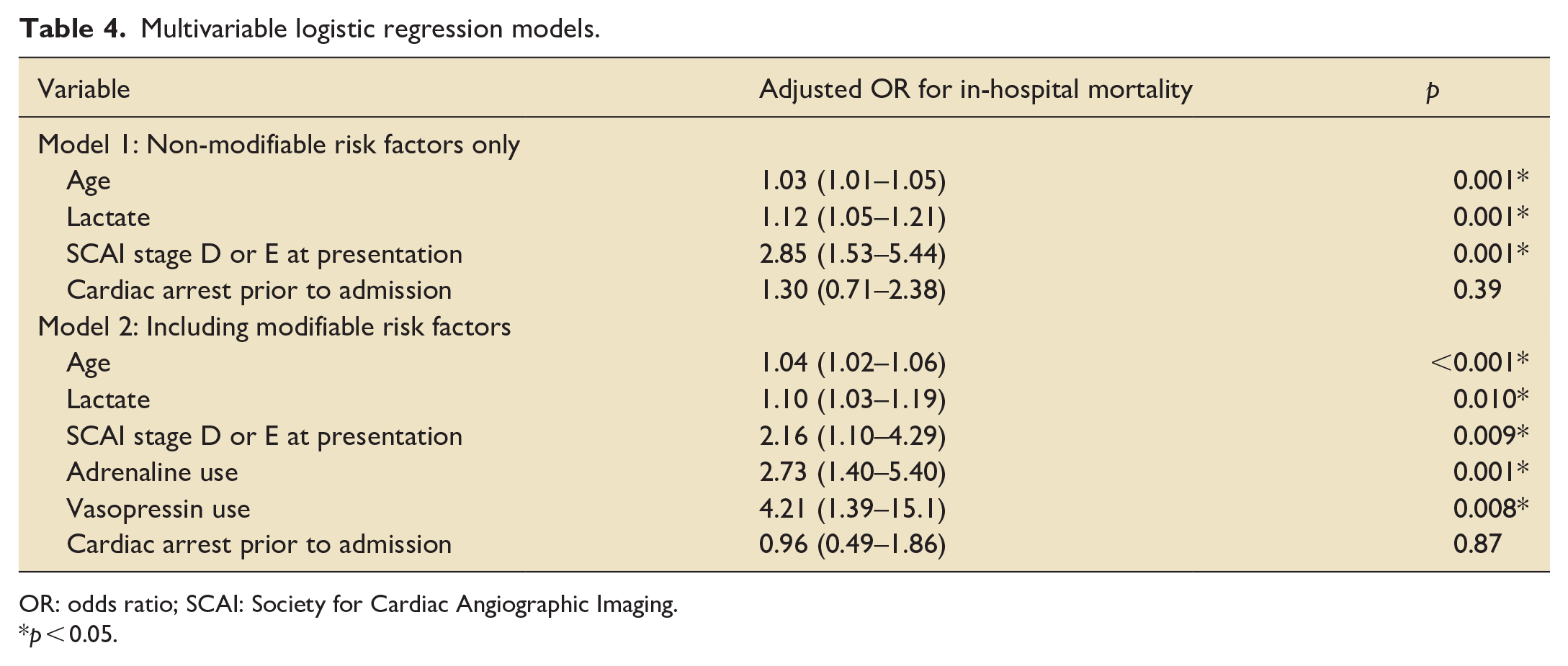
OR: odds ratio; SCAI: Society for Cardiac Angiographic Imaging.
p < 0.05.
Estimates of prevalence
After excluding patients from the pilot study and those admitted to a coronary care unit, 160 patients with cardiogenic shock were admitted to an ICU during the 6-month study period. Given there were 10,686 admissions to these ICUs during the preceding 12-month period, we estimate the annual prevalence of cardiogenic shock presenting to an ICU in Scotland to be 3.0% (95% CI 2.6%–3.5%). Prevalence varied by participating site with a median of 1.8% [interquartile range 1.4%–3.4%] and a range of 0.5%–6.2% (Supplemental Table 1). Of the five units with the highest prevalence, four (A, E, C and I) were primary percutaneous coronary intervention centres, three of which (A, C and I) also had cardiac surgical services. The other (K) was a district general hospital without tertiary cardiac services.
Three sites (B, F and H) were identified as potential outliers having reported significantly fewer patients than those of comparable size and case mix, that is, district general hospitals without pPCI or interventional cardiac services (3, 1 and 2 patients respectively), likely representing under-reporting. Sensitivity analysis with exclusion of these three units resulted in an estimated prevalence of 3.5%, median 3.0% [IQR 1.8%–4.5%].
Discussion
For the first time, this study provides a comprehensive description of the epidemiology of patients with cardiogenic shock in Scotland, providing insight on management and outcomes. It demonstrates several important findings. The estimated prevalence of cardiogenic shock in Scottish ICUs is 3%. This is challenging to benchmark internationally, as comparable cohort studies have either not reported a denominator2 –5 or focused exclusively on academic medical centres with specialised cardiac intensive care units. 6 It may also be an underestimate due to under-reporting, a limitation of the study design requiring the screening of admissions by site investigators. The presence of aetiologies other than acute myocardial infarction in almost half of included patients is comparable to US data, where prevalence of cardiogenic shock due to other aetiologies is increasing faster than that due to acute myocardial infarction. 7 Accordingly, clinicians and researchers should ensure that patients with all aetiologies are included in the design of clinical pathways and research studies for cardiogenic shock.
The primary outcome of 54% in-hospital mortality in our study is higher than comparable contemporary multicentre cohort studies from France (FRENSHOCK study, 26%), 2 the US (Cardiac Critical Care Trials Network, 32%), 6 Australia (44%) 3 and a large (n = 1000) single centre study from Germany (49%). 4 The observed in-hospital mortality of 54% in cardiogenic shock secondary to acute myocardial infarction in our study was also slightly higher than that of 50% in a multicentre study of the same population from Denmark. 5
The reasons for these differences are likely numerous and may include both under-reporting of less severe cases and differences in study design, patient population and treatment strategy. It is likely that this study under-reported patients treated in coronary care units with a lower severity of shock. That said, the proportion of patients in this study with SCAI grade D or E shock (the most severe) was 35% compared to 41% in the only large comparable study to report the SCAI grade. 4 The FRENSHOCK study, which reported mortality of 26%, included fewer patients with preceding cardiac arrest (10% vs 40% in our study) and who received invasive ventilation (38% vs 58%) and a greater proportion of patients from coronary care units (56% vs 24%), who are likely to be less sick than those admitted from ICU. 2
In addition to differences in study design and potential bias from under-reporting, it is also possible that there are differences in the population of patients with cardiogenic shock between Scotland and elsewhere contributing to the high observed mortality in this study. There are significantly fewer ICU beds per capita in Scotland 18 than in Germany, France or the US, 19 which may result in a higher threshold for critical care admission and hence a sicker patient population. Scotland has a significant burden of cardiovascular risk factors 20 and a high prevalence of social deprivation, which is known to influence critical care outcomes 21 ; this may also have contributed. Further, the absence of a formalised cardiogenic shock network or transfer and escalation pathway in Scotland may result in time delay to treatments and worse outcomes.
Our study demonstrated significant variation in the management of cardiogenic shock, specifically with regard to use of vasoactive medications and MCS. Adrenaline, noradrenaline and dobutamine were used in almost equal proportion as first-line therapy. While this may represent individualisation of therapy to patient physiology, varying institutional preferences likely also play a role. Coronary angiography was associated with lower mortality in univariable regression analysis, but not once severity of shock was included in the model; this may suggest that the patients who received coronary angiography were more stable at presentation than those who did not.
In our study, adrenaline use was associated with an increased risk of mortality in a multivariable model including preceding cardiac arrest and presenting severity of shock (lactate and SCAI stage). As in any observational study, unmeasured confounding may explain this association, as adrenaline tends to be used in sicker patients. However, adrenaline increases myocardial oxygen consumption and is associated with increased biomarkers of multi-organ failure in cardiogenic shock.22,23 In the only randomised controlled trial to date of adrenaline in CS, adrenaline led to a higher incidence of refractory shock and death compared to noradrenaline. 24 Current European Society of Cardiology and American Heart Association guidelines both recommend noradrenaline as the first-line vasoactive treatment in cardiogenic shock.25,26
In our study MCS use was not associated with survival in univariate analysis, however we have not formally adjusted for confounding by indication (i.e. patients receiving MCS in our study having a higher probability of death, even if this treatment was beneficial). The overall MCS use in our study of 30% is similar to other cohorts, but this largely consisted of IABP with only 5% of the whole patient cohort receiving ECMO. No patients received a peripheral ventricular assist device (pVAD). There is only one funded transplant and advanced MCS centre in Scotland and therefore local institutional preferences and policies are likely to have influenced this. Use of ECMO and pVADs is notably higher in contemporary French, German, Danish and US cohorts (ECMO 6%–20%, pVAD 5%–14%),2,4,6,27,28 although a mortality benefit of any specific MCS strategy in CS is yet to be demonstrated.
In summary, this study clearly demonstrates that cardiogenic shock in Scotland has a high mortality, and that there is significant variation in treatment. Given over half of included patients had shock caused by aetiologies other than acute MI, existing care networks for ST-elevation MI are likely inadequate for the cardiogenic shock patient. Cardiogenic shock networks have been introduced in some centres in the UK 29 and North America, 30 and while there is no certain evidence of improvement in outcomes, the ability to concentrate resources, provide rapid access to clinical expertise and potentially life-saving therapies is clearly attractive. Further work is needed to determine the ideal structure of a potential cardiogenic shock network in Scotland accounting for geography, distribution of existing expertise and access to relevant specialities and resource.
Limitations
This study had several limitations. The design of the study was non-systematic and prone to identification bias, as it relied on clinicians screening admissions at each participating centre and patients are likely to have been missed, especially at centres identified as outliers in terms of low reporting. As mentioned above, it is possible that there has been an over-estimate of mortality due to under-reporting of patients with less severe shock.
While efforts were made to include as many potentially prognostic factors in modelling, there is likely residual confounding from unobserved variables and therefore the conclusions that can be drawn from this – and, indeed, any observational research – are naturally limited. Follow-up was unavailable in four patients (2%), but this was a small proportion of the overall sample and overall follow-up was robust. We did not have the resource to collect any patient-centred outcomes regarding destination after hospital discharge or quality of life.
Conclusions
CS comprises approximately 3% of admissions to critical care in Scotland and has a hospital mortality higher than other contemporary cohorts. Significant variation in pharmacological treatment strategy was observed; use of adrenaline was associated with decreased survival in multivariable analysis including markers of disease severity.
Supplemental Material
sj-docx-1-inc-10.1177_17511437231217877 – Supplemental material for EPidemiology Of Cardiogenic sHock in Scotland (EPOCHS): A multicentre, prospective observational study of the prevalence, management and outcomes of cardiogenic shock in Scotland
Supplemental material, sj-docx-1-inc-10.1177_17511437231217877 for EPidemiology Of Cardiogenic sHock in Scotland (EPOCHS): A multicentre, prospective observational study of the prevalence, management and outcomes of cardiogenic shock in Scotland by Alex Warren, Philip McCall, Alastair Proudfoot, Stuart Gillon, Ahmad Abu-Arafeh, Angus John McKnight, Rosemary Mudie, David Armstrong, Evangelos Tzolos, John Andrew Livesey, Andrew Sinclair, Veronica Baston, Jonathan Dalzell, Deborah Owen, Lucy Fleming, Ian Scott, Alex Puxty, Matthew MY Lee, Fiona Walker, Simon Hobson, Euan Campbell, Michael Kinsella, Eilidh McGinnigle, Robert Docking, Grant Price, Alex Ramsay, Richard Bauld, Suzanne Herron, Nazir I Lone, Nicholas L Mills and Louise Hartley in Journal of the Intensive Care Society
Footnotes
Acknowledgements
The authors acknowledge the assistance of the Surgical Informatics Group at the University of Edinburgh, particularly Riinu Pius and Malcolm Cameron, for managing the RedCAP platform during the study, and all the bedside staff who provided care for the included patients.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Funding for the secure data storage platform (£1000) was obtained from an unrestricted educational grant for conducting epidemiological research in cardiogenic shock awarded to AP and AW by Abbott Vascular. Abbott Vascular had no part in the design, conduct, analysis or reporting of the research and have not seen the manuscript prior to submission. AP is supported by a Medical Research Council Clinical Academic Research Partnership Award reference: MR/W03011X/1. NLM is supported by a Chair Award, Programme Grant, and Research Excellence Award (CH/F/21/90010, RG/20/10/34966, RE/18/5/34216) from the British Heart Foundation.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
