Abstract

The use of platelet transfusions is increasing. Approximately 287,000 platelet transfusions were issued in the in the United Kingdom (UK) in 2021 compared with 269,000 in 2020. 1 After cancer services, intensive care units (ICUs) are amongst the largest users of platelets. 2 Until recently, this common practice has not been informed by data from randomised controlled trials (RCTs) in critically ill patients but extrapolated from other clinical settings (e.g. haematology/oncology).
In this editorial, we will first discuss tests for assessing bleeding risk, followed by a review of the evidence suggesting that platelet transfusions may be less effective and more harmful than previously thought. We will also describe current platelet transfusion practice in UK ICUs. Our focus is on prophylactic platelet transfusions, typically administered to non-bleeding patients with thrombocytopaenia in the belief that the benefits of reducing any bleeding (e.g. in the context of invasive procedures) outweigh the risks.
Thrombocytopaenia is common in critically ill patients, with 4–6% of patients having severe thrombocytopaenia (defined as a platelet count of <50 × 109/L) at admission and up to 12% at some point during their ICU stay.3,4 Common reasons include sepsis, liver failure, haematological malignancies and bleeding. 4 Thrombocytopaenia is associated with increased morbidity and mortality in critically ill patients, but it is unclear to what extent thrombocytopaenia mediates poor outcomes or simply reflects illness severity. 4 In light of this uncertainty and current patterns of use of platelet transfusions, two important research questions bear consideration: (i) ‘Are prophylactic platelet transfusions effective in reducing bleeding in critically ill patients?’; and (ii) ‘What harms are associated with a platelet transfusion in critical illness?’
Procedure-related bleeding in critical illness
In order to answer the first question, it is important to highlight that platelet count and other coagulation tests (e.g. prothrombin time) are poor predictors of peri-procedural bleeding. 5 All RCTs evaluating platelet transfusions have reported high persisting rates of bleeding in patients with thrombocytopaenia despite testing different policies of platelet transfusion (e.g. higher platelet count threshold or higher platelet dose). There is limited evidence to support a role for near-patient viscoelastic haemostatic assays (VHAs) such as thromboelastography (TEG) and rotational thromboelastometry (ROTEM) to guide transfusion therapy in some settings, particularly in the ICU. 6 The Implementing Treatment Algorithms for the Correction of Trauma-Induced Coagulopathy (ITACTIC) multicentre RCT compared standard Major Haemorrhage Protocols (MHPs) using conventional coagulation tests versus VHA-guided algorithms reported no difference in the primary outcome of patients who were alive and free of massive transfusion at 24 h. 7
Severe bleeding (requiring invasive intervention or blood transfusion) or life-threatening bleeding for low-risk procedures, such as central venous catheter (CVC) insertion, rarely occurs except through technical misadventure, with a reported rates of<1% in large cohort studies.8,9 One study reported a major bleeding complication rate of approximately 1% in patients with thrombocytopaenia undergoing paracentesis, and bleeding complications occurred in patients with platelet counts between 41–46 × 109/L. 10 In patients undergoing tracheostomy, bleeding (defined as bleeding requiring intravenous fluids or another intervention) occurred in 3.1% of patients. 11 These low rates of bleeding may, in part, be driven by the increasing use of ultrasound. 12 Observational studies suggest that prophylactic platelet transfusions do not reduce the rate of major bleeding in critically ill patients when compared with no platelets 13 and that platelet increments following a transfusion are highly variable, and often very limited. 14
Risks associated with platelet transfusions
Platelet transfusions have risks and are the most commonly implicated component associated with transfusion reactions. 1 Febrile non-haemolytic transfusion reactions and allergic reactions may occur at a frequency of 1 in 14 and 1 in 50 per-unit transfusions, respectively. 15 Although some of these reactions may often be seen as ‘minor’ by clinicians and may be masked in critically ill patients, recipients of platelet transfusion often say these episodes can be distressing, particularly when repeated. Sepsis from a bacterially contaminated platelet unit is the most frequent infectious complication from any blood product, 16 but again may be under recognised in critical illness. Platelets are now known to mediate many physiologically processes beyond haemostasis, including interactions with immunological and inflammatory pathways. 17 These observations may explain the findings of two recent RCTs. Critically ill neonates, randomly allocated to a liberal prophylactic platelet transfusion to prevent major bleeding, paradoxically had higher mortality and more frequent major bleeds than those allocated to a restrictive strategy (50 vs 25 × 109/L). 18 In a second RCT, adults with intracerebral haemorrhage who were receiving antiplatelet therapy had higher mortality rates and worse neurological recovery with liberal platelet transfusion. 19
What is current practice in ICUs?
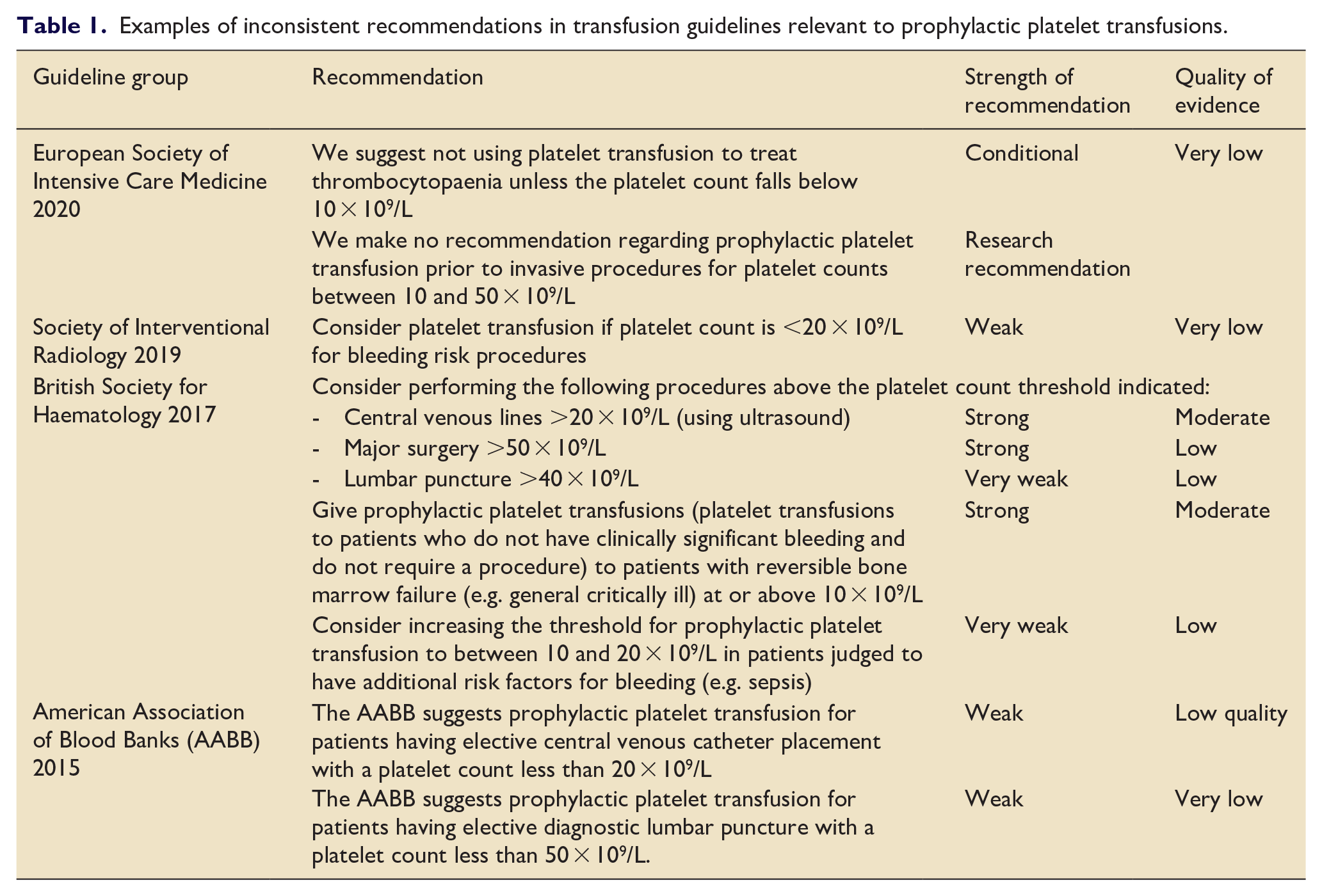
We previously conducted a survey of current UK clinical ICU practice across four commonly encountered clinical scenarios in non-bleeding, critically ill adults with thrombocytopaenia. 20 Data from 99 clinical staff covering 78 ICUs were available and responses are displayed in Figure 1. We found current UK platelet transfusion practice to be highly variable, a variability that has also been observed internationally. 21 The Intensive Care Study of Coagulopathy study, conducted across 29 ICUs in the UK in 2011, found similar patterns. 4 Many platelet transfusions occur at higher platelet thresholds than those recommended by guidelines, 22 but these recommendations are not based on high quality data and there is considerable variation between guidelines15,23 –25 (Table 1).
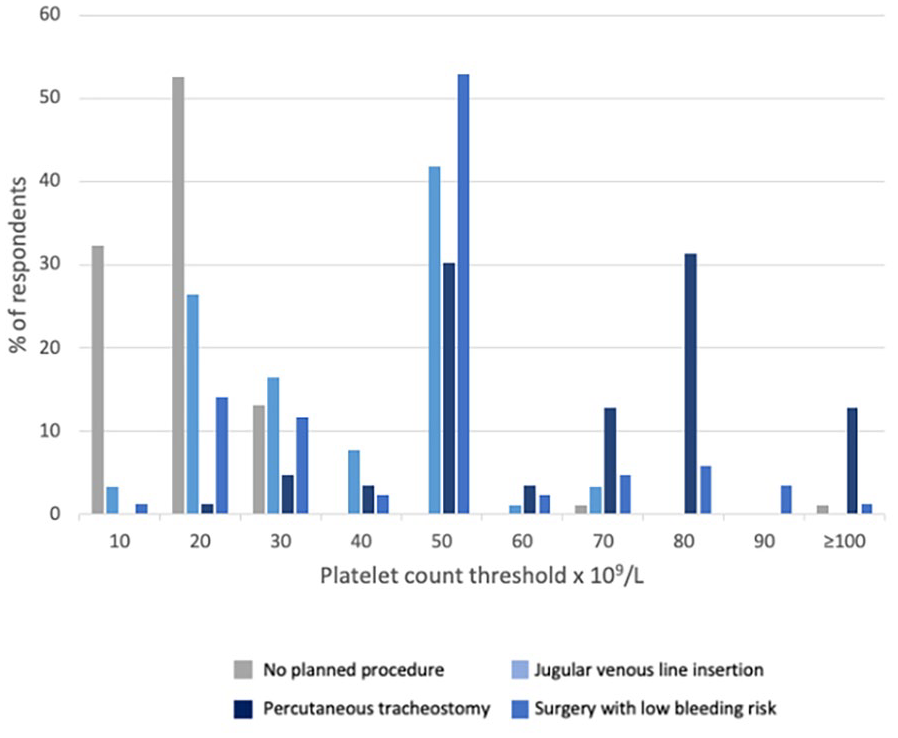
Prophylactic platelet transfusion thresholds without any planned procedures and prior to different procedures in UK ICUs (Reproduced with permission from John Wiley and Sons, 01 Aug 2023).
Examples of inconsistent recommendations in transfusion guidelines relevant to prophylactic platelet transfusions.
Until recently there were no RCT data on the use of prophylactic platelets in ICU. van Baarle et al. 26 conducted a multicentre, RCT where patients on either ICU or the haematology ward, with severe thrombocytopaenia (platelet count 10–50 × 109/L), were randomly assigned to receive one unit of prophylactic platelet transfusion or no transfusion before ultrasound guided CVC placement. In total, 338 patients were randomised, most in the haematology setting where underlying bone marrow failure is the likely cause of thrombocytopaenia. The authors found that withholding prophylactic platelet transfusion by adopting a lower platelet count threshold for transfusion pre-procedure was associated with a higher risk of bleeding. However, some limitations should be recognised. The increased risk of bleeding was largely driven by subclavian CVC insertion in haematology ward patients, which does not reflect practice in UK ICUs. There was no evidence of an increased bleeding risk in ICU patients. The majority of bleeding episodes were Grade 2 which includes ‘bleeding that results in minor interventions to stop, such as prolonged manual compression (>20 min)’. In a relatively small study (not powered to detect differences in outcomes other than bleeding), it is of note that ICU patients who received a platelet transfusion experienced numerically more acute lung injury, days in ICU and deaths, and there were two transfusion reactions to platelets. These outcomes may be more important to patients than non-severe bleeding.
Challenges in trial design
In summary, equipoise remains for the use of platelet transfusions in adult ICUs. Further trials are required, but investigators should reflect on:
The preferences and beliefs of treating physicians with a desire to reduce perceived risks of bleeding in patients with severe thrombocytopaenia: We have some insight into this issue from the findings of a randomised trial which aimed to evaluate a fresh frozen plasma (FFP) prophylaxis versus no prophylaxis strategy in nonbleeding critically ill patients, but which stopped early due to low recruitment. The authors reported that the most important barrier for patient inclusion was physician driven. 27 Many physicians felt that certain patients should be excluded from the trial (e.g. patients with heart failure, poor/worsening oxygenation or liver failure). A minority of physicians did not want to take the risk of randomisation to no FFP with the ensuing consequences should the patient experience a catastrophic haemorrhage, whilst other physicians felt there was no need for an FFP transfusion and did not want to subject patients to potential harms of transfusion.
Methodological limitations: There are real challenges on how to conduct an appropriate and adequately powered trial. All trials to date have applied conventional designs comparing one threshold to another (e.g. 10 vs 50 × 109/L) but may not identify an optimal threshold for all patient types or all procedures, and may also hinder physician buy-in. Should studies be superiority, equivalence, or non-inferiority study for different thresholds of prophylactic platelet transfusion? There are inherent difficulties in allocation concealment and blinding in such studies.
Outcomes: Is bleeding the best outcome measure, given its potential subjectivity and risks of transfusion, or should we measure ‘harder’ outcomes that encompass benefits and risks (e.g. mortality)? The low frequency of bleeding associated with low-risk procedures means that very large sample sizes would be required to detect any difference in RCTs.
It remains entirely plausible that that we are over transfusing some ICU patients, subjecting them to unjustified risks. However, the heterogenous nature of critically ill patients means that there could be distinct subgroups of critically ill patients in whom platelet transfusions may reduce bleeding and improve outcomes.
The threshold for platelets (T4P) trial
Current guideline recommendations are largely based on extrapolated data from RCTs in patients with haematological malignancy,28,29 which may have included small numbers of patients with co-existing critical illness (5%–10%). The need for research in platelet transfusions in critical illness has been supported by international transfusion and intensive care societies, 24 and the James Lind Alliance Blood Transfusion and Donation Partnership. 30
T4P is currently recruiting across UK ICUs and is an open-label, randomised, Bayesian adaptive, comparative effectiveness trials across five equally spaced-placed platelet thresholds (<10 to <50 × 109/L). The study is funded by an NIHR Health Technology Assessment grant (NIHR131822). Patients can be included if they are accepted for admission or already admitted to a participating ICU, require a low bleeding risk invasive procedure, with a platelet count <50 × 109/L, at any time from 24 h prior to ICU admission until ICU discharge. Patients will be randomised to one of five platelet count thresholds (10–50 × 109/L) below which they would receive a single adult dose of platelet transfusion administered before or during the procedure and subsequent included procedures during their ICU stay. Given the many effects of transfused platelets the study will primarily assess all-cause 90-day mortality, but will also collect data on bleeding, organ support requirements and any transfusion reactions. The study aims to recruit 2550 patients.
What should we do in the light of the uncertainty?
We suggest a personalised, risk-adapted approach that does not solely focus on platelet count. Consideration should be given to patient characteristics, the cause, severity and time course of thrombocytopaenia, likelihood of bleeding, response to any previous platelet transfusions and use of alternative agents for example, tranexamic acid. Effective treatment of the underlying cause (e.g. sepsis, immune thrombocytopaenia) will increase platelet counts. Use of ultrasound for relevant procedures (e.g. CVCs), along with an appropriate anatomical site (e.g. internal jugular vein for CVC) is recommended by NICE, to reduce the risk of bleeding. Clinicians should continue to report transfusion reactions and adverse events to haemovigilance systems. In particular, most haemovigilance systems fail to clearly report paediatric and neonatal transfusions where the rate of transfusion reactions may be more common proportionately relative to adults.
In conclusion, there is widespread variation in platelet transfusion practice in critically ill patients, and there is a pressing need for robust RCT evidence to guide practice. Multiple observational studies have demonstrated an association between platelet transfusions and worse clinical outcomes, supporting the clear need to protect patients from harm without evidence of benefits.
Footnotes
Acknowledgements
None
Collaborating authors
On behalf of the Threshold for Platelets (T4P) Investigators:
• Hayley Noble, Clinical Trials Unit, Intensive Care National Audit & Research Centre (ICNARC), London, UK
• Joanna Calder, Patient Representative
• Doug W Gould, Clinical Trials Unit, Intensive Care National Audit & Research Centre (ICNARC), London, UK
• Paul R Mouncey, Clinical Trials Unit, Intensive Care National Audit & Research Centre (ICNARC), London, UK
• David A Harrison, Clinical Trials Unit, Intensive Care National Audit & Research Centre (ICNARC), London, UK
• J Duncan Young, Nuffield Department of Clinical Neurosciences, University of Oxford, Oxford UK
• Matteo Quartagno, MRC Clinical Trials Unit, University College London, London, UK
• Timothy S Walsh, Department of Anaesthesia, Critical Care and Pain Medicine, Royal Infirmary Edinburgh, Edinburgh, UK; Edinburgh Critical Care Research Group, University of Edinburgh, Edinburgh, UK
• Richard Grieve, Department of Health Services, Research and Policy, London School of Hygiene and Tropical Medicine, London, UK
• Alexina Mason, Centre for Statistical Methodology, London School of Hygiene and Tropical Medicine, London, UK
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: PJW is Chief Investigator for T4P. AS and SJS are members of the current Working Party for the Association of Anaesthetists guideline on ‘Use of blood components and their alternatives’.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: All authors are co-investigators on the NIHR-funded T4P trial (NIHR131822).
