Abstract
Background and aims:
Sepsis is not only a leading cause of intensive care unit (ICU) admission but also one of the variables which affect outcomes of cancer patients. We aimed to assess the clinical characteristics, clinical course, mortality and risk factors associated with 30-day mortality in medical oncology patients admitted in a multi-disciplinary medical ICU.
Methods:
We conducted a retrospective analysis of 435 consecutive cancer patients admitted in medical ICU over a 28 months period. Patients were divided into two groups based on the presence of sepsis at the time of ICU admission. Data regarding baseline patient characteristics, clinical and laboratory data, need for organ support and 30-day mortality were collected. Sepsis patients were further classified as 30-day survivors and non-survivors and risk factors for mortality in these patients were determined.
Results:
Overall 30-day mortality was 57.8%. It was significantly higher in sepsis group patients (73.9%) as compared to non-sepsis patients (46.6%) (p < 0.001). Most common reason for ICU admission in non-sepsis group was respiratory distress (51.4%) followed by altered sensorium (28.4%). Presence of metastasis [odds ratio, OR: 3.89 (95% confidence interval, CI: 1.536–9.901)], high lactate [OR: 1.374 (95% CI: 1.024–1.843)] and need of invasive mechanical ventilator (IMV) support [OR: 7.634 (95% CI: 2.519–23.256)] or vasopressor support [OR: 3.268 (95% CI: 1.179–9.090)] were directly associated with 30-day mortality.
Conclusion:
Critically ill cancer patients admitted with sepsis had high mortality. Presence of metastasis, high lactate and need of IMV or vasopressor support was associated with worse prognosis in cancer patients admitted with sepsis in ICU.
Introduction
Sepsis is a major health problem worldwide and is associated with high mortality rates. The incidence rate of sepsis varies from 13.6% to 39.3% in the different regions of world. 1 The risk of sepsis in cancer patients has been estimated to be 10 times higher than that for non-cancer patients, with variations among cancer subtypes. 2 The risk is 7–8 times higher for haematological malignancies as compared to solid tumours. 3 Factors causing increased risk of sepsis in cancer patients include the qualitative and quantitative defects in neutrophils due to myelo-suppression and adverse effects of cancer therapy, non-selective cytotoxic activity of chemotherapy, infections from medical devices, such as stents, catheters, immunomodulatory effects of cancers, post-operative status, nutritional deficiencies and invasive intravascular access devices.
Mortality rates in septic patients range from 19.3% to 47.2%. 1 Cancer patients experience prolonged lengths of stay and higher morbidity and mortality as compared to general patients with sepsis. Sepsis patients with underlying neoplastic disease have a 30% higher risk of death, compared to non-cancer patients with sepsis. 4 Sepsis is not only a major cause for acute deterioration of cancer patients necessitating intensive care unit (ICU) admission, but also an independent predictor of mortality in these patients. 4 This increased risk of death may be precipitated by opportunistic infections, cytokines production by tumour, or by worsening organ dysfunction secondary to localised obstruction. 5
Most of the studies in critically ill cancer patients with sepsis are done in developed countries, and there is a dearth of studies about clinical characteristics, outcomes and factors associated with increased risk of mortality in cancer patients with sepsis and septic shock from developing countries. There is also a lack of studies comparing ICU course and outcomes of sepsis and non-sepsis cancer patients. In addition, most of the earlier studies have used the systemic inflammatory response syndrome (SIRS) criteria to define sepsis patients rather than the more recent consensus definition. 6 Therefore, we aimed to assess the clinical characteristics, ICU course, and 30-day mortality in cancer patients admitted with sepsis in a specialised oncology medical ICU in comparison to critically ill non-sepsis cancer patients admitted during the same time.
Materials and methods
This study was a single centre retrospective cohort study conducted in a tertiary care centre in India. We have an advanced specialised oncology medical ICU setup with 24-h on-site intensivist coverage and state-of-the-art facilities for organ support including invasive mechanical ventilation, advanced haemodynamic monitoring, continuous renal replacement therapy and extracorporeal membrane oxygenation.
Data were analysed from the records of the adult patients admitted in the medical oncology ICU during the 28 months period from January 2018 to April 2020. We evaluated all adult patients (>18 years) with a definite diagnosis of cancer at ICU admission. If the patient was readmitted to ICU more than once during his/her hospital stay, only the first admission was included in the study. Patients who had ICU stay of less than 12 h, post-operative patients (oncology patients admitted for elective surgeries), those in complete cancer remission for more than 5 years and those admitted from or discharged to another ICU, were excluded from the study. Patients were divided into two groups on the basis of presence of sepsis at the time of ICU admission. Sepsis and septic shock were defined according to the latest surviving sepsis consensus definitions. 6 Sepsis group patients were further classified as survivors and non-survivors on the basis of mortality after 30 days and risk factors for mortality in these patients were determined.
Data regarding the baseline patient characteristics, clinical and laboratory data, including comorbidities, type of malignancy, presence of metastasis, need for vasopressor, renal and invasive mechanical ventilation (IMV) support, and 30-day mortality were collected in a pre-designed proforma. Sequential Organ Failure Assessment (SOFA) score and Acute physiology and chronic health evaluation II (APACHE II) scores were calculated from the data obtained on the day of ICU admission.
Statistical analysis
The collected data were transformed into variables, coded and then entered in Microsoft Excel. Data were analysed and statistically evaluated using IBM SPSS-PC-25 version. Quantitative data was expressed in median and interquartile range (IQR) and depending on normality distribution difference between two comparable groups were tested by student’s t-test (unpaired) or Mann Whitney ‘U’ test. Qualitative data were expressed in percentage and statistical differences between the proportions were tested by chi square test or Fisher’s exact test, as appropriate. Univariate analysis and multivariable logistic regression were used to identify factors associated with 30-day mortality. Odds ratio (OR) along with respective 95% confidence intervals (CIs) were calculated. p Value <0.05 was considered statistically significant.
Results
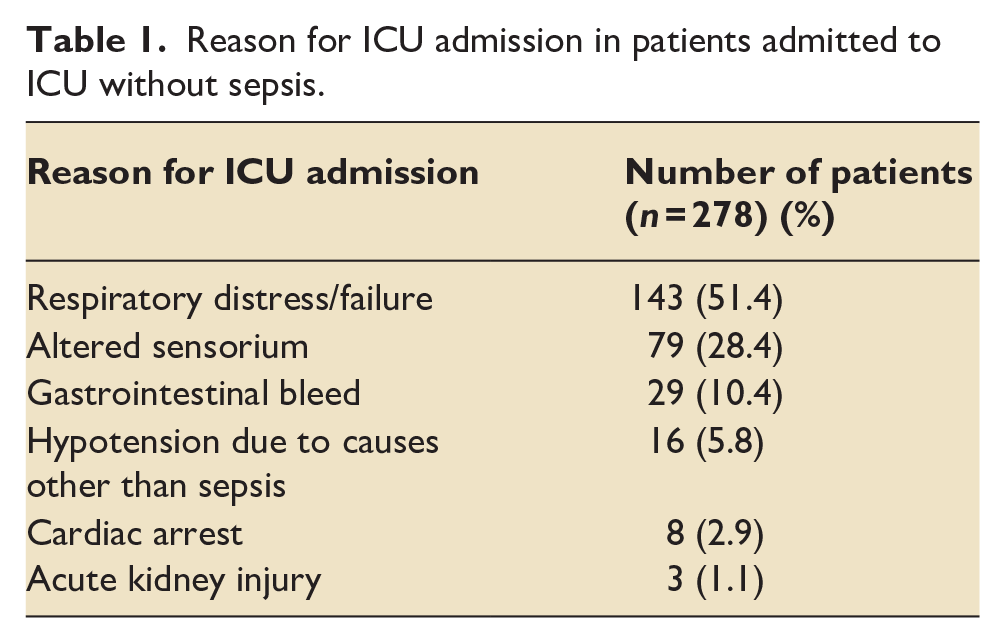
Data were analysed from 435 patients who fulfilled the inclusion criteria. Out of 435 patients, 157 (36.1%) patients were admitted in ICU with sepsis (sepsis group) and 278 (63.9%) patients were admitted in ICU with diagnosis other than sepsis (non-sepsis group). Reasons for ICU admission for patients admitted to ICU without sepsis are given in Table 1. Baseline characteristics of sepsis group patients were compared with non-sepsis group patients as given in Table 2. Overall, the 30-day mortality rate was 58.4%. It was significantly higher in sepsis group patients (73.9%) as compared to non-sepsis group patients (46.6%) (p value < 0.001).
Reason for ICU admission in patients admitted to ICU without sepsis.
Comparison of baseline parameters and outcome variables between sepsis and non-sepsis groups.
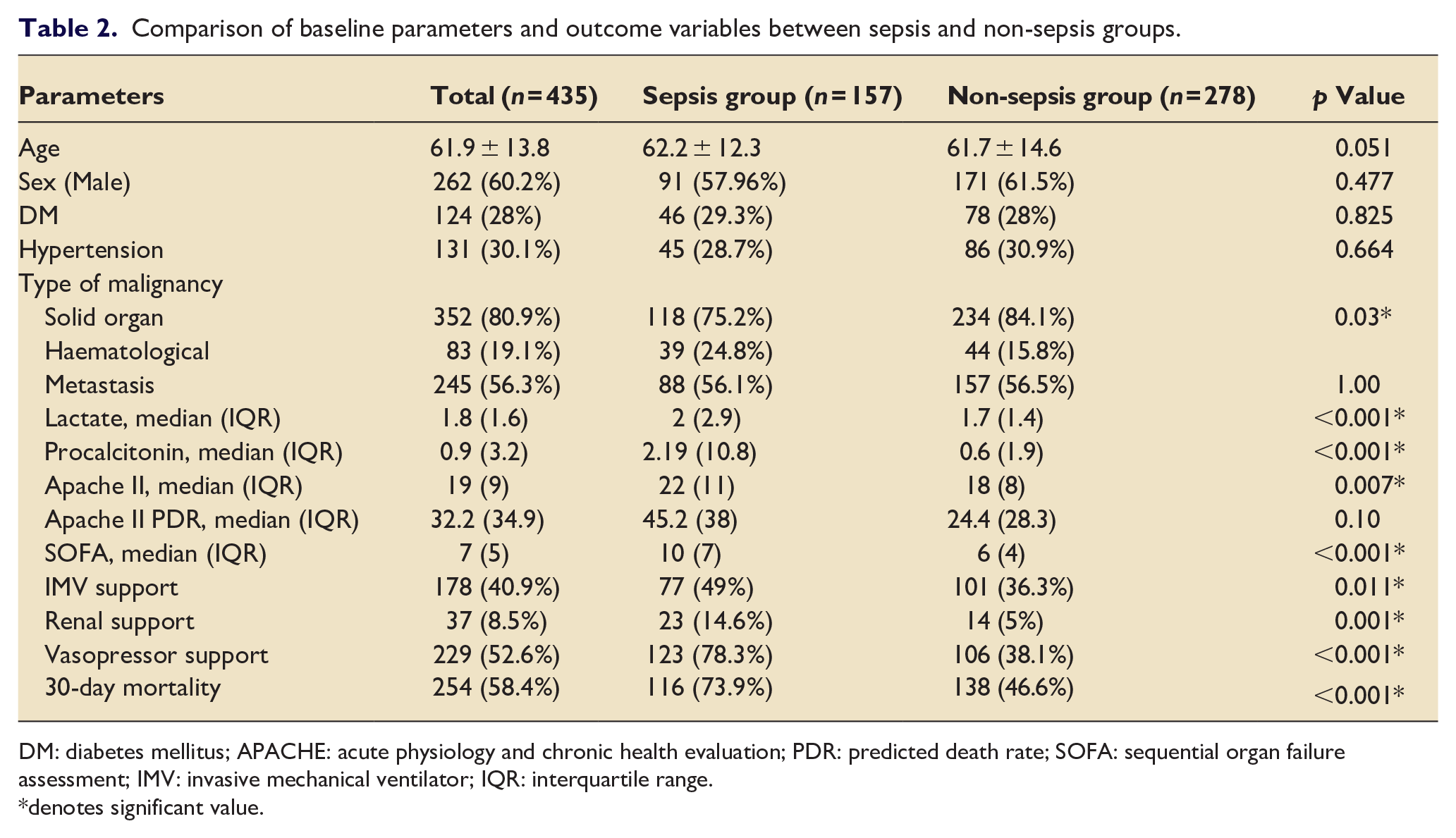
DM: diabetes mellitus; APACHE: acute physiology and chronic health evaluation; PDR: predicted death rate; SOFA: sequential organ failure assessment; IMV: invasive mechanical ventilator; IQR: interquartile range.
denotes significant value.
Comparison between survivors and non-survivors among the patients admitted with sepsis is shown in Table 3. Out of 157 patients with sepsis, 81 (51.6%) patients had septic shock at the time of ICU admission. However, presence of septic shock at admission was not associated with 30-day mortality. Seven factors were associated with higher 30-day mortality, these included absence of DM, metastasis, higher lactate, procalcitonin and APACHE II score and need of IMV support or vasopressor support during ICU stay. These factors were then analysed by multivariate analysis and only four factors; presence of metastasis, lactate and need of IMV support or vasopressor support, were found to be significantly associated with 30-day mortality (Table 4).
Comparison of baseline parameters and outcome variables between survivors and non-survivors in patients admitted with sepsis using univariate analysis.
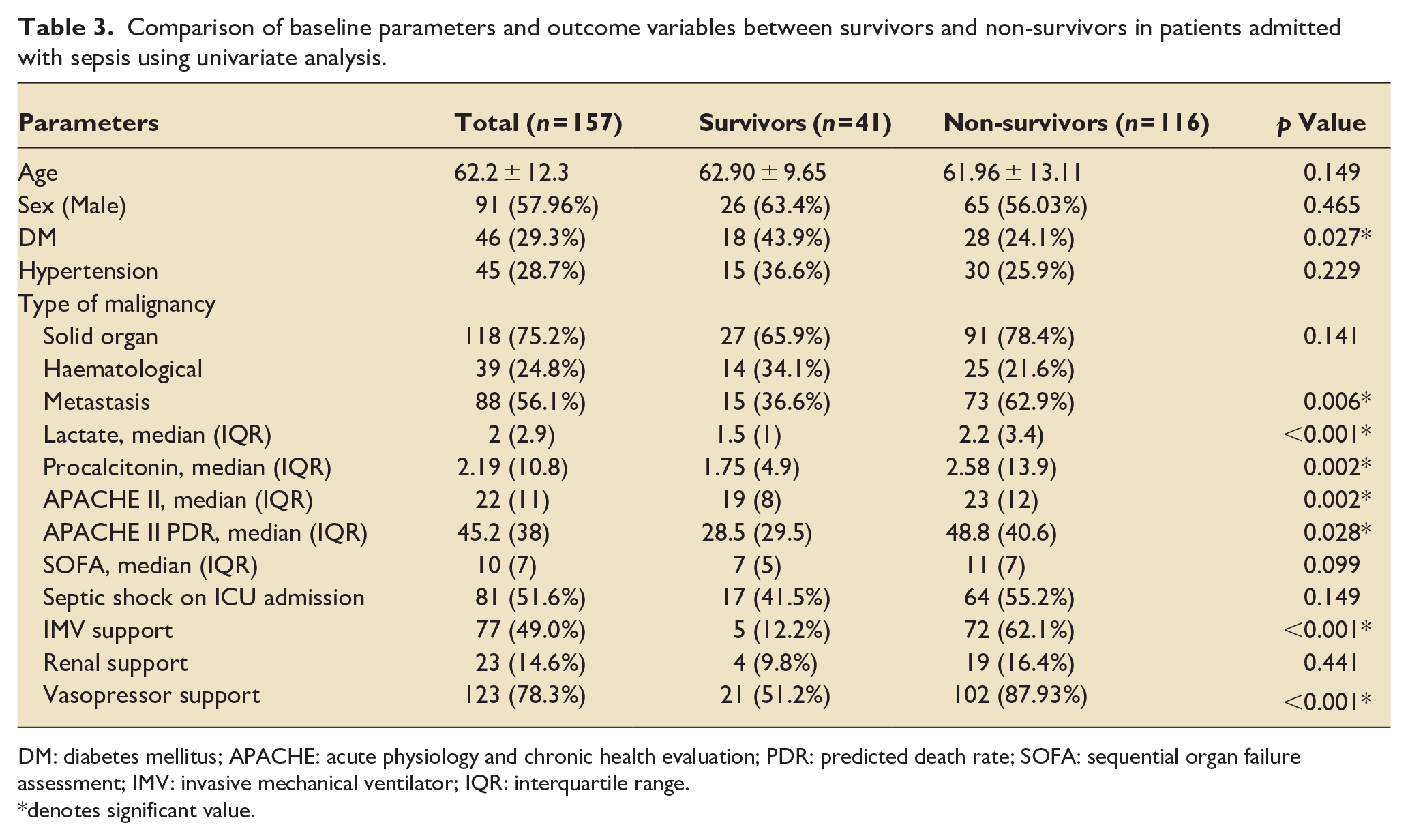
DM: diabetes mellitus; APACHE: acute physiology and chronic health evaluation; PDR: predicted death rate; SOFA: sequential organ failure assessment; IMV: invasive mechanical ventilator; IQR: interquartile range.
denotes significant value.
Multivariate analysis for 30-day mortality in oncology patients admitted with sepsis.
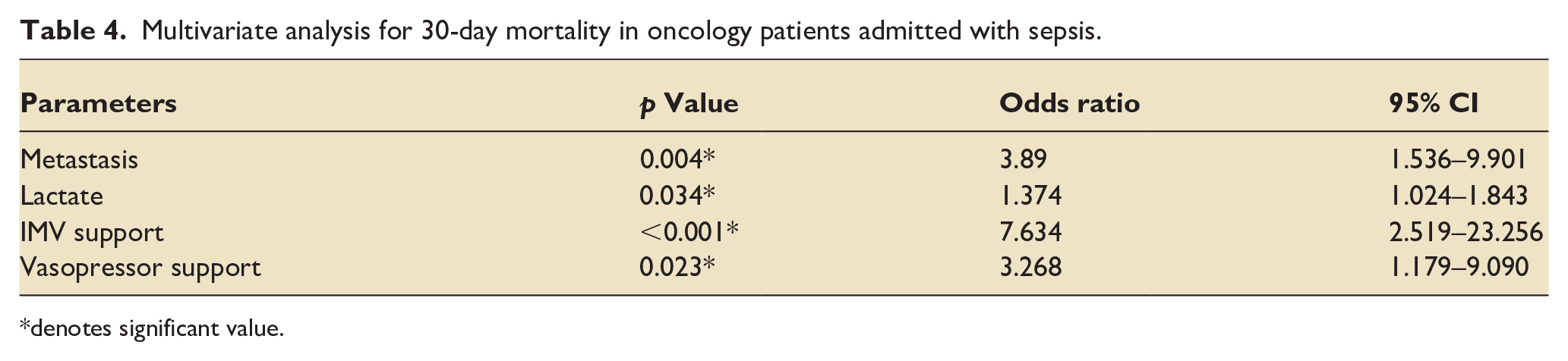
denotes significant value.
Discussion
Progression of disease or complications associated with cancer or chemotherapy may necessitate ICU admission of cancer patients. Even with advancements in cancer therapy and intensive care management, outcomes of these patients remain poor, and physicians may be sceptical for ICU admission of critically ill cancer patients because of poor their survival rates. Hence, it is imperative to identify risk factors for poor outcomes at the time of ICU admission, which may help us in initiating early aggressive care, for prognostication and also triaging of limited ICU resources.
Sepsis is not only a leading cause of ICU admission but also one of the variables which affect outcomes of the cancer patients. In our study of oncology patients, sepsis was the leading cause for ICU admission constituting 36.1% of ICU admissions. As compared to patients with other causes for ICU admission, oncology patients with sepsis had a higher incidence of haematological malignancies, higher levels of serum lactate and procalcitonin, higher severity of illness score and need for organ support.
Our overall 30-day mortality of 58.4%, was in accordance with previous studies who have reported the mortality between 30%and 77% in critically ill cancer patients.7–9 The 30-day mortality rate in our patients admitted with sepsis was 73.9%, which was significantly higher from the mortality rate of 46.6% observed in patients requiring admission for other causes. Other authors have also reported high mortality rates in similar patient cohorts.5,10A few studies reporting lower mortality rates have used older sepsis definitions,6,11,12 which has been shown to underestimate mortality. 13 As our patients were classified according to the recent sepsis-3 definition, it could explain higher mortality. In addition, our patient cohort had high rates of metastasis (56%), as compared to other studies (30%) 12 which again could be a cause of higher mortality rate. Presence of metastasis was significantly associated with 30-day mortality in our study. Previous studies have also shown that presence of metastasis is associated with poorer outcomes in critically ill cancer patients.12–14
Several risks factors have been recognised which may increase the risk of mortality in cancer patients admitted to ICUs. These include haematological malignancies, presence of metastasis, non-curative setting, severity of illness scores and need for organ support during ICU stay.12–18 In our study, presence of metastasis, high lactate levels and need for IMV or vasopressor support were associated higher 30-day mortality.
Lactate levels could be assessed at the bedside via point-of-care testing. High lactate level is an indicator of tissue hypoperfusion and a sign of organ dysfunction in critically ill patients. They have been shown to be associated with increased mortality in patients with suspected sepsis and sepsis.19,20 It is also an important component of septic shock definition, and is also useful in guiding resuscitation. 21 However, it is a non-specific biomarker and may be raised in many other clinical conditions in critically ill patients. 22 Cancer per se, is associated with abnormal lactate metabolism and hence, its utility in managing sepsis in cancer patients is not widely studied. Lactate was high in sepsis group patients as compared to non-sepsis group patients in our study cohort. It was also significantly raised in non-survivors as compared to survivors in patients with sepsis, which is similar to previous studies. It was directly associated with increased mortality in our sepsis group patients as shown in multivariate analysis and hence, it may be used as a prognosticating marker in cancer patients with sepsis.
Serum procalcitonin is considered a biomarker for the early diagnosis of bacterial infections. Its use as a marker for infection has been validated in numerous clinical situations, including cancer patients. However, it can be elevated when there is no infectious process, in particular in tumorous diseases like medullary thyroid cancer, lung cancer with neuroendocrine component or with liver metastases. It may also be a predictor of metastasis and advanced cancer in patients with no suspicion of BSI.23,24 In the present study, even though serum procalcitonin levels were significantly higher in sepsis group patients but were not independently associated with 30-day mortality.
Despite advances in management of sepsis, the outcome of cancer patients with sepsis is still dismal, and these patients have been reported to have two-fold higher mortality compared with non-cancer patients. Therefore, early recognition of poor prognostic markers in sepsis is the cornerstone to reduce mortality and morbidity in cancer patients. Judicious patient selection for ICU admission in critically ill cancer patients may improve the use of ICU resources by admission of patients who have favourable prospects for survival. Our results can help clinicians in triage processes for ICU admission and decisions related to patient’s care. Early palliative care has been shown to improve the quality of life in advanced cancer patients. A transition from restorative to palliative treatment upon failure should be taken in the ICU. However, the decision to make this transition depends on assessment of prognosis of patients.
Ours was a tertiary care private ICU with strict ICU admission and discharge policies where patients are admitted as per the discretion of the treating physician from the wards or the emergency room. Only those patients with advanced malignancy, on palliative care, who have ‘do-not-escalate’ or ‘do-not-intubate’ orders are not shifted to ICU. Care in ICU is primarily managed by the ICU physician with inputs from other specialities. Decision to discharge from the ICU is collectively taken by the ICU physician and the oncologist.
Limitations of our study include that our study was a single centre retrospective study so the data acquisition was prone to bias. We could not assess data on therapies for sepsis, control of sepsis source and processes of care. As a single-centre study our results may not be applicable to other centres with different populations which limit generalisation of our findings. SOFA score was higher in patients with sepsis, which could be understandably secondary to it being a part of clinical criteria to diagnose sepsis. Additionally, as we had used the recent sepsis-3 criteria for classifying patients, some of the patients in the non-sepsis group also may have had ‘infection not amounting to sepsis’. Also, we did not study the long-term outcomes of these patients.
Conclusions
To conclude, critically ill cancer patients admitted with sepsis have a higher mortality rate as compared to non-sepsis cancer patients. The presence of metastasis, high lactate level and need of IMV or vasopressor support may be associated with worse prognosis in cancer patients admitted with sepsis in ICU.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
