Abstract
Background
Current personal protective equipment (PPE) practices in UK intensive care units involve “sessional” use of long-sleeved gowns, risking nosocomial infection transmitted via gown sleeves. Data from the first wave of the COVID19 pandemic demonstrated that these changes in infection prevention and control protocols were associated with an increase in healthcare associated bloodstream infections. We therefore explored the use of a protocol using short-sleeved gowns with hand and arm hygiene to reduce this risk.
Methods
ICU staff were trained in wearing short-sleeved gowns and using a specific hand and arm washing technique between patients (experimental protocol). They then underwent simulation training, performing COVID-19 intubation and proning tasks using either experimental protocol or the standard (long-sleeved) control protocol. Fluorescent powder was used to simulate microbial contamination, detected using photographs under ultraviolet light. Teams were randomised to use control or experimental PPE first. During the simulation, staff were questioned on their feelings about personal safety, comfort and patient safety.
Results
Sixty-eight staff and 17 proning volunteers were studied. Experimental PPE completely prevented staff contamination during COVID-19 intubation, whereas this occurred in 30/67 staff wearing control PPE (p = .003, McNemar). Proning volunteers were contaminated by staff in 15/17 control sessions and in 1/17 with experimental PPE (p = .023 McNemar). Staff comfort was superior with experimental PPE (p< .001, Wilcoxon). Their personal safety perception was initially higher with control PPE, but changed towards neutrality during sessions (p < .001 start, 0.068 end). Their impressions of patient safety were initially similar (p = .87), but finished strongly in favour of experimental PPE (p < .001).
Conclusions
Short-sleeved gowns with hand and forearm cleansing appear superior to sessional long-sleeved gowns in preventing cross-contamination between staff and patients.
Background
Since the early stage of the COVID-19 pandemic, the safety, availability and accessibility of Personal Protective Equipment (PPE) has been a priority for healthcare professionals worldwide. National PPE guidance from infection control experts, including UK Health Security Agency, has evolved as our understanding of the epidemiology and pathophysiology of COVID-19 has grown, and has been largely based on potential routes of transmission.1,2 Whilst droplet spread may predominate, ‘Aerosol Generating Procedures’ (those that produce airborne particles, which can travel over a distance and cause infection if inhaled) mandate healthcare professionals to take a higher level of protection, ‘Level 2 PPE’. 3 This includes a filtering face piece 3 respirator (FFP3), visor, long-sleeved gown, apron and disposable gloves. Whilst facial protection is fairly consistent, the types of gown and the number of gloves worn varies considerably. Guidance is not definitive and there is scanty evidence for ‘best practice’.
As the mode of transmission via the respiratory tract has become clearer, advice on the protection required has been reduced from that of High Consequence Infectious Diseases such as Ebola. However, problems in PPE provision have generated understandable fear amongst healthcare professionals of contracting the virus through occupational exposure, making PPE a focus of significant staff anxiety and psychological stress throughout the pandemic. This makes any proposed changes in guidance which appear to ‘downgrade’ PPE difficult to implement.
During wave 1 of the COVID-19 pandemic, UK intensive care unit (ICU) COVID19 patients experienced increased rates of healthcare associated infections (personal communication, P Dean, PPE lead for National Emergency Committee for Critical Care, UK). Infection control experts suggested that sessional use of long-sleeved gowns by staff looking after multiple patients in cohorted areas was likely a significant contributory factor. In addition, due to adherence to PPE protocols, staff were not removing gloves and washing their hands between patient care episodes, which should be standard practice. One hospital reported a decrease in hand hygiene compliance and an increase of measured surface gram-negative bacteria (GNB) in the COVID-19 critical care area when staff were wearing long sleeved gowns sessionally. Subsequently the hospital changed its PPE guidance policy to replace long-sleeved gowns with short sleeve gowns. 4
A team of infection control, human factors, intensive care and education experts has collaborated to develop a new protocol for Level 2 PPE, aiming to reduce the risk of healthcare staff mediated transmission of multi-resistant organisms. The team recognised that any change in PPE must be demonstrated to be equally as effective at protecting staff from infection as current practice, and that the comfort and psychological safety of staff was important, particularly when PPE was worn for extended periods. Any changes must therefore be made adopting a Human Factors approach, taking into consideration perceived as well as actual changes in protection and hence feelings of vulnerability. For any change to be effective, engagement of staff in the understanding and the process of change is as important as the data obtained from the study. A simulation-based experiment was therefore designed, allowing us to compare the effectiveness of two Level 2 PPE protocols (control and experimental) at protecting patients from contamination through staff and patient contact and protecting staff from ‘residual contamination’ after doffing.
Methods
Objectives
The primary objective was to develop and test new protocols for Level 2 PPE, specifically designed to reduce the spread of pathogens between staff and patients in ICU, whilst maintaining a level of protection from COVID-19 equivalent to current protocols.
Outcome measures
The primary outcome of this study was contamination of staff and simulated patients with fluorescent powder when staff wore modified (experimental) Level 2 PPE in comparison to the current (control) Level 2 PPE.
The secondary outcomes were staff perceptions of comfort, personal safety and patient safety, when wearing experimental versus control PPE.
Study design
We conducted a dual-centre, non-blinded randomised cross-over trial in Oxford University Hospitals Foundation Trust (OUHFT), Oxford and Whittington Hospital, London. This was preceded by an uncontrolled cohort study of the development of the new PPE protocol, based on the recommendations for IDEAL Stage 2a and 2b studies. 5
Ethical approval for this trial was obtained from Oxford University Research Ethics Committee (CUREC) (Reference: R72882/RE001). The study was registered on clinicaltrial.gov (identifier: NCT04712045).
Participants were nurses, physiotherapists or doctors working in Adult Intensive Care Units at Oxford University Hospitals Trust and Whittington Health NHS Trust. Participants were recruited by an email invitation. Prior to enrolment, written informed consent was obtained, including specific reference to use of photography and video recording.
Development of Level 2 PPE protocol
Guided by infection prevention and control experts, the study team agreed on transitioning from a long sleeve to a short sleeve gown and from double gloves to single gloves. No changes were made to facial protection.
The new protocol stipulates that after any patient interaction, staff should remove their plastic apron and gloves, wash and dry their hands and forearms and don a new apron and gloves prior to attending another patient. This process was termed a ‘mini-doff’, to differentiate from the ‘full doff’ undertaken when leaving a COVID-19 area.
Prototype PPE doffing and mini-doffing protocols were designed and revised through an iterative co-design process following the recommendations for IDEAL Stage 2a studies
5
(see Figure 1). ICU staff were invited to test the prototype protocols, feeding back to the study team on their comfort and perceived safety and the ease of use of visual aids, until a stable version was established. Prototype Level 2 personal protective equipment doffing and mini-doffing guidance posters used during the simulation.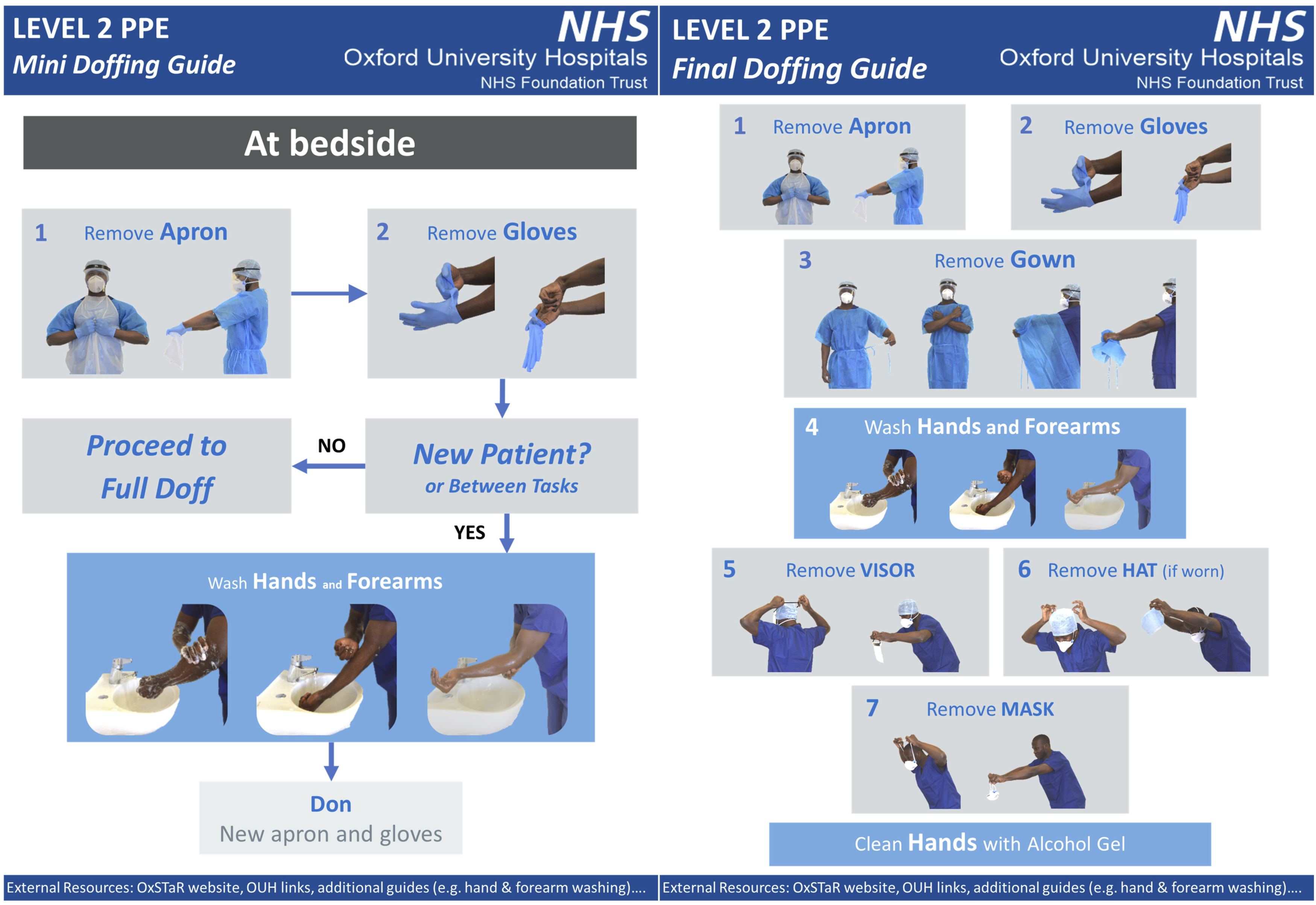
The performance of the two PPE protocols was evaluated right from the design phase with the use of an ultraviolet tracer in powder form. The powder (Glogerm, www.Hygienicsolutionsuk.com), which glows white under ultraviolet light, was a surrogate for pathogens which may be spread between staff and patients by physical contact (including COVID19 and hospital acquired multi drug resistant organisms). For the purpose of this study, the goal was complete decontamination of individuals using the proposed Experimental PPE with the associated doffing and mini-doffing techniques, to demonstrate that the protocol offered equivalent protection to staff from COVID19 as the control PPE protocol currently used. To achieve this required a hand and forearm washing technique that would completely remove any trace of the ultraviolet powder. During the design phase, the proposed Experimental PPE and doffing and mini-doffing protocols were shown to be effective at achieving complete decontamination, provided they were employed meticulously. It proved important to use stroking movements from the elbows distally, and not in the reverse direction, during both washing and drying, to ensure that contamination was not displaced above the elbows during the cleaning process. The inclusion of the forearms in the hand hygiene process was an addition to the standard WHO hand hygiene protocol, 6 not a replacement of this process. The hand and forearm washing technique taught to all the study participants and used throughout the study is demonstrated in this supplemental video.
To avoid bias due to learning curve effects, all subjects for the randomised study underwent a preliminary training session involving demonstration and repeated practice of both current and experimental PPE doffing and mini-doffing protocols (including the hand and forearm washing with the experimental protocol), until competence was demonstrated in both, following the principles of IDEAL Stage 2b studies. 5
Level 2 PPE content and protocols
Level 2 PPE Control
The ‘Control’ PPE protocol (current practice) in OUH consisted of FFP3 mask, hat, visor, long sleeved gown, plastic apron, long gloves (inner) and short gloves (outer). In the Whittington hospital single gloves were used. The apron and one pair of gloves were changed between patients.
Level 2 PPE experimental
The ‘Experimental’ PPE protocol consisted of FFP3 mask, hat, visor, short sleeved gown, plastic apron and short gloves (single pair). The “mini-doffing” protocol between patient contact included removal of apron and gloves, thorough washing of hands and forearms and donning of a fresh apron and gloves. The handwashing technique emphasised the need to cleanse and dry the arms with the hand hanging down, and all movements directed from the elbow distally, (see above).
Randomised study
After training, participants were divided into groups of 4 (5 in the Whittington), including at least one airway-trained doctor. Each group performed 2 simulated activities (COVID-19 intubation and proning) twice: once with control PPE and once with experimental PPE. The order in which the protocols were used was randomised in permuted blocks of four sessions, using a computer-generated random number method.
These tasks were selected to maximise ‘patient’ contact and prepare staff entering the second wave of the pandemic. They were conducted and debriefed according to usual simulation practice incorporating the procedural checklists of their respective hospitals
Simulation task 1 was oral endo-tracheal intubation of a simulated patient (a mannequin), with respiratory failure, secondary to COVID-19 pneumonia. Staff were also asked to change the patient’s gown and sheet to increase contact with the mannequin.
Simulation task 2 was turning a simulated patient (an actor) from the supine position (lying on back) into the prone position (lying on front), a standard therapy for patients with refractory hypoxaemia. Prior to proning, staff were asked to change the undersheet to increase contact with the patient.
The proning guideline requires five staff, so a member of the research team joined the OUH teams to facilitate this task, but was tasked with holding the patient’s airway during the proning and was not included in any data collection.
A powder (Glogerm, www.Hygienicsolutionsuk.com), which glows white under ultraviolet light, was used to compare the effectiveness of the control and experimental Level 2 PPE. The powder represented contamination of staff and patients with COVID-19 and organisms which may be spread by physical contact.
Bespoke plastic scoops (capacity 0.4 cm3) ensured standardisation of the quantity of powder applied by the study team, which was determined during the IDEAL Stage 2a development study. The dose chosen for Task 1 was designed to detect any transfer of powder to staff, but to avoid any visually detectable powder residue on the mannequin. The higher dose used in Task 2 was designed to ensure that persistent contamination would be detectably transferred from staff to volunteer patients and was clearly visible to staff in both Experimental and Control groups.
Prior to simulation task 1, powder was applied to the simulated patient (mannequin) - 2 scoops to the head, 3 to the ventral aspect of the arms, 2 to the chest, 1 to the gown and 2 to the blanket.
Prior to simulation task 2, powder was applied to the arms of the participants – 3 scoops to the ventral and 3 to the dorsal aspects of both hands and arms.
Power calculation
Sample size calculations were based on data analysis using the mid-P version of the exact McNemar’s test. Power was calculated by finding the percentage of 100,000 simulated datasets for which the null hypothesis would be rejected at the 5% level of significance with 90% power. This indicated that a minimum of 50 participants would be required.
Conduct of simulation sessions
The simulation sessions began with a briefing and mannequin introduction and proceeded according to the flowchart (see Figure 2). Simulation flow chart.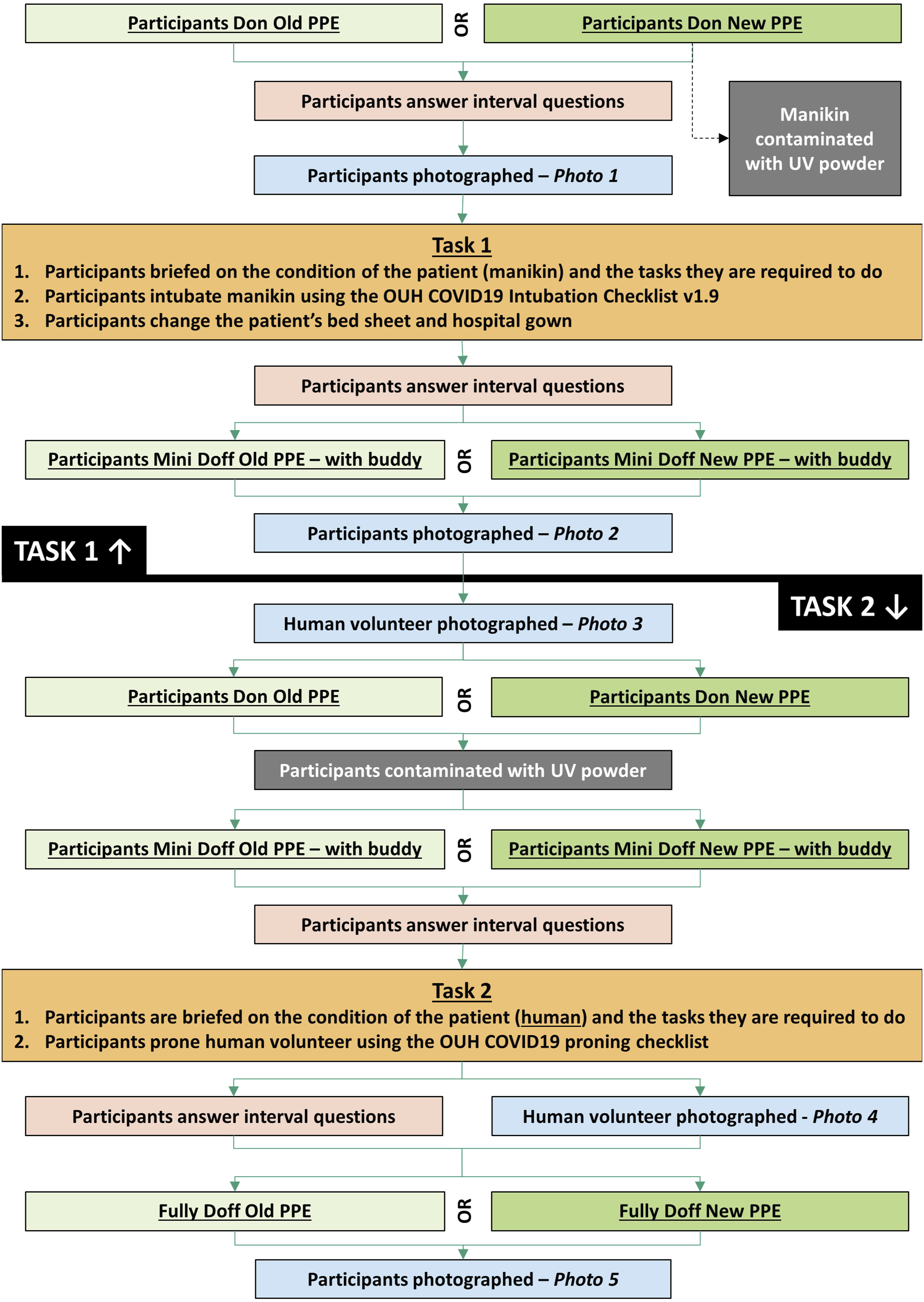
Participants donned the Control or Experimental Level 2 PPE (according to randomisation). A photograph was taken under UV light to assess for baseline contamination (expected to be zero). They completed Simulation Task 1 (intubation), then performed a ‘mini-doff’ and were photographed again, to assess the extent of contamination on their hands (or gloves) and forearms (or sleeves).
Having donned new apron and gloves, powder was applied to the participants forearms and they were asked to ‘mini-doff’ again, and don new apron and gloves, as if moving between patients. They completed Simulation task 2 (proning), and then undertook a ‘full doff’. A 3rd photograph was taken following this to assess for residual contamination. The 'proned’ actor wore fresh scrubs for each proning and was photographed before and after proning to assess for contamination.
A simulation debrief was performed after task 2, and the whole process was then repeated wearing the other PPE protocol.
Data collection
Questionnaires
Simple repeated questionnaires were used at 4 points during the simulation sessions: 1. Immediately after donning PPE; 2. Immediately after Task 1; 3. Immediately before Task 2; and 4. Immediately after Task 2. The questionnaires evaluated participants’ feelings about their personal safety, physical comfort and the safety of their “patient” on a 10-point Likert scale (See APPENDIX A).
Photography
Participants were photographed twice on each encounter, first facing forward with arms up and palms facing forward and second facing forward with palms facing backwards. The ‘proned’ patient was photographed before and after each proning exercise, anteriorly and posteriorly.
The camera, light and subject positions were standardised to ensure consistent data was obtained. The camera was positioned to allow the arms and torso to be captured. The UV lamp was placed close to camera height and as far from the participant as possible. Positioning markers for feet and index fingers ensured participants stood in exactly the same place and position in front of a black screen with a black and white chequerboard attached to facilitate analysis. It was important to photograph participants in the same order each time to aid identification for analysis.
Data Analysis and Statistics
Photographs were assessed by two independent observers, who then conferred to reach agreement on any discordant results. For participant photos, attention was focused on the forearm, where the two protocols differed. For the proning volunteer the whole body was studied. Photos were assessed as either showing contamination or not. Differences in the percentage of participants showing contamination were evaluated using McNemar’s test to compare baseline (pre-activity), post-intubation and post-full doffing contamination for the Experimental and Control PPE protocols in a modified intention to treat analysis.
Staff impressions of comfort, personal safety and patient safety were compared using a Wilcoxon Signed Rank test to explore the differences between the Experimental and Control groups at each time interval. Changes in staff views between the beginning and end of the study were evaluated using both Pearson and Spearman (ranked) correlation between score and task number. All tests were 2 sided.
Results
Ideal studies
The IDEAL stage 2a study was carried out using research team members and staff volunteers. Ideas to improve the protocol were tested in 4 sessions resulting in 5 iterative changes: 1. Washing all the way to the elbows was emphasised, as initially it was not consistently completed. 2. A surgical scrubbing technique was tried, but abandoned, as it left a ring of contamination just above the elbows. 3. The sleeves of the short-sleeved gown were rolled up to ensure they did not get wet. 4. A technique involving rinsing and drying from the elbows downwards towards the hands was adopted to deal with the elbow contamination and achieved 100% removal of contamination when properly completed. 5. Several iterations of the visual aids were produced until a satisfactory version was achieved.
The IDEAL Stage 2b study was conducted informally, assessing the performance of all participants during training in doffing and donning. Errors in washing technique decreased rapidly with practice and coaching and were virtually eliminated in most participants after 3 attempts. No formal analysis of learning curves was attempted.
Randomised trial
Thirteen simulation sessions were held at OUH and 4 at the Whittington. Four participants dropped out of sessions at OUH and had to be substituted by research team members, leaving 48 participants at OUH and 20 at the Whittington (who used a team of 5 people per session). Each session involved an actor who was proned for the proning task; 13 at OUH and 4 at the Whittington. All proned patient data was used in the analysis.
Contamination results
Sixty eight participants and 17 proned actors were studied. One participant in the Control group was excluded as no photos could be found except the baseline picture, leaving 67 participants for analysis (see Figure 3). Two participants had detectable contamination at baseline wearing the Experimental PPE, and none wearing the Control PPE. After the Intubation scenario 30 of 67 participants had detectable contamination wearing the Control PPE (on the sleeves of the retained gown) and none wearing the Experimental PPE (p < .001, McNemar). After proning by participants wearing Control PPE, contamination of the ‘patient’ was detected in 15 of 17 episodes, compared to 1 of 17 episodes for the Experimental PPE (p < .001, McNemar). At final doffing, 18 of 67 participants in the Experimental group showed contamination compared to 7 of 67 in the Control group (p = .012). Figure 4 shows examples of contamination of a participant post intubation using the Control PPE (A), a participant post proning (B), and the proned ‘patient’ post proning (C and D). Results of the presence of contamination as shown by UV photography. The error bars show the 95% confidence interval for the percentage contamination, calculated by Wilson’s method. Example photos of participants using Control personal protective equipment: (a) - contamination of participant post intubation, (b) - participant post proning, (c) and (d) – ‘patient’ post proning.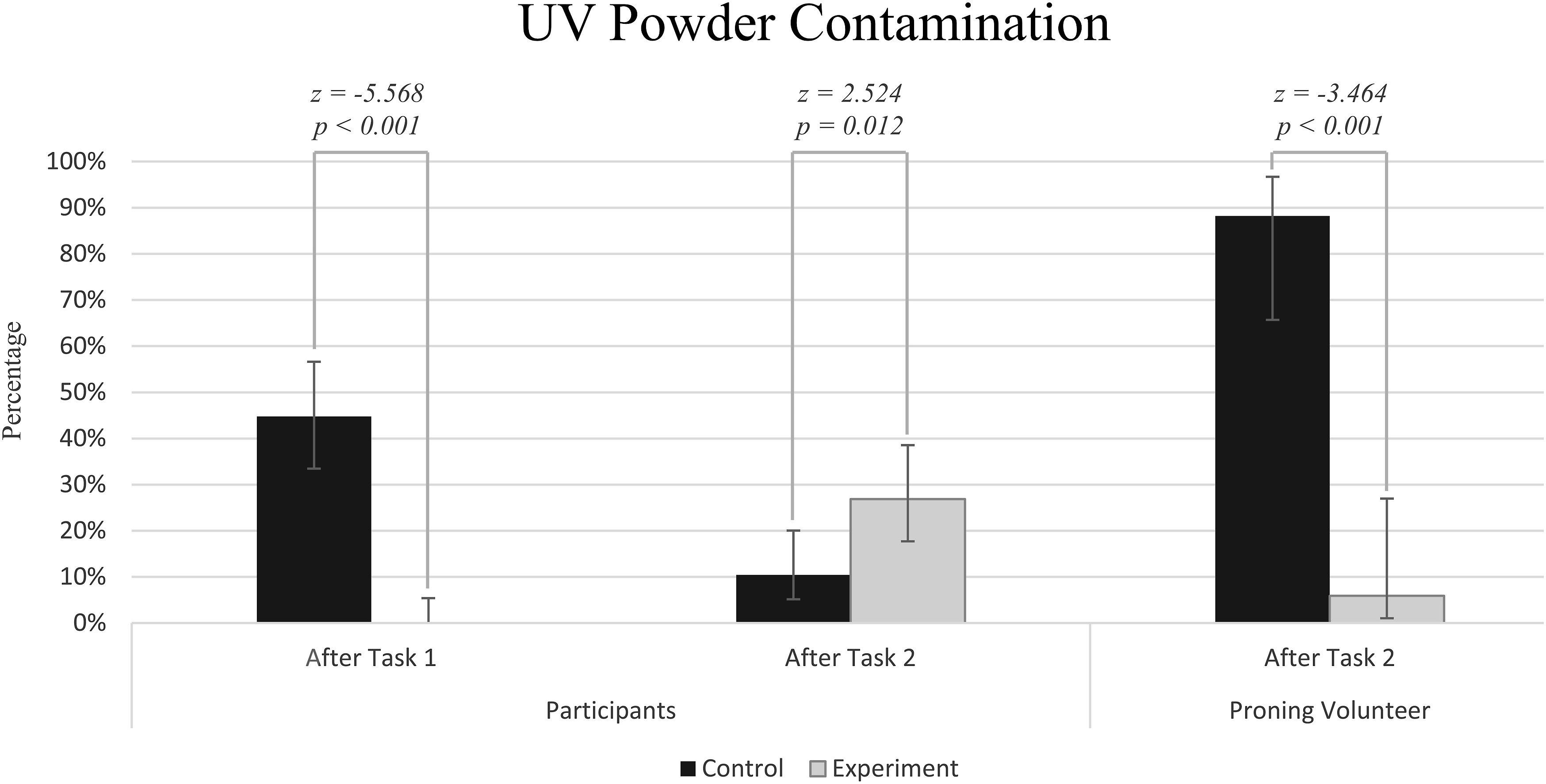

Questionnaire results
Results from the statistical analysis of the questionnaire data are presented in Figure 5. Feelings of personal comfort were consistently better in the Experimental PPE than in the Control PPE (p < .001, Wilcoxon, for all conditions). Personal safety was scored lower with the Experimental PPE before and after the first task (p < .001, Wilcoxon), but the difference between the groups decreased as participants gained experience of the Experimental equipment (p = .002, Spearman). The participants’ impression of patient safety remained consistent for the Experimental PPE with no discernible difference in the first half of the simulation (before task 1 p = .952, after task 1 p = .004, Wilcoxon), whereas the impression of patient safety for the Control PPE decreased throughout, with the Experimental PPE strongly preferred from by the end of the session (p < .001, Spearman). Results of the Physical Comfort, Personal Protection and Patient Protection Impression Questionnaire.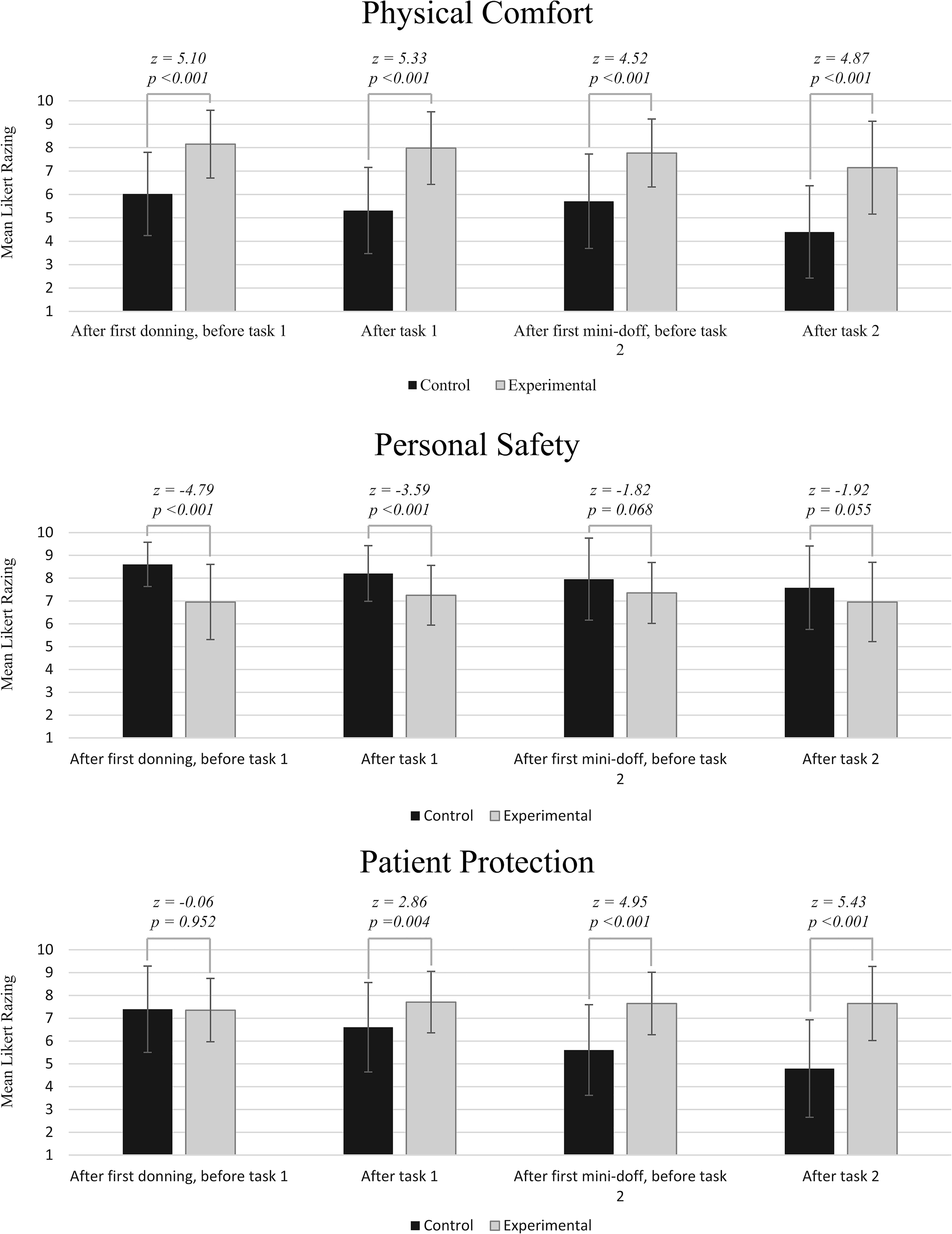
Discussion
Our study suggests that use of short sleeved gowns, with hand and forearm hygiene between patients, is as effective as sessional use of long-sleeved gowns at avoiding staff contamination, and could considerably reduce transmission of nosocomial infection between patients.
A simulation study was performed due to staff concerns, ethical concerns and the statistical power challenges of a study in a clinical setting. An initial development and learning phase based on the IDEAL Framework was used to avoid bias or disruption of the study due to learning curve effects or experimental protocol changes. Randomisation of the order that Level 2 PPE was worn was performed because teams were faster and more accurate on their second attempt, a well-recognised phenomenon in simulation work. 7 The simulation scenario learning objectives fulfilled the immediate educational priorities for ICU staff entering the 2nd wave of the COVID-19 pandemic and the format was consistent with our usual practice, led by an experienced trained facilitator.
The results demonstrated a very large reduction in the probability of acquired contamination on the forearm and hand areas for the Experimental PPE protocol after the ‘mini-doff’ had been performed, compared with the Control PPE protocols using long-sleeved gowns. The proning simulation confirmed that staff contamination was readily transferred to patients during procedures requiring physical contact, and was greatly reduced (from 88% to 6% of simulated pronings) using the Experimental PPE protocol. The contamination at baseline of 2 participants in the Experimental PPE group is likely to have come from these individuals’ participation in the previous set of tasks or from self-contamination from the study environment. Given that no contamination was detected on any participants in the Experimental PPE group after the first task and mini-doff were complete, this baseline contamination was clearly removed during the hand-arm washing process and hence would not have influenced the results. The discordant finding that staff wearing Experimental PPE had more residual contamination following the final full doff after the proning exercise than with Control PPE seems likely to be an artefact caused by the combination of three factors. First, an excess of powder was deliberately used in the proning task to ensure that any transfer from staff to patients was detected. Second, proning requires staff to push their forearms under the patient, potentially pushing any residual powder up towards the elbow. Third, as noted above, if the washing technique was not performed carefully, contamination above the area washed was commonly found. All photographs of the 18 cases of participant contamination post-proning with Experimental PPE, showed a ring of contamination at the elbow, with only very slight contamination elsewhere in one or two cases. This was consistent with the experience of the research team during development of the hand and arm washing protocol, whereby the elbow was the site that was more challenging to clear completely of powder and led to the technique whereby arms were washed with the hands in the dependent position and with water and soap applied from the elbow distally. We do not know the extent to which the behaviour of the powder reflects that of a microorganism in terms of its propensity to be pushed along the skin surface. It should be noted that staff self-contamination was a secondary measure in this experiment, which was principally focused on evaluating transfer of contamination from the staff to the patient, and that, in the task specifically focused on contamination of staff by the patient, no cases of failure of the Experimental PPE regime occurred. Nevertheless, to establish the superiority of the Experimental protocol with respect to staff protection, may require further work. Human Factors principles suggest that reliance on precise attention to detail in routine but complex tasks, is likely to lead to significant failure rates.8,9 Although very satisfactory as a simulation exercise, the washing protocol used here as a demonstration might be impractical between patients in clinical practice in a busy ICU. Alcohol-based hand sanitisers are recognised as highly effective substitutes for soap and water cleansing,10–12 and might both reduce the time required to sanitise hands and arms and make the experimental regime less dependent on meticulous technique. However, since the effect of such sanitisers on the powder used in this study was to cause it to adhere more firmly to the skin, an alternative surrogate for the virus would be necessary for use as a tracer to evaluate the effectiveness of alcohol gel, such as a non-pathogenic organism whose viability could be tested after exposure.
Any regime which involves frequent application of either soap or alcohol to skin will cause problems for some staff with eczema or other skin conditions, and for these the continued use of long-sleeved gowns seems the only viable option.
There is clear evidence from both national data on bloodstream infections and outbreak studies, that the change in infection prevention and control protocols during the first phase of the pandemic, which included the sessional use of long-sleeved gowns, was associated with an increased incidence of invasive healthcare-associated bloodstream infections.13–16
Personal protective equipment is considered to be a key component of strategies to prevent transmission of infection, however, guidance for using it is largely based on theoretical rationale, because there is a paucity of epidemiological evidence to demonstrate its efficacy in protecting staff and patients. 17 PPE is recommended if exposure to infection cannot be adequately controlled by other, more effective measures in the Hierarchy of Controls e.g. elimination, engineering, administrative controls. However, as demonstrated by the adoption of the PPE routines for High Consequence Infectious Diseases for the care of groups of patients with COVID-19 in the early stages of the pandemic, there are practical challenges and dangers associated with its use which need to be considered when developing guidance. Transmission of COVID-19 occurs by direct or indirect contamination of mucous membranes with respired droplets or the inhalation of virus on respired aerosols. PPE is required to protect the oropharyngeal mucosa from exposure to respiratory particles. Other control measures, such as hand hygiene, are also important to prevent indirect transfer to mucous membranes. This study has demonstrated how PPE has the potential to act as a vector of infection between patients but that safer options are available. Staff comfort was significantly better wearing the Experimental PPE, and their confidence in both their personal safety and that of their patient improved steadily through the study. This suggests that testing the Experimental PPE in ICU settings is merited and introducing the new PPE via simulation training may overcome the reluctance of staff to adopt the change.
Limitations
The washing technique used was novel, having been developed during the IDEAL study prior to the randomised trial. Whilst relatively easily learned (see Methods and Results), there was a gap of several weeks in some cases between training and the simulation study, which could have resulted in poorer performance in those with a wider gap. Whilst fluorescent powder is widely used in research to simulate a range of particles, including microorganisms, it is not possible to determine the extent to which the powder reflects the movement of microorganisms. 18 However, this model clearly demonstrates that cross-contamination, both to and from PPE, is possible and provides a plausible explanation for the increased cross-transmission of a wide range of microbial pathogens which occurred between ICU patients during the COVID pandemic.
The fluorescent powder was white in ambient light and was visible when used in excess (Simulation Task 2) although more difficult to detect in the smaller dose used in Simulation Task 1. This could have influenced participants behaviours, making them more meticulous than usual in doffing and cleansing techniques. However, both control and study PPE groups were exposed to the same conditions so this should not have influenced the behaviour of one group over the other. The analysis of photographs could not be blinded, as the observer could see the gowns worn, but the binary choice between ‘any contamination’ and ‘none’ by independent observers was very clear and effectively objective. The Hawthorne effect, where subjects improve their performance when under observation, is unlikely to have affected our findings for the same reason.
A stepped wedge design trial to stage the introduction of the new PPE protocol clinically might facilitate both high quality evaluation of the new PPE protocol and rapid adoption. As a minimum evaluation requirement, any introduction of the new protocol should be prospectively evaluated as a cohort study using an IDEAL 2b format.
Supplemental Material
Footnotes
Acknowledgements
We thank Dr Katie Jeffery and Lisa Butcher (Microbiology & Infection Control, OUHFT) for helpful advice and Dr David Garry, Lyn Bennett and Trevor Venes (Clinical Lead, Matron and Nursing Education lead for ICU, OUHFT) for their strong support and help with recruitment. Thanks to the staff of the OxSTaR simulation centre (Oxford University). At the Whittington Hospital we thank Anezka Pratley (CRN), Ana Alvaro, Karen Johnston and the staff of the Simulation Centre and Julie Singleton (Lead Infection Prevention and Control Nurse) whose idea for using short-sleeved gowns in ICU informed this study. Thanks also to Professor Hugh Montgomery (UCH and Whittington Trust) for his encouragement and facilitation. The study was initiated in response to discussions at the National Emergency Critical Care Committee, where Peter McCulloch represents the Chartered Institute of Ergonomics and Human Factors.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This study was supported by the Oxford University Covid Response Fund. Dr Ibrahim is supported by a Maimonides Research Fellowship.
Supplemental Material
Supplemental material for this article is available online.
APPENDIX A
APPENDIX A1
APPENDIX A2
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
