Abstract

Introduction
Among patients admitted to hospital with COVID-19 in the UK, 10% develop severe hypoxemic respiratory failure managed with invasive mechanical ventilation (IMV). 1 Much interest has focused on non-invasive strategies to avert progression to IMV. UK guidelines recommend the use of continuous positive airway pressure (CPAP), including in patients for whom IMV is not appropriate. 2 However, other nations have recommended against the use of CPAP, 3 and within the UK, CPAP use has varied widely (personal communication).
The greatest burden of COVID-19 disease is carried by older patients with comorbidities, many of whom are deemed unsuitable for IMV and critical care. However, it is unclear whether they might benefit from CPAP. The RECOVERY-RS trial is investigating the efficacy of CPAP and high-flow nasal oxygen (HFNO) in severely hypoxic patients with COVID-19, but will not complete until late 2021, and excludes patients unsuitable for IMV. 4 Current evidence is limited to cohort studies of heterogeneous patient groups, with no published data focussing on patients for whom CPAP is the ceiling-of-care.5–7
Physicians caring for such patients, and those involved in planning the delivery of CPAP services, must balance any potential benefits of CPAP against its burden on patients, families, staff, and services. Therefore data in this patient population are urgently needed.
Methods
We included all patients commenced on CPAP as a ceiling-of-care for treatment of respiratory failure due to COVID-19, on the wards of two large UK hospitals. Data were collected during the first 2 months of CPAP provision for COVID-19 at these sites, (26/3/20 – 25/5/20). Patients were excluded if they were suitable for escalation to IMV, or received CPAP in a critical care setting.
Suitability for CPAP was based on local clinical guidelines; these recommended considering CPAP if FiO2 ≥40% (site 1) or ≥35% (site 2) was required to maintain SpO2 ≥92% (or 88% if risk of CO2 retention). Guidelines at both sites recommended assessing clinical frailty scale (CFS) to inform individualised decision making, with CFS ≤ 6 a suggested threshold for treatment. CPAP was delivered using air-driven SleepCube (DeVilbiss) and A40 (Philips Respironics) machines, with oxygen entrained to a face mask interface, as per national guidance. 2
Data about patient and CPAP factors, prior to and during CPAP treatment, were collected. Their relationship with 30-day mortality was tested for statistical significance using standard non-parametric methods. Institutional approval was granted to conduct this service evaluation, so research ethics approval was not required.
Results
70 patients were included; all were followed up to 30 days, or to hospital discharge if this was beyond 30 days. The median age was 76 years [IQR 69–80], with median CFS of 5 [IQR 4–6]. Initial CPAP settings used were pressure 5cmH2O [5–10] and O2 flow 10 L/min [10–15].
30 days after CPAP initiation, 21/70 (30%) patients were alive. Unadjusted comparison of demographic and clinical variables between survivors and non-survivors showed no statistically significant differences (Table 1). Changes in vital signs to 24 hours after CPAP initiation also showed no significant differences (Figure 1).
Patient demographics, comorbidities, investigations, and outcomes.
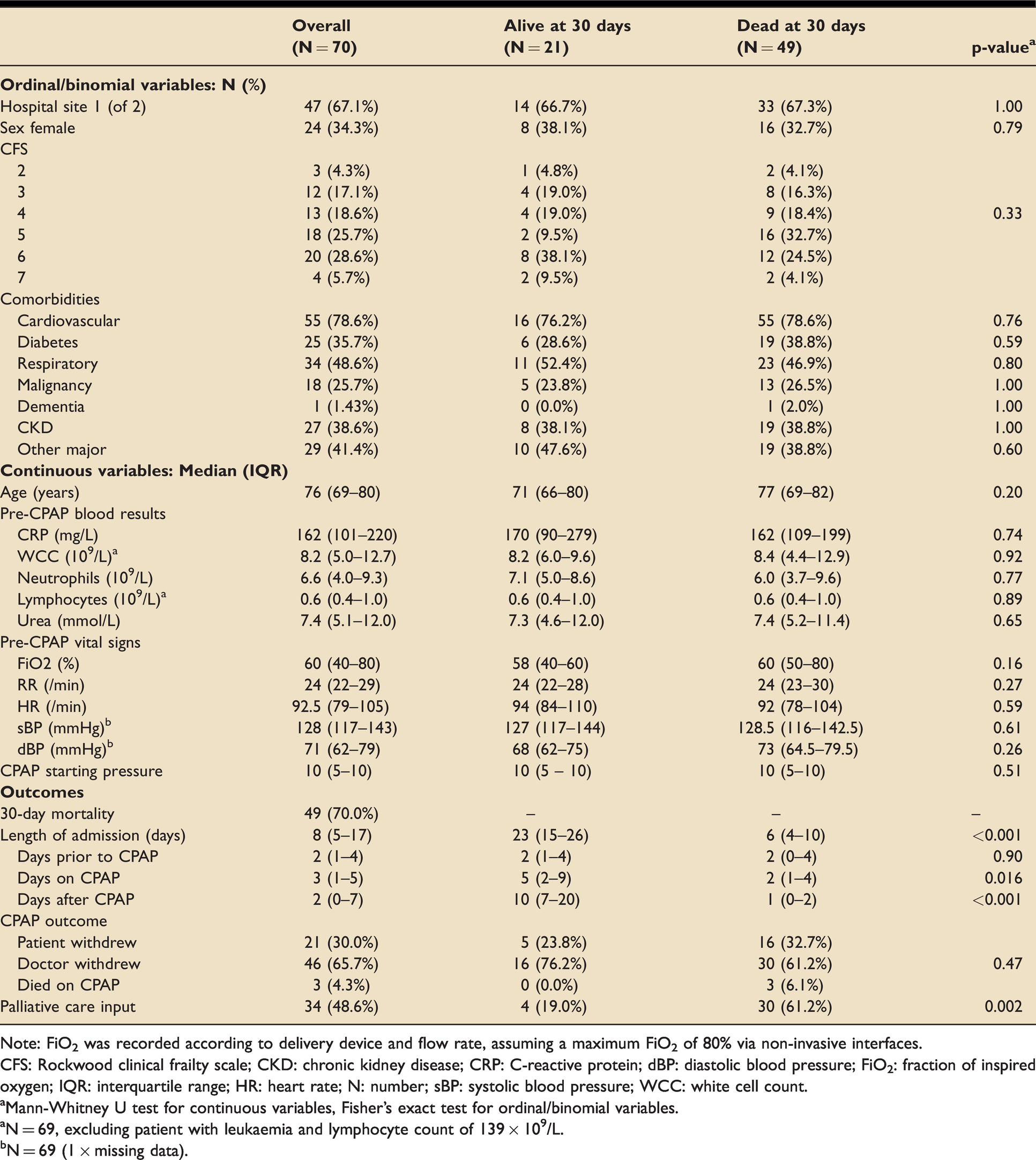
Note: FiO2 was recorded according to delivery device and flow rate, assuming a maximum FiO2 of 80% via non-invasive interfaces.
CFS: Rockwood clinical frailty scale; CKD: chronic kidney disease; CRP: C-reactive protein; dBP: diastolic blood pressure; FiO2: fraction of inspired oxygen; IQR: interquartile range; HR: heart rate; N: number; sBP: systolic blood pressure; WCC: white cell count.
aMann-Whitney U test for continuous variables, Fisher’s exact test for ordinal/binomial variables.
aN = 69, excluding patient with leukaemia and lymphocyte count of 139 × 109/L.
bN = 69 (1 × missing data).
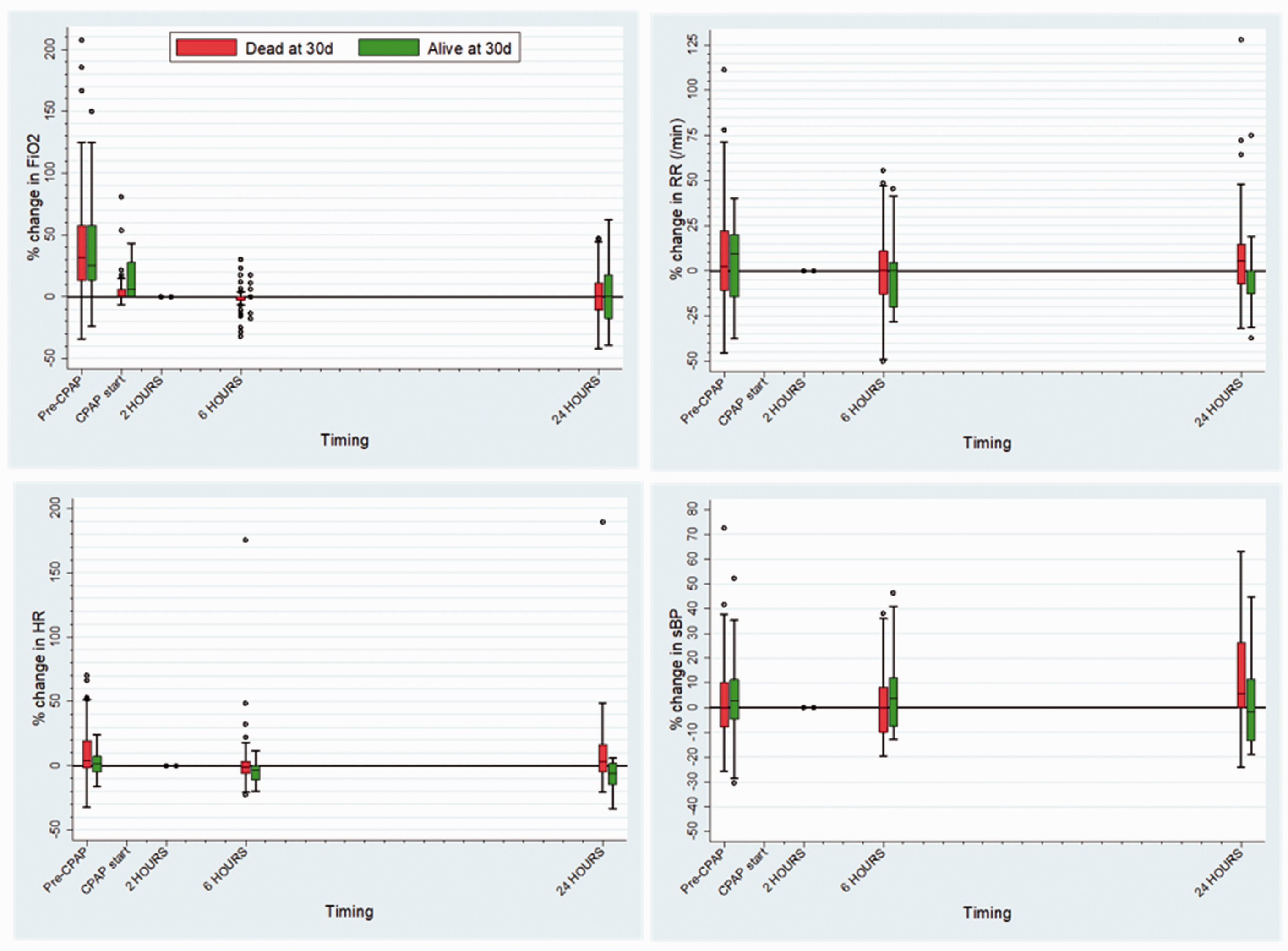
Box and whisker plots showing trends in patient observations and CPAP settings as percentage change per patient, with 2-hour CPAP values defined as baseline (matched).
Survivors spent a median 23 [15–26] days in hospital, with 5 [2–9] of those days on CPAP. 3 patients (4.3%) died on CPAP and 21 (30%) chose to discontinue it; the remainder discontinued CPAP according to clinical decisions (either weaning or futility).
Discussion
We evaluated two hospitals’ experience of offering CPAP as a ceiling-of-care treatment for COVID-19. There was high mortality among this relatively frail cohort with a high burden of comorbidity. Baseline patient characteristics were similar between survivors and non-survivors. The notable rate of patient-initiated discontinuation of CPAP implies a substantial burden of treatment. CPAP also prevents any visits from relatives, and may potentially increase the risk of staff infection and emotional distress.
Limitations of this study include its relatively small sample size and lack of a control group. However, this is among the largest reported cohorts of patients receiving CPAP as a ceiling-of-care in this context, and the only one focussing solely on this group.5–7 Other strengths include the use of clinically relevant outcomes across two hospital sites.
Given the ongoing widespread use of CPAP despite a lack of evidence, characterisation of this patient group is urgently needed. Further work should include larger studies, comparison with other management options, and exploration of the physical and psychological effects of CPAP on patients and staff. While the efficacy of CPAP in this context remains unproven, and its potential adverse effects unquantified, deciding whether, and in whom, to offer it remains a matter for local protocols, clinical judgement, and careful shared decision-making with patients.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
