Abstract
A 70 year old man, who had recently travelled in rural Iraq, presented with fevers, rigors, and developed multiorgan failure. An extensive range of diagnostic tests was undertaken in an attempt to identify the cause. He was treated with multi-organ support and a number of antibiotics. Critical illness in the returning traveller presents a number of challenges on the ICU: obtaining adequate history, the potentially broad differential diagnosis, the requirement for numerous and sometimes specialised investigations and risks of infection transmission to staff and other patients. Travellers are more often elderly, more likely to have comorbidities and immunosuppression whilst global disease patterns are changing. Particular consideration should be given to unusual infections and venous thromboembolic disease from prolonged immobility whilst in transit, alongside more commonly encountered diseases. Antimicrobial resistance may be encountered and appropriate infection control is essential for the protection of patients, staff and others. Specialist support is available in the UK via the Imported Fever Service, especially for High Consequence Infectious Diseases. Consideration of non-infectious causes of fever and critical illness in returning travellers is also warranted. Crucially, a multidisciplinary team approach with thorough information gathering, repeated clinical review and judicious use of investigations are essential for optimal patient care.
Case
A 70 year old man with type two diabetes mellitus and peripheral vascular disease presented to the emergency department (ED) with fever, rigors and dyspnoea. The patient reported his symptoms had been present for a week, after returning from travelling overseas. As he spoke little English and a translator was not immediately available, a more thorough history could not be obtained. He had a blood pressure of 80/40 mmHg, a heart rate of 115 beats per minute, a temperature of 39.1°C, and a prolonged capillary refill time of 4 s. His chest was clear, his heart sounds were normal, his abdomen was soft and non-tender with no organomegaly and normal bowel sounds. There were no rashes. An arterial blood gas revealed normal gas exchange but a base excess of −12 mmol L−1 and a lactate of 6 mmol L−1. He received a fluid challenge, co-amoxiclav and clarithromycin for sepsis of unknown origin.
Treatment and management
Because of persistent hypotension and lactic acidosis, he was referred to critical care. He rapidly developed hyperactive delirium and required intubation and ventilation for his own safety. Following intubation, a repeat chest x-ray showed bilateral infiltrates suggestive of acute respiratory distress syndrome (ARDS). Vasopressor support and renal replacement therapy were instituted. Blood, urine and sputum cultures, respiratory tract virus polymerase chain reaction (PCR), and a series of malaria screens, tests for HIV and hepatitis B and C were requested.
The next day, the patient’s family provided collateral history. The patient had returned to the UK seven days previously from a month-long trip to rural Iraq, where he had attended multiple gatherings with individuals from across the Middle East, stayed in local houses and come into contact with agricultural animals (including camels) on at least one occasion. Prior to this, he had had no travel outside Western Europe since moving to the UK from Iraq 30 years ago.
Given this new information raised the possibility of influenza, Middle Eastern Respiratory Syndrome (MERS) and Crimean-Congo fever, isolation measures were escalated. A repeat physical examination was undertaken looking for wounds or bite marks, rashes, adenopathy, organomegaly or other features that might provide clues to the underlying diagnosis. His antibiotics were changed empirically to meropenem, levofloxacin and oseltamivir, to widen the antimicrobial spectrum.
Over the next three days, the patient had marked vasoplegia, requiring vasopressin 0.06 units min−1 and noradrenaline at 0.8 µm kg−1min−1, and developed septic cardiomyopathy necessitating and adrenaline infusion at 0.3 µm kg−1min−1. The patient went on to develop progressive digital ischaemia, thrombocytopaenia and deranged liver function tests, and so several epidemiologically appropriate serological and molecular tests (including leptospirosis, dengue, malaria, Q-fever, Middle-Eastern respiratory virus, influenza A and B, Hepatitis A, B, C and E, Crimean-Congo Fever, Brucella serology, meningococcal/pneumococcal PCR) were requested. A CT neck, chest, abdomen and pelvis was undertaken, looking for infectious foci or non-infective causes of fever, but this did not reveal any relevant abnormality. Echocardiography revealed globally impaired left and right ventricles, but no evidence of endocarditis. Specialist input from an Infectious Diseases consultant was sought, who liaised with Public Health England’s Imported Fever Service.
A total of six sets of extended blood cultures, the battery of serological and molecular tests above and the vasculitis screen were negative. Oseltamivir was stopped on the exclusion of influenza and 10 days of meropenem were given, with termination of the antibiotic in line with clinical improvement in the patient’s condition and multiple negative bacterial cultures. The patient’s cardiovascular support was weaned over a week, but he had developed ICU-acquired weakness and a prolonged respiratory wean was required to liberate him from mechanical ventilation. His digital ischaemia progressed to dry gangrene. He was discharged after a month on ICU for ongoing rehabilitation. A causative organism was never identified.
Discussion
Febrile illnesses in returning travellers or individuals usually resident overseas are common presentations to healthcare services. Gastrointestinal and respiratory infections are the most common, and many patients (especially the elderly) may have community acquired infections from their home area. However, given increasingly affordable overseas travel and international migration,1–3 unusual infections are becoming more common, whilst travellers are now more frequently elderly, and/or have complex comorbidities than in the past. Disease patterns and vectors are changing and outbreaks, such as Zika, Ebola and the global SARS-CoV-2 pandemic, illustrate the risks.3,4 The development of critical illness further complicates patient care as relevant history may be more difficult to obtain, investigations may be more difficult to interpret and nosocomial infection may be difficult to differentiate from primary disease later in the illness course. 2 Appropriate history, examination and investigations need to be performed in parallel with resuscitation and treatment (see Figure 1). Consideration needs to be given to how treatable the disease is, whether this is an unusual (or indeed emerging) pathogen and whether it is significant to public health.
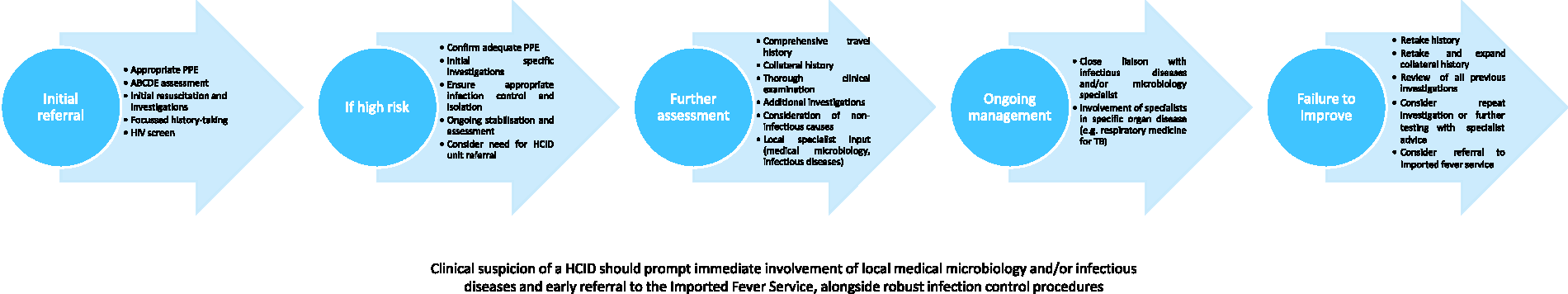
Clinical suspicion of a HCID should prompt immediate involvement of local medical microbiology and/or infectious diseases and early referral to the Imported Fever Service, alongside robust infection control procedures.
History-taking
The travel history is essential to produce a comprehensive and realistic differential diagnosis. 2 It is essential to know exactly where patients have travelled, down to the region or city, modes of transport, points of stop-over, the nature of their accommodation, food and beverages consumed, travel vaccination, bed-net and anti-malaria prophylaxis use, and use of antimicrobials whilst abroad. Contact with animals (especially bites), foodstuffs, local infection outbreaks, local healthcare, exposure to freshwater or seawater, or participation in unusual activities such as cave exploration, triathlon or adventurous activities allows due consideration of appropriate pathogens. 1 For example, this patient, who had stayed in a rural area with basic accommodation and contact with local farm animals, had a much higher likelihood of being unwell with a zoonosis compared to a diplomat resident in modern accommodation in a major city.
Numerous activities undertaken whilst travelling risk the acquisition of infection. Travel is associated with brief sexual encounters, use of sex workers, inadequate access to sexual health services and/or barrier contraception, increased alcohol consumption, illicit drug use and other activities which may expose patients to disease transmission. 5 Family members may not know or choose not to disclose such information, therefore a high index of suspicion and consideration of such diseases is essential. Furthermore, sexual encounters occur in both elderly and young individuals and diseases may manifest after many years without symptoms (e.g. syphilis and HIV), so sexually transmitted infection should not be excluded solely on the basis of age. 1 Screening for HIV and other potential causes of immunosuppression is essential, as HIV substantially increases the differential diagnosis for infectious illness and in itself will require treatment.
Immunosuppressed patients, whether due to HIV, other immune disorders or secondary to medical therapy (e.g. transplant recipients) may also have unusual presentations, atypical symptom complexes and incubation periods and potentially indolent or more severe infections than the immunocompetent. 1 Patients receiving immunosuppression therapy now travel more often and therefore imported infections in this cohort are more frequent. 3
Investigations
Investigations are necessarily wide-ranging. However, having a sensible clinical rationale for particular tests, especially serology, is important. Serological tests have a false positive rate: if numerous tests are requested with no realistic prospect of the patient having acquired the disease, a positive test will delay finding the actual diagnosis, trigger inappropriate treatment and risk adverse reactions, and have both cost and staffing implications.1,3,6 For example, syphilis serology may be falsely positive in patients with rheumatological conditions or certain vasculitidies, pregnancy, parenteral drug abuse and chronic infections (including HIV, infective endocarditis and mycobacterial disease). 7 In the case above, when testing for viral haemorrhagic fevers (VHF), there was a reasonable risk of Crimean-Congo fever, but testing for others, such as Marburg, was not indicated based on epidemiological risk.
As well as unusual diseases, illnesses may be explained by community acquired infections, either acquired before or after travel. 1 Home and imported illnesses may be different, for example, the viruses that cause pneumonia are similar in temperate and tropical regions, whereas Klebsiella pneumoniae frequently causes community acquired bacterial pneumonia in the tropics. 8 Furthermore, there are numerous infections which occur infrequently in developed nations but are common in less developed countries 1 or where infrastructure has been damaged following natural disaster or war. 9
High consequence infectious diseases
Few tropical infections have a high risk of person-to-person transmission, but a number require stringent infection control measures.3,6,10 High Consequence Infectious Diseases (HCID) are a set of acute infectious illnesses with a high fatality rate, able to spread in the community and healthcare settings which require enhanced individual, population and system responses to be managed safely and effectively. 6 Specific infections are listed in Table 1.
HCIDs as agreed by Public Health England and NHS England HCID Programme. 6

Infection control is an essential consideration in these patients: a large proportion of deaths from SARS-1 were in healthcare workers who acquired their infection from the patients they were treating. 11 Patient isolation should be started immediately in the ED and continued on the ICU. Personal protective equipment should be used by staff and visitors. Patients in ICU carry a high risk of transmission of infection as they undergo invasive procedures (including intubation and central line insertion) and may require several staff to manage them (e.g. for rolling). 12 Procedural interventions should be minimised. Essential procedures should take place at the bedside where possible to limit spread from patient transport. 13 However, overall risk needs to be carefully assessed: during the Ebola outbreak in West Africa, one unit used central venous access for some patients to both provide intravenous access and avoid the need for repeated phlebotomy, 14 whilst other units undertook conventional procedures. Care needs to be taken in disposal of fluids, including renal dialysate fluid, to prevent viral transmission.13,15
In some cases, such as suspicion of VHF, careful selection of blood tests to rule in or out particular diagnoses may need to be considered, as blood tests will require high levels of laboratory containment, and undertaking a large number of tests is both laborious and risks accidental viral spread.6,13 Support for diagnosis of such patients is available in the UK from the Imported Fever Service, which should be sought via a hospital’s local microbiologist or infectious diseases specialist (www.gov.uk/guidance/imported-fever-service-ifs). This service can also provide support for the clinical care of illnesses in the returning traveller more generally, and again can be accessed via hospitals’ medical microbiology and/or infectious diseases specialists.
Public Health England recommends consideration of VHF in patients with a fever of 37.5°C or more, or fever in the previous 24 h, developing within 21 days of leaving an endemic area, extensive bleeding or bruising, or additional risk factors (see Table 2). 16 Differentiation of VHF from sepsis associated with disseminated intravascular coagulation (such as meningococcal sepsis) may be especially challenging. 3 Patients should be isolated in a side room (ideally negative pressure), and staff should undertake hand hygiene, wear double gloves, fluid repellent gown, full length plastic apron, head covering, fluid repellent footwear, full face shield or googles, and a fluid repellent FFP3 respirator. If the malaria test (the main differential diagnosis of possible VHF) is negative, then the local infectious diseases consultant should consider discussion with the Imported Fever Service and arranging urgent testing VHF (including appropriate sample containment for safe transport). 6 If positive for a VHF or other HCID, transfer to a national centre for ongoing treatment will be arranged.6,10
Additional risk factors for VHF. 16

In the UK, there are designated High Level Isolation Units (HLIU) for patients with HCID, with additional beds at supporting units. 6 HLIU use the Trexler isolation system (essentially a flexible tent around the patient, with negative pressure and a number of half-suits projecting into the tent), allowing interventions (including critical care support) to patients with a high degree of protection to staff. 10 Major limitations of this system include limited availability, minimal rapid access to patients and a lack of scalability in the event of an outbreak. The UK ambulance services and Royal Air Force can transport patients with this level of isolation.10,16 Healthcare can be delivered on a larger scale whilst minimising risk to staff using effective personal protective equipment of staff. 14 However, the use of machines with exposure to blood (e.g. renal replacement therapy or extracorporeal life support) may present an unacceptable risk of transmission to other patients. 10 In the event of an epidemic outbreak of some HCIDs, some groups advocate the curtailment of such treatments, due to risk of transmission and likely overwhelming of hospital services. 10
Multi-resistant organisms and infection control
Patients may have received medical or dental treatment overseas to standards below those expected in developed nations, with regards to skin preparation, environmental and equipment decontamination and antibiotic prophylaxis. The overuse of antibiotics in many parts of the world promotes multidrug resistance. Admission to healthcare facilities in these countries is frequently associated with the acquisition of multidrug resistant organisms, 13 including methicillin-resistant Staphylococcus aureus, extended-spectrum beta-lactamase producers, carbapenemase producing enterobacteriaceae, extensively drug resistant Pseudomonas and multi-drug resistant organisms such as Acinetobacter baumanni. 13 Such organisms are associated with high mortality and options for antibiotics are limited. Clinicians may have to use agents with unfavourable side-effect profiles (for example, nephrotoxicity). 13 The antibiotic susceptibility of a familiar organism acquired overseas may well differ from that of that seen in developed nations (for example, imported Salmonella and Shigella have been reported with extensive drug resistance).2,13 The ICU is a nodal point in the hospital: infection outbreaks may be spread not only to other critically unwell patients, but to other wards by discharged patients. 12 Furthermore, both ICU staff undertaking outreach to other areas of the hospital and visiting specialty teams entering the ICU, may act as vectors for infection transmission. 12
Multidrug resistant (MDRTB) and extensively drug resistant (XDRTB) tuberculosis is increasing worldwide, with 20% of isolates demonstrating resistance to at least one antibiotic. High risk areas include Russia and Central Asia, the Indian Subcontinent, China and South East Asia and Sub-Saharan Africa. Infection control measures include negative pressure side rooms with adequate ventilation, appropriate respiratory filter masks and powered air purifying respirators for high risk procedures (such as bronchoscopy and intubation). 17 Laboratory staff should be notified of the potential for MRDTB or XDRTB to ensure proper measures are undertaken to minimise risk to personnel. 3
Infection following combat or terrorism
Individuals injured in combat or by terrorist incidents who have been repatriated for ongoing treatment may be exposed to unusual organisms from environmental contamination from the weapon or environmental fragments, such as Acinetobacter baumannii. 18 Tetanus prophylaxis should be ensured. 18 Injuries from suicide bombings risk blood borne virus transmission from missile fragments travelling through multiple casualties and contaminating wounds. Patients should be screened for blood borne viruses and offered appropriate post-exposure prophylaxis and/or vaccination. 19
Acute respiratory distress syndrome
The majority of ARDS in the returning traveller is caused by community acquired pneumonia. 8 Alongside normal pathogens, consideration should be given to Legionella pneumophilia, Avian influenza viruses, Hanta virus pulmonary syndrome, MERS, COVID-19, Burkholderia pseudomallei and other Gram negative organisms, especially with antibiotic resistance. 8 The current SARS-CoV-2 outbreak has provided ICUs across the world with experience of large numbers of patients with ARDS and the challenges of patient triage, investigation and management, resource limitation (from equipment and drugs to personal protective equipment and oxygen supply). A comprehensive review of this specific disease is beyond the scope of this article. ARDS may also be seen in travellers as a complication of malaria and following aspiration due to alcohol intoxication. 3 Occasionally, decompression sickness in those who have undertaken scuba diving can present several days after diving, including following long-haul air travel. 20 Although high altitude pulmonary and cerebral oedema are both serious illnesses in those travelling to over 2500 m, resolution is usual following descent, so is only seen in critical care facilities at such altitudes. 21
Non-infectious causes of fever
Consideration of non-infectious causes of fever is also essential.1,22 Key disease groups are listed in Table 3. Indeed, in modern cohorts, non-infectious causes account for 65–83% of patients with pyrexias of unknown origin (although only a minority of these will be overseas travellers). 22 In the above case, thorough clinical, radiological and laboratory testing was undertaken looking for malignancy, as there was a high probability of this as a differential diagnosis, given his age. Occasionally poisoning with organophosphates or envenomation from snakes, spiders and sea animals may cause fever. 23 There is increasing use of positron emission tomography-computed tomography imaging for “hot spots” of occult infection, malignancy or inflammatory processes. 1 Finally, despite thorough clinical assessment and extensive investigation, in a substantial proportion of patients (reported as high as 51%), such as the case presented, no cause is identified. 24
Conclusion
Imported infectious diseases are likely to be an increasing problem and patients will frequently require critical care. Thorough history and examination, appropriate targeted investigations and multidisciplinary involvement of infectious disease specialists, microbiologists, laboratory staff and other specialists is crucial. A high index of suspicion is required for both highly infectious diseases and organisms with extended antibiotic resistance. Finally, non-infectious causes of pyrexia should also be sought.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.

