Abstract
Introduction
The age of patients admitted into critical care in the UK is increasing. Clinical decisions for very-old patients, usually defined as over 80, can be challenging. Clinicians are frequently asked to predict outcomes as part of discussions around the pros and cons of an intensive care unit (ICU) admission. Measures of overall health in old age, such as the clinical frailty scale (CFS), are increasingly used to help guide these discussions. We aimed to understand the characteristics of the very-old critically unwell population in the UK and the associations between frailty and outcome of an ICU admission in our healthcare system (National Health Service, NHS).
Methods
Baseline characteristics, ICU interventions and outcomes (ICU- and 30-day mortality) were recorded for sequential admissions of very old patients to UK ICUs as part of the European VIP 1 and 2 studies. Patient characteristics, interventions and outcome measures were compared by frailty group using standard statistical tests. Multivariable logistic regression modelling was undertaken to test association between baseline characteristics, admission type and outcome.
Results
1858 participants were enrolled from 95 ICUs in the UK. The median age was 83. The median CFS was 4 (IQR 3–5). 30-day survival was significantly lower in the frail group (CFS > 4, 58%) compared to vulnerable (CFS = 4, 65%) and fit (CFS < 4 68%, p = .004). Sequential organ failure assessment (SOFA) score, reason for admission and CFS were all independently associated with increased 30-day mortality (p < .01).
Conclusion
In the UK, frailty is associated with an increase in mortality at 30-days following an ICU admission. At moderate frailty (CFS 5–6), three out of every five patients admitted survived to 30-days. This is a similar mortality to septic shock.
Introduction
The mean age of admission to critical care in the United Kingdom (UK) is 60, and over 40% of admissions are aged 65 and over. 1 Caring for older people is therefore an integral part of critical care medicine. Part of this role is helping patients and their families make decisions about their care. For the very-old, usually defined as over 80, 2 this can be particularly challenging. This age group’s relatively shorter life-expectancy brings sharply into focus the question of what adverse effects of a critical care admission are tolerable for each individual. Decisions about care for any patient necessitates careful evaluation of the potential benefits of a therapy and its risks of failure or harm. In order to fully communicate the balance between these competing forces to patients and their families, clinicians frequently derive probable outcomes for an individual, from data about the populations they care for. However, data in the very-old age group is lacking, with ‘elderly’ care studies often having a lower age limit between 60 and 70 years.3,4 Chronological age alone is an insufficient predictor of outcome from critical illness, as there is often significant variation in the physical and physiological reserve of older people. 2 A preferred descriptor of the physiological changes associated with ageing is frailty. This term refers to both the cumulative physiological decline associated with age and the dramatic effect of even minor insults that accompany increasing frailty. 5 There is growing awareness that measures of frailty such as the clinical frailty scale (CFS) 6 may be useful to inform decision on care by helping to predict likely outcome. Indeed, guidelines to deal with case surge in the COVID-19 pandemic from the National Institute for Health and Care Excellence (NICE), advocated using a CFS ≥ 5 as indicative of likely poor outcome from critical care admission as part of an assessment of severely unwell patients with the disease.7,8 Utilising these scores outside of the patient groups and care settings they were validated in has recognised limitations, but mirrors the evolution of other scoring systems in critical care. For example, the sequential organ failure assessment (SOFA) score 9 was originally developed to assess population-level morbidity and mortality in critical care, but its use has subsequently evolved to predict individuals at high risk of mortality, most notably in the recently updated sepsis diagnostic criteria.3,10 As yet, neither CFS nor SOFA score have been shown to provide valid and reliable risk prediction for the very old who are admitted to intensive care.
Recently, two large prospective observational studies of very old intensive care patients (VIP-1 and -2) have reported their findings of the outcomes of very old (over 80 years of age) patients admitted to intensive care units (ICUs).11,12 These studies recruited participants from over 300 critical care units in 21 countries across Europe. Results from these studies confirmed the associations between increasing frailty and 30-day mortality. In addition, they found regional variations in the characteristics of patients admitted to ICU. This is perhaps unsurprising, given the recognised variation in healthcare provision across the continent, 13 but does raise an important question about how the clinician can best interpret findings from across healthcare systems and apply them to their own and consequently help inform decisions about care.
In our view, it is necessary to understand the characteristics of the very-old critically unwell population in our own healthcare system (National Health Service, NHS). As such, we have undertaken an analysis of participants in the VIP-1 and -2 studies from ICUs in the UK. Using similar methodology to a recently published analysis of patients from Germany, 14 the aim of this work is to describe the characteristics of very-old patients admitted to the ICU in the UK and determine whether the associations between frailty and sickness severity scores and outcome found in the continental evaluation hold true for the UK.
Methods
Study participants and data collection
Participants included in this analysis were enrolled by a UK critical care unit to one of two prospective observational studies of very-old intensive care patients (VIP-1 and -2).11,12 Briefly, participating critical care units in the UK enrolled consecutive admissions of patients over 80-years old in a 3-month period in 2016–17 (VIP-1) or a 6-month period in 2018–19 (VIP 2). VIP-2 included only unplanned admissions; VIP-1 also included elective admissions (e.g. following major surgery). Ethical approval for this work was provided by the NHS Health Research Authority (REC references 16/LO/1642 and 18/WA/0193), and the studies were registered on clinicaltrials.gov (IDs: NCT03134807 and NCT03370692). Participant consent was obtained prospectively where possible and assent for participation sought from relatives where patients lacked capacity. Ethical approval was granted to include those participants who died prior to consent being obtained.
Demographic data for each participant included age, sex and reason for admission to critical care. Clinical frailty scale was calculated for each participant, based on pre-morbid state (i.e. frailty prior to admitting condition). 6 Assessment of CFS was undertaken by a healthcare professional or member of the research team at each study site. Information to calculate CFS was provided by the patient, a relative or carer or the patient record. Participants were categorised as fit (CFS < 4), vulnerable (pre-frail) (CFS = 4) or frail (CFS > 4). 12 Sickness severity was measured by SOFA score in the first 24-h. 9 Provision of life-supportive measures such as ventilation (including non-invasive), vasoactive drugs and renal replacement therapy was recorded, along with documentation of whether life-supportive care was withheld or withdrawn. Outcome was measured as critical care survival and 30-day survival from critical care admission.
Statistical analysis
Patient characteristics, interventions and outcome measures were compared by frailty group using standard statistical tests (Chi-squared, Kruskal–Wallis and Fisher’s exact). Uni- and multi-variable logistic regression modelling was undertaken to test association between baseline characteristics (age, gender, CFS, SOFA and type-of admission [unplanned vs planned] and outcome). Statistical analyses, tables and plots were undertaken and created using R: A language and environment for statistical computing (version 3.6.3). 15 These statistical analyses match those undertaken on the subset of participants from German critical care units by Muessig et al. 14
Results
Participant characteristics by reason for admission.
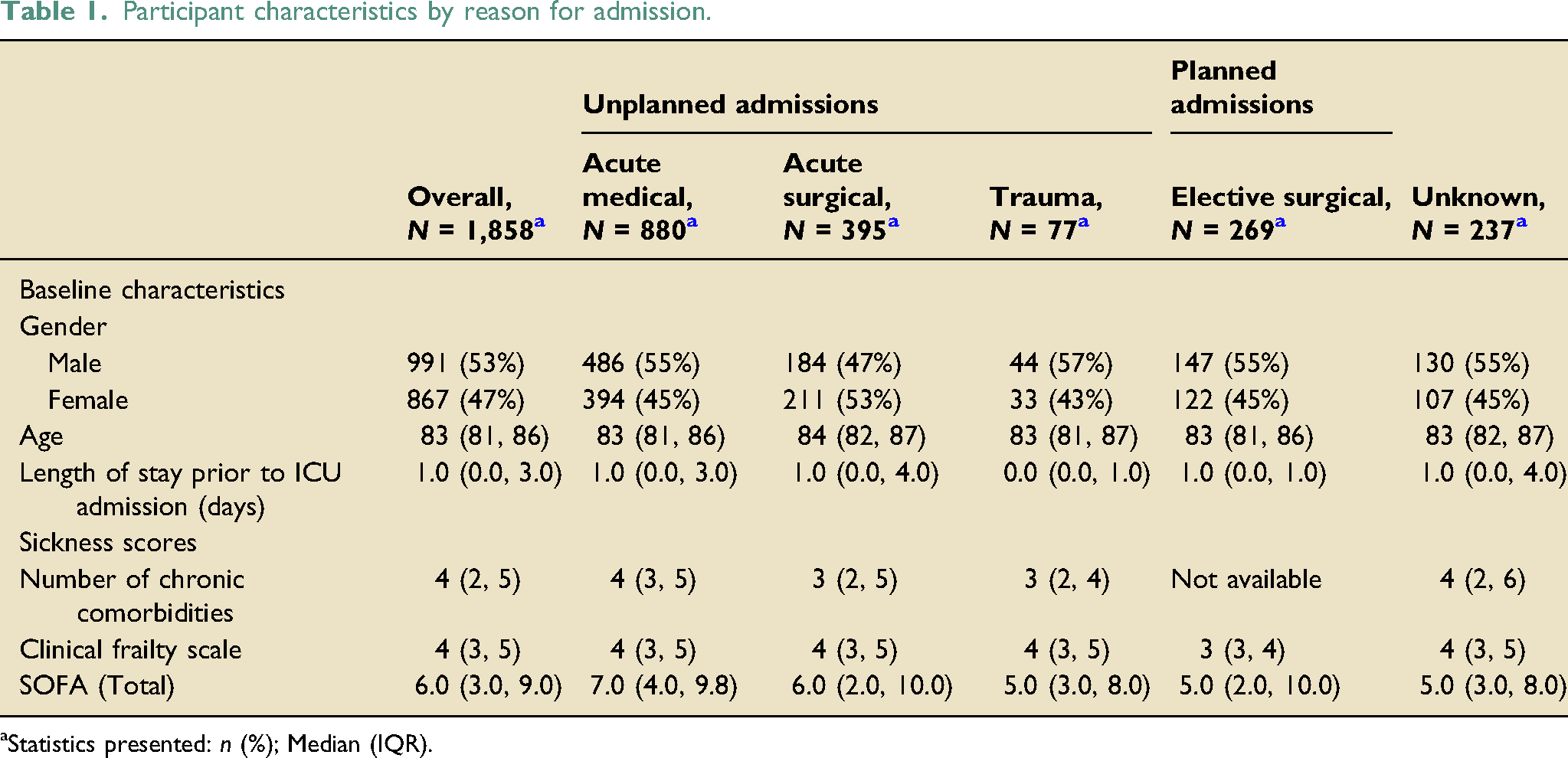
aStatistics presented: n (%); Median (IQR).
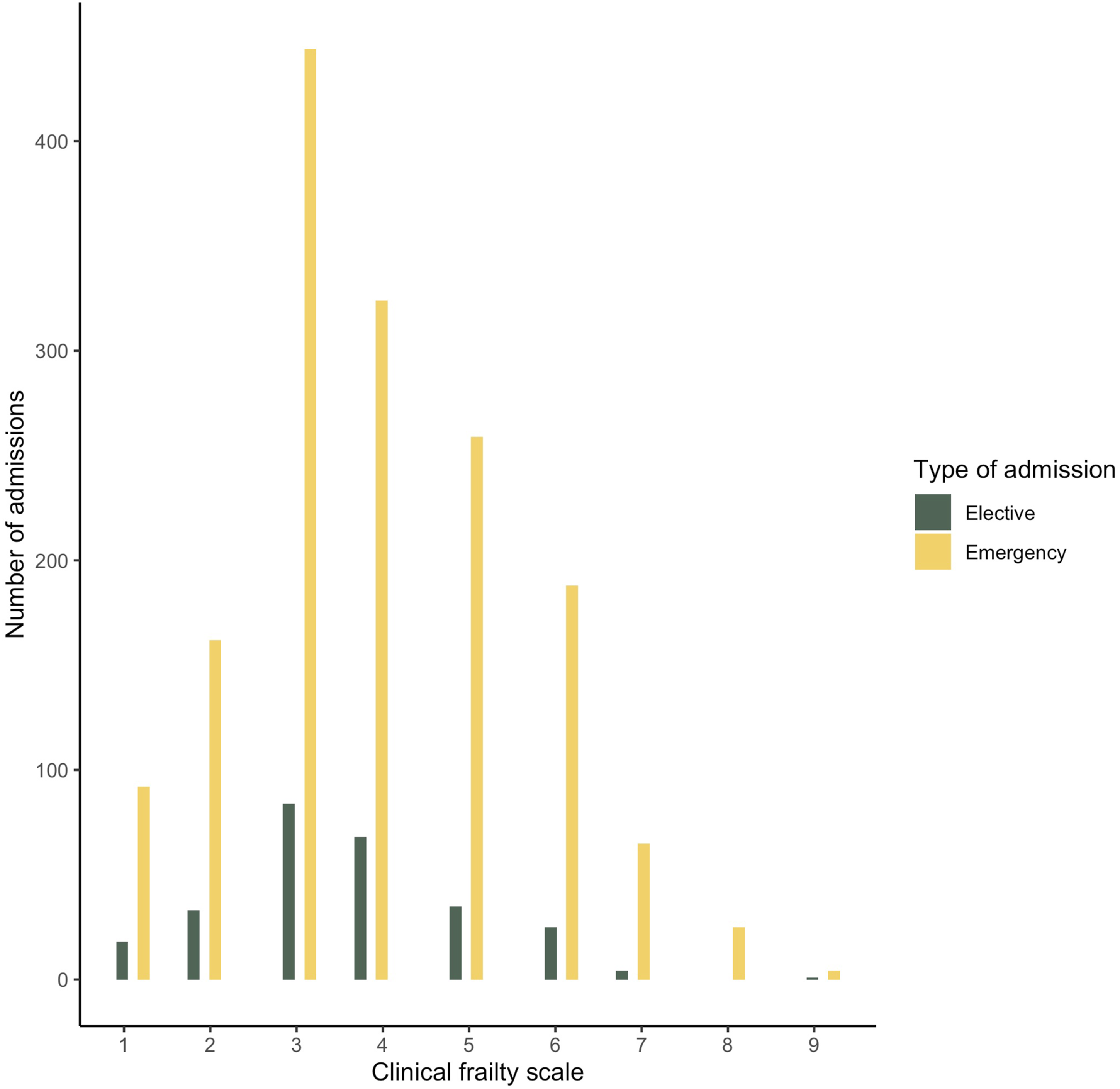
Number of admissions by clinical frailty scale. Distribution of clinical frailty scale amongst very-old-patients (over 80-years) admitted to UK ICUs.
Patient resource utilisation and outcome by frailty group (unplanned admissions).
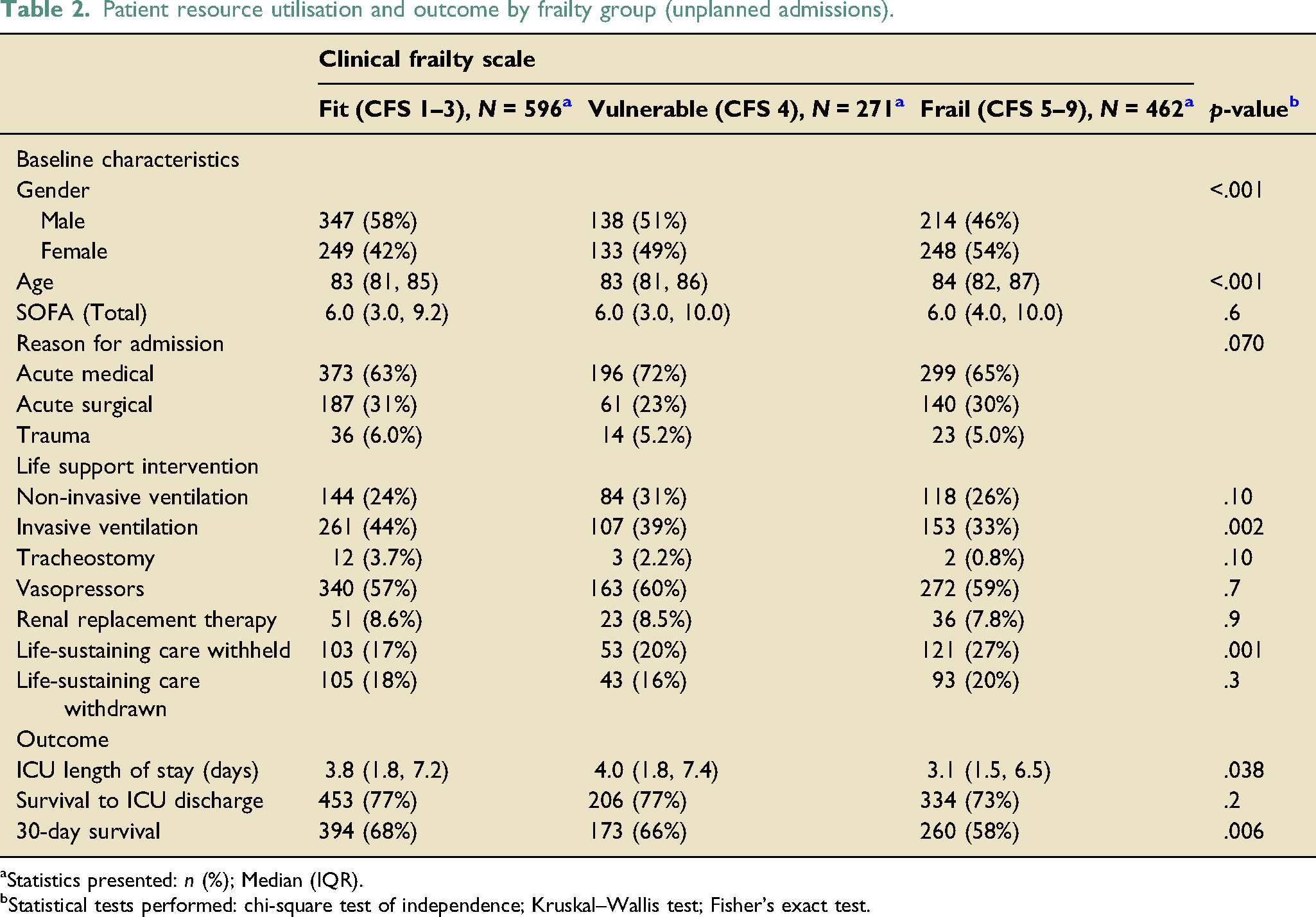
aStatistics presented: n (%); Median (IQR).
bStatistical tests performed: chi-square test of independence; Kruskal–Wallis test; Fisher’s exact test.
Patient resource utilisation and outcome by frailty group (elective admissions).
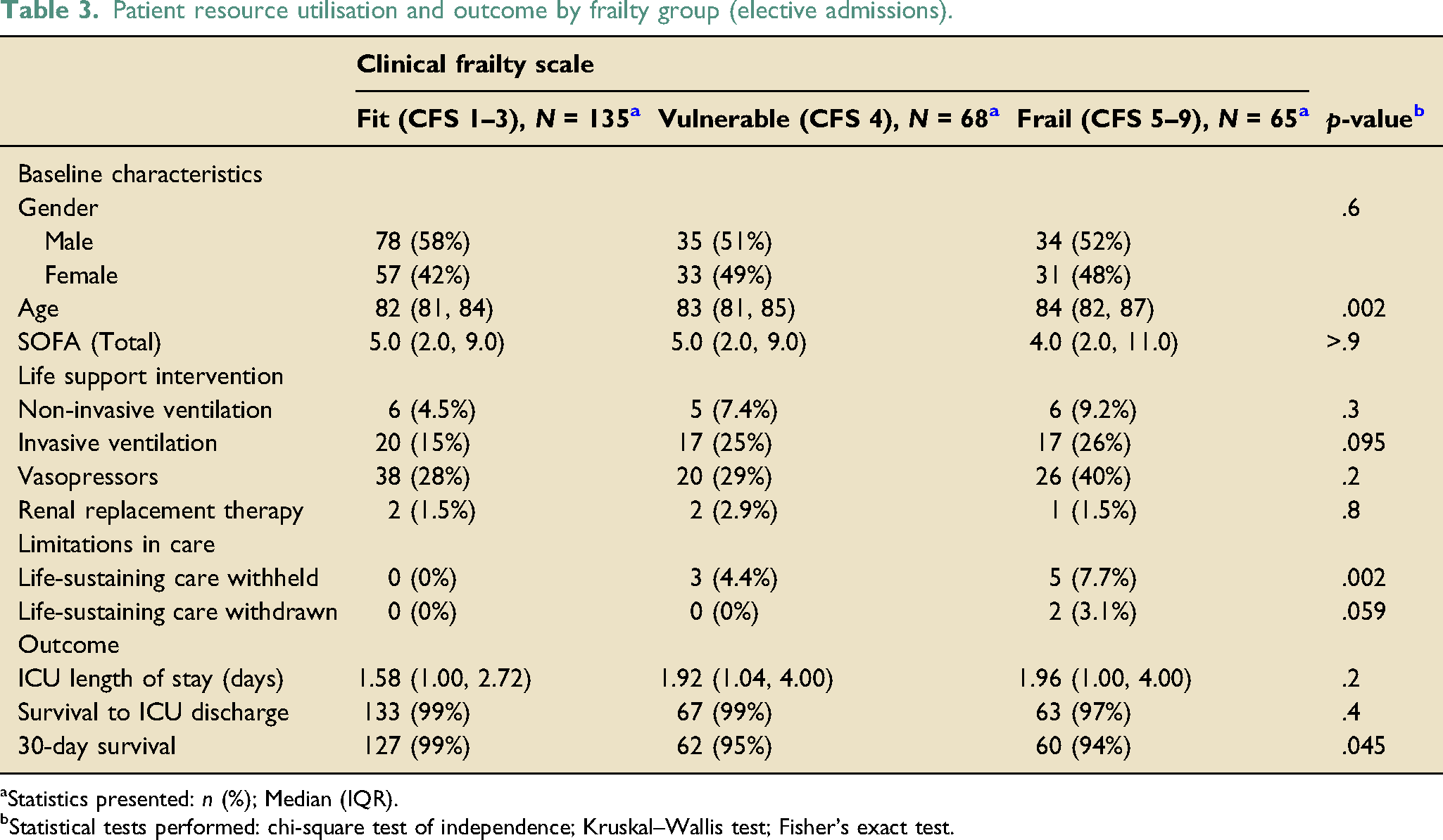
aStatistics presented: n (%); Median (IQR).
bStatistical tests performed: chi-square test of independence; Kruskal–Wallis test; Fisher’s exact test.
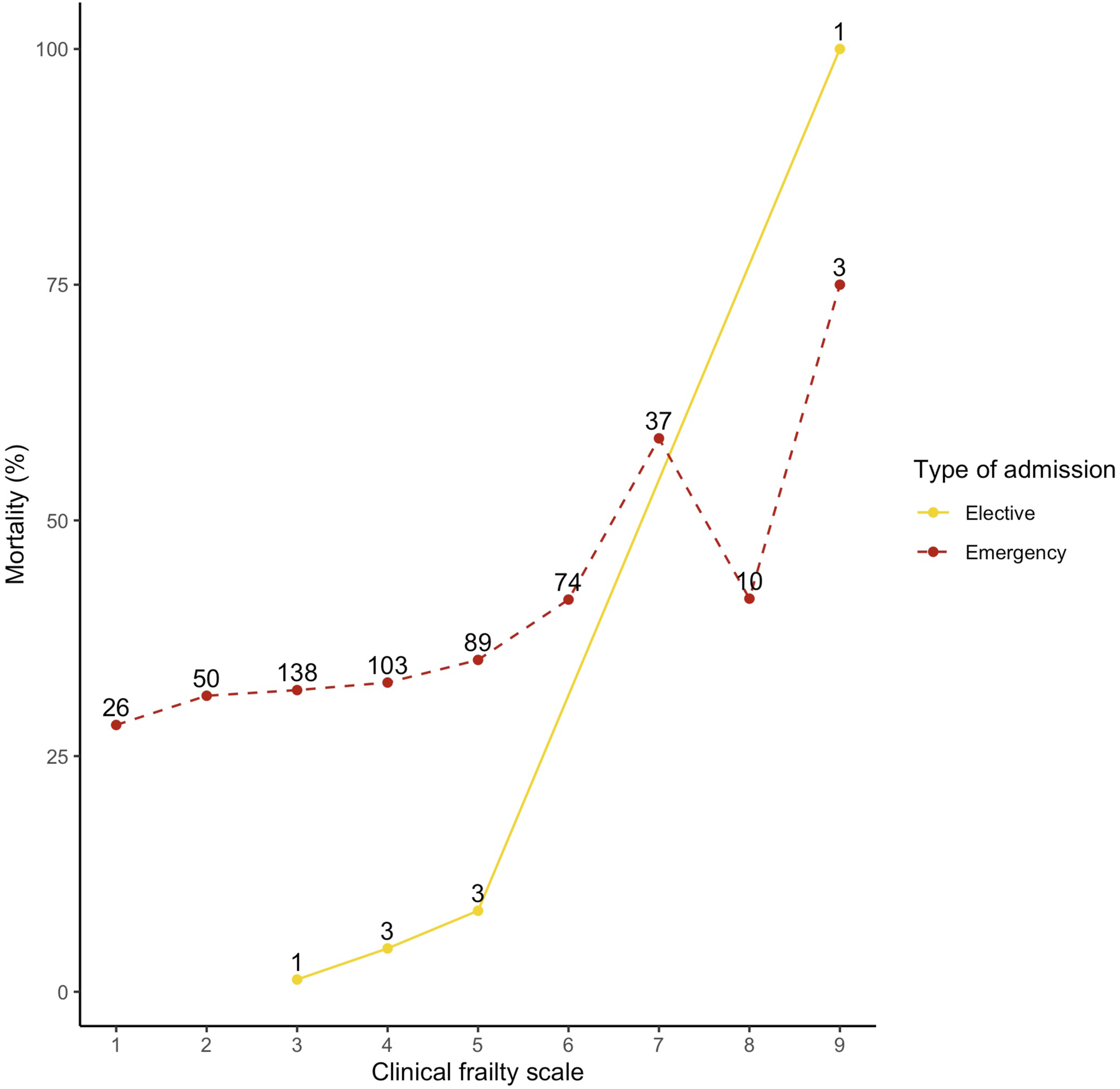
30-day mortality by clinical frailty scale. Percentage mortality 30-days following ICU admission by frailty scale. There is a clear increase in mortality with increasing frailty for both unplanned and elective admissions. Number (n) of deaths at each clinical frailty scale indicated by numbers next to data points.
The characteristics and outcomes for participants with a planned admission (Table 3) were broadly similar across all frailty categories. Elective admissions were less likely to receive invasive ventilatory support than unplanned admissions (21% vs 38–49%, p < .001), with a similar finding for vasoactive drugs (32% vs 50–61%, p < .001). 30-day survival was ≥ 94% across all frailty categories. There was a similar trend of increasing mortality with increasing frailty group (Figure 2), but numbers were small.
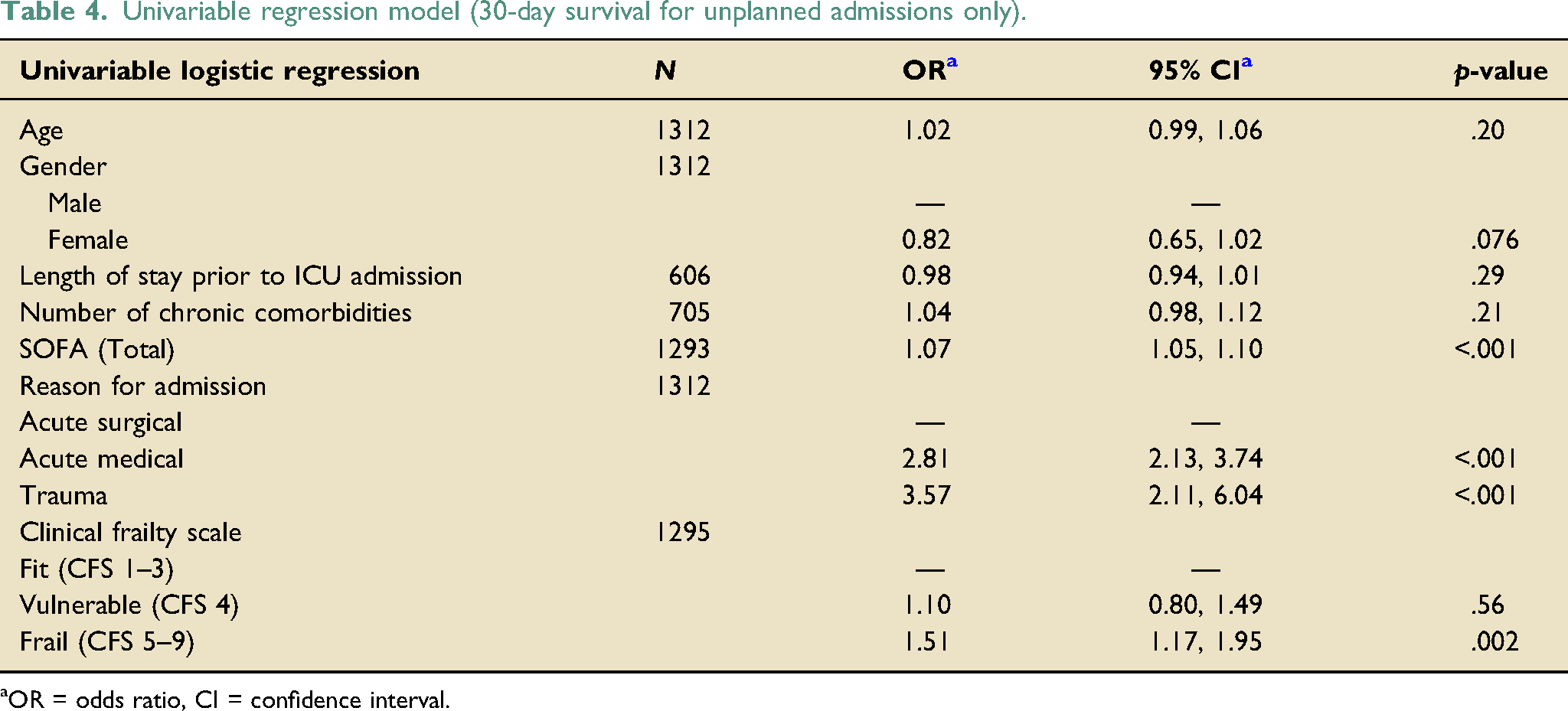
Univariable regression model (30-day survival for unplanned admissions only).
aOR = odds ratio, CI = confidence interval.
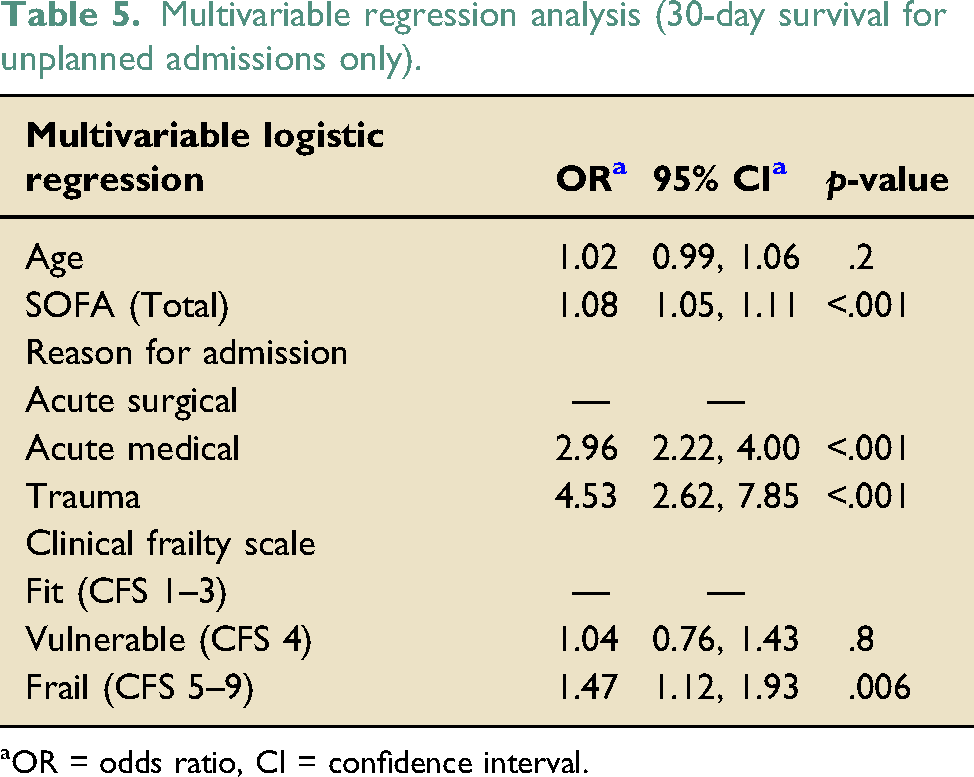
Multivariable regression analysis (30-day survival for unplanned admissions only).
aOR = odds ratio, CI = confidence interval.
Discussion
We have presented detailed data on the largest very-old cohort of patients admitted to critical care in the UK, both in terms of number of patients and number of ICUs. Data was provided on over 1800 patients from 95 critical care units. With a median age of 83 years, these data provide a good snapshot of the use of critical care by the very old (>80 years) across the country and healthcare system (NHS). This cohort covers the full remit of critical care provision, including medical, trauma and surgical, planned and unplanned admissions. With an increasing prevalence of admissions of the very old, clinicians frequently find themselves trying to explain the risks and benefit of an ICU admission to patients, their relatives and carers. These data may help inform those decisions, as well as future resource plans.
These data show a balance of sexes admitted to critical care. This does not mirror population data on sex from the 2011 census in England, which showed that just 37% of people over 80 are men. 16
At 30-days, frailty as measured by the CFS was associated with increased mortality independent of illness severity (as measured by SOFA score). The odds of survival at 30-days reduced with increasing frailty (supplementary Table 1), although numbers were smaller in the higher CFS groups. The lower 30-day survival is perhaps unsurprising in the frail group of participants. However, with three out of every five frail admissions surviving at least 30 days, it is unclear how these data will help inform decisions to admit or continue therapy. A recent guideline for admission into ICU published by the NICE during the ongoing coronavirus (COVID-19) pandemic suggested that CFS of 5 is the threshold at which clinicians should start to consider if the harms will outweigh the benefits of critical care organ support. 7 Survival rates for CFS 5 and 6 at 30 days post-ICU admission in this study were 63% and 59%. respectively. Interestingly, this is a similar mortality to septic shock, 10 where early and aggressive intervention is recommended. 17 This may add weight to the argument that mortality is not always a helpful standalone outcome measure, but some may argue that these findings support a raising of the CFS threshold at which the benefits of a critical care admission are questioned and at the very least a time limited trial of treatment may be suitable for patients with this level of frailty. With 30-day survival of just 40% with CFS of 7 and 25% for CFS of 9 (supplementary Table 1), the importance of using the full range of the CFS rather than just frail/non-frail is apparent. These findings are similar to the work by Darvall et al. that showed a significant association between CFS and mortality in only the severe/very severely frail (CFS 7 and 8) in their retrospective study of ICU admissions for pneumonia in Australia and New Zealand. 8 In the most recent update to COVID-19 guidelines, NICE continue to advocate the use of CFS in guiding decision making in the elderly, although the specific reference to CFS ≥ 5 has been removed. 18
In this study, associations between frailty and mortality are mirrored in both resource utilisation and decisions to limit life supportive treatment. Although this finding may be unsurprising, it perhaps may indicate that decisions on whether or not to provide life-supportive therapies could be made earlier in the ICU stay, if not prior to admission. This is particularly true for those with a higher clinical frailty scale (CFS > 6) where more than one in three patients had some limitations in care. A frailty assessment of the very old at time of ICU referral could prompt and help inform discussions on potential therapies. As noted in our regression analysis, increasing SOFA score was also associated with increased mortality. A further potential avenue for triage therefore may be to use assessment of frailty alongside acute sickness severity. Figure 3 shows a heatmap of 30-day mortality by SOFA and frailty scale, with trends towards higher mortality (darker colour) with higher SOFA and frailty. We do not propose cutoff or threshold values, but these trends could help to inform discussions with patients or their representatives. Heatmap showing percentage mortality at 30-days referenced to SOFA and clinical frailty scale. Number of participants shown in white text in each reference square. Darker colours represent higher mortality. Plot shows trend to higher mortality with increase in either or both scores. A combination of chronic and acute frailty scores like these could be used to inform discussions around benefits and risks of ICU admission and associated treatments.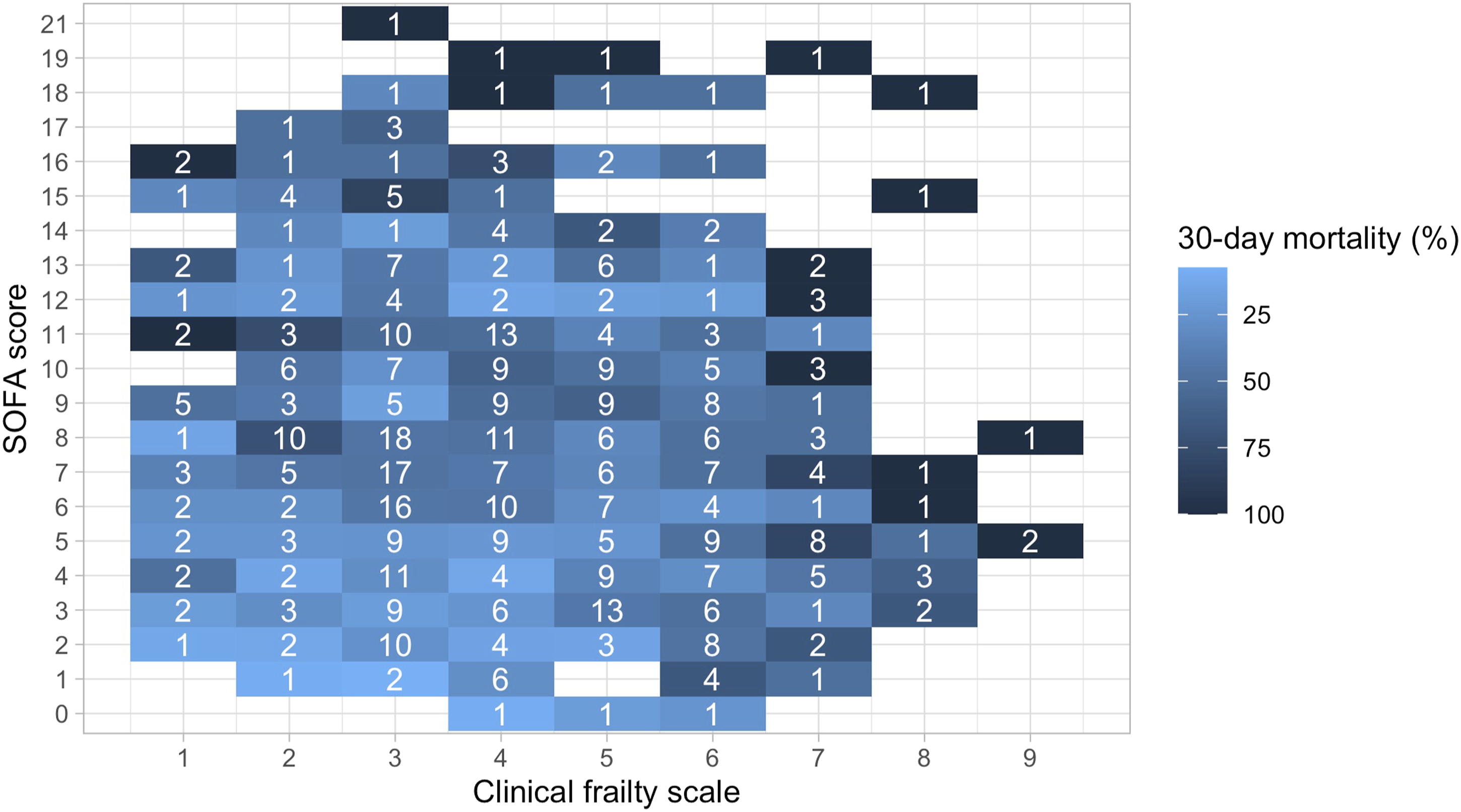
In keeping with other studies investigating the associations between frailty and outcomes from critical illness, it would be helpful to have a comparison with those patients not admitted to critical care (i.e. where critical illness exists but where admission to an ICU has not occurred as it is felt not in the patient’s best interests). Given the potential physical and psychological trauma associated with a critical care admission, this comparison would provide important information on the benefit of care on an ICU over continued care on a general hospital ward for those in the most-frail groups where decisions on escalation of therapy are particularly challenging. Similarly, details on patients provided with non-invasive ventilation or high flow oxygen outside of the ICU (e.g. on a respiratory ward) are also missing from these data and could provide a useful comparator in future studies. In addition to longer-term mortality data (e.g. 90-days), this work is also missing quality of life and disability outcome measures. The chance of short-term survival alone may not be the most important factor for patients and their families or carers who are making complex decisions about treatment during critical illness at this stage in their lives. Patients may be more interested in understanding how likely it is they will achieve an outcome with a quality of life they find acceptable. McNelly et al. showed that functional capacity is reduced for all survivors of critical illness 18-months after discharge in a cohort (n=41, mean age 55 years) of participants in the MUSCLE study. 19 Other work has suggested that frailty can help to predict functional outcome, although these data are often derived from slightly younger age groups and healthcare systems outside of the UK.20–24 In a single centre study in Holland, Geense and colleagues recently showed that 42% of patients with an unplanned critical care admission had increased frailty 12-months after discharge in all-age groups admitted to critical care. 22 Interestingly, survivors with higher baseline frailty scale at admission were found to have reduced frailty 12-months later. Survivorship and selection bias (with significant loss to follow-up) may confound this finding, again demonstrating the difficulty and complexity of research aimed at determining predicting factors for critical care outcome. In a cohort of older patients in the United States of America (USA) (n = 216 ICU survivors, mean age 84), Ferrante et al. found higher disability scores at 6-months in patients who were frail at admission to critical care, with nearly 60% nursing home discharge for frail patients. 24 However, data from this cohort includes critical care admissions from 1998–2014; so, whether the findings are generalisable to current cohorts or outside the USA is unknown. Finally, we must acknowledge that many clinicians already use frailty, either informally or with scores like CFS, when making decisions about care. The associations we have found in these data are likely to be influenced by this.
Conclusions
In the very-old cohort of patients admitted to critical care in the UK, frailty is associated with an increase in mortality at 30-days. The odds of death increased with increasing frailty as measured using the CFS. At moderate frailty (CFS 5–6) three out of every five patients admitted survived to 30-days, a similar survival rate to septic shock. Associations between frailty and death were found to be independent of sickness severity (SOFA) score. These findings support the use of the CFS to aid patients, caregivers and clinicians making decisions about critical care provision for very-old patients.
Supplemental Material
sj-pdf-1-inc-10.1177_17511437211050789 – Supplemental Material for The clinical frailty scale – does it predict outcome of the very-old in UK ICUs?
Supplemental Material, sj-pdf-1-inc-10.1177_17511437211050789 for The clinical frailty scale – does it predict outcome of the very-old in UK ICUs? by Dagan O Lonsdale, Liting Tong, Helen Farrah, Sarah Farnell-Ward, Chris Ryan, Ximena Watson, Maurizio Cecconi, Hans Flaatten, Jesper Fjølner, Christian Jung, Bertrand Guidet, Dylan de Lange, Wojciech Szczeklik, Johanna M Muessig and Susannah K Leaver in Journal of the Intensive Care Society
Footnotes
Acknowledgements
We would like to acknowledge the significant contribution of the following individuals involved in running these studies at the participating sites. Jason Cupitt, Emma Stoddard, Steve Rose, Aimi Collins, Irina Grecu, Richard Partridge, Christian Frey, Beverley Stidolph, Michael Reay, Karen Reid, Michael Spivey, Karen Burt, Coarlie Carle, Alan Pope, Nicola Butterworth-Cowin, Marcela Vizcaychipi, Laura Martins, Clare Bolger, Nicky Cullum, Yolanda Baird, Kerry Barnes, Carole Boulanger, Nicola Paver, Ritoo Kapoor, James Douglas, Mark Puulletz, Sarah Williams, Patricia Williams, James Wood, Miriam Davey, Phillipa Wakefield, Ashley Quinn, Amanda Cowton, Nikki Collings, Clare Bolger, Ingeborg Welters, Karen Williams, Georgina Randell, Katie Stammers, Jenny Lord Jordi Margalef, Madhu Balasubramaniam, Jenny Anderson, Alison Lewis, Agnieszka Kubisz-Pudelko, Rachel Savine, Rebecca Gale, Maria Faulkner, Laura Parry, Richard Pugh, Victoria Garvey, Tom Daubeny, Emily Bevan, Deise Griffin, Mupudzi McDonald, James Nathan, Reena Khade, Daniel Martin, Helder Filipe, Mason Pannell, Amy Collins, Kiran Salaunkey, Philip Bastone, Sally Pitts, Katie Bowman, Reni Jacob, Richard Innes, Patricia Doble, Rosie Reece-Anthony, Babita Gurung, Ben Creag-Brown, James Doyle, Nanci Doyle, Richard Savigne, Laura Montague, Rachel Oguntimehin and Carlos Castro Delgado.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The ESICM supported VIP-1 with a research award. Free support for running the electronic database and was granted from Aarhus University, Denmark. Financial support for creation of the e-CRF and maintenance of the database was possible from a grant (open project support) by Western Health region in Norway 2018.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
