Abstract
FUSIC haemodynamics (HD) – the latest Focused Ultrasound in Intensive Care (FUSIC) module created by the Intensive Care Society (ICS) – describes a complete haemodynamic assessment with ultrasound based on ten key clinical questions: 1. Is stroke volume abnormal? 2. Is stroke volume responsive to fluid, vasopressors or inotropes? 3. Is the aorta abnormal? 4. Is the aortic valve, mitral valve or tricuspid valve severely abnormal? 5. Is there systolic anterior motion of the mitral valve? 6. Is there a regional wall motion abnormality? 7. Are there features of raised left atrial pressure? 8. Are there features of right ventricular impairment or raised pulmonary artery pressure? 9. Are there features of tamponade? 10. Is there venous congestion? FUSIC HD is the first system of its kind to interrogate major cardiac, arterial and venous structures to direct time-critical interventions in acutely unwell patients. This article explains the rationale for this accreditation, outlines the training pathway and summarises the ten clinical questions. Further details are included in an online supplementary appendix.
Introduction
The Intensive Care Society’s (ICS) Focused Ultrasound in Intensive Care (FUSIC) sets UK training standards, competencies and accreditation for the use of ultrasound in critically ill patients. This article outlines the concept of a new module, called FUSIC Haemodynamics (FUSIC HD), which is designed to give bedside clinicians the tools to make a complete haemodynamic assessment using ultrasound. FUSIC HD builds on existing FUSIC modules – FUSIC Heart (formerly known as FICE), FUSIC Abdomen and FUSIC Lung – and is structured around ten key clinical questions to be answered at the bedside of a haemodynamically unstable patient. It is the first accreditation system of its kind to include the entire circulation – cardiac, arterial and venous – for haemodynamic assessment. Such a complete assessment is not possible with clinical examination, cardiac output monitors or other imaging modalities alone. Ultrasound is uniquely placed as a non-invasive and comprehensive tool that can be used rapidly by the bedside clinician. The hope is that FUSIC HD will open new doors to how critically ill patients are assessed and treated. Further details, including tables of views and measurements, a full data set, and normal and abnormal values are available in the supplementary online appendix.
Both the British 1 and American Societies of echocardiography 2 have published guidelines on performing a comprehensive echocardiogram. FUSIC HD practitioners are encouraged to read these documents to understand the differences between haemodynamic assessment and comprehensive echocardiography. Governance standards and recommendations can be found in ‘Guidelines for the Provision of Intensive Care Services V2’. 3
Rationale for a new accreditation system
A FUSIC Heart study is effective at detecting major cardiac pathology at the bedside. However, beyond its scope exist many life-threatening diagnoses that are notoriously difficult to make clinically but readily detectable with ultrasound. So, it is natural for clinicians who have completed FUSIC Heart accreditation to wish to extend their skillset to detect these. Level 2 providers are not widely available out-of-hours, and Level 2 accreditation is a demanding process that few clinicians have resources to achieve. Consequently, some practice beyond their basic accreditation without supervision, which represents clinical risk. We have designed FUSIC HD to bridge this gap and enable clinicians to progress their ultrasound skillset within the safety of a governance process. While advanced echocardiography accreditation systems already exist, these focus exclusively on the heart and include highly specialist competencies that are less relevant to haemodynamic assessment. By contrast, FUSIC HD evaluates the entire haemodynamic system and focuses on the most important clinical questions that need answering in critically ill patients. Clinicians are ideally suited to integrate imaging and clinical context. Understanding both is key, which makes FUSIC HD potentially so powerful.
Whilst a FUSIC HD examination is a comprehensive assessment of haemodynamics, it is not a comprehensive echocardiogram. Practitioners should understand the limitations this presents and have a low threshold for referring a patient to a Level 2 practitioner. On the other hand, a comprehensive echocardiogram does not include all views and measurements done in FUSIC HD, meaning it cannot replace a complete FUSIC HD study. The abdominal views are only for assessing blood flow in specific vessels; they are in no way meant to replace abdominal imaging for other reasons.
We acknowledge the importance of transoesophageal echocardiography (TOE), but chose not to include this in FUSIC HD for two reasons: firstly expertise isn’t yet widespread in the UK outside cardiac centres, and secondly we are currently working with stakeholder organisations to develop a specific training pathway for focused TOE.
An outline of the accreditation process can be seen in Table 1. Before undertaking HD training, candidates will already have collected logbooks of 50 basic heart, 30 lung and 25 abdominal scans, and been assessed as competent in these areas. Given the scope of FUSIC HD, we believe that a further 50 training scans strikes the right balance between effectiveness and achievability. However, this will be kept under constant review. Competence will be checked by logbook review, pathology questions and a practical examination. Full details can be found on the ICS website.
The FUSIC HD accreditation process.
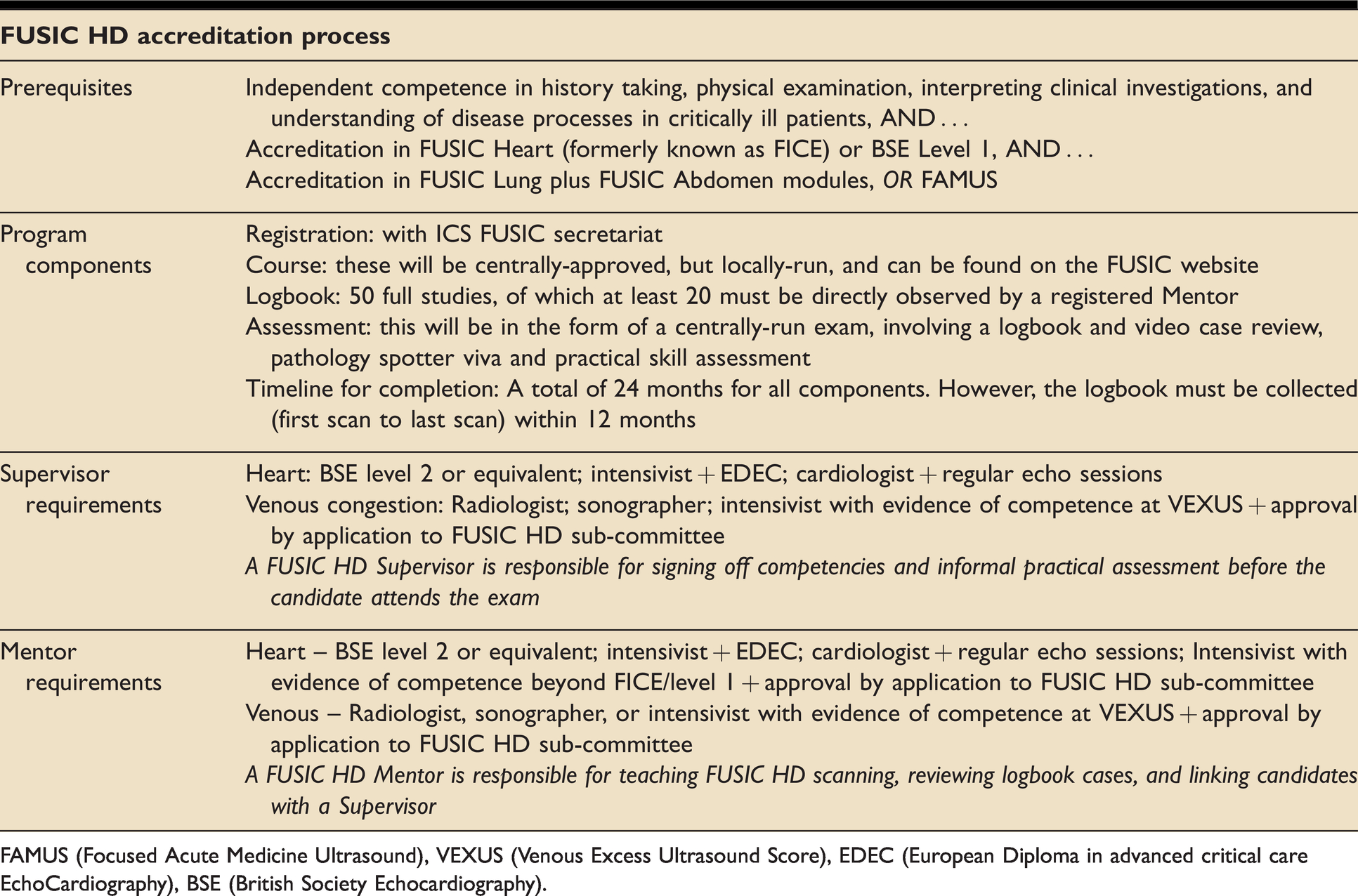
FAMUS (Focused Acute Medicine Ultrasound), VEXUS (Venous Excess Ultrasound Score), EDEC (European Diploma in advanced critical care EchoCardiography), BSE (British Society Echocardiography).
The ten clinical questions
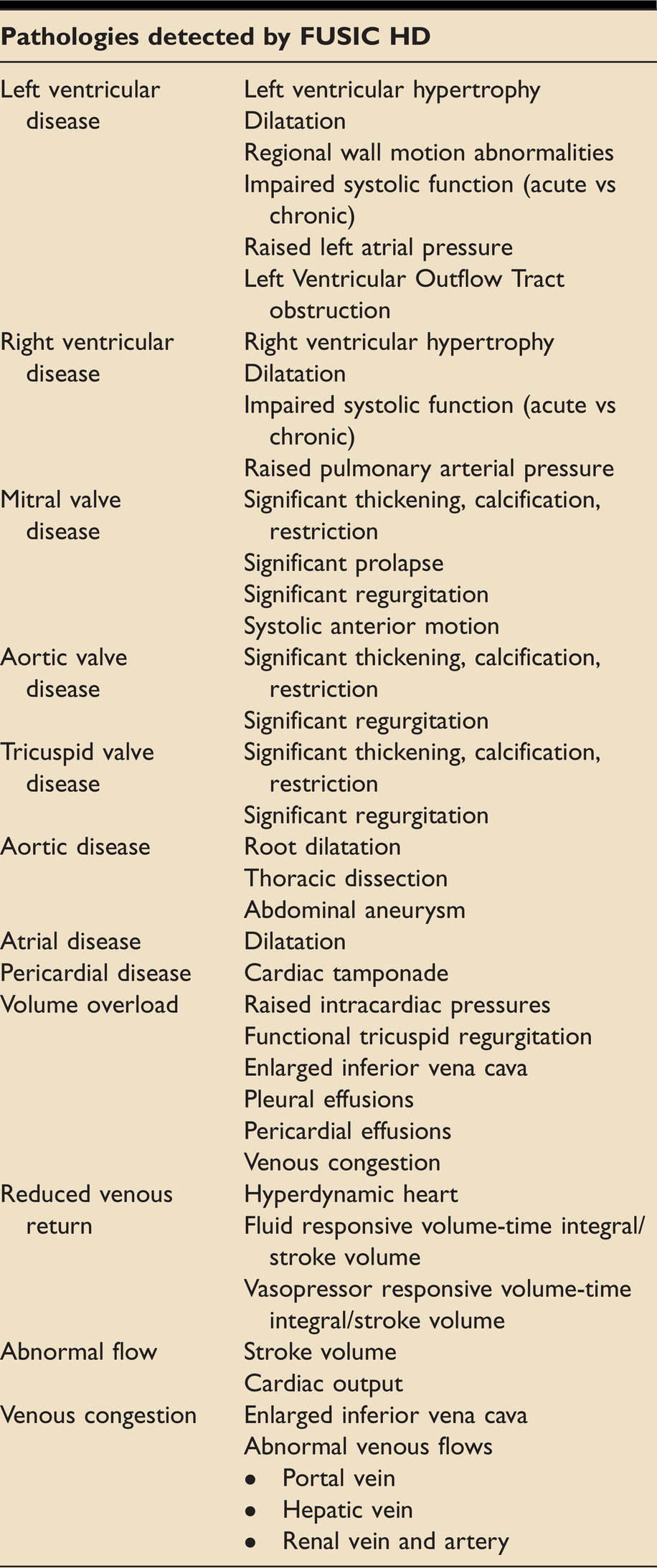
FUSIC heart, a prerequisite to learning FUSIC HD, is designed to answer five clinical questions: is the left ventricle dilated and/or significantly impaired? Is the right ventricle dilated and/or significantly impaired? Are there features of low venous return? Is there a pericardial effusion? Is there a pleural effusion? FUSIC HD enables the clinician to answer ten more. These are listed in Table 2 and explained in more detail below. Pathologies that can be detected with a FUSIC HD scan are listed in Table 3.
The clinical questions FUSIC Heart and FUSIC HD are designed to answer.

The pathologies detectable with a FUSIC HD examination.
Q1. Is stroke volume abnormal?
Detecting and treating shock is one of the most important roles of an intensive care clinician. Despite this, published evidence demonstrates that it is remarkably difficult to identify a low cardiac output state clinically. 4 Calibrated continuous stroke volume monitors exist, but these are rarely found in locations that patients are referred from. Ultrasound can accurately quantify stroke volume (SV) using left ventricular outflow tract (LVOT) volume-time integral (VTI) and LVOT diameter measurements, and has the advantage of being portable.5,6 Any error in LVOT diameter measurement will be squared in SV calculation. When quantification isn’t necessary, measuring LVOT VTI alone is a quick and useful surrogate of SV and its trends.
While SV and left ventricular ejection fraction (LVEF) are related, both must be interpreted in context of the following haemodynamic factors: left ventricular (LV) chamber size and function, preload and afterload, degree of inotropic support, and clinical context (gross valvular abnormality, sepsis etc). The impact of mechanical ventilation on ventricular loading must also be taken into account.
Because LVEF can change so significantly with disease progression or resolution, and because of the experience and time required to measure it accurately, FUSIC HD does not recommend quantifying this. However, a FUSIC HD practitioner will be able to visually assess LVEF and grade LV function (into normal, impaired and severely impaired) accounting for all the haemodynamic factors mentioned above.
Q2. Is stroke volume responsive to fluids, vasopressors or inotropes?
A clinician needs to know whether or not an intervention – fluid bolus, vasopressor or inotrope – will improve stroke volume. A FUSIC Heart study will have indicated a high or low fluid tolerance profile based on evaluation of LV and right ventricular (RV) size and systolic function, but it cannot answer this. The commonest cause of haemodynamic instability in a critically ill patient is low venous return from reduced stressed venous volume, caused by either vasoplegia or hypovolaemia. Unfortunately, ultrasound cannot distinguish between these two causes as both share the same appearances (a small, hyperdynamic heart). Administering empirical fluid therapy in vasoplegia is less effective than vasopressors 7 and risks being counter-productive through fluid overload, which causes significant harm. 8
Preload responsiveness can be predicted with ultrasound by measuring respiratory variations in LVOT VTI and diameter of the inferior vena cava. 9 To be valid, a number of preconditions must be met: sinus rhythm, mechanical ventilation with no spontaneous breaths, tidal volumes of at least 8ml/kg, normal intra-abdominal pressure, and an intact thorax. In addition, a failing right ventricle can result in a false positive. 10 These caveats often render these methods unusable. 11 In such cases, LVOT VTI can be measured before and after a fluid challenge, a passive leg-raise or starting a vasopressor infusion.
Q3. Is the aorta abnormal?
Aortic dissection carries a high mortality and is frequently overlooked. As such, ultrasound can be pivotal to making this diagnosis and enabling life-saving treatment. With small modifications from standard echo and abdominal windows, ultrasound can be used to image the entire aorta, from its root to bifurcation, making this an important target in a patient with undifferentiated shock. A FUSIC HD level practitioner should regard aortic dissection as a rule-in, not rule-out, diagnosis. The presence of an intimal dissection flap confirms the diagnosis. Flow in the aorta should be pulsatile using colour Doppler. Continuous flow suggests a shunt; turbulent flow suggests dissection. Associated features of aortic root dissection include a pericardial effusion and aortic regurgitation. A dilated aorta, anywhere along its course, should prompt referral for an expert scan and/or cross-sectional imaging.
Q4. Is the aortic valve, mitral valve or tricuspid valve severely abnormal?
Some acute valvular pathology requires immediate surgical intervention, while chronic valvular pathology can have dramatic effects on the way the heart handles different loading conditions. Therefore, the clinician needs to know if haemodynamically significant valve disease is present in a critically ill patient.
Quantifying valve disease in a critically unwell patient is a highly complex task that requires considerable expertise in echocardiography, well beyond the scope of a haemodynamic study. Instead, FUSIC HD utilises visual inspection with 2D and colour Doppler to detect features of severe valvular obstruction (marked leaflet thickening, calcification and immobility) and/or regurgitation (large, highly turbulent, upstream colour signals), and empower clinicians to refer in a timely fashion. With regard to aortic stenosis, rather than measuring valvular gradients, which has many pitfalls, FUSIC HD uses spectral Doppler only to measure Dimensionless Index (a marker that is relatively independent of blood flow, ventricular loading, body size). 12 With regard to valvular regurgitation, practitioners should be able to take ventricular function and loading conditions into account, as well as distinguishing functional (valve leaflets stretched apart by chamber dilatation) from non-functional (intrinsic disease of the valve apparatus) states.
Infective endocarditis is another important diagnosis but one that cannot be ruled out with transthoracic echocardiography. However, a FUSIC HD practitioner should have the appropriate knowledge to recognise obvious vegetations and refer to a Level 2 expert for a more detailed assessment.
Q5. Is there systolic anterior motion of the mitral valve?
Basal septal hypertrophy, common in the elderly population, can raise the velocity of blood as it is ejected through the LVOT. This is compounded by hyperdynamic states, involving low preload and/or afterload, that cause the left ventricle to become smaller than usual at end-systole. As blood velocity rises within a given radius, its pressure falls. When severe enough, this can suck the mitral valve leaflets into the LVOT during systole, causing acute mitral regurgitation (MR) and dynamic LVOT obstruction: a condition known as systolic anterior motion (SAM).
It is important to be able to identify SAM because it can cause shock and acute cardiogenic pulmonary oedema, mimicking left ventricular failure, and lead to potentially dangerous treatment with diuresis and vasodilators. Conversely, treatment of SAM is aimed at minimising inotropy, optimising preload and afterload, and maximising diastolic filling time. Therefore, early detection is essential.
Q6. Is there a regional wall motion abnormality?
Evaluating global systolic function is a core FUSIC Heart skill. Assessing regional wall motion requires further experience. But it is an important means of identifying patents with ischaemic heart disease who might benefit from coronary intervention. Wall thickening is a better gauge of regional function than endocardial movement as an akinetic segment may be moved because it is attached to another moving part. In systole, normal endocardial excursion is >5 mm and normal wall thickening is >30%. 13 Based on this, wall motion is described as normokinetic, hypokinetic, akinetic, dyskinetic or aneurysmal.
When hypo/akinetic segments are limited to a certain distribution, usually relating to coronary artery territories, they are known as regional wall motion abnormalities (RWMAs). RWMAs can be seen early in ischaemia, sometimes even before ECG changes are present, so being able to detect them can help the intensive care clinician make a timely diagnosis. LV walls have been divided up into 17 segments, with different segments being seen in various standard echo views. 14
Non-ischaemic RWMAs also exist – notably stress cardiomyopathy, which causes symmetrical RWMAs that extend beyond coronary territories. Stress cardiomyopathy is an important diagnosis to the intensivist because its treatment includes discontinuation of any beta-agonists, which may seem counter-intuitive when presented with clinical signs of poor ventricular function. While there are characteristic features of stress cardiomyopathy with ultrasound, it is not possible to formally distinguish it from ischaemic cardiomyopathy without further investigations. Urgent referral to a cardiologist should be made.
Q7. Are there features of raised left atrial pressure
Being able to identify raised left atrial (LA) pressure is of key importance in critically ill patients. Ultrasound can help clinicians distinguish between cardiac and non-cardiac pulmonary oedema, avoid injudicious IV fluid administration, monitor the response to fluid administration or removal, and predict weaning failure. 15 However, rather than quantifying LA pressure, FUSIC HD aims to identify the likelihood of it being high, low or indeterminate in keeping with ASE guidance. 16 Our simplified algorithm is shown in Figure 1. It does not aim to quantify left ventricular diastolic function, which is more complex and esoteric.
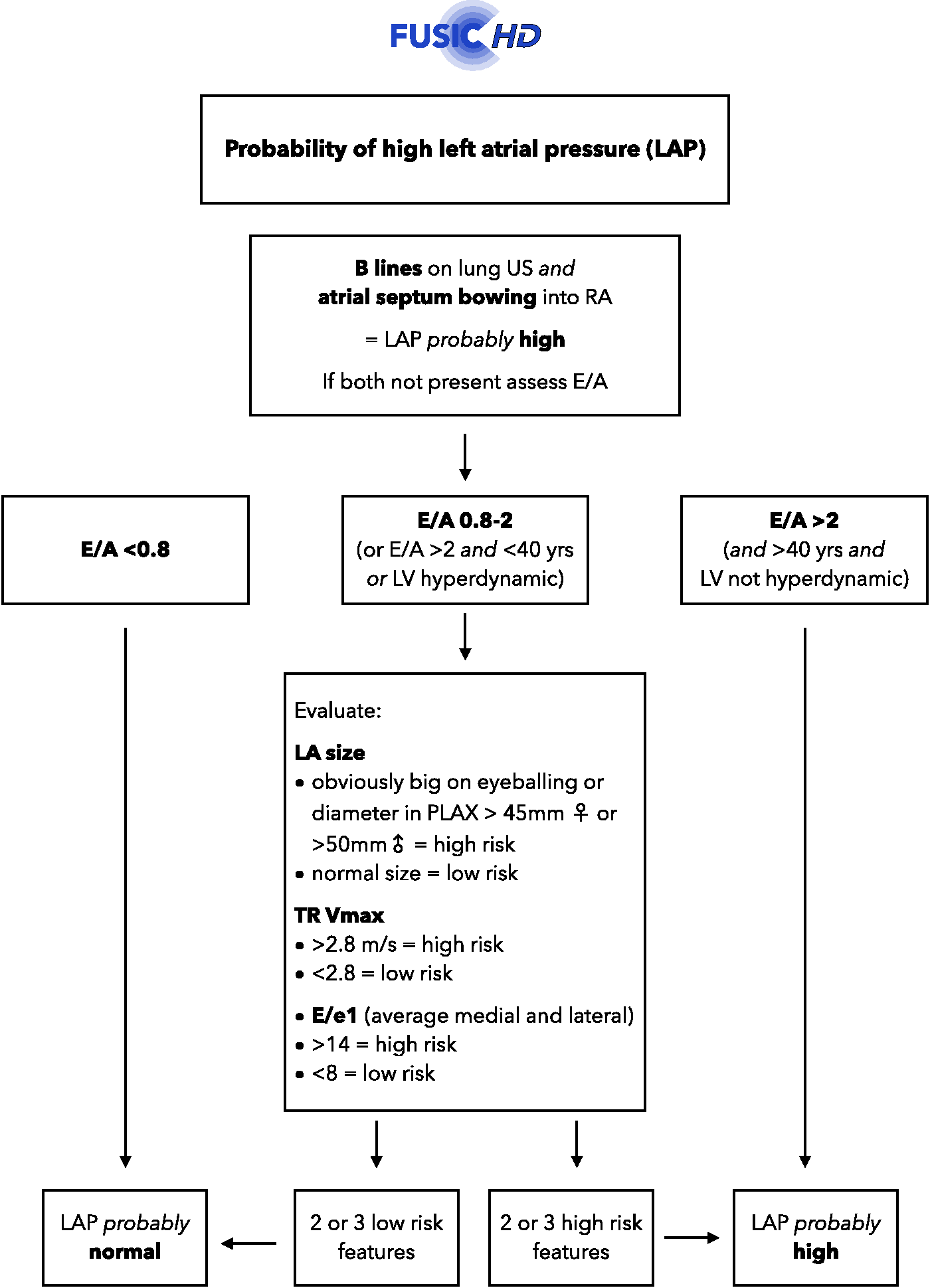
A pragmatic algorithm for estimating LAP, adapted and simplified from ASE guidelines.
The presence of B lines on lung ultrasound and bowing of the inter-atrial septum into the right atrium throughout the cardiac cycle make raised LA pressure almost certain. More detailed qualitative assessment of LA pressure can be done by measuring early mitral valve (MV) inflow velocity (E and A waves), which classifies the patient into a low, indeterminate or high risk profile. An indeterminate risk profile requires further measurement of LA size with 2D, TR Vmax with pulsed-wave (PW) Doppler, and early LV diastolic lengthening (e’) with tissue Doppler imaging (TDI).
Q8. Are there features of right ventricular impairment or raised pulmonary artery pressure?
RV impairment can be a significant contributor to haemodynamic instability and is independently associated with increased ICU mortality. 17 RV function is assessed in FUSIC HD by visually estimating RV area change and free wall movement, and measuring longitudinal movement and velocity of the TV annulus using M mode and tissue Doppler, respectively.
Knowing whether the patient has raised pulmonary artery (PA) pressure is key to understanding or predicting a patient’s haemodynamic response to, say, instigating mechanical ventilation or a fluid challenge. As such, it is a key question for FUSIC HD. The cornerstone of PA pressure assessment is identifying TR using colour Doppler, then measuring TR Vmax. 18 This velocity is used to risk stratify patients for raised PA pressure. 19 Most ventilated patients have some degree of TR. There are a number of caveats surrounding TR Vmax measurement. It will underestimate PA pressure in the presence of poor RV function, severe TR (in the presence of a large regurgitant orifice) and misalignment between the TR jet and CW Doppler. A singly long cardiac cycle (long R-R time) can overestimate velocity. The relationship between TR Vmax and PA pressure is invalidated by the presence of pulmonary stenosis.
Increased PA pressure can be acute or chronic, and the right ventricle can be pressure or volume overloaded, or both. Differentiating between each of these these using echo can be challenging. Assessment of TR Vmax, RV function, the thickness of the RV free wall, and the shape of the interventricular septum in systole and diastole is required.
Q9. Are there ultrasound features of cardiac tamponade?
FUSIC Heart practitioners can identify pericardial and pleural effusions, and distinguish between them. FUSIC HD goes further to evaluate whether or not the pericardial effusion is consistent with cardiac tamponade. While tamponade is a clinical diagnosis, certain echo features are characteristic.
A pericardial effusion with any right sided chamber collapse should prompt high suspicion of tamponade. Cardiac chambers are affected during their relaxation, when they are at their lowest pressures. And those with the lowest pressure are usually affected first. Hence, the right atrium is first to collapse in ventricular systole, followed by the right ventricle in diastole. Left-sided chamber collapse is a late phenomenon. Raised right atrial pressure leads to a dilated, non-collapsing inferior vena cava and other signs of venous congestion. Trans-mitral/tricuspid variation in flow velocities occur but should should not be relied upon in mechanically ventilated patients.
These features are not necessarily present when there is regional tamponade, which can be missed even by experienced operators. Tamponade cannot therefore be excluded post-cardiac surgery by an apparently normal HD study. Expert input is always required if this is suspected.
Like cardiac tamponade, a tension pneumothorax can cause obstructive shock and haemodynamic instability. A quick assessment for lung sliding can rule this out in a matter of seconds.
Q10. Is there venous congestion?
High venous pressures reduce organ blood flow, causing organ dysfunction and injury, and have been linked specifically with acute kidney injury 20 and post-operative delirium. 21 Central venous pressure (CVP) measurements of >8mmHg have been shown to be particularly harmful. 22 However, patients do not always have CVP monitoring and the absolute CVP value does not necessarily predict whether venous flow is affected. If CVP is unavailable, or if the effect CVP is having on the venous system is in doubt, then ultrasound should be used.
The method adopted for FUSIC HD – the venous excess ultrasound (VExUS) score (see Table 4) – has high specificity for predicting acute kidney injury, outperforming CVP and isolated inferior vena caval (IVC) measurements. 23 High venous pressures first manifest on ultrasound as IVC dilatation, then as flow abnormalities in the great veins when assessed with PW Doppler. 24 The hepatic vein, portal vein and renal veins are key targets for assessment. Figure 2 illustrates normal and abnormal venous Doppler patterns.
The Venous Excess UltraSound (VExUS) score.

IVC = inferior vena cava, S = systolic Doppler wave, D = diastolic Doppler wave, PI = pulsatility index.
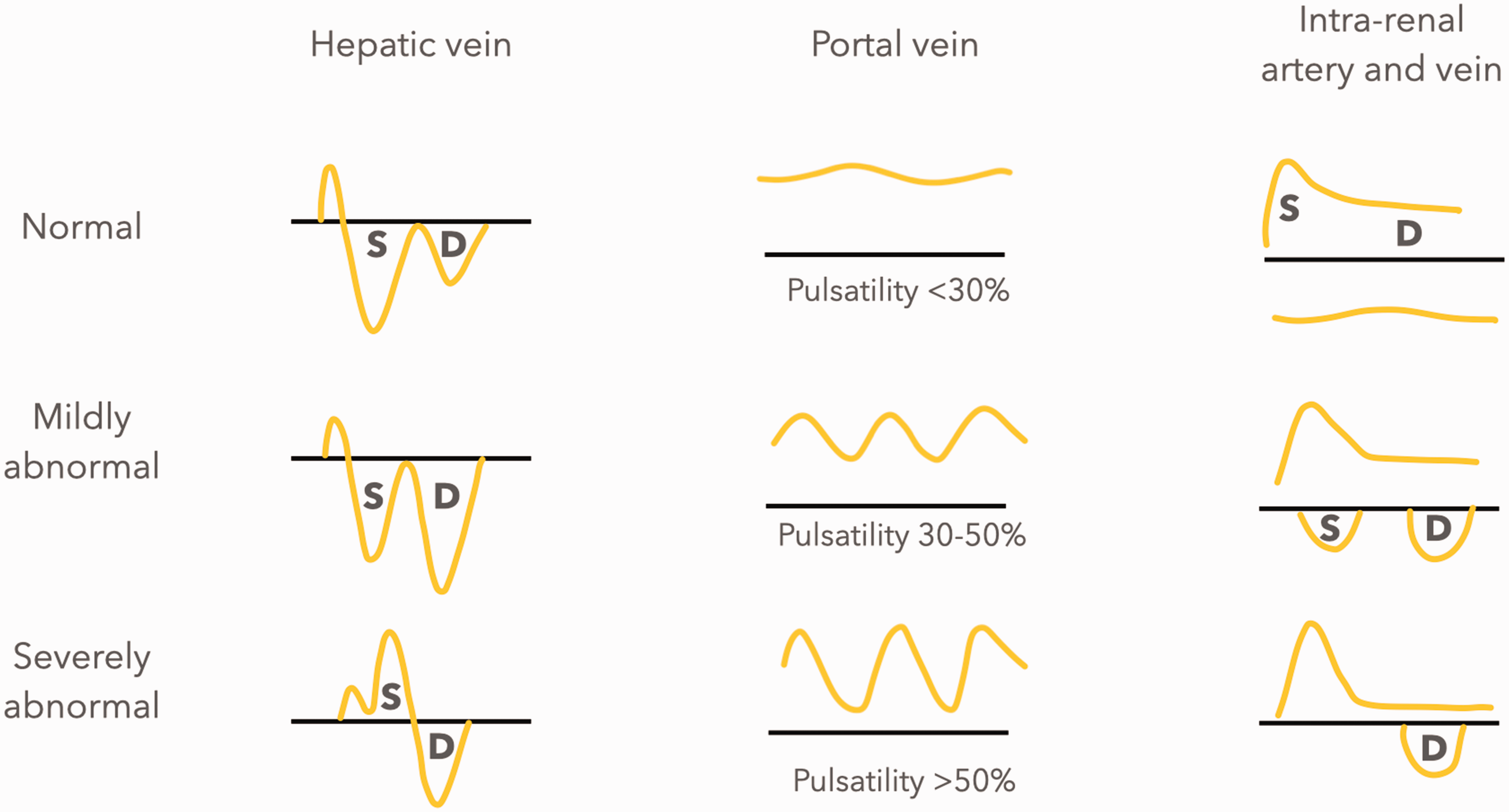
Normal and abnormal venous waveforms in VExUS scoring.
Heart disease, lung disease and volume overload are all causes of venous congestion, as high pressure anywhere downstream of the veins will result in high venous pressures. Causes therefore include LV failure, MR, pulmonary disease, RV failure, TR and pericardial effusions – all of which can be detected with ultrasound. Iatrogenic fluid overload is also a common cause in critically ill patients. Indications for venous congestion assessment include assessment of volume status, acute kidney injury, delirium, newly deranged LFTs, guidance of fluid removal, heart failure and a CVP >8 mmHg.
Performing the scan
Clinicians undertaking FUSIC HD training are expected to already be accredited at the level of FUSIC Heart, Lung and Abdomen, which provide the fundamentals on which a full FUSIC HD scan is built. FUSIC Heart teaches 4 basic cardiac views while FUSIC HD introduces additional views. These can be seen in Table 5.
Additional views obtained in FUSIC HD compared to FUSIC heart.
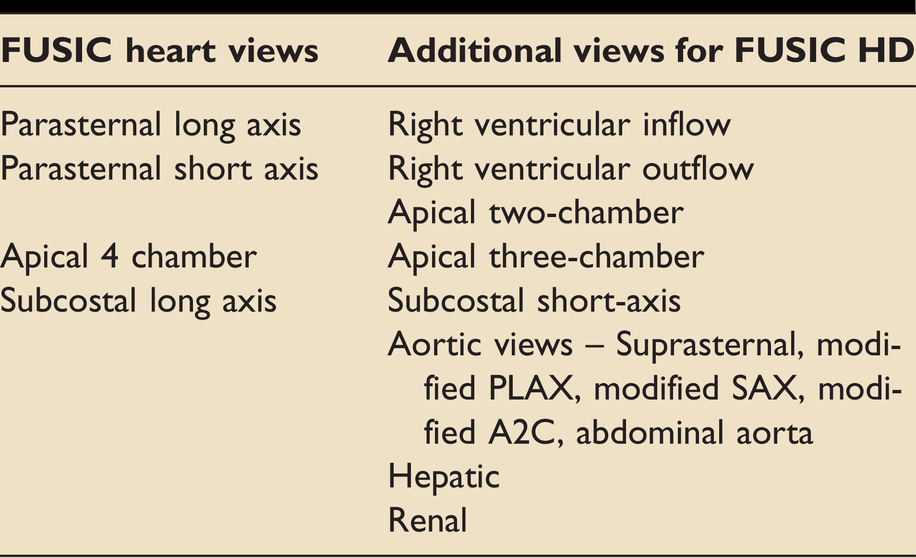
The views, measurements and calculations required for a full FUSIC HD scan are listed in table 1s in the supplementary online appendix. Table 2s provides a step by step sequence of the suggested order of image acquisition with details of probe placement, example images, technical settings, measurements and calculations to be performed, and what to look for in each view. Several of the aortic views are modified standard echo windows. The operator can obtain these views whilst on the relevant echo view or perform them separately. If an aortic dissection is suspected it would be advisable to perform these separately.
Readers are encouraged to look at the FUSIC section of the Intensive Care Society website for e-learning resources. Comprehensive lists of normal and abnormal values for echo measurements have been published by both the BSE 25 and ASE 26 . These have been simplified for FUSIC HD and, combined with other important measurements, can be seen in Table 3s in the supplementary online appendix.
Conclusion
FUSIC HD is a method by which the whole haemodynamic system can be assessed with ultrasound. It allows the bedside clinician to answer the key haemodynamic questions that matter when treating critically ill patients, many of which are difficult to answer by other means. It provides a structured framework and competencies, and enables accreditation by supervised practice, logbook collection and assessment by examination. Comprehensive details, including more detailed explanations of the clinical questions and how to perform a FUSIC HD scan, can be found on the ICS website.
Supplemental Material
sj-pdf-1-inc-10.1177_17511437211010032 - Supplemental material for FUSIC HD. Comprehensive haemodynamic assessment with ultrasound
Supplemental material, sj-pdf-1-inc-10.1177_17511437211010032 for FUSIC HD. Comprehensive haemodynamic assessment with ultrasound by Ashley Miller, Marcus Peck, Tom Clark, Hannah Conway, Segun Olusanya, Nick Fletcher, Nick Coleman, Prashant Parulekar, Jonathan Aron, Justin Kirk-Bayley, Jonathan Nicholas Wilkinson, Adrian Wong, Jennie Stephens, Antonio Rubino, Ben Attwood, Andrew Walden, Andrew Breen, Manprit Waraich, Catherine Nix and Simon Hayward in Journal of the Intensive Care Society
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
