Abstract
The arrival of the COVID-19 pandemic in early 2020 threatened to overwhelm the NH ability to provide sufficient critical care support to patients in the UK. In response to a rapid rise in cases in March 2020, the UK Government issued a call to industry to rapidly design and develop additional ventilators to expand the UK’s capacity for mechanical ventilation. Three NHS consultants working in conjunction with TTP Plc (The Technology Partnership), were at the forefront, evolving the Government brief and developing a safe and effective ventilator, the CoVent™, in less than 5 weeks. The project demonstrates the ability of physicians to guide industry and pool knowledge and resources to rapidly develop and evolve technology in the face of a national emergency. This article discusses key aspects of the design process, highlights the unique human factors and engineering aspects of undertaking this amidst the coronavirus pandemic. Overall we demonstrated that when industry, healthcare and regulatory bodies collaborate and communicate efficiently, huge progress can be made in a fraction of the usual timescales.
Introduction
The COVID-19 pandemic caused by the SARS-CoV-2 virus placed unprecedented demands on health services around the world, in particular critical care. The first autochronous case in the UK with no direct or indirect travel links was confirmed on the 28th February 2020 and the first COVID-19 death on the 5th March 2020.
The scale of the pandemic, combined with the high proportion of patients requiring mechanical ventilation, presented the possibility of insufficient availability of ventilator equipment, even in advanced healthcare systems. The UK was in an especially precarious situation as it has a relatively small number of critical care beds for the size of population when compared to many other European healthcare systems.1,2
On the 13th March 2020, in response to growing concerns over the lack of ventilators, the UK Government called on manufacturers and engineering companies to work together to design and produce devices to support the NHS (National Health Service). Following this, on the 18th March, the Government issued the specification for a Rapidly Manufactured Ventilator System (RMVS), although the key operating parameters were subject to change.
Of the many companies that answered the call, TTP Plc (The Technology Partnership), developed and tested the CoVent™ in under 5 weeks; a process that would normally take around 5 years. The clinical lead of the TTP project (MG) was advised by frontline clinicians, Consultant Anaesthetists (MW and JF) and a Consultant Intensivist (MS), from the Royal Free Hospital, London. This clinical input drove many of the development decisions of CoVent, including the device risk management (led by DS) and human factors (led by DL).
This article discusses the design process, highlighting the unique clinical, human factors and engineering aspects of undertaking this amidst the coronavirus pandemic.
COVID-19: Clinical aspects
Critically ill patients with COVID-19 typically present with profound hypoxaemia, high fever and bilateral pulmonary infiltrates on chest radiography. Despite the severity of their respiratory failure many of these patients present with minimal respiratory distress, known as ‘silent hypoxia’. 3 Because of the degree of hypoxaemia, perceived speed of disease progression and fear of viral aerosolisation, patients were frequently intubated and ventilated relatively early in the evolution of the disease.
At the outbreak of the pandemic, a key principle was to avoid ventilator associated lung injury (VALI) by adopting established guidelines associated with the ventilation of patients with Acute Respiratory Distress Syndrome (ARDS). 4 Our initial experience and early information from Italy and China suggested that a high positive end expiratory pressure (PEEP) strategy would be most beneficial in conjunction with lung protective ventilation parameters in line with the pre-existing ARDSNet recommendations. 5
With increased experience we found the disease to be much more complex than anticipated. Many patients transitioned to a low respiratory compliance that presented problems with maintaining lung protective ventilation whilst achieving adequate gas exchange. Ventilation in the prone position was found to be helpful in these patients but a high proportion of patients developed additional COVID-related complications during their intensive care admission.6,7
Our approach to patient management evolved during the pandemic as a result of our experiences with ventilator associated lung injury and subsequent air leaks. Along with many centres, 8 we adopted a strategy of lower PEEP and peak airway pressures using early proning to facilitate this. Many patients with more severe COVID-19 required prolonged mechanical ventilation and eventual tracheostomy insertion to facilitate weaning from respiratory support.
Ventilator specifications
The UK Government estimated in early March 2020 that the NHS would need to increase access to ventilators from the existing 5,000 to around 30,000 and this demand would be met by procurement of existing devices and rapid development of novel machines. However, the supply of pre-existing ventilator designs was heavily limited by supply chain issues preventing the required massive upscaling of production in the short term.
Initial ventilator specification
The initial ventilator specification from the UK Government was published on the 14th March 2020 and underwent revision and refinement by the Medicines and Healthcare products Regulatory Agency (MHRA) and clinicians in the following days. The initial technical requirements reflected expectation that a large number of ventilators would be required within a very short timeframe, and as a result the specification only focused on key life-saving features. Hence, the specification outlined that the devices should have two settings for oxygen/air mix and tidal volume as well as an adjustable respiratory rate, but not necessarily an ability to provide variable inspiratory:expiratory ratios, PEEP or support spontaneous breathing.
Importantly, the ventilator had to be constructed principally from readily sourced ‘off-the-shelf’ components with a low risk UK supply chain.
Clinical priorities for ventilator development
Discussions from 16th March 2020 onwards between MW, MS and TTP Plc focussed on refining the initial Government specification based on our clinical expertise and knowledge, especially that gained from the early COVID-19 critical care literature.9,10 Additionally, we anticipated that later in the critical care admission it would be necessary for a ventilator to support spontaneous ventilation as respiratory weaning proceeded, in light of the frequently prolonged disease course. The ventilator would therefore require the ability to provide inspiratory pressure support and continuous positive airway pressure (CPAP) as an essential component rather than desirable.
Oxygen shortage
We had already identified in our own institution that oxygen supply may well become compromised due to overwhelming demand. Limitations on supply to patients was not just due to inadequate oxygen reserves, but also due to a limitation on maximum flow rate deliverable by the oxygen manifold. We therefore made recommendations for a circle rebreathing system to be incorporated into the ventilator in order to minimise oxygen requirements, to as low as 250 ml/min, depending on the patient’s basal oxygen consumption and circuit leak. We fed the results of our recommendations back to the Government.
Revised ventilator specification
On the 18th March 2020 the MHRA published a revised ventilator specification, for devices to be approved under an exceptional use authorisation, and stated that ‘it is proposed these ventilators would be for short-term stabilisation for a few hours, but this may be extended up to 1-day use for a patient in extremis as the bare minimum function. Ideally it would also be able to function as a broader function ventilator which could support a patient through a number of days, when more advanced ventilatory support becomes necessary.’
Technical requirements were changed to reflect the needs of the patients and included:
11
Must have mandatory ventilation (for the deeply sedated and paralysed patient); Optional pressure support mode for those patients breathing to some extent themselves; Plateau pressure should adapt to achieve volume and be limited to 35 cm H2O; Peak pressure should be no more that 2 cm H2O greater than plateau pressure; Ideally there should be a mechanical failsafe valve that opens at 40 cm H2O; PEEP range 5–25 cm H2O adjustable in 5 cm H2O increments.
Furthermore, on 10th April 2020, based on clinical experience, the guidance was revised to include the necessity for closed endotracheal suctioning and a greater emphasis on spontaneous breathing modes. 12
Ventilator development
The CoVent™ project team was assembled within 24 hours of the Government’s call and quickly grew to over 140 members. Medical device development mostly follows a waterfall model whereby an initial specification phase is followed by concept generation, detailed design and prototyping, before moving on to verification testing and scaling for manufacture. This often takes years. Given the urgency of the project, these tasks were conducted in parallel. By necessity, the pneumatics, mechanics, user interface and electronics of the ventilator were all designed simultaneously before the first MHRA device specification had been released on 18th March 2020.
First iteration
An initial prototype was built and bench tested, before being shown (virtually) to the clinicians (Figure 1). Simulated lungs were used to mimic various clinical scenarios. Pressure, flow and volume waveforms were generated. Results of the bench testing were then fed back to the clinicians who further refined the ventilator design. The device was able to deliver adjustable pressure-controlled breaths with variable PEEP, respiratory rate, inspiratory:expiratory ratio and fraction of inspired oxygen (FiO2).

Rapid evolution of prototypes – Monday 16 March, Tuesday 17 March, and testing during Week 3 (left to right).
Simultaneously, preparatory regulatory documentation and instructions for use were being drafted, without confirmation of the final device design. Prototype units were being developed and built at the same time as the production line was being laid out.
User interface development
MW and MS worked closely with the TTP Plc human factors team to develop the user interface. The clinician’s expertise with both critical care ventilators and the clinical demands of COVID-19 patients, resulted in over 15 iterations of the user interface in less than two weeks, each with incremental improvements. Initial iterations were made using digital animations and photos, before moving to physical mock-ups and prototypes. Consequently, many of the usability issues that might be difficult to anticipate had already been overcome by the time hardware implementations had been built (Figure 2).
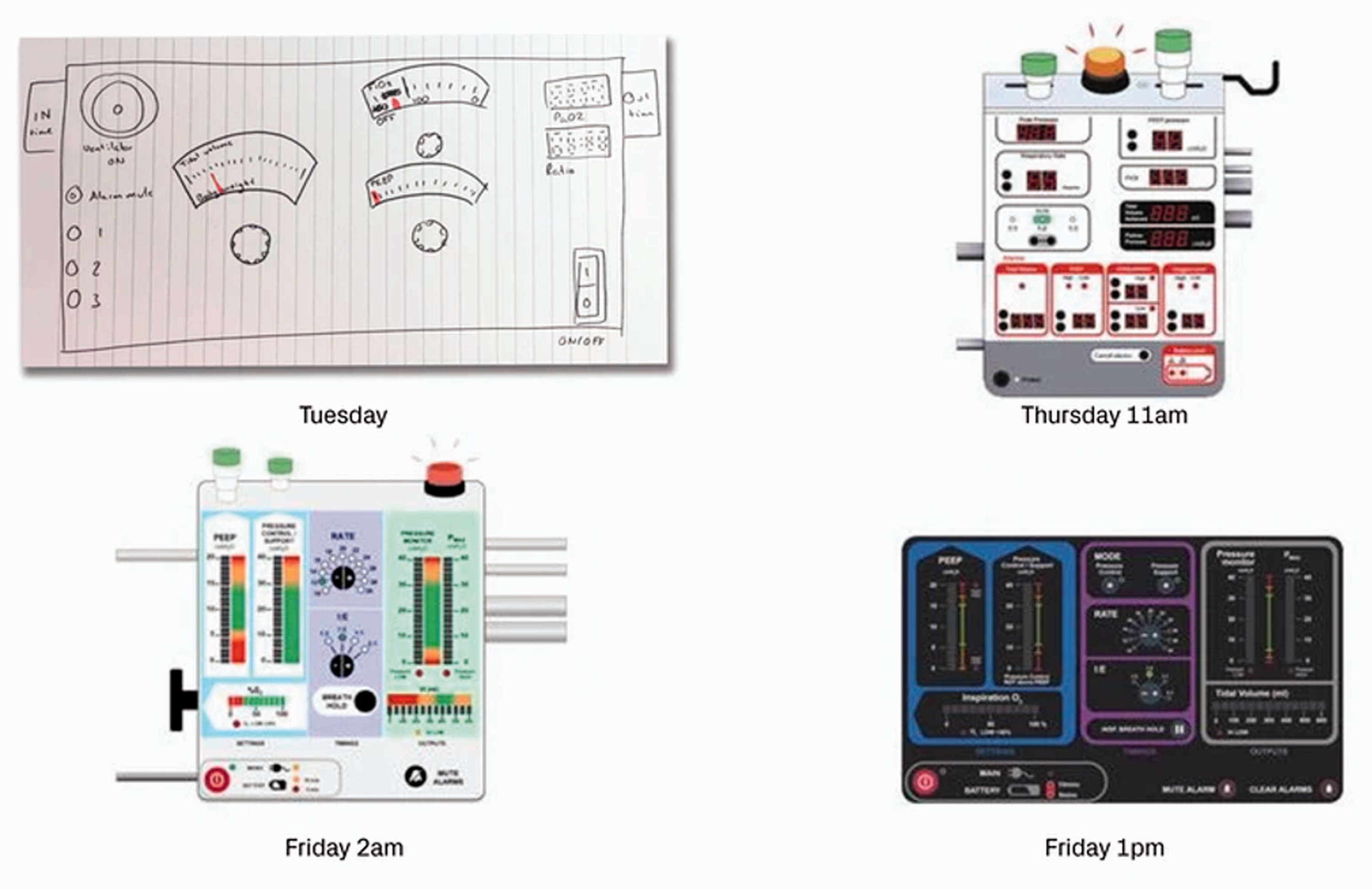
Rapid development of the user interface between Tuesday 17 March and Friday 20 March 2020.
Second iteration
By the end of March, as the prolonged duration of ventilation required in COVID-19 became known, adapting the ventilator to incorporate a spontaneous breathing mode became important. Subsequently, on 10th April, the MHRA added patient-triggered breathing as a necessary part of the specification. Clinical, hardware, firmware, human factors and electronics teams came together to create a modified system within 48 hours, although this was a much more complex task than the provision of mandatory ventilation.
In addition, the revised guidance also required compatibility with closed endotracheal suctioning, necessitating a pneumatic design change to prevent the generation of negative airway pressures during suctioning.
First-in-human trial development
While it did not ultimately take place, as the number of ventilators required by the Government was not as high as initially forecast, a phase 1 clinical trial was designed to assess whether the device provided reasonable safety and effectiveness in COVID-19 patients. A pragmatic approach to trial development, led by JF, was required with condensed timelines, aiming for recruitment, intervention and data collection within 72 hours. A sequential cohort, non-randomised, non-blinded, non-controlled single-centre study in ten COVID-19 patients was developed. The primary outcome was all-cause mortality at 24 hours following commencement of trial ventilation. A range of secondary outcomes were included, principally the maintenance of predetermined physiological parameters within first 48 hours of ventilation, the incidence of clinical complications and ventilator malfunction. The inclusion criteria focussed on severely unwell adult patients (paO2:FiO2 < 200 kPa) and the study excluded patients with cardiovascular complications, existing pneumothorax, pre-morbid lung disease or evidence of haemophagocytic lymphohistiocytosis. A range of safety components were incorporated into the study design, including the immediate availability of a conventional ICU ventilator should malfunction of the CoVent™ occur.
Agile and parallel working
The development of the CoVent™ happened without a traditional line management structure, instead by permitting highly distributed authority and responsibility. The success of this method of working was due to tight coordination and simultaneous execution of tasks by experts in their field (Figure 3). Akin to a high performing sports team, groups joined forces to solve problems but also worked autonomously and independently with common purpose.
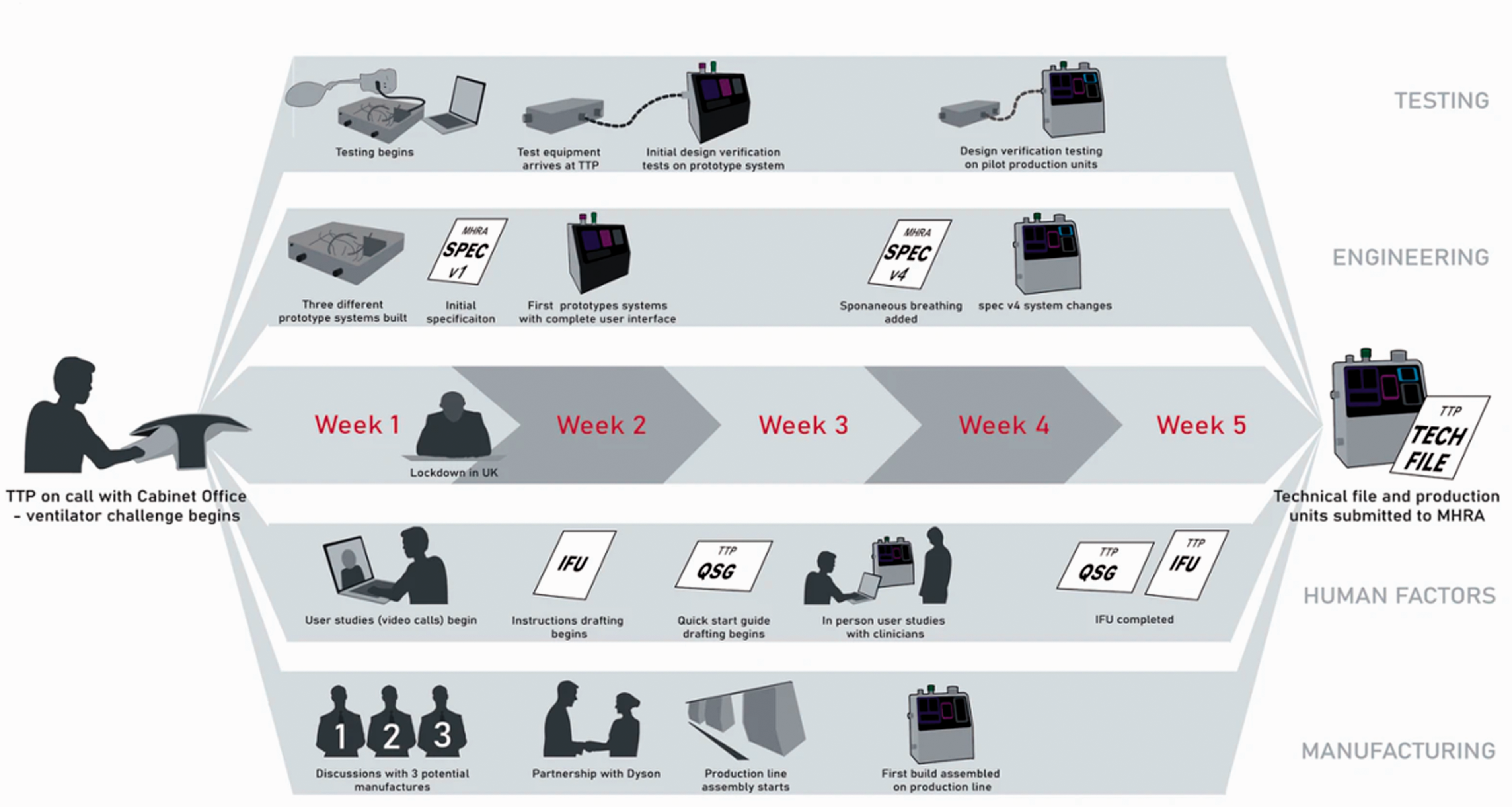
Parallel streams during the development of CoVent, TTP's rapid manufacture ventilator for COVID-19 patients.
Clear and immediate communication within the team was key to ensuring that individuals had clear goals so as to make the right decisions. Early on in the project, 23 ‘single points of contact’ were designated for the multiple work streams (ranging from ‘clinical’ to ‘mechanical development’ to ‘risk management’). Decisions were taken by those with the most immediate knowledge, and the highly parallel nature of the process made it easier to identify the knock-on effects of a single decision as early as possible within the design phase while they were still easy to change.
Managing risk and uncertainty
Early on in the project there was significant uncertainty around the specification and, as discussed, it continued to evolve. In order to make rapid progress it was critical for the clinicians to have significant input into what ventilator features were likely to be must-haves rather than should-haves. As MS, MW and JF became more familiar with the history and management of COVID-19 and fed this forward, the TTP Plc team were able to iterate the ventilator design in real time.
This process was maintained throughout the development, with almost daily communication, and facilitated the translation of clinical requirements into actual technical requirements, as well as rapid decision making with respect to the overall goal.
Even with such stakeholder input, there were still many risks which at the outset of the design process were unknown and impossible to quantify. To manage this, the design team adopted a principle known as Redundancy Based Design (RBD). Multiple independent concepts were pursued through the concept development stage, which were deliberately selected to have different risk profiles. For example, a pneumatic system using solenoid valves was progressed in parallel with a mechanical bellows system using motors. This mind-set continued throughout the development and meant that as the clinical need evolved or unknown risks became apparent, we were able to pivot and quickly adapt to the new situation by drawing on some of the other device concepts.
Getting it right first time vs. fail fast and often
Medical devices are often developed following a right first time approach since they must not have a significant number of faults that arise in the field. This mind-set generally flows through development, with significant effort going into analysis and testing, and proving the design at each stage.
In contrast, for the CoVent™ project, we accepted that some level of programme risk needed to be taken if the urgent timeline was to be met. This is as opposed to device risk: most of all, the ventilator needed to be safe, adhering to design specified in standards like IEC 62366-1:2015 13 and ISO 14971:2019. 14 Despite the short timescales the human factors team followed this approach rigorously. Having engaged clinicians on-hand meant that clinical input could be sought to a greater than usual extent for other aspects of the design process such as review of hazard analysis and user error modes. This input was essential to getting early and authoritative input that helped prioritize engineering efforts to design out potential sources of use error. Consequently, many of the usability issues that might be difficult to anticipate had already been overcome by the time hardware implementations had been built.
Within three days of the kick-off call, teams were testing integrated prototype systems made from off-the-shelf parts on simulated lungs, and this rapid testing and iteration carried on throughout the project. Such rapid testing helps to quickly flush out the unknown unknowns, which often can have a significant effect on timelines. Ventilators have validated in vitro test procedures that simulate real-world clinical performance. This meant that within the first week of development, the team procured ventilator flow analysers to be able to benchmark performance. As a result, we were able to perform many formal verification tests, where the design is assessed against all aspects of the specification, throughout the development cycle as opposed to only late in the development.
Conclusions
The COVID-19 pandemic placed unprecedented demands on the NHS including an anticipated need for critical care ventilators that far exceeded the supply. In response to the Government’s call, industry worked with key stakeholders in previously unimaginably rapid timescales to develop, test and produce ventilators that exceeded the brief.
The CoVent™ project was extraordinarily challenging for a number of reasons, not least because the clinicians involved had to balance working at the critical care coal-face during the pandemic with the need to provide timely input into the project. However, this also provided a distinct advantage as the requirements of the device were changed based on rapidly accumulating clinical experience. The project development necessarily involved a number of key internal and external stakeholders each providing input that necessitated often very rapid responses and sometimes changes to the device design. This was demanding on the project team and in some cases involved going ‘back to the drawing board’, often out of hours. Good communication was fundamental to this.
The success of the of the project was in part due to the fact that the normal ways of working in medical device development were superseded with innovative and agile techniques that evolved to enable rapid development whilst simultaneously managing the risks. As our article describes, a positive and trusting team culture was generated from the outset that allowed coordinated parallel working, ensuring that the device could be designed, tested and manufactured in a very short space of time.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
