Abstract
The use of lower tidal volume ventilation was shown to improve survival in mechanically ventilated patients with acute lung injury. In some patients this strategy may cause hypercapnic acidosis. A significant body of recent clinical data suggest that hypercapnic acidosis is associated with adverse clinical outcomes including increased hospital mortality. We aimed to review the available treatment options that may be used to manage acute hypercapnic acidosis that may be seen with low tidal volume ventilation. The databases of MEDLINE and EMBASE were searched. Studies including animals or tissues were excluded. We also searched bibliographic references of relevant studies, irrespective of study design with the intention of finding relevant studies to be included in this review. The possible options to treat hypercapnia included optimising the use of low tidal volume mechanical ventilation to enhance carbon dioxide elimination. These include techniques to reduce dead space ventilation, and physiological dead space, use of buffers, airway pressure release ventilation and prone positon ventilation. In patients where hypercapnic acidosis could not be managed with lung protective mechanical ventilation, extracorporeal techniques may be used. Newer, minimally invasive low volume venovenous extracorporeal devices are currently being investigated for managing hypercapnia associated with low and ultra-low volume mechanical ventilation.
Introduction
Acute respiratory failure is one of the common indications for admission of patients to intensive care. Most of these patients require mechanical ventilation to assist in management of respiratory failure. Mechanical ventilation that was used in the past was aimed at maintaining blood gasses at normal ranges. This often required high inspiratory pressures that were subsequently shown to worsen lung injury and respiratory failure. 1 A strategy of reducing inspiratory pressures on mechanical ventilation appeared to reduce the mortality. 2 The current standard of care in treating patients with acute hypoxic respiratory failure is to use low tidal volume and low inspiratory pressure mechanical ventilation. 3 One of the effects of such ventilation strategy is development of hypercapnia and hypercapnic acidosis.
The effects of hypercapnia and acidosis in critically ill patients are not clearly established. Some clinicians believe hypercapnic acidosis to be protective by itself independent of low volume ventilation and may aid in reducing the lung injury and mortality. 4 Indeed, they have hypothesised that inducing hypercapnia by supplemental carbon dioxide (CO2) may be beneficial in critically ill patients with acute respiratory failure. 5 To the contrary, other clinicians consider hypercapnic acidosis to be harmful with possible increase in mortality and morbidity.3,6 This uncertainty appears to be based on data from animal experiments or clinical studies with small sample sizes. These factors also limit the validity and generalizability of these studies.
Over the recent past, several large studies evaluated hypercapnia and hypercapnic acidosis in various clinical settings. Our group investigated the effects of hypercapnia and hypercapnic acidosis during the first 24 h of intensive care admission in over 250,000 mechanically ventilated patients. 7 In this study, the adjusted hospital mortality of patients with compensated hypercapnia and hypercapnic acidosis was significantly higher than those patients who had normocapnia and normal pH. Furthermore, the adjusted odds of hospital mortality increased with increasing hypercapnia. 7 The study by Nin et al. 8 investigating the effects of hypercapnia during the first 48 h of intensive care admission in patients with acute respiratory distress syndrome (ARDS) showed that severe hypercapnia, as defined by PCO2 > 50 mmHg, was independently associated with increased ICU mortality, and higher complications including barotrauma, renal dysfunction, and cardiovascular dysfunction.8,9 Hypercapnic acidosis was also found to be independently associated with increased mortality in various diagnostic categories of mechanically ventilated patients including acute cerebral injury, trauma, sepsis, cardiac, respiratory, gastrointestinal and renal causes of admission to ICU.10,11 From these data, it appears that severe hypercapnia especially when associated with acidosis should be avoided or actively managed.7,8,10,12 We performed a review of published literature to identify the therapeutic options that are currently available to manage acute hypercapnia in critically ill patients requiring invasive mechanical ventilation.
Search strategy
MEDLINE via PubMed (from inception to June 2019) and EMBASE (from inception to June 2019) were searched. The search was performed using the following exploded medical subject headings and text words ‘CO2’, ‘hypercarbia’, ‘hypercapnia’, ‘acidosis’, ‘critically ill’ or ‘critical care’ or ‘management’ or ‘treatment’ in isolation and in combination without restrictions. Studies including animals or tissues were excluded from the review. We also searched bibliographic references of relevant studies, irrespective of study design with the intention of finding relevant studies to be included in this review.
Summary of interventions available for the treatment of hypercapnia.
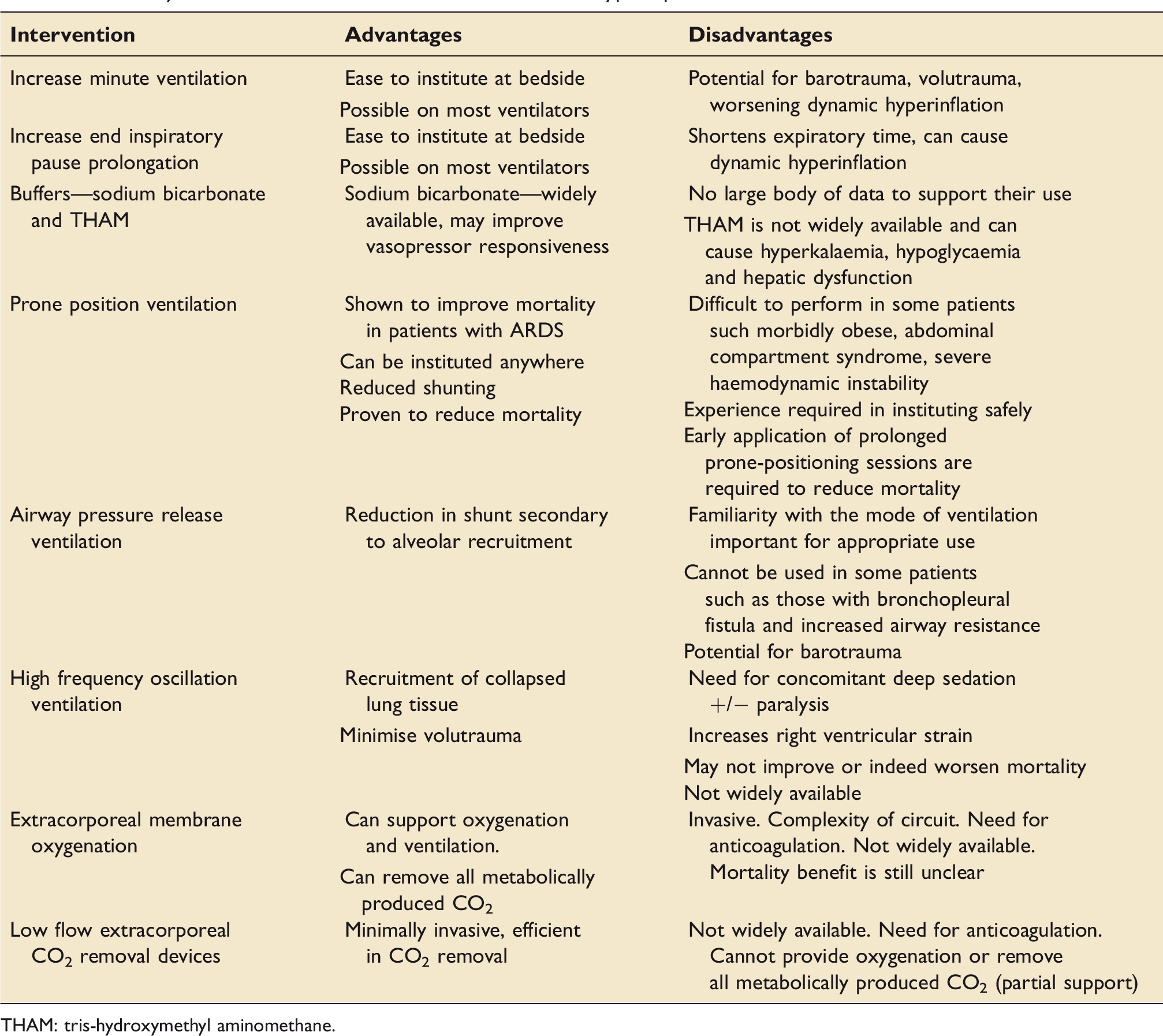
THAM: tris-hydroxymethyl aminomethane.
Mechanical ventilation
The efficacy of lower tidal volume ventilation to improve mortality was initially shown in observational studies. 2 This was considered to be due to a reduction in lung injury with low tidal volumes as well as the associated hypercapnia with lower tidal volume ventilation. In randomised controlled trials, a strategy of low volume ventilation when associated with hypercapnia actually demonstrated a trend towards increased mortality. 6 However, when low volume ventilation was performed while ensuring normocapnia the mortality was significantly reduced. 3
Optimal management of mechanical ventilation should remain the primary modality of prevention or correction of hypercapnic acidosis.
Increasing minute ventilation
Changes in delivery of conventional mechanical ventilation including optimising the settings such as increasing respiratory rate (and minute ventilation) as recommended by the ARDS network 3 may help in management of hypercapnic acidosis.
Increasing respiratory rate and minute ventilation, however, involves a trade-off with higher minute ventilation needing greater delivered power of mechanical ventilation. Higher mechanical power has been associated with increased risk of in-hospital mortality using data from two separate ICU databases. 13 Mechanical power can be measured using dynamic pressure volume curve in volume targeted ventilation mode, recorded during tidal ventilation. 14 Mechanical power can be calculated at the bedside and comprises of all the ventilator related variables that contribute to ventilator induced lung injury (VILI) (tidal volume, respiratory rate, driving pressure and airway resistance). 14 The increase in respiratory rates may, however, be associated with dynamic hyperinflation and right ventricular dysfunction without clearance of hypercapnia. 15 While increasing respiratory rate, one must be mindful of the impact on other ventilatory variables (inspiratory flow rate and expiratory time) as well as on mechanical power to achieve best possible balance between restoring normocapnoea and potential for VILI. 16 Interpretation of ventilator waveforms to adjust the respiratory rate may aid in avoiding ventilator associated lung injury. 17
Reducing dead space ventilation
Dead space ventilation causes hypercapnia and was shown to be associated with an increased mortality. 18 Techniques that could reduce dead space include alteration in the ventilation circuit and strategies to reduce physiological dead space are relatively simple to implement in patients with hypercapnic respiratory failure. Catheter mounts are routinely used to connect the Y-piece to the endotracheal tube, mainly to prevent accidental extubation. Modifications to the ventilator circuit such as connecting Y-piece directly to the endotracheal tube to reduce the dead space in ventilator circuit. 19 Changes to the circuit in mechanical ventilation including removal of heat and moisture exchanger and using heated humidifier was also shown to reduce hypercapnia without the need for increase in the need for increasing tidal volumes or the respiratory rate.
Reduction in physiological dead space
End-inspiratory pause prolongation was shown to increase clearance of hypercapnia in ARDS patients. 20 By increasing end-inspiratory pause prolongation from 0.1 to 0.7, Bermeo et al. demonstrated a significant decrease in PaCO2 from 54 ± 9 to 50 ± 8 mmHg. 20 They showed that the decrease in PCO2 was due to a reduction in physiological dead space. 20 The diffusion of CO2 during respiration is time dependent. End inspiratory pause prolongation increases the time available for alveolar gas exchange of CO2 and hence its elimination. It must, however, be noted that end inspiratory prolongation may be associated with potential adverse effects such as an increase in intrinsic positive end expiratory pressure (PEEP) and inversion of inspiration to expiration (I/E) ratio that can increase mean airway pressure and cause dynamic hyperinflation.
Lung recruitment and PEEP titration
PEEP is aimed to maintain recruitment of the lung regions opened during previous inspiration. 21 Optimum PEEP can reduce physiological dead space by recruiting atelectatic lung tissue, while excessive PEEP can increase dead space by over distension of the alveoli. With regional heterogeneity in diseased lung both over distension and recruitment could happen at the same time, requiring the clinician to strike a delicate balance between the two phenomena units.
However, deploying ‘open lung strategy’, lung recruitment manoeuvres with PEEP titration intended to improve oxygenation and reduce VILI, are shown to cause severe hypercapnia during the first 24 h of institution. 22 The strategy of lung recruitment manoeuvre and PEEP titration according to the best respiratory–system compliance was recently investigated by Cavalcanti et al. in a large multinational, multicentre randomised controlled trial. 22 This study showed a higher 28-day and 6-month all-cause mortality in patients who were treated with lung recruitment. Higher mortality noted with lung recruitment was attributed to several factors including changes in driving pressure and lung over distention, breath stacking, need for neuromuscular blockade and haemodynamic compromise. 22 It is important to note that in the lung recruitment group, hypercapnia and acidosis during the first hour of randomisation was observed, which was shown to be associated with higher mortality in mechanically ventilated patients.7,8
Buffers in the management of hypercapnic acidosis
The use of buffers in the management of hypercapnic acidosis remains controversial. 23 Sodium bicarbonate and Tris-hydroxymethyl aminomethane (THAM) were both used in clinical practice to buffer hypercapnic acidosis.3,24,25 Kallet et al. 25 demonstrated THAM in improving arterial pH and base deficit, with a reduction in PCO2 that could not be fully accounted for by ventilation. Weber et al. investigated the use of THAM in ARDS patients where permissive hypercapnia was implemented for 2 h aiming for a target PCO2 of 80 mmHg. In their randomised controlled trial of 12 patients with ARDS, the use of THAM buffering attenuated depression of myocardial contractility and hemodynamic alterations during rapid permissive hypercapnia institution. 24 The ARDS network trial recommended the use of sodium bicarbonate when pH was lower than 7.1.3 However, bicarbonate infusions should not be administered to patients who are hypoxemic and or having lactic acidosis.
Some patients with severe ARDS will be hypercapnic in spite of best possible conventional ventilation. In such patients, other modalities in addition to conventional ventilation may be required. These include APRV, prone position ventilation and high frequency oscillatory ventilation
Airway pressure release ventilation
APRV entails continuous positive airway pressure at a high level, with intermittent time cycled release, to maintain alveolar recruitment and lung volume. Patients can breathe spontaneously, independent of the phase of respiration, through a biphasic positive pressure circuit. 26 This allows movement of posterior muscular part of the diaphragm increasing distribution of ventilation to dependent posterior lung regions, improving ventilation perfusion matching, as compared to movement of anterior tendinous region of diaphragm during controlled mechanical ventilation. APRV was initially described as a spontaneous mode of ventilation to treat patients with acute lung injury with aim of maintaining lower airway pressure and to allow unrestricted spontaneous ventilation. There are some reports suggesting that APRV may prevent progression of acute lung injury in high-risk trauma patients. 27 APRV was also shown to be effective in reducing CO2 as well as improving oxygenation without increasing minute ventilation 28 in conjunction with a reduction in peak and mean airway pressures. Improvement in gas exchange with APRV is related to the reduction in dead space ventilation. The use of APRV, however, is not widespread and this mode is not available in all commercially available ventilators. 29 Furthermore, the improvement in survival with this mode of ventilation remains to be evaluated. Higher mean airway pressures and low release time reduce the applicability of APRV in patients with conditions such as bronchopleural fistulae, raised intracranial pressure, right ventricular dysfunction and with prolonged expiratory time constants. Patients who cannot breathe spontaneously because of neuromuscular paralysis or diaphragmatic weakness do not benefit from this mode either.
Prone position ventilation
Prone position reduces the heterogeneity of ventilation of dorsal to ventral lung regions as compared to supine position, increasing homogeneity of stress and strain as a result. 30 By virtue of ventral de-recruitment exceeding dorsal recruitment, or reduction in hyper inflated ventral regions, prone position ventilation can result in reduction in dead space. Indeed, prone position ventilation was shown to decrease hypercapnia in patients with ARDS 31 especially with those who are responders to prone position ventilation. 31 Prone ventilation was also shown to improve oxygenation and improve mortality in patients with ARDS. 32 Prone position ventilation aids in recruitment of previously excluded alveoli reducing the dead space ventilation.
High frequency oscillation ventilation
HFOV is a ventilatory technique where using an oscillatory pump breathing frequencies of 180–900 breaths/min (3–15 Hz) are used with very small tidal volumes (1–4 ml/kg) at a constant airway pressure, potentially reducing volutrauma, decreasing anatomical dead space, and improving ventilation-perfusion matching. HFOV was used as a mode of lung protective ventilation. The recent studies on adult patients with ARDS showed no significant benefit and with possible increase in mortality. 33 It may be of benefit in-groups of patients with more severe ARDS (PaO2/FiO2 < 64 mmHg) where hypercapnia is unresponsive to conventional ventilation. 34 Friesecke et al. reported the use of HFOV in patients with hypercapnic acidosis not responsive to conventional ventilation. 35 In their cohort of 26 patients, 24 patients responded to HFOV with improvement in hypercapnia and acidosis at 24 h of initiating HFOV. It is, however, important to note that the routine use of HFOV was not recommended due to lack of mortality benefit and a potential for harm with the use of HFOV as compared to conventional ventilation with low tidal volumes.
Some patients may have severe hypercapnia that could not be managed with mechanical ventilation alone. In such patients, extracorporeal CO2 removal needs to be considered.
Extracorporeal management of hypercapnia
Extracorporeal management include ECMO and other newer low flow devices specifically introduced to support clearance of CO2.
Extracorporeal membrane oxygenation (ECMO)
ECMO provides oxygenation, ventilation and cardiac assist. Oxygenation with a membrane lung in venovenous ECMO is mainly dependent on the blood flow and CO2 clearance is primarily dependent on the fresh gas flow.
Clinical application of extracorporeal CO2 removal was first reported in an observational study by Gattinoni et al. 36 in an uncontrolled group of patients with severe ARDS. They reported encouraging results in patients with severe ARDS by using this technique as a strategy to ‘rest’ the lungs. The survival rate of the treated patients in this study was 49%. The blood loss was, however, significant (average blood loss 1800 ± 850 ml/day). Subsequently, Morris et al. 37 conducted a randomised clinical trial comparing pressure controlled inverse ratio ventilation with an extracorporeal CO2 removal technique in patients with ARDS. However, no significant difference in survival was found between the mechanically ventilated patients and those treated with the extracorporeal CO2 removal. While there may be several important reasons for this, one of them may be that the concept of lung protective ventilation at that time was not as well established as in the current practice of mechanical ventilation. The use of ECCO2 removal did not gain much acceptance due to complexity, costs and implications of intervention including high blood flow rates, large cannulas and systemic anticoagulation with its associated potential complications. More recently, the use of ECMO had gained momentum with improvements in technology, advances in intensive care practice especially of the anticoagulation while patients were managed on extracorporeal circulation. CESAR trial, 38 incorporating such advances in equipment and clinical practice investigated the use of ECMO in ARDS patients with hypoxic or hypercapnic acidosis with respiratory failure. The results of CESAR showed that an ECMO-based management protocol improved survival without severe disability. However, only a small proportion (about 5%) of patients in this trial had hypercapnic acidosis and the results may not support the routine use of ECMO in patients with hypercapnic acidosis. An observational study on use of ECMO in Influenza A associated ARDS showed that when ECMO was used as rescue therapy, the ICU mortality was 23%. 39 More recently, EOLIA trial, investigated the efficacy of ECMO in patients with ARDS, and did not show a survival advantage with ECMO. 40 This trial again had a relatively small number of patients (<20%) treated with ECMO for hypercapnic respiratory failure. These results may not therefore be generalisable in managing patients with hypercapnic acidosis. Furthermore, the availability of ECMO is limited to very few centres.
Low flow extracorporeal CO2 removal devices
Over the last two decades several less invasive extracorporeal devices were evaluated as alternatives to ECMO support. These are less invasive and less complex devices that may be used to treat hypercapnia and acidosis that may be associated with low tidal volume lung protective ventilation. Most of these less invasive devices provide partial extracorporeal support where they are efficient in clearing CO2, but do not provide significant oxygenation. The cannulas used to access blood vessels are smaller and require minimal anticoagulation similar to renal replacement therapy circuits.
Some of the devices evaluated for extracorporeal CO2 removal include arteriovenous devices that are pumpless and venovenous low flow devices that include a pump to assist in extracorporeal circulation. These devices with pump may be of low flow (300–500 ml/min) or high flow (800–1000 ml/min). 41
Interventional lung assist (ILA) is a sophisticated pumpless extracorporeal arteriovenous CO2 removal device that is driven by the patient’s cardiac output and therefore, do not require extracorporeal pump assistance. ILA was shown to be effective in clearing hypercapnia when tidal volumes as low as 3 ml/kg predicted body weight were used. 42 However, the need for arterial cannulation increases morbidity with complications such as limb ischaemia and arterial pseudoanneurysms. 42
Low flow venovenous devices such as hemolung and prismalung appear to be more promising in being minimally invasive (avoiding arterial cannulation). Initial results mainly from case reports43,44 and feasibility observational studies45,46 appear encouraging. Extracorporeal CO2 removal at low flow (300–500 ml/min) and high flow (800–1000 ml/min), was recently investigated in prospective, multicenter international phase 2 study (the SUPERNOVA study). 47 In this study, the investigators aimed to facilitate ultra-protective ventilation (tidal volume of 4 ml/kg of ideal body weight and a plateau pressure of ≤25 cm H2O) in 95 patients with moderate ARDS. The results of this study showed that ultra-protective ventilation was achieved within 8 h of instituting ECCOR in 78% and in 82% of the patients within 24 h. These results suggest that ECCOR could facilitate ultra protective ventilation avoiding severe hypercapnia in majority of patients. Randomised controlled trials are currently ongoing (REST) 48 to further define the efficacy of these low flow venovenous extracorporeal CO2 removal devices in instituting ultra protective lung ventilation that may lead to a reduction in mortality.
Conclusions
The options to manage hypercapnic acidosis include modifications to mode of mechanical ventilation to enhance CO2 clearance as well as buffers to normalise pH. In patients where hypercapnic acidosis could not be managed with mechanical ventilation, extracorporeal techniques may be used. The newer low flow extracorporeal devices are minimally invasive and are effective in clearing hypercapnia. These devices are currently being investigated in randomised controlled trials to further define their role not only in clearing hypercapnia but to reduce mortality.
Footnotes
Authors’ note
Institution where the work was performed: The Australian and New Zealand Intensive Care Research Centre, Department of Epidemiology & Preventive Medicine, School of Public Health and Preventive Medicine, Monash University.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
