Abstract
Background
Studies consistently report deranged sleep in patients admitted to intensive care unit. Poor sleep has harmful physical and cognitive effects, and an evidence-based intervention to improve sleep is needed. It is, however, difficult to measure sleep in the intensive care unit. ‘Gold standard’ monitoring (polysomnography) is unsuitable for usual care.
Methods
We collected concurrent sleep data from electroencephalograph recordings, activity monitoring, and nurse- and patient-completed Richards-Campbell Sleep Questionnaires (RCSQ).
Results
Electroencephalograph data (n = 34) confirm poor sleep. Individual bouts last approximately 1 min and around 2 h of sleep overnight is common. Correlation between electroencephalograph, self-report, nurse-report, actigraphy and overall activity score is low (ρ = 0.123 (n = 24), 0.127 (n = 22), and 0.402 and − 0.201 (n = 13), respectively). Correlation between nurse and patient assessment is limited (ρ = 0.537 (n = 444)).
Conclusions
No current method of sleep monitoring seems suitable in the intensive care unit. However, to facilitate comparison across studies, the patient-completed RCSQ seems the most meaningful measure.
Introduction
Sleep is important for recovery and general wellbeing. 1 This is particularly true of patients admitted to hospital. 2 Poor sleep has harmful effects on patients, both physical and cognitive, 3 and there is a strong interest and need for a well-designed, evidence-based intervention to improve sleep and, by default, sleep monitoring on the intensive care unit (ICU).3,4
The ‘gold standard’ for sleep assessment is polysomnography (PSG) 5 and a number of small-scale studies have successfully measured ICU patient sleep using PSG. All of these studies have indicated abnormal sleep patterns in patients being treated on the ICU. 6 PSG is impractical for routine use in the ICU environment where patients have complex care needs, and the proximity of a range of electronic devices can cause interference with the recordings.6–10 Reliable methods to enable simple routine monitoring of sleep in the ICU are needed.
The Richards-Campbell Sleep Questionnaire (RCSQ) is designed for patient use in the ICU and is a composite measure of sleep derived from five visual analogue scales (VAS). This is generally considered the most clinically useful sleep observation tool for use in the intensive care unit.11,12 A nurse-completed version is available for when patients cannot complete it themselves. 13
Simplified PSG units with fewer connecting electrodes are available for general use (Sleep Profiler, Advanced Brain Monitoring Inc., Carlsbad, CA, USA). Wrist-worn activity monitoring devices are also commonly used to determine sleep patterns in the general population. Given the impracticalities of widespread PSG in ICU, we designed a study to investigate the feasibility of using these simpler devices to measure sleep in the ICU population. Metrics from these devices can be tested against both patient- and nurse-completed RCSQ questionnaires, with the intention of determining if such equipment has a role in ICU sleep measurement.
Methods
For the purposes of familiarising ourselves with the equipment, and to collect baseline comparator sleep data from healthy adults, we recruited 27 volunteers prior to recruiting patients from the ICU. Approval for this preliminary study was given in March 2014 by the University of Oxford Medical Sciences Inter-divisional Research Ethics Committee (ref: MSD-IDREC-C1-2014-033). All volunteers provided written informed consent to take part in the study.
The subsequent observational study had two primary objectives. The first was to use electroencephalograph (EEG) monitoring to objectively assess the duration and quality of sleep in patients admitted to the ICU. The second was to compare sleep results from other, more practical and cost effective, means of measurement. We collected concurrent sleep data using a wrist-worn activity monitor (MotionWatch 8, CamNtech Ltd, Papworth Everard, UK) and the Richards Campbell Sleep Questionnaire. 14
This was a multi-site observational study. We aimed to recruit approximately 500 non-sedated/ventilated patients to assess nurse- vs. patient-completed RCSQ correlation (cohort 1: questionnaire only). This was a convenience sample, as it was not possible to determine a statistically accurate sample size from existing available data. Due to the practicalities of equipment and software availability, patients from one site (Oxford University Hospitals NHS Foundation Trust (OUH)) were recruited to wear the EEG and activity monitoring equipment as well as complete the RCSQ (cohort 2: questionnaire plus electronic monitoring).
We proposed to recruit 50 patients to wear the EEG monitor. This was a convenience sample size based on other similar studies reported in the literature. A sub-set of these patients also agreed to wear the activity monitor. We also intended to recruit about 10 patients to the EEG monitoring arm who were sedated and ventilated. This was to assess if sleep patterns for sedated patients are similar to those for patients who are not sedated.
Unless patients were being palliated, or language barriers were present, all patients admitted to the ICU for at least one overnight stay were eligible. If patients could not wear a monitor, for example for reasons of their condition, anatomy, or care requirements, they completed the questionnaire only. All patients with capacity gave written informed consent at the point of recruitment. For patients without capacity, favourable opinion was sought from an appropriate relative acting as ‘consultee’.
In addition to sleep measures, we also collected demographic data and information on delirium screening, pain scoring and medications for pain and sedation for the day before and after the monitored night.
Simple statistics describe the population. Correlation analysis and tests of agreement were performed to compare the sleep efficiency index as recorded by the EEG monitor with the RCSQ scores reported by the patient and the nurse. Further correlation analyses and tests of agreement were performed to compare activity-determined sleep patterns as recorded by the wrist-worn monitor against the EEG monitor data and the RCSQ scores. All p values are two sided; proportional data were compared using chi squared tests; continuous data were analysed by t-tests/ANOVA or a suitable non-parametric alternative where necessary.
Results
Patient demographics (cohort 1).
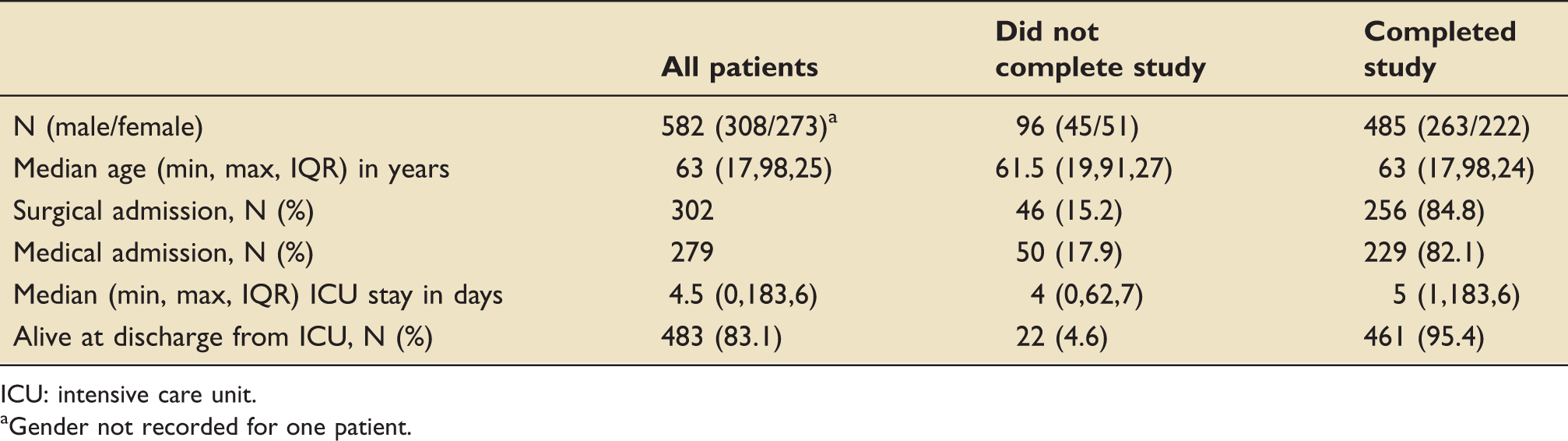
ICU: intensive care unit.
Gender not recorded for one patient.
Patient demographics (cohort 2).

ICU: intensive care unit.
There were no significant differences between demographics of patients recruited from different sites.
Continuous data were not normally distributed; therefore, median and IQR are quoted. Of the 45 patients recruited from OUH, 34 wore the EEG monitoring overnight. From almost 611 h of recording, approximately 16% was categorised by the software as ‘invalid’ and unsuitable for analysis. The median sleep per patient overnight was 2 h, 6 min (IQR; 3.5 h). Of this time, 7 min (5.6%), 13 min (10.2%) and 1.5 h (69.2%) were spent in rapid eye movement (REM) sleep, slow wave sleep (SWS) and non-REM (stages 1 and 2) sleep, respectively. Median (IQR) individual bouts of sleep were 1 min (45 s).
For the three patients who were sedated/ventilated while they wore the EEG monitor, their total recorded sleep times were 12:38:30, 00:28:30 and 09:46:30. Of this time, 04:53:30 (39%), 00:02:00 (7%) and 00:33:00 (6%) was spent in REM sleep, 01:53:30 (15%), 0 (0%), and 04:19:00 (44%) was spent in SWS, and 10:45:30 (85%), 00:26:00 (90%) and 04:12:30 (43%) in non-REM stages 1 and 2.
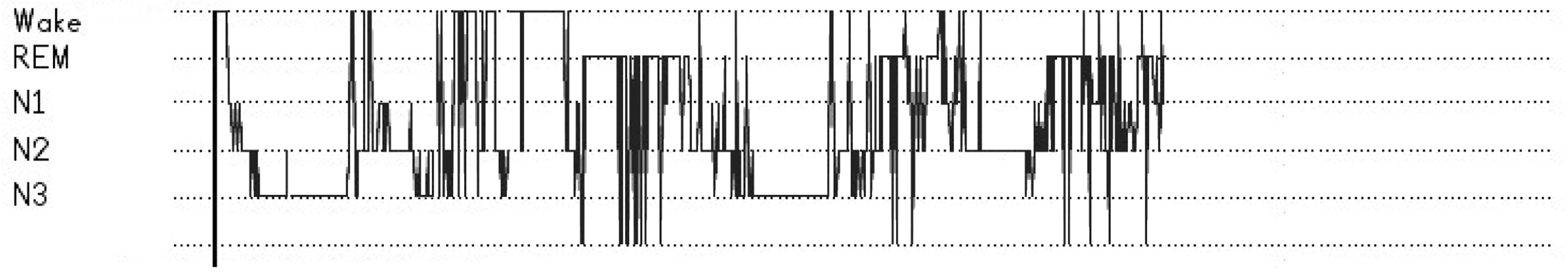
Figures 1 and 2 illustrate the difference between sleep experienced by a patient in the ICU and a healthy volunteer. The healthy volunteer spent much more time in both REM and slow-wave sleep and experienced fewer awakenings per hour.
A night’s sleep report from the EEG device for a patient in the ICU. A night’s sleep report from the EEG device for a healthy volunteer.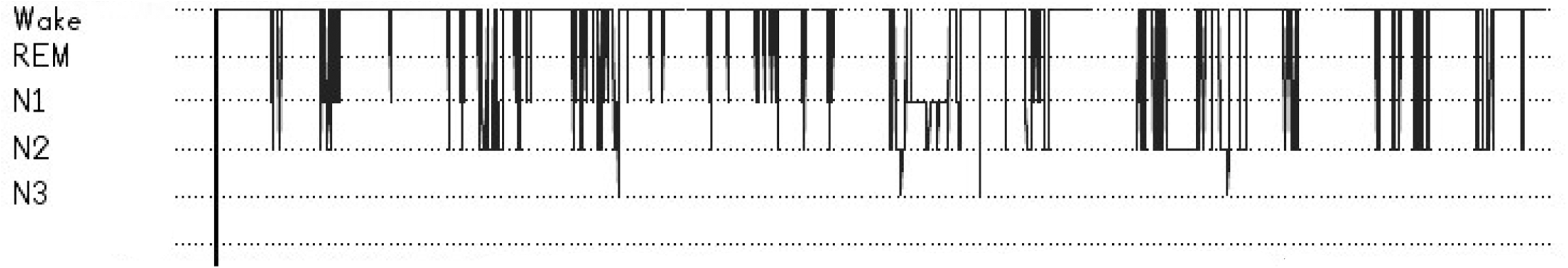
Patients and nurses who completed the RCSQ did not always complete every question. N values for analysis sets therefore vary. Correlation (Spearman’s Rho) analyses between the EEG monitoring and nurse- and patient-completed total scores from the RCSQ are ρ = 0.127 (n = 22) and ρ = 0.123 (n = 24), respectively.
Of the 34 patients who wore the EEG monitor overnight, 13 also wore the activity monitor. Non-parametric correlation for EEG vs. actigraphy measured duration of sleep = 0.402. Correlation of EEG measured duration of sleep and overall actigraphy score = −0.201.
The RCSQ is a continuous visual analogue scale that reports sleep assessment scores between 0 and 100. Both nurses and patients recorded maximum (100) and minimum (0) scores when assessing sleep. The mean (95%CI, SD) score reported by patients was 53.1 (50.5–55.7, 27.9). The mean (95%CI, SD) score reported by nurses was 61.0 (58.6–63.4, 25.7). The overall correlation between nurse and patient completed RCSQ was ρ = 0.537 (n = 444). See Figure 3 for scatter plot of results. Individual RCSQ question correlation results were lower than the combined mean (see Table 3).
Plot of nurse and patient RCSQ results. Correlation results for nurse- vs. patient-completed RCSQ.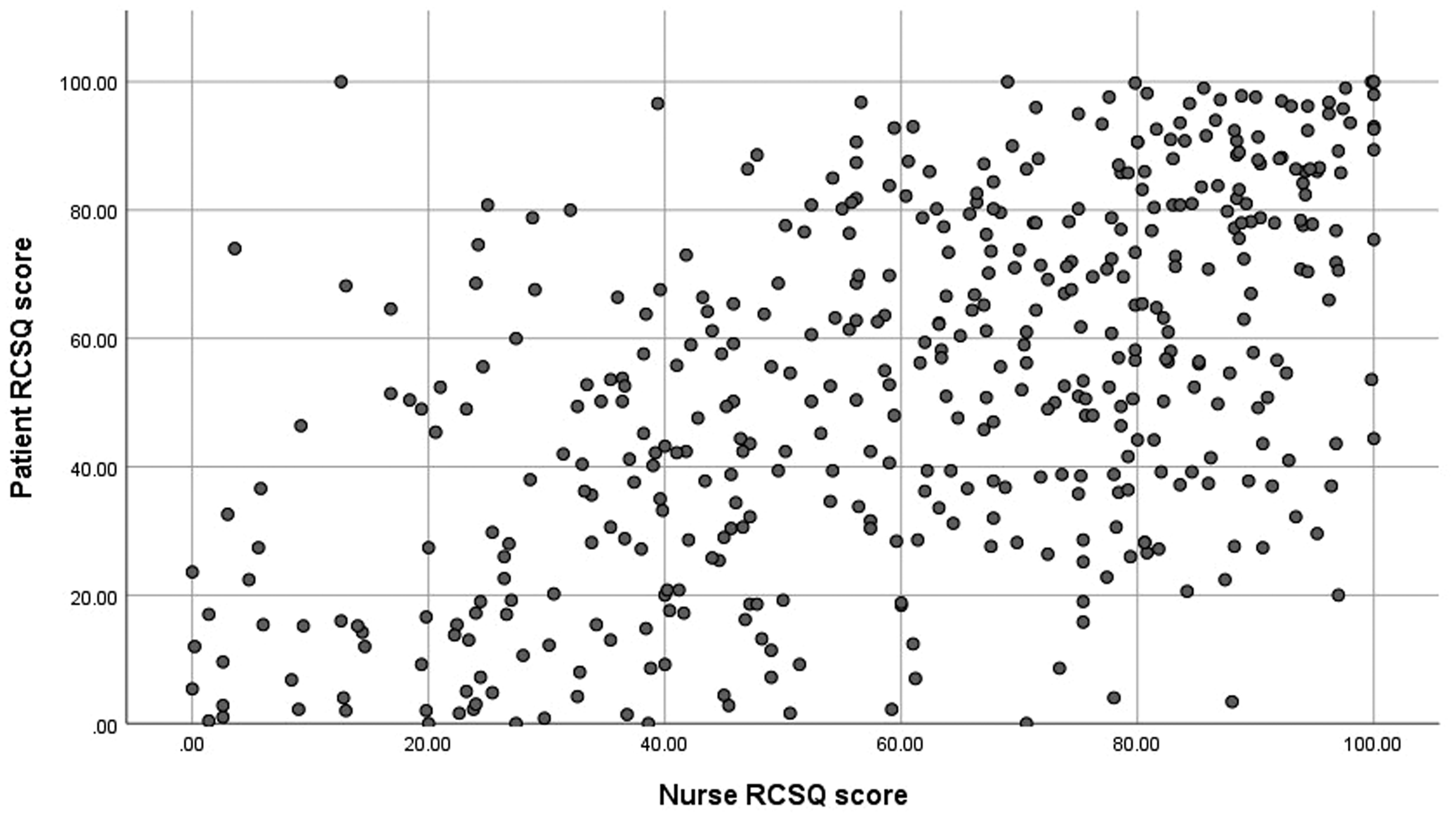

The highest pain scores in the previous 12 h were reported at baseline, and at the beginning and end of the monitored night. Completion rates were good at 95.3%, 87.7% and 88.9%, respectively. The Confusion Assessment Method for the Intensive Care Unit (CAM-ICU) was completed at baseline and the day following the monitored night. Completion rates were 97.7% and 91.6%, respectively. Two patients tested positive for delirium at baseline and two (different) patients tested positive the following day.
Linear regression analysis found no relationship between either analgesic or sedation medication given to patients, or their delirium status and their reported sleep either before or after the monitored night.
Although we used relatively simple EEG monitors with just three leads, it became apparent quite quickly that the system is still too complex, too cumbersome and too expensive to use on all patients routinely.
Discussion
It is clear from all the information available that patients admitted to the ICU experience very poor sleep. Results from this study add to the body of evidence, confirming that sleep is short and highly fragmented with inadequate rapid eye movement or deep sleep. This is consistent with other studies that have measured sleep in this population and which have been summarised elsewhere.6,8
The duration and quality of sleep indicated by the EEG results in this study are comparable to other studies that have used more complex PSG systems to measure patient sleep in the ICU.9,15,16 The monitoring equipment we used was able to recognise and interpret 84% of the data recorded from these patients. In theory, this indicates that the simplified EEG monitoring devices are suitable for use in the ICU; however, it is relatively expensive, requires expertise to use, is not 100% reliable in the environment and patient tolerance is an issue with almost a quarter of patients being unable to wear the monitor overnight. A small-scale study (n = 5) suggests that single channel EEG data could be a plausible means to estimate at least depth of sleep in the ICU. 17 This form of monitoring may be better tolerated by patients but has limitations in measurement capabilities.
We only recruited three patients to wear the monitor who were sedated/ventilated. Their sleep recordings were quite different from each other. With such a low number and no clear pattern to the data, we can make no generalisations about sleep duration or quality for these patients.
We conclude that EEG and actigraphy correlate poorly, and this is consistent with existing data. 18 Actigraphy can be a useful alternative to EEG monitoring for hospitalised patients outside of the ICU. 19 However, unusual activity patterns seen in ICU patients seem to render standard sleep detection algorithms inadequate, leading to a tendency to overestimate sleep in the ICU population. 16
Although the RCSQ is widely regarded as the best option for measuring patient sleep in the ICU,8,11 it is only suitable for patients who have the cognitive abilities to complete it. Nurses can use the same questionnaire to assess patient sleep. Prior to this study, three small-scale studies (N = 31, 20 33, 21 and 151, 22 respectively) had published correlation results between nurse- vs. patient-completed RCSQ. These data were conflicting. It was unclear from these studies whether nurse-completed questionnaires are a realistic interpretation of patient sleep. This much larger, multi-site study has conclusively demonstrated that the nurse- and patient-completed RCSQ are not interchangeable. In line with other third-party reporting of subjective measures (e.g. pain23,24), these study results indicate that it is likely that nurses will report better sleep (both in quality and quantity) than patients themselves. This should be taken into consideration when interpreting results from the RCSQ.
A number of studies have now demonstrated that neither full (20 lead) PSG nor actigraphy are reliable practical measures of sleep for the ICU patient population. We have also demonstrated that even simplified EEG monitoring is unlikely to be suitable for usual care.
Limitations
We acknowledge the high withdrawal rate from the electronic monitoring arm, and significant numbers of nurses and patients from this arm did not complete their RCSQ. This affected the analyses, as from 45 patients recruited to the study we were able to correlate paired data for just 24 patients. Also, only 13 patients wore the actigraph in addition to the EEG monitor; therefore, our denominator for these analyses is much lower than we had expected. We did not formally collect reasons for failure to complete the study, but we believe that the majority of the 16.5% of patients who did not complete their questionnaires were discharged to the ward between agreeing to take part in the study and being able to complete their sleep assessment. This was always a risk with this cohort of patients as because they need to be cognitively alert to self-assess their sleep, they represent some of the less sick patients in the ICU. We cannot guarantee that nurses did not collude with patients when completing their RSCQ but believe that if they had, the correlation values would be higher.
Conclusions
This study demonstrates clearly that nurse- and patient-completed RCSQ results are not interchangeable. With limited correlation between the EEG monitoring and any other verified measure of sleep, we are also hesitant to recommend any method of sleep monitoring for use in the ICU. However, to maintain consistency in reporting in a way that enables useful comparison across studies, we believe that the patient-completed RCSQ is the most reliable measure of sleep in the ICU. Where self-reporting is not possible, we propose that nurses complete the RCSQ on behalf of their patients but that care is taken when interpreting results.
Authors’ note
The datasets used and/or analysed during the current study are available from the corresponding author on reasonable request.
Footnotes
Acknowledgements
We would like to thank all the patients and their nurses who completed their sleep questionnaires during this study. We would also like to thank the NIHR Local Clinical Research Network for their support, in particular the research staff at each participating site who recruited patients and collected data for the multi-site phase of this study.
Authors' contributions
JLD – co-applicant on NIHR grant, devised the study and wrote the protocol, managed and co-ordinated the study, analysed the data, and wrote the first draft of the article; MB, PE, SV, LH – advised on study direction as members of the study advisory committee (LH also co-applicant on NIHR grant); JDY – overall supervision of project and NIHR grant holder. All authors have read and had the opportunity to comment on and revise the article. All authors approve the final version of the article.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical approval
This study received ethical approval from NREC South Central Hampshire A in June 2014 (ref: 14/SC/0164). Standard informed consent procedures applied.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This paper presents independent research funded by the NIHR under its Research for Patient Benefit (RfPB) Programme (Grant Reference Number PB-PG-0613-31034). The views expressed are those of the author(s) and not necessarily those of the NHS, the NIHR or the Department of Health.
