Abstract
Ventilated patients are at risk of acquiring ventilator-associated pneumonia. Various techniques are available for diagnosing ventilator-associated pneumonia including bronchoalveolar lavage, protected specimen brush and non-directed bronchoalveolar lavage. There is a paucity of evidence regarding the safety profile of these techniques, particularly non-directed bronchoalveolar lavage. This service evaluation aimed to establish whether non-directed bronchoalveolar lavage is a safe procedure. A prospective service evaluation of non-directed bronchoalveolar lavage on our adult intensive care unit was undertaken by a senior physiotherapist trained into carrying out the procedure, measuring pre- and post-procedure vital signs including heart rate (HR), tidal volume (VT), systolic blood pressure (SBP) and pulse oximetry (SpO2). Eighty-five episodes in 41 patients were included in the evaluation. There was a statistically significant difference between pre- and immediately post-procedure recordings for all vital signs measure. HR (min-1), means (SD) 87.1 (16.4), 91.5 (16.5), 87.5 (15.9), 87.7 (15.7) respectively pre, immediately, 5 min after and 30 min after procedure (P < 0.01). SBP mmHg, means (SD) 133.9 (26.1), 142.1 (25.6), 136.9 (25.3), 134.8 (23.4) pre, immediately, 5 min and 30 min after procedure (P < 0.01). VT mL, median (range) 0.523 (0.118–1.180), 0.512 (0.131–1.05), 0.519 (0.104–0.95), 0.534 (0.110–1.080) each pre, immediately, 5 min and 30 min post procedure (P < 0.05). SpO2 %, median (range) 98 (89–100), 100 (96–100), 98 (92–100), 97 (90–100) again each pre-, immediately post, 5 and 30 min post-procedure time-points (P < 0.0001). The statistically significant difference was not detected between pre-, 5 or 30 min post-procedure time-points. None of the changes observed were clinically significant and no untoward events happened to any of the subjects included. Non-directed bronchoalveolar lavage is a safe and inexpensive procedure that can be carried out easily in an intensive care setting by a trained physiotherapist, avoiding the need for invasive bronchoscopy.
Introduction
Ventilator-associated pneumonia (VAP) is defined as pulmonary infection that occurs in mechanically ventilated patients at least 48 h after endotracheal intubation. 1 The lung is the most common site of nosocomial infection in critically ill patients. 2 In England and Wales, approximately 12,000 patients develop VAP annually resulting in approximately 3600 deaths and an estimated associated cost to the National Health Service of £300 million.3,4 A wide range of pathogens, including difficult-to-treat organism such as Pseudomonas aeruginosa, may cause VAP. 5 Antimicrobial resistance is increasingly identified in critically ill patients with VAP and is associated with poor clinical outcomes.6,7 Excess antibiotic use in patients with VAP is associated with increased antimicrobial resistance and mortality.7,8 In contrast, a strategy of antibiotic de-escalation is associated with reduced mortality. 9 Therefore, early identification of the pathogen responsible for VAP facilitates stewardship of antibiotics, which should reduce the emergence of antimicrobial resistance and improve the overall outcomes.
Microbiological culture of lower airways secretions from critically ill patients remains central to the diagnosis of VAP. 1 Samples may be acquired by bronchoscopically directed lavage or brushings, or by non-directed (without a bronchoscope) bronchial lavage (NBL), also known as mini-bronchoalveolar lavage. 10 Formal bronchoscopy requires a high degree of operator proficiency, relatively complex equipment and is associated with complications. 11 Common complications include transient desaturation, which can occur in up to 10% of critically ill patients undergoing bronchoscopy and bronchoalveolar lavage (BAL). 11 Furthermore, a bronchoscope is not only costly to purchase and maintain, but also carries a risk of cross contamination, and the cost of reprocessing is complex and expensive.11.
Samples obtained via NBL are effective for the diagnosis of VAP, with a sensitivity of 50% and specificity of 95% compared with post-mortem histological confirmation of pneumonia. 12 Good concordance has been demonstrated when comparing NBL samples with culture samples from both protective specimen brushing and conventional bronchoalveolar lavage.13,14 However, there is a paucity of evidence supporting the used of NBL in critically ill patients in UK critical care units. Perkins et al. 15 reported a single centre experience of 14 patients, which showed minimal physiological disruption following NBL although patients did require increased sedation to facilitate NBL collection. A handful of similar small studies have demonstrated NBL to be safer than bronchoscopy and BAL in critically ill patients inducing less variation in both minute volume and heart rate 14 and less frequent and fewer significant transient desaturation.16,17 In these studies, collection of the NBL was performed by either clinician or respiratory therapists.
The aim of this project was to evaluate the changes in cardiovascular and respiratory physiological measures resulting from the collection of an NBL in critically ill patients. Multiple patient episodes from a single patient have been included as repeated intervention is required for a surveillance procedure. Finally, NBL collection was performed by an appropriately trained physiotherapist, three times a week, as part of our clinical standard, to facilitate early recognition of VAP.
Method
We conducted an observational service evaluation of physiotherapist-performed NBL in critically ill patients managed in our critical care unit between January 2016 and April 2016. As per our standard unit practice, surveillance NBL was performed three times per week in patients mechanically ventilated for longer than 48 h either via endo-tracheal tube or tracheostomy, by a senior physiotherapist. NBL was not performed in patients with any of the following: (a) fraction of inspired oxygen (FiO2) > 0.85; (b) positive end expiratory pressure (PEEP) > 10 cm H2O; (c) prone position, (d) thrombocytopenia with platelet count < 50 × 109 L−1; (e) desaturation during routine endotracheal suction; (f) agitation; and (g) bronchospasm.
The procedure for NBL has been previously described in detail but briefly comprises the following key steps:
18
Pre-oxygenated (FiO2 1.0 for 2 min), prior to carry out the technique. Disconnection from the ventilator circuit and rapid instillation of 20 mL 0.9% wt/vol saline into the lung using a suction catheter and 50 mL bladder syringe. Suction catheter introduced immediately after instillation via the airway device until resistance felt and then withdrawn 2 cm. Fluid was aspirated and the patient's ventilator circuit reconnected.
Ventilator settings (other than FiO2) and the level of sedation were not changed during the procedure, only a bolus of FiO2 was administered prior to carrying out NBL. No further bolus of pre-oxygenation administered. Levels of sedations were not altered in any of the patients for the procedure. The time of disconnection from ventilator and to the end of procedure lasted an average of 7 s.
Microbiological culture was performed on the aspirated fluid sample. Heart rate (HR), systolic blood pressure (SBP), tidal volume (VT), pulse oximetry (SpO2) and FiO2 were recorded immediately prior to collection of the NBL, immediately after the intervention and at 5 min and 30 min post intervention. Significant desaturation was defined as a reduction in oxygen saturation below 88% at the pre-defined time-points or a greater than 10% reduction in SpO2. Pre-defined time point was stipulated at 89%, as this was the lowest value of pulse oximetry of any subject included in the study. Recording of baseline pulse oximetry was taken prior to administering FiO2 for 2 min. The procedure always started after the oxygen bolus was administered. A change of 20% from baseline (immediately pre-procedure) was deemed clinically significant for haemodynamic variables.12,15
The D'Agostino and Pearson omnibus normality tests were used to assess Gaussian distribution. For Gaussian distributed variables the mean and standard deviation are presented. Repeated measures analysis of variance (RMANOVA) with Geisser–Greenhouse correction was used with the Dunnett multiple comparison test for post hoc comparisons. Non-parametrically distributed variables are presented with median, interquartiles and range and were analysed using Friedman repeated measures analysis with the Dunn paired test for post hoc comparisons. Significance was defined at P < 0.05 (two-sided) including corrections for multiple comparisons. Statistical analysis was performed using Prism 7.03 (GraphPad Software Inc., San Diego, CA, USA).
Results
Patient characteristics and underlying diagnosis. Data are summarised as number (%) or mean (range).
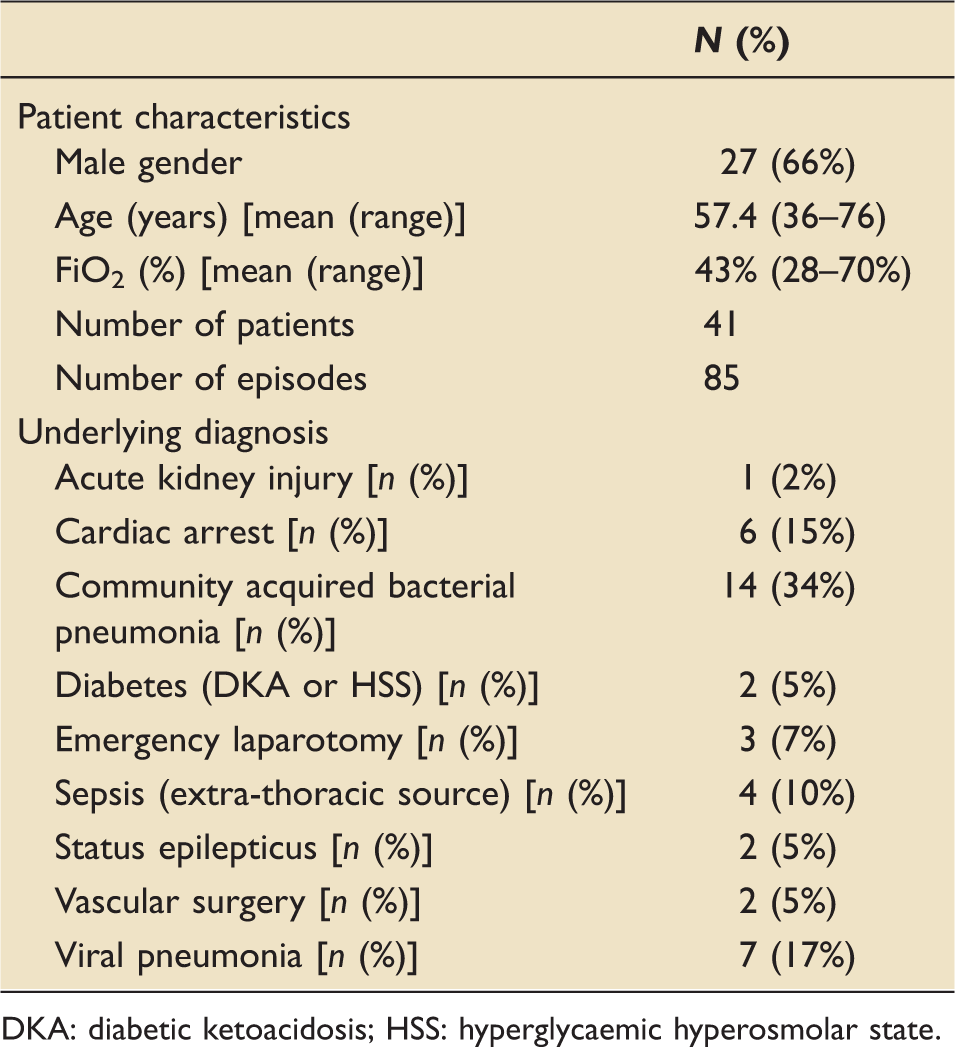
DKA: diabetic ketoacidosis; HSS: hyperglycaemic hyperosmolar state.
Effect on heart rate
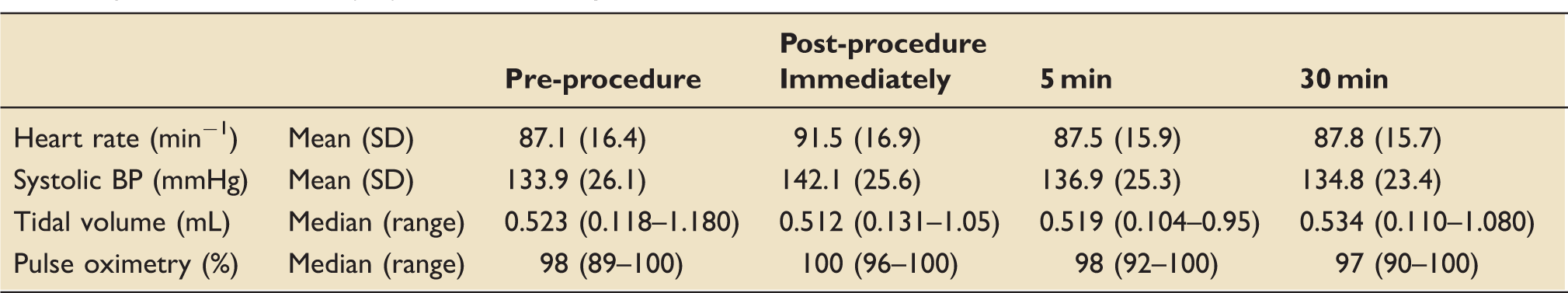
Data are presented in Table 2 and Figure 1. There was a significant difference between time-points (<0.01). Dunnett multiple comparison test showed a significant increase (+4.4 beats per minute) between the pre- and immediately post-procedure time-points (P < 0.01). There was no significant difference between the pre- and the 5 min and 30 min post-procedure time-points.
Tidal volume prior to and following non-directed bronchial lavage. [Median (range)]. Heart rate, systolic blood pressure, tidal volume and pulse oximetry prior to and following non-directed bronchial lavage. Data are presented as mean (SD) or median [range].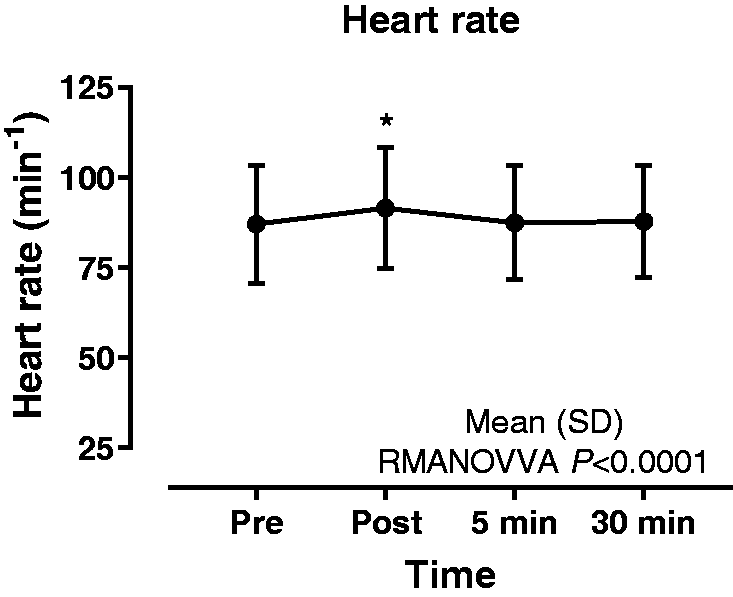
Effect on systolic blood pressure
Data are presented in Table 2 and Figure 2. There was a significant increase (+8.2 mm Hg) between time-points (P < 0.01). Dunnett multiple comparison test showed a significant difference between the pre- and immediately post-procedure time-points (P < 0.01). There was no significant difference between the pre- and the 5 min and 30 min post-procedure time-points.
Systolic blood pressure prior to and following non-directed bronchial lavage. [Mean (SD)].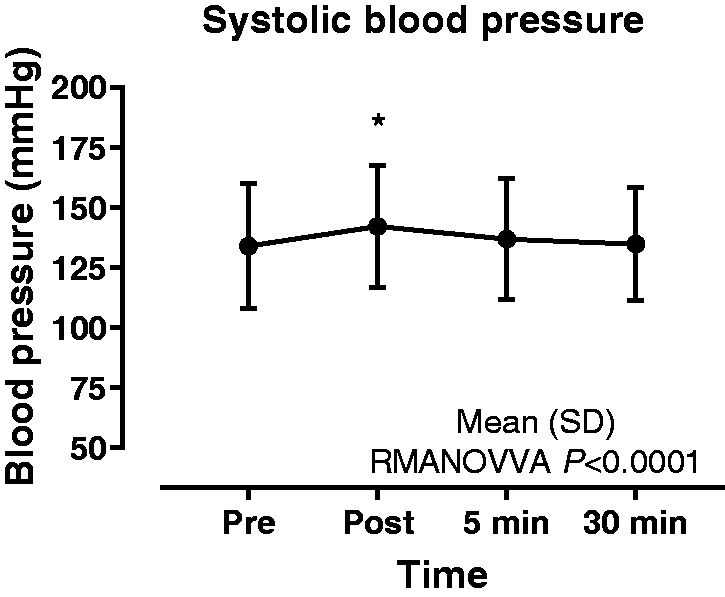
Effect on tidal volume
Data are presented in Table 2 and Figure 3. There was a significant difference between time-points (P < 0.01) with Dunn multiple comparison test showing a significant increase (+0.22 L) between the immediately post- and 30 min post-procedure time-points (P < 0.01). There was no significant difference between the pre- and the immediately post-, 5 min or 30 min post-procedure time-points.
Heart rate prior to and following non-directed bronchial lavage. [Mean (SD)].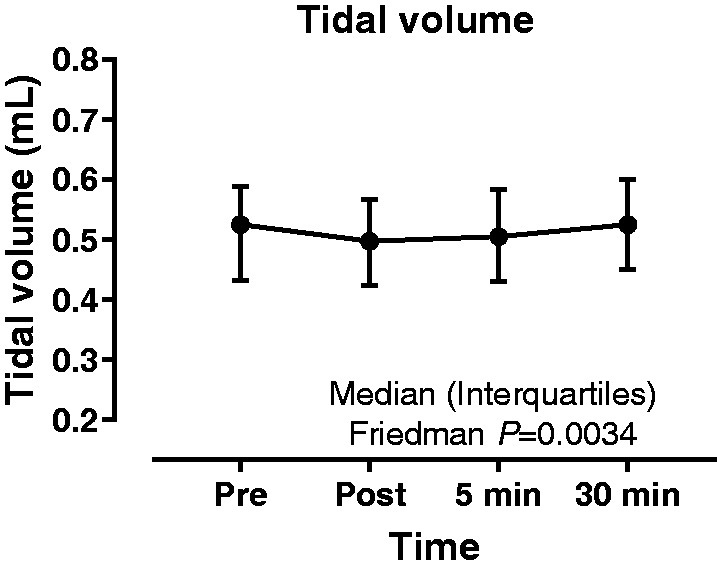
Effect on pulse oximetry
Data are presented in Table 2 and Figure 4. Clinically significant desaturation was not observed in any patient. Friedman test demonstrated a statistically significant difference between time-points, P < 0.01, with Dunn post hoc test showing a significant increase (+4%) between the pre- and the immediately post-procedure time-points (P < 0.01). There was no significant difference between the pre- and 30 min post-procedure time-points. No adverse events occurred during any of the procedures.
Pulse oximetry prior to and following non-directed bronchial lavage. [Median (range)].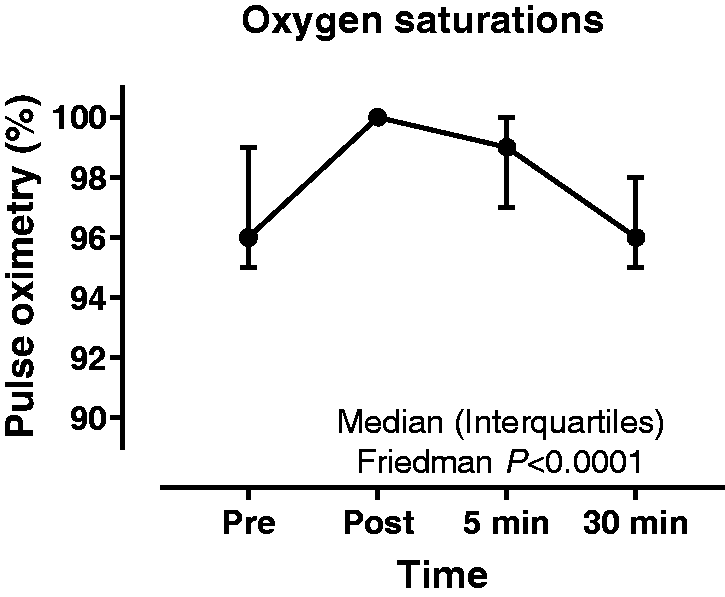
Discussion
Our study demonstrates that NBL is a safe method for sampling the lower respiratory tract in critically ill patients and can be undertaken by appropriately trained physiotherapists. No episodes of significant change in HR, SBP, VT and SpO2 were observed at 30 min post-procedure when compared with pre-NBL observations. A minimal increase in both HR and SBP was observed immediately post-procedure with HR and SBP returning to pre-NBL rates within 5 min. Our finding that NBL does not cause significant desaturation is in keeping with previously published studies, which describe desaturation occurring in less than 2% of critically ill patients. 17 A transient, but statistically significant, reduction in VT was observed, with recovery to tidal volume to baseline levels within 5 min of NBL collection. Median VT was reduced by 33 mL or 6% of baseline tidal volume which is not clinically significant and within the generally accepted tolerance of 20% for safety of a procedure. 15 We hypothesise that reduction in VT occurs due to the loss of positive end-expiratory pressure related to accessing the airway. Further support for the safety of NBL is evidenced by the cardiovascular stability seen following the procedure.
Our evaluation has some potential limitations. Subjects with high ventilator requirements, such as a high fraction of inspired oxygen or high levels positive end-expiratory pressure, were excluded from the evaluation. Additionally, subjects placed in a prone position were also excluded due to technical difficulties in carrying out the procedure. Potential poor tolerance of the NBL procedure would be expected in these patients but it is likely that BAL would not be collected in the patients for the same reasons. However, our results suggest that NBL is well tolerated and indeed there may be a role for NBL in patients receiving a higher fraction of inspired oxygen or high levels positive end-expiratory pressure. Further studies are required in this area, with prolonged follow up times, providing safety data differences between NBL and BAL.
Recent guidance from the Infectious Diseases Society of America and the American Thoracic Society suggest that non-invasive sampling is the recommended methodology to diagnose VAP based on the review of the current literature. 1 Due to the paucity of evidence for NBL only endotracheal aspiration is included in these guidelines. Endotracheal aspiration had the lowest sensitivity of all the techniques used to sample lower respiratory tract secretions. NBL should offer the improved diagnostic performance of BAL with improved safety.13,14 Further studies are required to establish the role of NBL as both a surveillance tool and in the diagnosis of VAP. It is also worth pointing out that the material used to carry out NBL costs pennies and is single use only, in comparison with the elevated cost and specialised maintenance incurred by using a bronchoscope. 19
This study has demonstrated that NBL is a safe and well-tolerated method for sampling the lower respiratory tract of critically ill patients. Non-directed lavage can be performed safely by appropriately trained physiotherapist, avoiding risks of cross contamination, which can occur using a bronchoscope. 20 Further studies are required to demonstrate the improved diagnostic performance of NBL when compared with BAL and to establish the safety of NBL in patients requiring very high levels of ventilator support.
Footnotes
Acknowledgement
This project was supported by researchers at the NIHR Manchester Biomedical Research Centre.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
