Abstract
A 79-year-old critically ill woman presented with remarkable prolongation of activated partial thromboplastin time and thrombin time combined with high levels of anti-factor IIa activity 26 days after coronary artery bypass grafting. Coagulation disorder was associated with severe bleeding. Cause of coagulopathy was accidental administration of argatroban in an unknown dosage. Clearance of argatroban was significantly prolonged because of a liver function disorder related to septic multiorgan failure. Argatroban reversal was performed with prothrombin complex concentrate.
Introduction
In this study, we present the case of a critically ill patient with acute coagulation disorder due to accidental administration of argatroban. Written informed consent was obtained from a patient’s legal surrogate. Identification of the underlying cause of a coagulation abnormality is still challenging in a critically ill patient because of prolonged and unknown half-life of drugs in patients with severe multiorgan failure. Furthermore, management of argatroban-induced bleeding is complicated because of the lack of evidence for safe and effective anticoagulant reversal.
Case report
A 79-year-old Caucasian woman was admitted to the intensive care unit (ICU) because of acute renal failure following coronary artery bypass grafting two days before.
Initial examination revealed a cooperative and fully orientated woman with respiratory failure, spontaneous breathing with a continuous positive airway pressure mask and hemodynamic stability despite atrial fibrillation. She presented with acute renal failure and anuria. The calculated SAPS II score was 56 and the APACHE III score 71 at admission to ICU.
Medical history was remarkable for obesity (BMI 30.6), severe arterial hypertension, insulin-dependent diabetes mellitus type II, chronic renal failure, cholecystolithiasis, hypercholesterolemia and restrictive ventilation disorder.
Her premedication included antihypertensive medication (amlodipine, urapidil, rilemenidine, nebivolol, ramipril), psychotropic drugs (citalopram, amitryptiline), antidiabetics (gliquidone, sitagliptin, insulin), hydrochlorothiazide, nicorandil, atorvastatin, pantoprazole and acetylsalicylic acid.
The patient was living alone with support from friends and a social worker. She complained about angina pectoris during exertion for one year. Three-vessel coronary heart disease was diagnosed and she underwent an aortocoronary bypass operation with left internal mammary artery and three venous bypass grafts. The operation was uneventful. Immediately postoperative she was transferred to a postoperative cardiac care unit. Because of acute renal failure requiring haemodialysis she was transferred to an ICU Class III on the second postoperative day. Immediately after admission to ICU Class III haemodialysis was started.
Time course of PT, aPTT, TT, anti-factor IIa – activity, argatroban concentration and substitution of coagulation factors.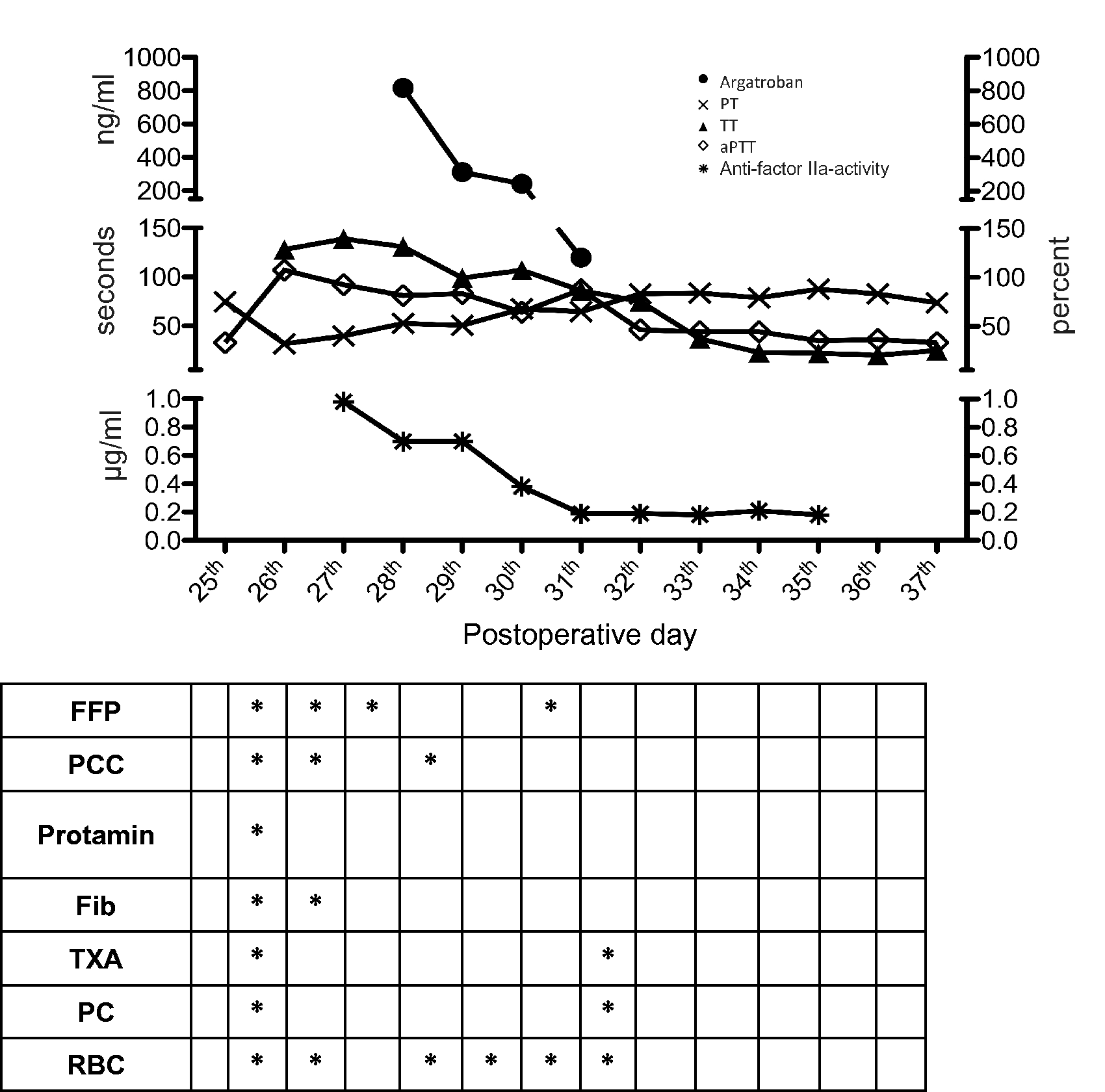
Reintubation was necessary because of severe acute respiratory distress syndrome, sepsis from a sternal wound infection and development of severe rhabdomyolysis. She had remarkable hyperbilirubinemia, presumably as a consequence of postoperative congestive hepatopathy (peak direct bilirubin: 18.56 mg/dl). Because of signs of cholecystitis in sonography a percutaneous transhepatic gallbladder drain was inserted. On the 26th postoperative day, routine morning laboratory tests revealed an activated partial thromboplastin time (aPTT) of 63 s (26–37). Prothrombin time (PT) was 46% (70–130); platelets 92 G/l (150–380) and fibrinogen 231 mg/dl (210–400) (Figure 1). Anticoagulation therapy with low molecular weight heparin (LMWH) had not been prescribed for a few days because of the intervention and a low number of platelets. She had no prior history of bleeding diathesis. All solutions for the continuous infusion pumps were prepared anew to exclude medication error. Blood sampling was repeated, but aPTT still increased (peak aPTT: 107 s). Meanwhile, bleeding via the endotracheal tube, in the field of the sternal wound dehiscence and at the injection sites as well as macrohaematuria were observed. Three units (U) of red blood cells (RBC) were administered. Diagnostic assessment of the bleeding patient was processed as follows (Figure 2).
Diagnostic algorithm in case of prolonged activated partial thromboplastin time.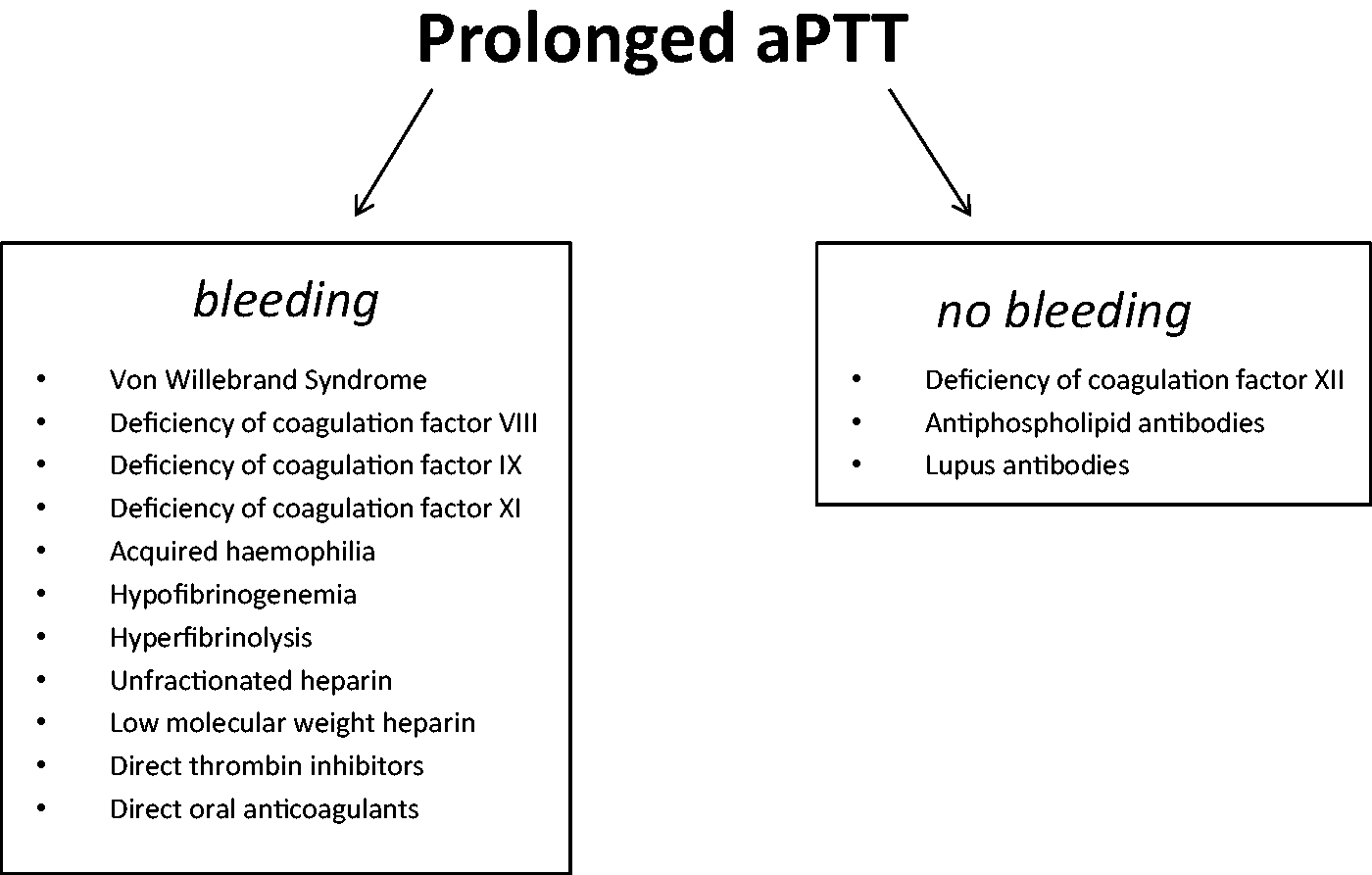
Coagulation factor testing was performed. Factor XI was significantly decreased: 7% (70–120). In the mixing test no inhibitor was found against factor XI.
Direct thrombin inhibitors interfere with coagulation assays. Halbmayer et al. report that direct thrombin inhibitors induce falsely decreased results for factor VIII, IX, XI and XII activities. 1
Acquired haemophilia due to fibrin glue containing bovine thrombin 2 was unlikely because fibrin glue with human thrombin (Tisseel ©) was used during the operation and no inhibitors were found in the mixing test.
Congenital haemophilia A or B seemed unlikely because she had no prior history of bleeding diathesis. Coagulation factors VIII and IX were within the normal range or slightly decreased: Factor VIII 106% (70–150), Factor IX 43% (70–120).
There was no evidence of acquired von Willebrand Syndrome: vWF activity 630% (48–173); vWF antigen 616% (58–174); vWF ratio (RC:AG) 1.0 (0.7–1.5); vWF: collagen-binding activity 616% (40–250).
Fibrinogen was 231 mg/dl (210–400) measured by Clauss method and 380 mg/dl (200–400) measured by an immunological assay. Hyperfibrinolysis was excluded by rotational thromboelastometry (ROTEM) analysis: normal level of lysis index after 30 and 60 min.
The patient had no medication with platelet inhibitors and platelet function test (Multiplate®) showed normal findings. Anticoagulation therapy with LMWH had not been administered for eight days. Anti-factor Xa activity was slightly increased at 0.09I U/ml. Clotting time (CT) in HEPTEM was not prolonged. INTEM – CT and EXTEM – CT were much more prolonged in comparison to aPTT and PT, meaning that INTEM – CT did not correlate with aPTT and PT with EXTEM – CT. Anti-factor IIa activity was determined and remarkable elevated. Prolonged aPTT and TT together with elevated anti-factor IIa activity made iatrogenic administration of direct thrombin inhibitors the most likely cause of the patient’s coagulopathy.
Massive bleeding was treated with 8 g fibrinogen concentrate, 9.600 IU prothrombin complex concentrate (PCC), 20 IU fresh frozen plasma (FFP), 2 IU platelets, 9 IU RBC and 2 g tranexamic acid from postoperative day 26 to 32. Bleeding decreased on postoperative day 29. Anti-factor IIa activity was < 0.18 ug/ml for the first time on postoperative day 30. Subsequently, aPTT and TT reached approximately normal values and bleeding stopped. The obscure coagulation disorder resolved.
On postoperative day 53, 27 days after onset of the coagulation disorder, the patient died because of intracerebral bleeding. Blood coagulation parameters showed no abnormalities on the day of death and the autopsy revealed no evidence of aneurysm.
Post mortem blood samples from the patient were analysed in a specialized laboratory abroad. The results confirmed argatroban (Argatra®, Mitsubishi, Germany) in the blood samples taken on the days with prevalent coagulation disorder (postoperative days 27, 29 and 30), proving that coagulopathy was caused by accidental administration of argatroban. Argatroban was not detected in any other blood samples, including those taken on the day of death.
Discussion
Argatroban is a direct synthetic thrombin inhibitor. After discontinuation of argatroban anticoagulation parameters drop from therapeutic levels to baseline within two to four hours according to the package insert. 3 In our case report aPTT remained >60 s for five days.
Paediatric patients with elevated bilirubin (secondary to cardiac complications or hepatic impairment) have 80% lower argatroban clearance than do paediatric patients with normal bilirubin levels. 3
Argatroban is excreted primarily in the feces, presumably through biliary secretion. 3
Our patient had total bilirubin 8.61 mg/dl at the onset of the coagulation disorder, which increased to 16.05 mg/dl over the next five days.
Saugel et al. described that the mean recommended argatroban dose in ICU patients with multiple organ dysfunction syndrome (MODS) significantly correlated with serum bilirubin (r = −0.739) and postulated that hepatic metabolism in ICU patients with sepsis and MODS may be influenced by reduced cardiac output, circulatory distributory failure, and/or disseminated intravascular coagulation, resulting in decreased hepatic perfusion. 4
Keyl et al. determined plasma concentrations of argatroban in critically ill cardiac surgery patients and found that argatroban plasma half-life was markedly prolonged in these patients and related to total bilirubin concentration. 5
Argatroban is eliminated predominantly by the liver and may be used without relevant dose reduction in patients with renal impairment. 3 Only limited data are available concerning elimination of argatroban by hemodialysis suggesting that continuous veno-venous hemodialysis has little role in increasing argatroban elimination and does not significantly affect argatroban plasma – half-life.5–7
Post-mortem the cause of coagulopathy was revealed: argatroban was iatrogenically administered to the patient. Argatroban is a clear, colourless to pale yellow solution that has to be diluted to final concentration. If the constituted solution is not clearly labelled, it is not possible to differentiate it from sodium chloride. How the medication error happened in the current case remains unclear.
According to Wheeler et al., the most common causes of an isolated aPTT prolongation are unfractionated heparin, hirudin, argatroban or human recombinant activated protein C. They report that heparin as a cause for prolonged aPTT can be ruled out with normal TT. In our patient TT was 128 s (15–21). Furthermore, Wheeler et al. suggest adding heparinase in vitro in case of suspected contamination with heparin. 8 In our case report HEPTEM – CT was within the normal range. A direct thrombin inhibitor (Argatroban) was suspected causing coagulopathy because of high levels of anti-factor IIa activity combined with prolonged aPTT and TT. Direct verification of argatroban in the patient’s blood was not possible at our center and was performed later by a specialized laboratory abroad.
Up to now, no specific antidote for argatroban reversal is available. 9 The package insert recommends discontinuing argatroban immediately and analysing aPTT and other coagulation parameters. 3 Yee et al. report the case of a critically ill man who received an overdose of argatroban without bleeding complications and improving coagulation parameters after 9 IU FFP. 10 In our case, the dose of administered argatroban remains unknown and the patient was treated with 8 g fibrinogen concentrate to maintain normal fibrinogen levels, 20 IU FFP, 2 IU platelets, 9 IU RBC and 2 g tranexamic acid as an unspecific and general treatment. For reversal of argatroban we administered a high-dose of 9.600 IU PCC (114 IU/kg body weight) to stop bleeding.
We here report on accidental administration of an unknown dosage of argatroban with massive bleeding and massive transfusion in a critically ill patient suffering from multiorgan failure. Because of the prolonged clearance of argatroban in this patient, it was even more challenging to determine the cause of coagulopathy. We demonstrate how to proceed in the case of unclear coagulopathy with prolonged aPTT and present a successful clinical approach for bleeding diagnosis in a critically ill patient. Moreover, to our knowledge this is the first reported successful reversal of argatroban with PCC.
Footnotes
Consent
Patient consent was given for the publication of this report.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Dietmar Fries has received study funding, honoraria for consultancy and board activity from Astra Zeneca, AOP orphan, Baxter, Bayer, BBraun, Biotest, CSL Behring, Delta Select, Dade Behring, Edwards, Fresenius, Glaxo, Haemoscope, Hemogem, Lilly, LFB, Mitsubishi Pharma, NovoNordisk, Octapharm, Pfizer, Tem-Innovation. Agnes Oberhuber, Benedikt Treml, Ingo H. Lorenz, Friesenecker Barbara and Griesmacher Andrea have no involvements that might raise the question of bias in the work reported or in the conclusions, implications, or opinions stated.
