Nebulized heparin (10,000 units) in conjunction with albuterol and a mucolytic was associated with a significant decrease in the initial duration of mechanical ventilation, and an increase in ventilator-free days during the first 28 days post-inhalational injury (IHI) in adult patients. Level of evidence: 4 (individual case–control study with limitations)
Appraised by: Katherine Laver, Bethan Barker and Tomasz Torlinski
Citation: McIntire A, Harris S, J Whitten, et al. Outcomes following the use of nebulized heparin for inhalation injury (HIHI Study). J Burn Care Res 2017; 38: 45–52.
Lead author: Allyson McIntyre; allyson.mcintire@eskenazihealth.edu
Three-part clinical question:
Patients: Adults admitted within 48 h of injury, requiring mechanical ventilation, and with an inhalational burn injury confirmed by bronchoscopy.
Intervention: Nebulised heparin 10,000 units given 4-hourly for seven days (or until extubation if sooner), alternating with nebulised 0.5% albuterol (a beta-agonist) and either 20% N-acetylcysteine (NAC) or 4% sodium bicarbonate 4-hourly.
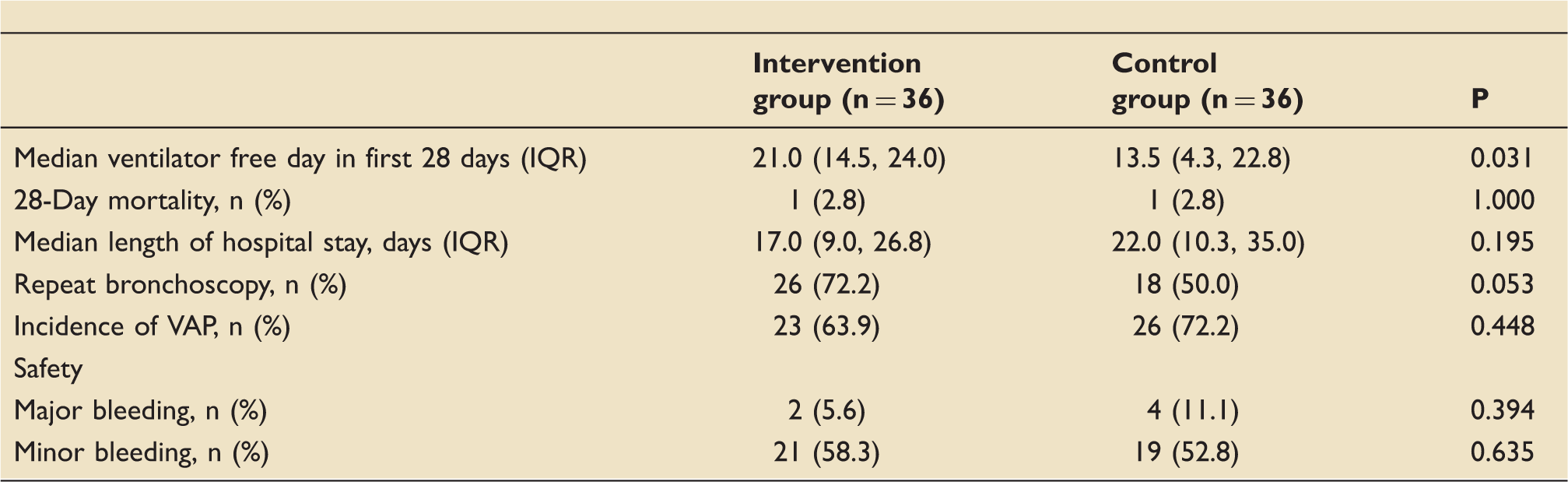
Outcome: The primary outcome was to determine if nebulised heparin decreased the duration of initial mechanical ventilation. Secondary outcomes were the difference in lung injury scores and ventilator-free days in the first 28 days, 28-day mortality, length of hospitalisation, incidence of bronchoscopy, incidence of ventilator associated pneumonia (VAP), and rate of bleeding events.
The study:
A retrospective single-centre case–control study, consisting of an intervention group of 36 patients who received the protocolised treatment described above, matched case by case by total percentage surface area burn and age, to 36 patients treated before heparin protocol implementation.
The study patients:
Eligible: Adult patients admitted within 48 h of injury, between 1 June 2014 and February 29 2016, with IHI confirmed by bronchoscopy and requiring mechanical ventilation.
Included: 36 cases (age >18 years).
Subgroups: Patients who did not die and were not discharged while still on the ventilator were analysed as a sub-group, but this was not a pre-specified sub-group analysis. An analysis of data for the sub-groups that received either nebulised NAC or sodium bicarbonate as a mucolytic is not presented.
Exclusion criteria: Patients mechanically ventilated <24 h, deaths <72 h following admission, terminal weaning for reasons other than burn/IHI, heparin allergy or previous heparin-induced thrombocytopenia, pulmonary haemorrhage within the last three months, known bleeding disorder, pregnancy or breastfeeding.
Control group: Historical patients (n = 36) with IHI presenting between 1 January 2000 and 29 February 2016, prior to heparin protocol implementation. Patients were matched on a case-by-case basis on size of burn (percentage of total body surface area, %TBSA) and age. Matched patients were within approximately 5% TBSA and 5% age of one another.
The evidence:
Secondary outcomes:
IQR: interquartile range; VAP: ventilator associated pneumonia.
EBM questions:
Do the methods allow accurate testing of the hypothesis? Unclear, as no defined hypothesis presented. The objective was “to determine if nebulized heparin 10,000 units improves lung function and decreases mechanical ventilation duration, mortality, and hospitalization length in IHI with minimal systemic adverse events.” The study was powered for the detection of difference in initial duration of mechanical ventilation based on an a priori calculation from a previous study, but was not powered for secondary analyses. The study was un-blinded and not randomised. The cases and controls were matched on the basis of %TBSA and age, although not on anatomical burn location or co-morbidities. From the information provided, it would appear to be a valid study although with significant limitations.
Do the statistical tests correctly test the results to allow differentiation of statistically
s
ignificant results? Yes, we believe so.
Are the conclusions valid in light of the results? Unclear. The primary outcome was only statistically significant when data were re-analysed for a second time excluding patients who died or were discharged on the ventilator. There was a statistically significant increase in ventilator-free days during the first 28 days, but there were no differences in any of the other secondary outcomes, although the authors admit that the study was not powered to detect these. Potential differences in patient treatment (other than heparin treatment) due to the length of recruitment are not accounted for in the analyses.
Did results get omitted? Yes. During the study period 100 patients were screened for eligibility in the heparin group but only 36 were included. Reasons for exclusion included not receiving heparin or receiving an inappropriate dose, not having an inhalational injury, or meeting previously declared exclusion criteria. Thirty-six historical controls were identified, although there is no mention of any prior exclusion to this group of patients. Two patients from the heparin group and three patients from the control group died or were discharged on a ventilator; data were analysed twice to adjust for this. Only after the exclusions did patients receiving nebulized heparin demonstrate a statistically significant decrease in median duration of initial mechanical ventilation compared with the control group.
Did they suggest further areas of research? Yes. The authors suggest that a large prospective, multi-centre, placebo-controlled trial using an IHI severity tool is needed to look into the safety and effects of nebulized heparin 10,000 units. A full economic analysis looking at cost savings for reduced duration of mechanical ventilation versus cost of nebulised heparin was also recommended.
Did they make any recommendations based on the results and were they appropriate? Yes, however, the authors suggest that their results should be interpreted with caution because it was a single-centre retrospective study. They recommended that while further studies are conducted, other centres implementing an inhalational injury protocol should administer all three nebulised agents until differences in outcome between heparin, and heparin with a beta-agonist and mucolytic, can be demonstrated. This is, however, a questionable suggestion as the benefit of nebulised beta-agonists in IHI in the absence of bronchospasm is unclear, and recent trials of intravenous beta-agonists in acute lung injury have found that they are unlikely to be beneficial and may worsen outcomes.
1
Is the study relevant to my clinical practice? Yes. As a tertiary burns centre with burns intensive care beds, we are often called to review, admit or treat patients with IHI. We already have a burns and IHI protocol
2
in place which includes nebulised heparin 5000 units and nebulised N-acetylcysteine or carbocisteine liquid. A beta-agonist is only administered to patients with bronchospasm. It should also be noted that in this study patients with an isolated IHI (with no cutaneous burns) admitted to the pulmonary intensive care unit were not included in the analyses as they did not receive nebulized heparin. This was due to provider preference, lack of perceived benefit, and overall change in culture with adopting a new protocol. This means results cannot be directly extrapolated to this patient population.
What level of evidence does this study represent? 4. This is an individual case–control study with significant limitations.
What grade of recommendation can I make on this study alone? C.
What grade of recommendation can I make when this study is considered with other available evidence? C. The results of this study do not correlate with other studies demonstrating improvements in lung function outcomes or a reduction in mortality with the use of nebulised heparin, but it did demonstrate a reduction in time spent on a ventilator; albeit in a single centre. A well-designed and suitably powered randomized control study would be required to improve on this grade of recommendation.
Should I change my practice because of these results? No. Due to the limitations of this study, we would not recommend changes based on this publication alone. However, Elsharnouby et al.
3
demonstrated that administration of 10,000 units heparin was associated with lower median lung injury scores and lower duration of mechanical ventilation, with no significant difference in coagulation parameters, length of ICU stay or mortality. Holt et al.
4
failed to demonstrate significant improvements in primary outcomes in IHI patients who received 5000 units of nebulised heparin. Based on these three studies, we will consider changing our regional protocol heparin dose from 5000 to 10,000 units.
Should I audit my current practice? Yes. We plan to audit our adherence to and outcomes from our current protocol for IHI. If needed, we will re-consider changing to a higher dose of nebulised heparin in the next edition of our regional guidelines.