Abstract
Acetic acid is an organic acid available in concentrations from 2 to 80%. Whilst lower concentrations of 2–6% are more commonly used as the table top condiment, vinegar, much stronger solutions are regularly used in Eastern Europe as food preservatives and cleaning solutions. Oral ingestion of greater than 12% has been reported to cause haemolysis, renal failure, shock and death. Most reported cases of deliberate or accidental poisoning are from Russia and Eastern Europe in the 1980s, with very little currently in western publications. We present the case of a female patient who attempted suicide by drinking 250 ml of 70% acetic acid. Her widespread gastrointestinal injuries were managed conservatively, and despite suffering extensive upper airway and renal complications, she was successfully decannulated and discharged home after a prolonged intensive care and hospital stay.
Case
A 59-year-old female was admitted to the intensive care unit (ICU) after deliberately ingesting 250 ml of 70% acetic acid. She had presented to the accident and emergency department stridulous, tachycardic (HR: 115 BP: 108/58), with a Glasgow Coma Scale (GCS) of 15 and was acidaemic (pH 7.2, base excess −10, lactate: 4.9 mmol/l). Direct nasendoscopy was performed which revealed swollen laryngeal structures, with haemorrhagic and sloughy looking arytenoid cartilages. She was brought to theatre for intubation where a Grade IIb view was obtained.
She was initially resuscitated using the advance life support (ALS) algorithm with intravenous fluids and small qualities of vasopressor to target a mean arterial pressure (MAP) >70 mmHg to aid renal and gastric perfusion.
Interpretation of the patient’s initial biochemistry was challenging due to severe haemolytic interference with baseline blood samples. Urinary catheterisation revealed frank haematuria. She subsequently became anuric, developed an acute kidney injury (AKI) (creatinine: 440 mmol/l (baseline creatinine: 50 mmol/l) urea 36 mmol/l (baseline 3 mmol/l)) was commenced on inotropic support and, after consultation with the National Poisons Information Service, continuous venovenous haemofiltration (CVVH) was established. Despite a brief, but significant rise in her liver function tests (alanine aminotransferase (ALT) peaked at 1799 IU/L by day 3) by day 10, these had returned to a normal baseline with no apparent compromise in her coagulation studies.
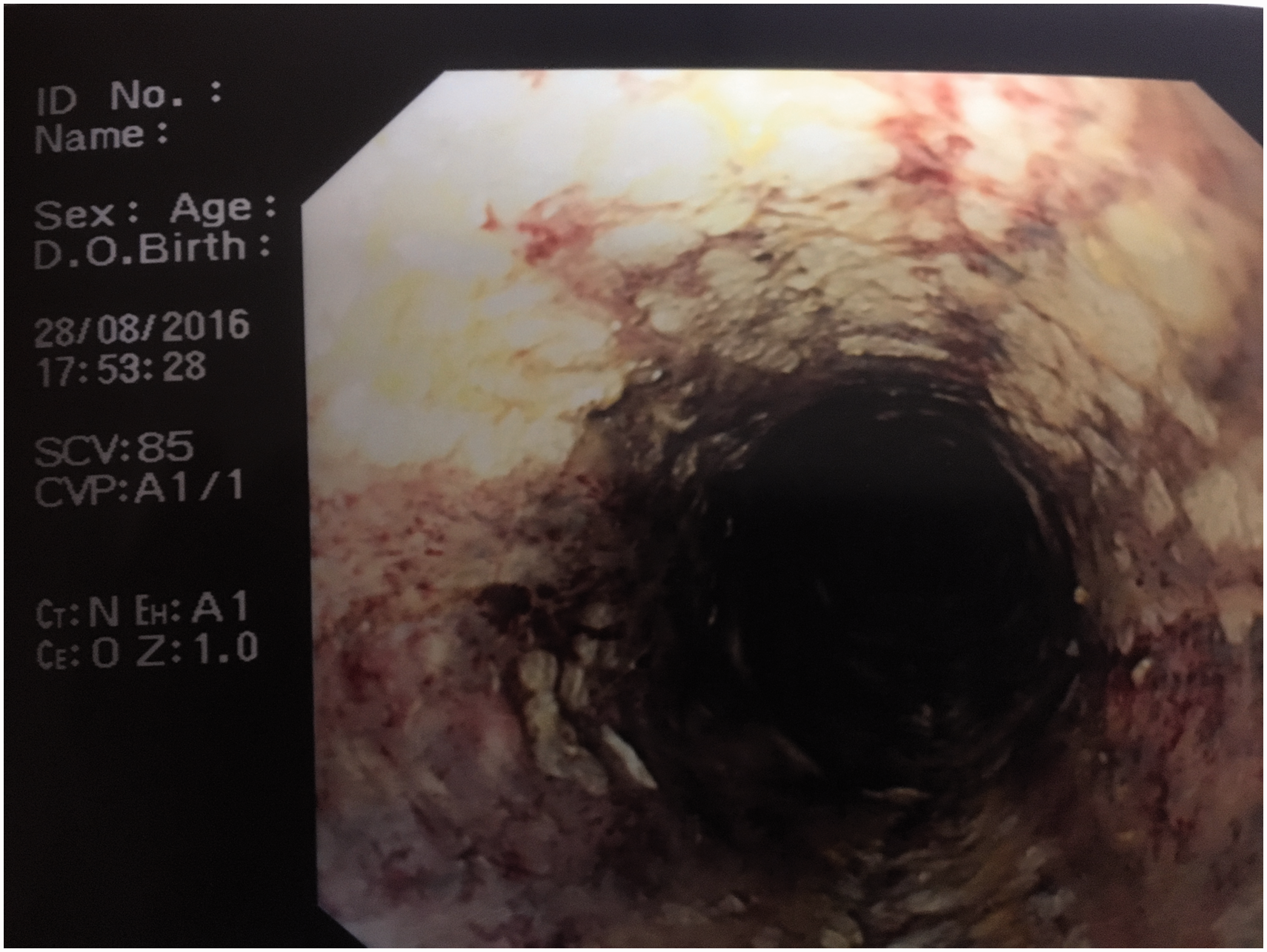
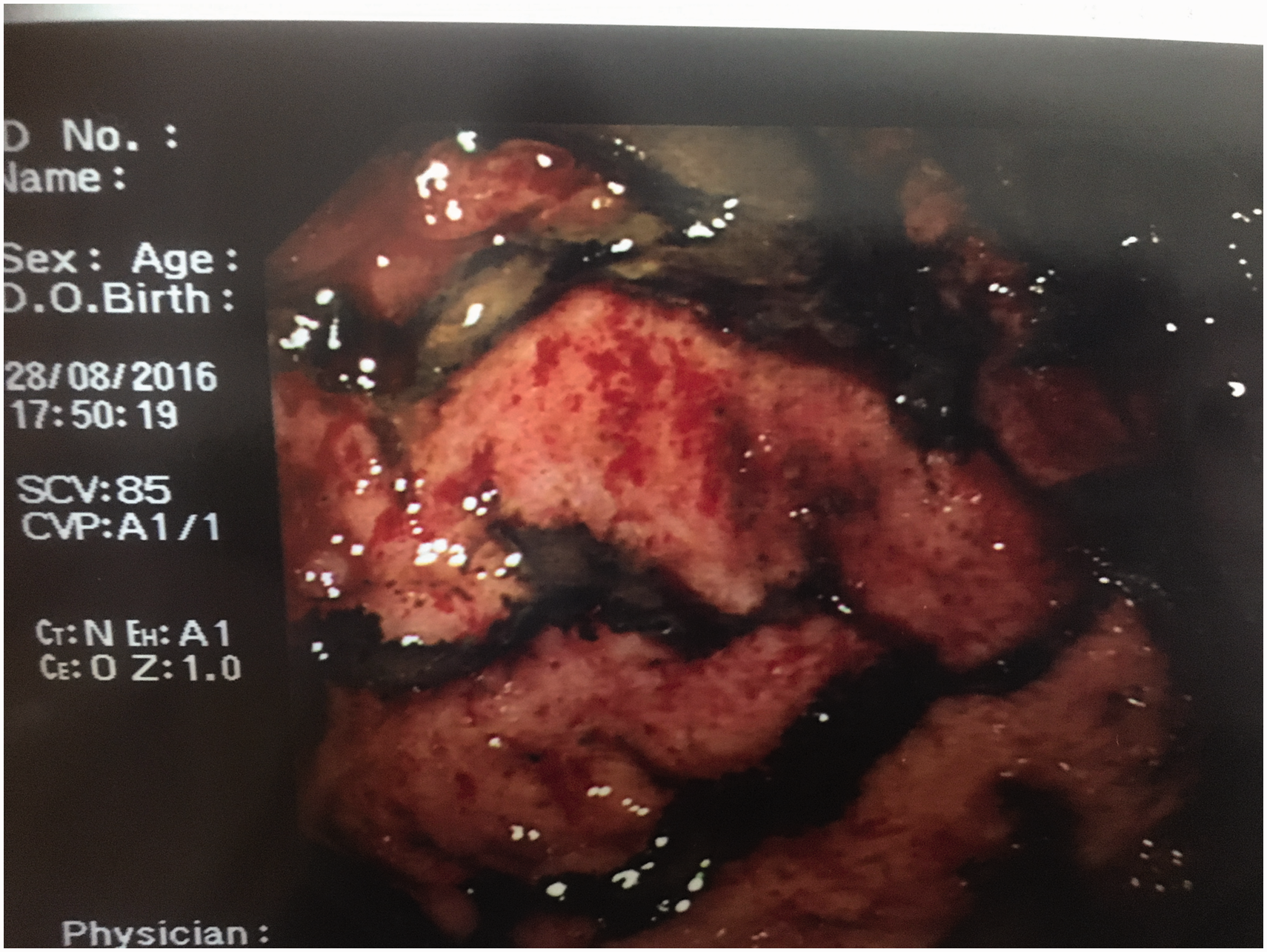
Although CT imaging revealed no evidence of viscous perforation, oesophago-gastroduodenosopy (OGD) demonstrated extensive oesophageal necrosis (Zargar grade IIIb) with contact bleeding and further, less widespread, necrosis of the stomach (Figures 1 to 3).
Proximal oesophagus showing extensive circumferential necrosis. Distal oesophagus. Contact bleeding and erythema antrum of stomach.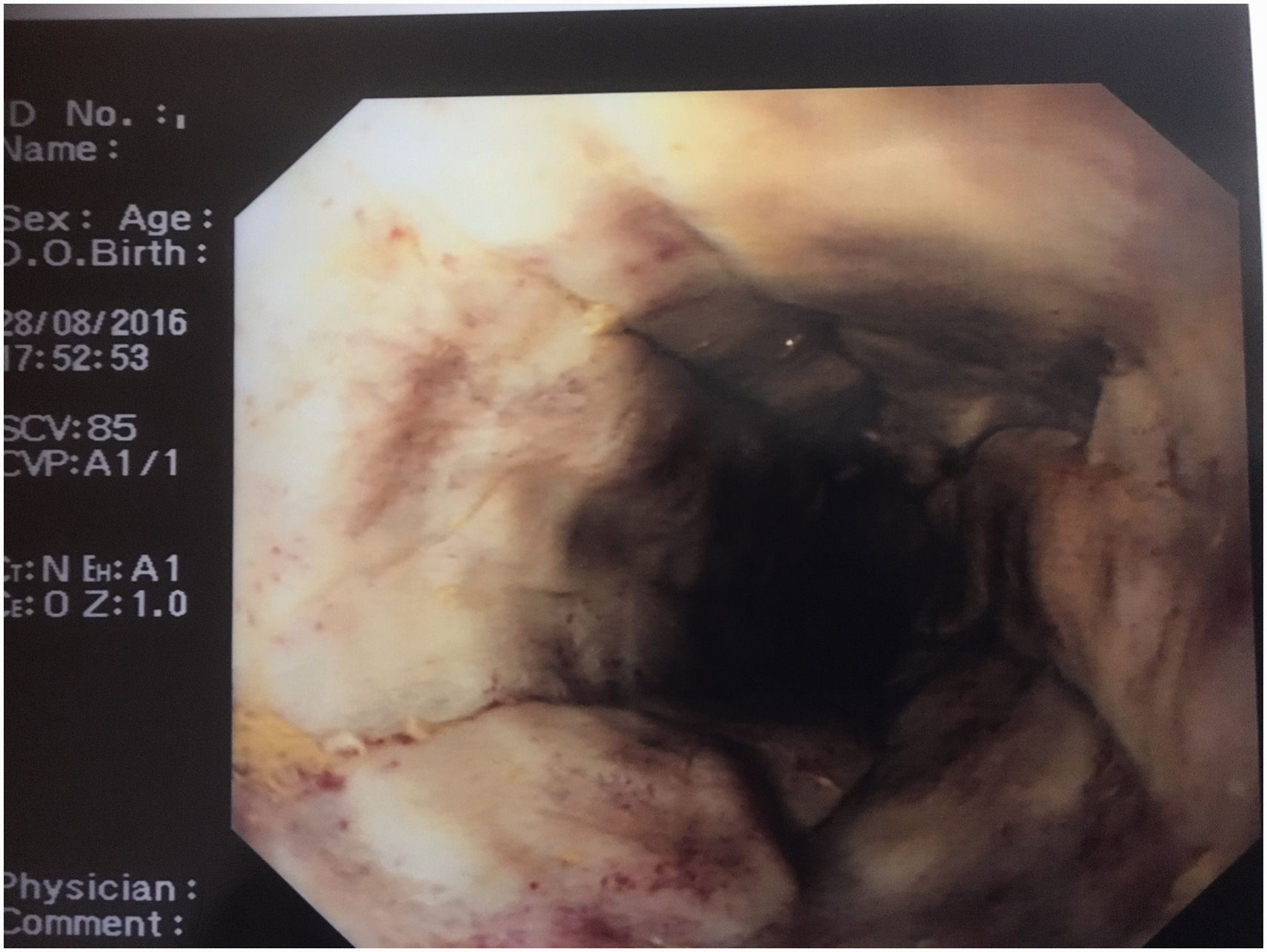
After discussion with the surgical team, a conservative approach was adopted for the management of her oesophageal and gastric injuries; total parental nutrition (TPN) was instituted on day 3, the patient remained nil by mouth and all nasogastic/nasojenunal tubes were avoided for the length of her stay. Episodes of malaena were treated with blood transfusions and a short course of tranexamic acid.
High-dose intravenous proton pump inhibitors were administered for the first month of her hospital admission. Empirical antibiotic use was avoided, with antimicrobial therapy only commenced after discussion with local microbiology consultants when there was evidence of ongoing supra-added infection – ventilator-acquired pneumonia (VAP).
A repeat OGD was performed on day 19 to reveal a significant improvement in the oesophageal epithelium with no evidence of stricture or tissue breakdown. Speech and language assessment was performed and the patient was commenced on small qualities of clear fluid with continued supplemental TPN.
Her renal function gradually plateaued to a new baseline creatinine and urea of 275 mmol/l and 39 mmol/l, respectively. Her urine output improved to 30–40 ml/h and CVVH was successfully discontinued on day 27.
Her stay in ICU was complicated by several VAPs which contributed to a protracted respiratory wean via a percutaneous tracheostomy. The patient also required slowly reducing doses of dexmetatomidine and opiate analgesia to manage episodes of agitation and pain. Repeat nasendosopy was performed on several occasions during her stay to monitor resolution of her laryngeal and arytenoid injuries. The tracheostomy tube was removed on day 34 and the patient was discharged to the ward for continued renal, psychiatric and physiotherapy support.
Discussion
Acetic acid is a clear, colourless organic acid with a pungent, vinegar-like odour. It has a diverse range of uses, reflecting the breadth of available concentrations. Its most common form found for domestic use is that of dietary vinegar (2–6%) 1 ; however, highly concentrated solutions of up to 80% are frequently used in Eastern Europe and Russia in the production of homemade vegetable preservatives and cleaning solutions. 2 Consequently, its ease of access and high potency make it one of the most common forms of self-harm in Eastern Europe with mortality rates as high as 20%.
Industrial use of acetic acid includes etching metals, fabric dye production and leather tanning. 1 Some of its properties have also been harnessed for medical purposes, specifically in the treatment of hepatocellular carcinoma, 3 diagnosis of cervical cancer 4 and for the treatment of box-jelly fish stings. 5
Exposure to concentrations as low as 10% has been reported to cause permanent damage to eyesight in animal studies, 5 whilst those exceeding 12% result in corrosive and systemic effects of skin loss, damage to mucosal membranes, haemolysis, renal failure, acidosis, shock and ultimately death. 6
Brusin and Krayeva 7 examined 400 cases of highly concentrated acetic acid in one of the only large-scale case reviews published in the literature. All patients ingested a minimum of 70% concentration, with volumes ranging from 10 to 250 ml (60 ml median). The most frequently observed complications included haemolysis (55%), renal failure (35%) and pneumonia (27%). Overall mortality was reported as 21%, but in those patients who ingested >100 ml, this almost doubled, to 40%. This significant increase is almost certainly due to the greater severity of gastrointestinal (GI) damage and subsequent greater morbidity associated with higher volumes of acid ingestion.
Zargar’s classification of mucosal injury by caustic substances. 10

Reproduced from Zargar SA, Kochhar R, Metha S, et al. The role of fibreoptic endoscopy in the management of corrosive ingestion and modified endoscopic classification of burns. Gastrointest Endosc 1991; 37: 165–169 with permission from Elsevier.
Whilst the link between the severity of oesophageal damage and mortality appears relatively unrefuted in the literature, the management of such injuries is less so. A choice remains between a conservative (non-surgical approach) verses early surgery and resection. Overt signs of perforation, peritonism and intra-peritoneal air all warrant further investigation by CT and almost certain laparotomy. 7 Some authors advocate early laparotomy as being advantageous in patients who have ingested large quantities of caustic substances. It is suggested that these patients are more likely to go on to develop necrosis, perforation and subsequent significant morbidity if not immediately presenting with signs of perforation on admission.11,12
Surgery itself, specifically oesophageal resection, in the context of caustic burns is not without significant risk and carries a major burden with long-term complications and morbidities. Thus, the management of patients presenting with severe injury without overt viscous perforation is more controversial. 5 In-depth analysis of this ongoing debate is beyond the scope of this review; however, it is of note that laboratory findings of renal failure, disseminated intravascular coagulation and acidosis have been postulated as criteria for acute surgery.12,13
The use of high-dose proton pump inhibitors has been shown in one study to improve oesophageal healing after ingestion of caustic material. 14 Whilst to date there does not exist any large controlled trials confirming this observation, current consensus appears to support their use if well tolerated and there are no contraindications. 15
As with any critical care patient, nutritional support is paramount to recovery. Placement of nasogastic tubes (NGT) after caustic oesophageal injury should be considered on a case-by-case basis. There are authors who advocate the avoidance of early NGT insertion due to the risk of iatrogenic oesophageal perforation, 16 the potential for tubes to contribute to the development of strictures 17 and the potential to act as a sources of infection 12 to a mucosa already stripped of its protected epithelium. In contrast, the benefits of early enteral feeding with regard to improved gastric and immunological health have been demonstrated elsewhere. 18 We suggest close collaboration between intensivists, gastroenterology and nutritional teams to assess the risk–benefit of NGT placement. Initiation of oral intake should be undertaken after further OGD assessment and in conjunction with speech and language teams to evaluate safe swallowing capabilities.
In addition to the severity of gastric injury, the development of systemic toxicity is reported to have high mortality rates. A number of studies report rapid progression to renal, hepatic failure and subsequent disseminated intravascular haemolysis and death shortly after ingestion with acetic acid.1,19 In the Brusin review, nearly 60% of the 105 patients who developed pneumonia as a consequence of acid ingestion died, 7 whilst mortality approached 40% in those with systemic complications in patients studied by Chisishev. 2
Haemolysis and renal failure is reported in all cases of acute acetic acid poisoning. The exact mechanism remains unclear, though it is likely related to a combination of direct renal tubular damage by the acid itself and the effect of myoglobin deposition secondary to erythrocyte haemolysis.2,20 Some authors have reported good outcomes following early plasmapheresis, thereby directly removing the caustic substance and preventing it from causing prolonged tubular damage. The benefits of one single session of plasmapheresis versus multiple courses have yet to be investigated. The authors do suggest, however that, for cases where there is ongoing haemolysis, repeated sessions of treatment may prove beneficial. 1
Hakenbeck et al. 21 in the 1980s were able to successfully use haemodialysis to treat a patient who had ingested 200 ml of 80% acetic acid, thus leading others to extrapolate that CVVH therapy may have a role in the management of similar patients. A similar benefit was reported by Russian authors who suggested that morality was reduced by nearly 30% in those patients undergoing early haemodialysis and death within 24 h of poisoning was reduced by almost 9%. 22
The findings of these earlier studies appear to further support Brusin’s outcomes for patients developing renal failure post acetic acid ingestion. Mortality for the 35% of patients who developed renal failure increased as severity of injury increased – using the RIFLE criteria, mortality was as high as 58% in those classified as ‘Failure’ and 64% as ‘Lost’. 7 Early use of renal replacement therapy is therefore increasingly accepted as one of the more important therapies to minimise morbidity and mortality in this cohort of patients.
Conclusions
Whilst not a frequently encountered method of poisoning, ingestion of concentrated acetic acid presents the intensivist with a complex array of multi-organ dysfunctions. Although early OGD may be beneficial in prognosticating, we have shown that prompt and aggressive renal replacement therapy combined with a conservative approach to oesophageal injury may result in beneficial long-term effects.
Key managerial learning points
Early assessment of upper airway patency with preparation for potential difficult intubation – consider nasendoscopy with senior ear, nose and throat support. Baseline blood tests should include full blood count, urea and electrolytes, liver function tests and clotting screen – it may be prudent to inform laboratory that suspected haemolysis is highly likely. Assess the degree of oesophageal injury with CT and OGD (gold standard). Classification using Zargar grading may be beneficial in planning future management. Assess the need for urgent surgery – e.g. oesophageal/gastric perforation. Early involvement of gastroenterology and upper GI surgeons. Establish renal replacement therapy as soon as practically possible. Proton pump inhibitors appear to offer improved healing and should be commenced. Caution with the use of nasogastic feeding tubes – we suggest early use of TPN and enteral nutrition only established once evidence of oesophageal healing with close collaboration with nutrition and gastroenterology teams. Speech and language team are also valuable in aiding assessment of swallowing abilities. Antibiotic cover as guided by local protocols and ongoing signs of overt infection/sepsis. We suggest avoidance of empirical treatment.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
