Abstract
Following the implementation of citrate anticoagulation for continuous renal replacement therapy, we evaluate its first year of use and compare it to the previously used heparin, to assess whether our patients benefit from the recently reported advantages of citrate. We retrospectively analysed 2 years of data to compare the safety and efficacy of citrate versus heparin. The results have shown that 43 patients received continuous renal replacement therapy with heparin, 37 patients with citrate. We found no significant difference in metabolic control of pH, urea and creatinine after 72 h. Filters anticoagulated with citrate had significantly longer median lifespan (33 h vs 17 h; p = 0.001), shorter downtime (0 h vs 5 h; p = 0.015) and less filter sets per patient day (0.37 vs 0.67; p = 0.002). Filters anticoagulated with heparin were commonly interrupted due to clotting (50% vs 16.4%), whereas filters anticoagulated with citrate were often stopped electively (53.4% vs 24.6%). Patients on heparin filters had significantly higher APPTs, some at potentially dangerous levels (>180 s), whilst patients on citrate filters had significantly higher levels of bicarbonate. Therefore, we conclude that citrate is superior in terms of safety and efficacy, with longer filter lifespan. It has become our first line anticoagulant for continuous renal replacement therapy.
Background
Renal replacement therapy (RRT) is an important supportive therapy used in critically ill patients who develop acute kidney injury (AKI). It is a therapy that has been widely reviewed and discussed, but despite significant improvement in technology, the mortality for patients with acute dialysis-dependent renal failure remains greater than 50%. 1 For this reason, an evaluation of how it is delivered is important.
In RRT, blood is taken from the patient and passed through an extracorporeal circuit, where waste products and water are removed by convection (±diffusion) across a semi-permeable membrane. The risk of blood clotting necessitates the use of anticoagulation to keep the extracorporeal circuit patent. Filter clotting poses a major problem in RRT contributing to blood loss and the requirement for blood transfusions, as well as a reduction in therapy delivery due to circuit ‘down-time’.2,3 These observations were the foundation of several subsequent studies comparing the safety and efficacy of different anticoagulants; in particular comparing heparin and the relatively new citrate system.
Until recently, systemic heparin remained the most common anticoagulant used for continuous renal replacement therapy (CRRT). 4 Heparin works by binding to antithrombin III, increasing its effects by 1000-fold and inhibiting the clotting factors Xa and thrombin (IIa). 1 It is relatively easy to use and monitor and provides adequate extracorporeal anticoagulation. However, its anticoagulant effects extend to the patient, posing an increased risk of bleeding and the development of heparin-induced thrombocytopenia (HIT).1,5 Citrate works by chelating calcium, which is required for activation of the clotting cascade. Normal serum calcium levels are maintained by a replacement infusion, preserving the coagulation process within the patient while acting as a regional anticoagulant, thus avoiding the systemic anticoagulation effects seen with heparin. Citrate is metabolised by the tricarboxylic acid pathway in the liver, skeletal muscles and renal cortex, producing bicarbonate. If it is not metabolised sufficiently, citrate may accumulate, causing hypocalcaemia and metabolic acidosis. On the contrary, metabolic alkalosis may occur if too much citrate enters the circulation and is metabolised to bicarbonate.5,6
In October 2014, Hampshire Hospitals NHS Foundation Trust introduced the use of citrate in one of its two critical care units. The decision was based on studies suggesting citrate may be superior to heparin with an increased filter survival time, reduced risk of bleeding and reduced number of blood transfusions.7–9 While randomised controlled trials (RCTs) and meta-analyses demonstrate the advantages of citrate over heparin, they also discuss potential disadvantages, such as increased complexity of the protocol, as well as potential metabolic and electrolytes disturbances and possible higher financial costs. 10 This is likely why citrate has been slow to gain popularity with less than 10% of all patients on CRRT for AKI receiving citrate in a worldwide survey in 2007 11 and none of the UK units using citrate in 2008. 4 Moreover, meta-analyses of RCTs question the power of these trials, as well as the generalisability of the results due to the great heterogeneity of the studies in terms of modalities, machines and filters used, patient groups and protocols used.12,13 For this reason, we felt it was important to review local practice in light of the recent changes; to identify potential problems and false perceptions with the use of citrate, outside the controlled research setting.
The main objectives were to:
Evaluate the safety and efficacy of the CRRT delivered in our unit in light of a recent change in practice Compare the two systems – citrate vs heparin
Method
CRRT delivery
The need for introduction and cessation of CRRT was decided by the treating physician, in a closed 14 bedded District General Hospital ICU with 24 h Consultant Intensivist cover; admitting an average of 379 level 3 and 667 level 2 patients per year. CRRT was provided using the PrismaFlex system in pre-dilution mode with ST150 filter-sets and Gambro 5 L fluid bags, as per standardised protocols (Prismasol 4 or Haemosol BO with heparin protocol; Prismocitrate 18/0, Prism0cal B22 and Prismasol 4 with citrate protocol). The heparin protocol included continuous veno-venous haemofiltration (CVVHF) or haemodiafiltration (CVVHDF) with a target effluent dose of 25–45 ml/kg/h. It involved protocolised heparin dosing, including an initial bolus of 0–5000ius, and subsequent target Activated Partial Thromboplastin Time (APPT) of 75–120 s. The regional citrate protocol included continuous veno-venous haemodiafiltration with an effluent dose 25–45 ml/kg/h. It included a prescription that allowed titration of citrate and calcium gluconate infusion according to filter calcium levels (target 0.25–0.35 mmol/L) and patients’ ionised calcium (target 0.99–1.2 mmol/L), respectively.
Study design
A retrospective analysis was conducted on data collected in routine care over a 2-year period. This included all patients on CRRT with heparin over a 1-year period (October 2013–September 2014) and another year’s worth of data using the citrate protocol (following the introduction of citrate in October 2014). Data were collected through analysis of medical and nursing notes, blood results and observation charts. A data collection tool was devised to compile demographic information and specific outcome measures including:
Efficacy: (i) extra-corporeal circuit (i.e. filter) survival time
(ii) metabolic control (pH, urea and creatinine)
Safety: (i) trend of haemoglobin concentration (Hb) during CRRT
(ii) severe derangement of coagulation profile (APPT > 120)
(iii) number of units of packed red cells transfused with no other identifiable cause other than CRRT
(iv) metabolic issues resulting from the use of citrate
Participants/sample
Participants included all patients receiving CRRT between October 2013 and September 2015, as identified from the Intensive Care National Audit and Research Centre (ICNARC) database for the critical care unit in the Basingstoke North Hampshire Hospital. Patients who received anything other than heparin or citrate were excluded from analysis.
For extra-corporeal circuit life-span analysis, the sample consisted of all the circuits anti-coagulated with heparin or citrate over the study period.
Statistics
Data were inserted into a database on SPSS v22 and this software was used for statistical analysis. A variety of descriptive statistics were used to summarise data for both groups. Median values are presented as most data were non-normally distributed and the Mann–Whitney U test was used to compare groups. Filter survival data were analysed using Kaplan–Meier plots and a log-rank test used to compare the median circuit life of the two groups.
Ethical issues
Individual patient consent was not required as this was a retrospective analysis of data collected for routine care. While NHS ethical approval was not necessary, the study was registered as a service evaluation project with the local Research Department and Clinical Governance office. As this project was work towards partial fulfilment of a University Degree, it was also registered on the University Ethics and Research Governance Online (ERGO) and the study protocol peer-reviewed and approved by the ethics committee. Patients’ confidentiality and anonymity was safeguarded at all times.
Results
One-hundred and three patients required RRT between October 2013 and October 2015. Seven of these patients had vital data missing and were therefore excluded.
Number of patients per anticoagulation protocol and number of filters for heparin and citrate groups.
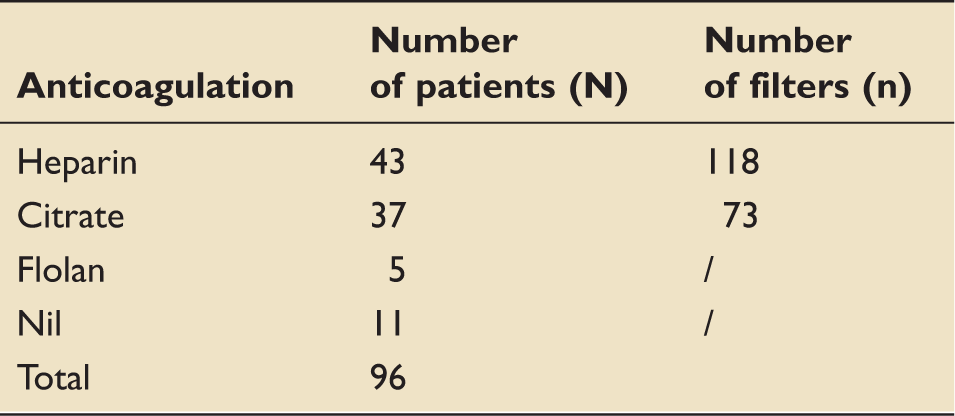
Out of the 43 patients on the heparin protocol, 15 had multiple episodes of RRT (10 had two episodes and 5 had three episodes – new treatment episode defined as therapy restarting after being off for more than 24 h). Out of the 37 patients on citrate protocol, 10 had two episodes of RRT.
Only the first episode of RRT was included for analysis of blood results. Patients who had a change in their anticoagulation mid-episode were excluded in order to keep efficacy data true to the anticoagulation in question.
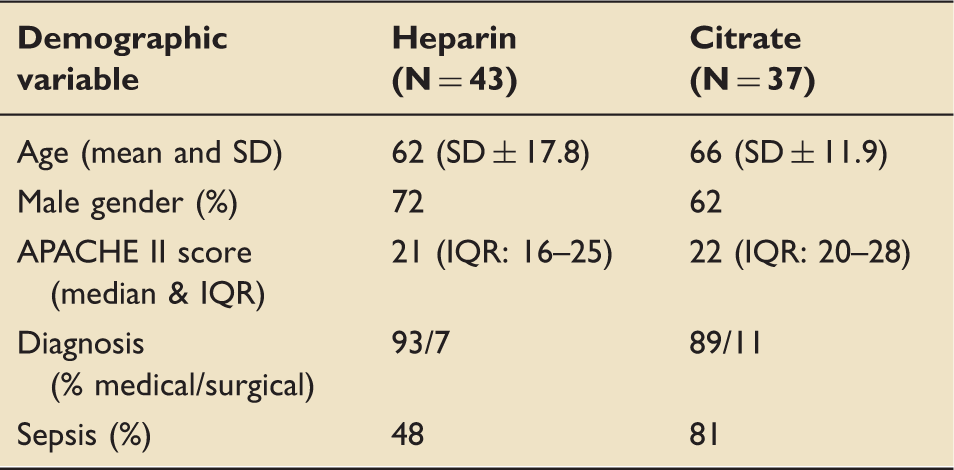
Patient demographic data
Demographic data.
Efficacy
For the purpose of this evaluation, efficacy was defined as
the filters’ ability to clear metabolites in particular clearance of urea and creatinine and filter life span, including filter downtime and filter sets per patient per day, to give a sense of filter wastage and disruptions in treatment delivery.
Metabolic control
Analysis of blood results: Values presented as medians (standard deviation).
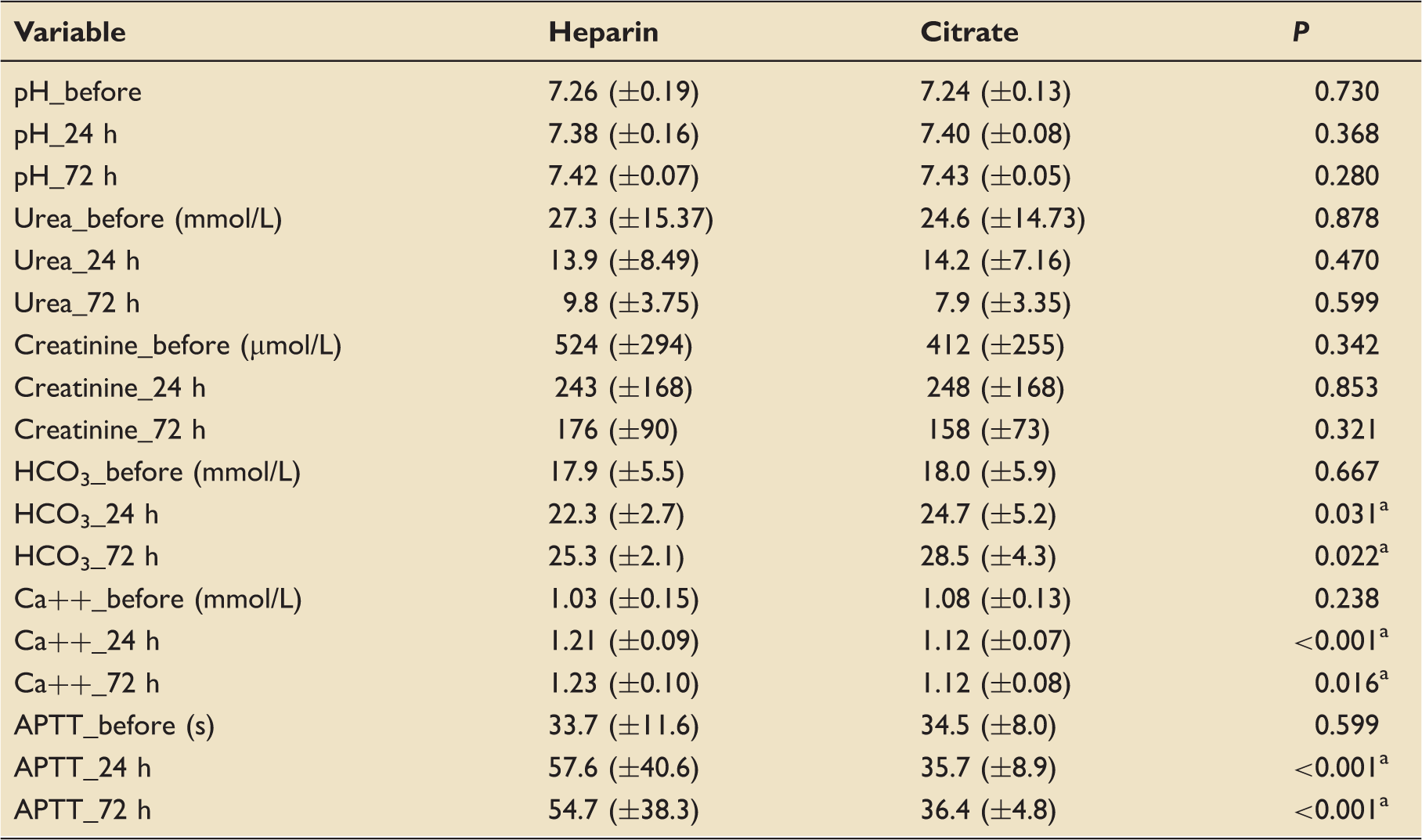
Significant P value (<0.05).
Filter life span
Filter lifespan.

Values presented are medians with minimum and maximum life-span in hours. P value < 0.05 = significant.
Percentage of filters that were stopped and for what reason.

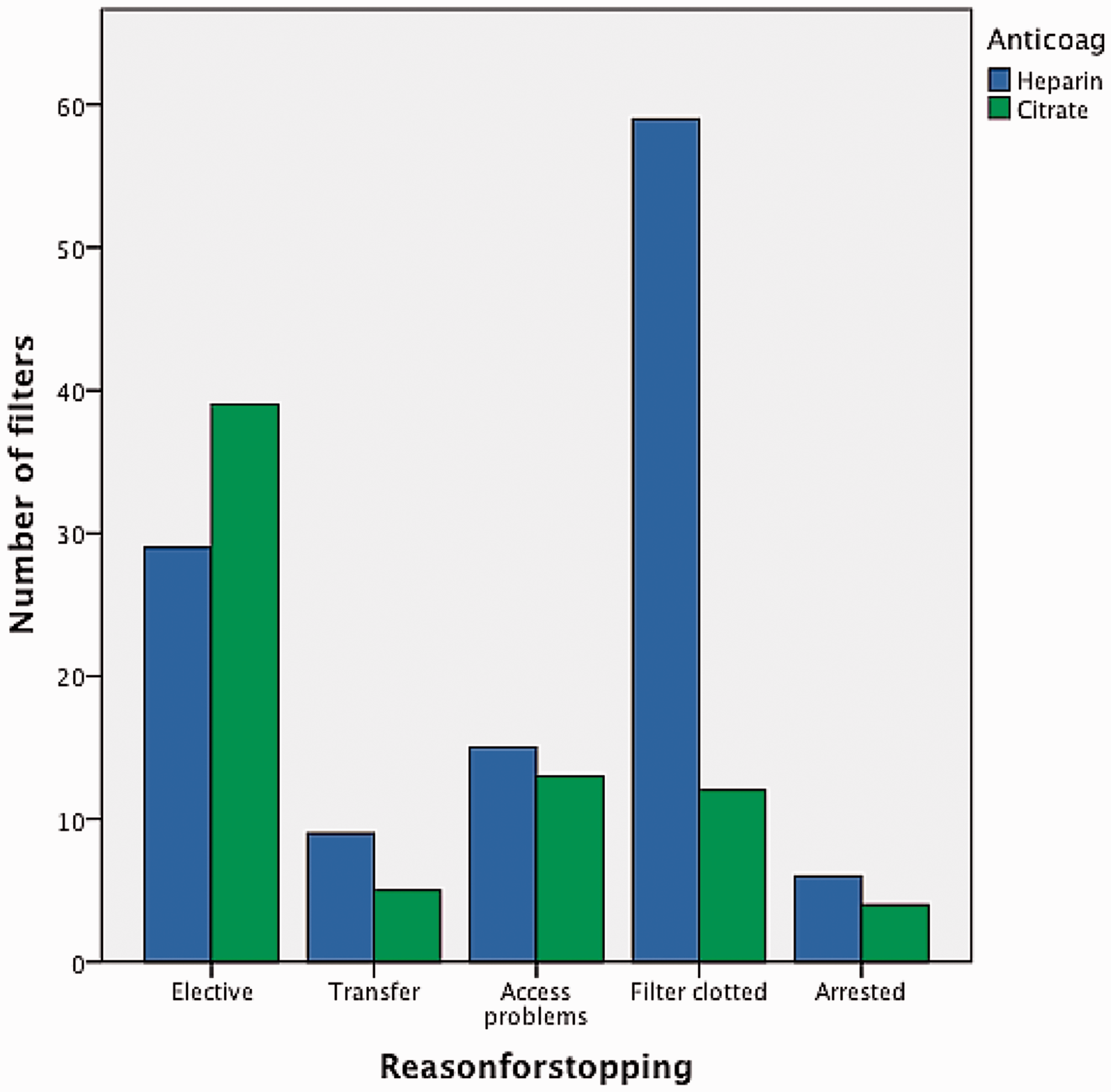
Reasons for stopping filter (presenting absolute number of filters).
Blood and filter-sets wastage due to clotting was looked at in more detail using the Kaplan–Meier survival analysis. This particular analysis does not take into account repeated events for the same patient. Therefore, only the survival probability of the first filter for each patient was analysed. This was initially done for all five reasons for stopping the filter and repeated with censors for analysis of clotted filters only. The groups were then compared by log rank tests (Figure 2). In each of these analyses, citrate was found to be superior to heparin for filter lifespan.
Kaplan–Meier curves illustrating filter life-span for all reasons for stopping (top) and life-span for clotted filters only (bottom).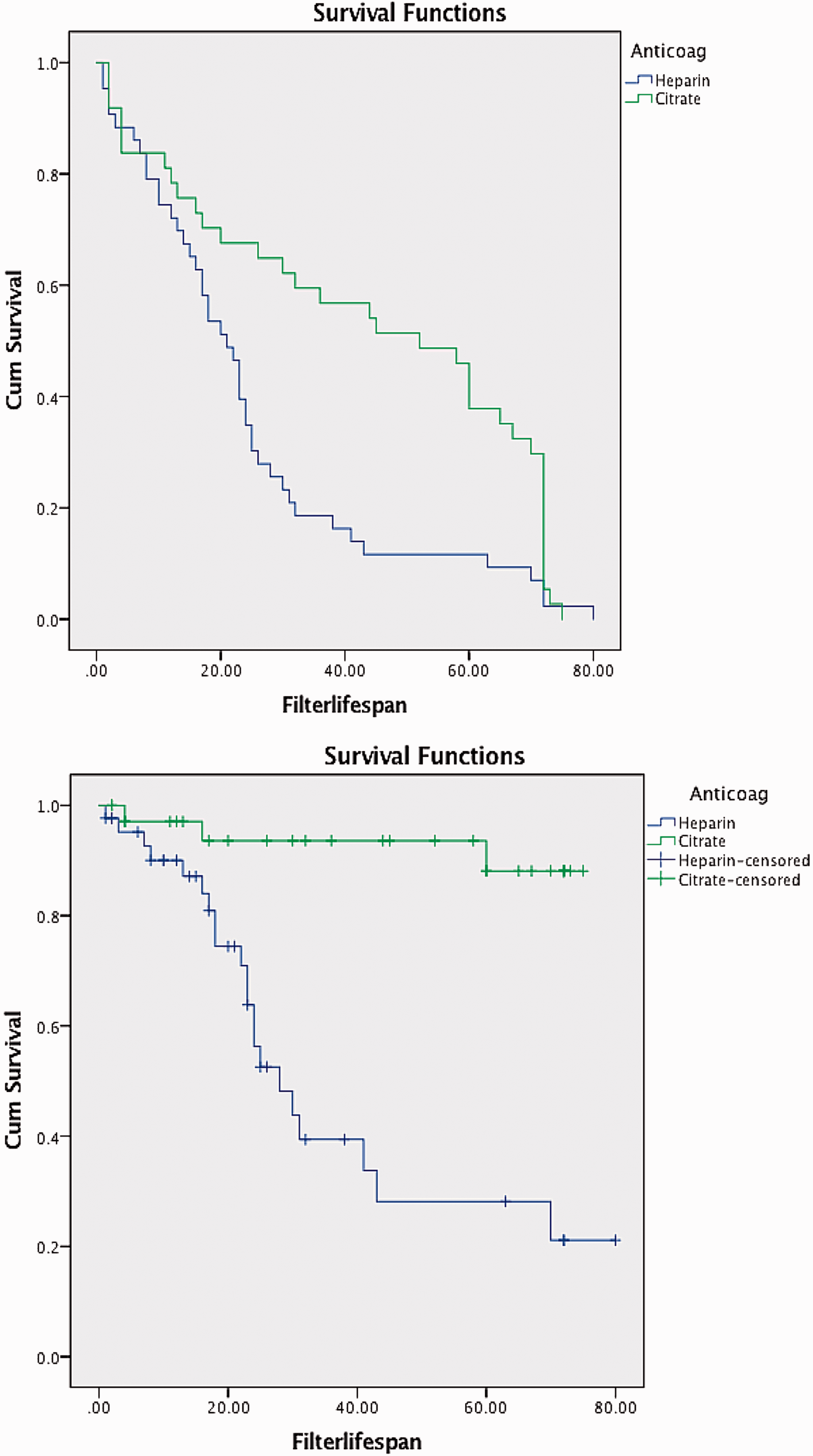
Filter downtime and filter sets per patient per day.
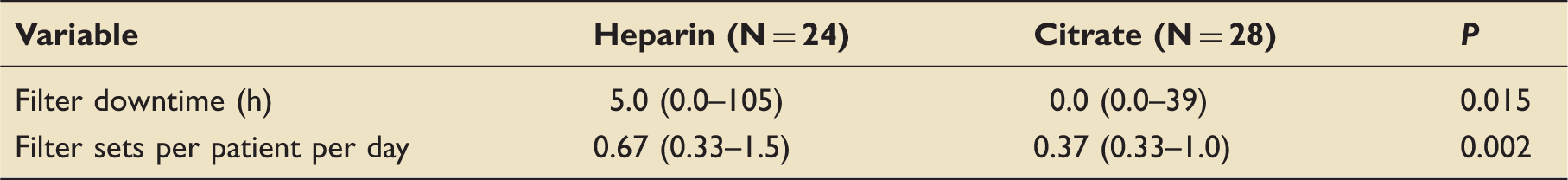
Values presented are medians with minimum and maximum ranges. P values < 0.05 = significant.
Haemoglobin trend for heparin and citrate at baseline, and after 24 and 72 h of filtration (standard deviation).
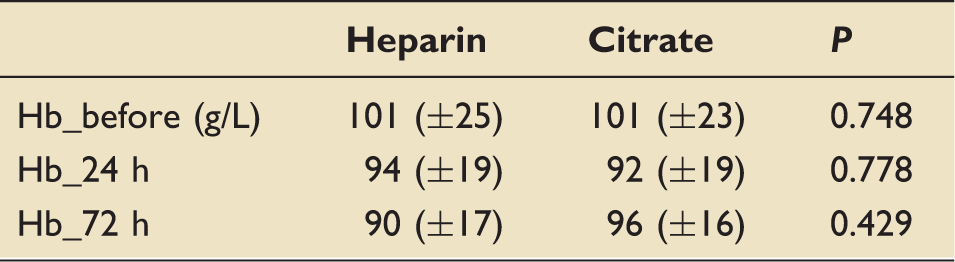
Safety issues
Heparin
Out of the 43 patients, 10 (4%) on heparin had recorded APPTs of >180 s, some of the patients with repeated readings at this level. Eight patients on heparin had their anticoagulation stopped or changed. Generally, this was due to a clinician’s concerns of risk of bleeding (N = 4), thrombocytopaenia (N = 2) or repeated clotting of the filter (N = 2).
Citrate
Filtration with citrate was discontinued on three occasions. One of these was because the patient required systemic anticoagulation with heparin for atrial fibrillation (AF). On the other two occasions citrate was stopped due to clinicians’ concerns of citrate accumulation warranted by persisted metabolic acidosis. In both cases, patients had liver dysfunction (one patient had acute liver failure with ALT > 300, and the other had liver cirrhosis confirmed on imaging with ALT > 100). Unfortunately, there were no calcium ratios recorded for either of these patients.
On review of ionised calcium levels during filtration with citrate, calcium ions seem to be maintained at safe levels (>1 mmol/L) with no obvious recorded adverse events related to low ionised calcium. Thirteen patients (5%) had documented alkalosis with pH > 7.45 and bicarbonate > 26 mmol/L.
Trend in haemoglobin and blood transfusions
An obvious downward trend in level of haemoglobin (Hb) was observed in both groups. While the drop is more pronounced in the heparin group, there was no significant difference in Hb levels up to 72 h (Table 7).
There were more recorded blood transfusions in the heparin group than in the citrate group during the first treatment episode (10 units vs 0 units). The correlation between the number of filters used and the number of blood transfusions required was explored using Spearman’s rho. This found a significant but moderate, positive correlation between these two variables: the more filters used the higher the number of blood transfusions (r = 0.4222, p ≤ 0.001).
Discussion
This retrospective study evaluates the impact of regional citrate anticoagulation on safety and efficacy of CRRT immediately after its implementation on a general intensive care unit, compared with pre-existing systemic heparin anticoagulation. Most of our findings are comparable to previous observational studies and RCTs.
Efficacy
Our results showed no significant difference in the metabolic control of urea and creatinine, suggesting equivalence in efficacy. This finding is similar to results from RCTs that looked at urea clearance as a measure of efficacy.8,14 In contrast, some studies found a significant difference in urea levels by the end of the CRRT, with lower levels of urea in the citrate group compared to the heparin group. This was attributed to longer filter lifespans with citrate and longer filter downtimes with heparin.7,15
Other studies measured treatment dose delivery and/or filter downtime as a marker of efficacy and concluded that CRRT with citrate is more efficient than heparin as there are less disruptions in treatment delivery.5,16–19 In this study, we evaluated both metabolic control and filter downtime. It was interesting to find that, whilst delivery of treatment with citrate was better with a significantly lower filter downtime, it did not correspond to a significant difference in urea and creatinine clearance. This is possibly due to the higher rates of predilution fluid used on the pre-blood pump to deliver the citrate anticoagulant, as set by the filter protocolised software, which haemodilutes the blood in the filter, reducing the concentration gradient and therefore reducing the efficiency of filtration. Furthermore, the retrospective design of this study meant that the blood results included in the analysis were not taken at exactly 24 and 72 h, which may have affected our results.
Filter life span
Filter patency is critical when delivering CRRT. Our data found that filter lifespan with citrate was significantly higher compared to heparin. This corresponds to findings from several observation studies10,15–17,20–22 and RCTs,5,7,8,18,23,24 where a wide variation of filter survival times are reported, which is thought to be related to the variety of protocols used in different studies.12,13,25 In contrast, two studies found no difference in filter life and a recent meta-analysis suggests that filter life is prolonged only when using particular filtration modes. 26
Premature clotting causes disruption of CRRT and potentiates the need for blood transfusion.2,3 Using the Kaplan–Meier model, we demonstrated that the probability of the first filter failing due to premature clotting was significantly reduced with citrate. This echoes findings of other studies that used similar models.7,10,16,18,22 Additionally, a review of the reasons for stopping CRRT showed the percentage of filters stopped due to transfer needs, access problems or sudden death were similar across both citrate and heparin protocols. However, there was a noticeably higher percentage of heparin filters stopped due to clotting. This was a common observation in similar evaluation studies, which again, reinforces citrate’s superiority in prolonging filter life whilst reducing blood and filter wastage.10,22
Safety issues
Inevitably, the safety concern with using systemic heparin as anticoagulation is the fact that the patient is anticoagulated as well as the filter, which increases the risk of bleeding. In this evaluation, we found a number of patients had worryingly high levels of APPT (>180 s) and more patients on heparin required a change in anticoagulation, due to bleeding, risk of bleeding or repeated clotting of filters. There is a fine balance between maintaining filter patency and patient safety; as previously noted, maintaining target APTTs can be difficult. 22
Citrate is not without its own set of challenges and safety concerns. It is fair to say that, this being our first year using citrate, there was some justifiable apprehension in using citrate in patients with liver dysfunction due to potential accumulation. 27 Despite our relative inexperience with the protocol, citrate was only discontinued on two occasions due to clinicians’ suspicion of accumulation in the context of liver dysfunction. Unfortunately, in both these cases calcium ratios (ratio of total calcium/ionised calcium), a recognised measure of potential accumulation, were not calculated. The lack of documentation of calcium ratios has been an important observation from this evaluation study and highlighted as a safety concern, with the recommendation for further staff education.
Similar to other studies,7,8,15,28 metabolic derangement with a significantly higher level of bicarbonate and a significantly lower ionised calcium were prominent observations in the citrate group, a reflection of the pharmacological properties of citrate. No adverse events were reported.
Trend in Hb and blood transfusion rates
Whilst an obvious downward trend in Hb was observed in both groups, this seemed to be more prominent in the heparin group. Interestingly, all the patients who required a blood transfusion had treatment episodes that lasted more than 3 days with multiple filter-set changes within that episode. This manifested in a positive correlation between the number of filters used and blood transfusion rates, similar to a previous study. 2 As they allude, the weak relationship between the two suggests that other confounding factors may be at play such as erythropoietin deficiency and bone marrow suppression due to severe illness, amongst others.
Cost implications
A full cost analysis was not done, but the reduction in filters per patient per day and the potential reduction in blood transfusions both have reduced cost implications when using citrate. 6 Potentially, citrate also reduces valuable nursing time spent in repeatedly taking down and setting up filters due to premature clotting. 29
Limitations
The retrospective nature of this study is its biggest limitation, as the quality of the data analysed is heavily reliant on the quality of the nursing and medical documentation. The blood tests were not taken at the exact 24 and 72 h of treatment; rather most times, the results used for blood analysis were from blood tests taken closest to these times allowing up to 10 h difference. In an attempt to look for a correlation between blood transfusions and number of filter sets, we excluded blood transfusions given for obvious reasons such as bleeding or post-op patients. This would have certainly introduced an inclusion/exclusion bias. Last, confounding factors that can potentially affect efficacy such as filtration modalities and flow rates were not controlled.
Going forward
These results have been used to inform changes in anticoagulation protocols as well as highlight areas that require further education, such as the use of citrate in liver dysfunction and refractory shock, and monitoring for signs of accumulation. At the time of writing, both heparin and citrate protocols are being reviewed by a team of senior nursing colleagues and medical consultants. We plan to re-evaluate certain aspects of this study after implementing these changes.
To our knowledge, there has not been a survey on anticoagulation practice across the UK since 2008, when none of the UK units were using citrate. 4 It would be interesting to review how many units have now adopted citrate following published guidelines in 2012 suggesting citrate as first line anticoagulation. 30
Conclusion
Whilst acknowledging the limitations of this study, we found that implementing a citrate protocol resulted in longer filter-life, less incidences of premature clotting, less risk of bleeding and potentially a reduction in blood transfusion rates. We recognise the challenges of implementing a new system but our evaluation demonstrates that citrate is safer, and most likely more efficient than heparin. Over the last 18 months, as we slowly gained confidence with the citrate protocol, we have moved on from using citrate for patients with increased risk of bleeding to using it as first line anticoagulation for CRRT.
Footnotes
Acknowledgements
The authors acknowledge Dr Sean Ewings, Research Fellow, University of Southampton, Medical Statistician, Southampton General Hospital for his advice on statistical analysis.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
