Abstract
An 18-year-old female inpatient on a neurosciences intensive care unitwith new onset super-refractory epilepsy became hypoglycaemic 48 h after commencing co-trimoxazole. She had been placed on this for prophylaxis against Pneumocystis jiroveci infection in the context of significant immunosuppression with high-dose corticosteroid therapy. In order to maintain glucose control, she required a continuous infusion of 10% dextrose at rates of 15–25 ml/h. Recurrent attempts to wean this were limited by further hypoglycaemia, until she spontaneously regained normoglycaemia after 73 days. This case report will discuss this unusual case of refractory hypoglycaemia, and the proposed pathophysiology of hypoglycaemia related to co-trimoxazole therapy.
Keywords
Background
Pneumocystis jiroveci pneumonia is a life-threatening condition associated with significant immunosuppression. As this can be life-threatening, it is advised that patients receiving over 20 mg prednisolone for over one month should be commenced on prophylactic antibiotics. 1 Co-trimoxazole (combination of trimethoprim and sulfamethoxazole in a 1:5 ratio) is first line for prophylaxis due to its efficacy. 1 However, it carries a high incidence of adverse effects, including hypoglycaemia.2,3
Hypoglycaemia is defined as plasma glucose levels low enough to put the patient at risk of harm. 4 This is generally considered to be below a threshold of 3.0 mmol/L in healthy individuals. 4 Hypoglycaemia poses immediate risk to patients (which in severe cases include seizures and death) but is also associated with an increased all-cause mortality rate (36.6% vs. 19.7% in critical care patients). 5
Co-trimoxazole-induced hypoglycaemia is poorly understood and rarely described in medical literature, but it can cause significant morbidity, as described here. 3 This case describes a neurosciences intensive care unit (NICU) patient who required a 10% dextrose infusion to maintain normoglycaemia for 73 days after administration of co-trimoxazole. This case is unusual due to the long duration of hypoglycaemia after stopping co-trimoxazole (47 days), and she was taking medications usually associated with hyperglycaemia (high-dose prednisolone). The aetiology of her status epilepticus remains unknown and will not be discussed here.
Case presentation
Patient's medication list at time of hypoglycaemia onset.

Her plasma glucose was routinely measured every 4 h. Her first measured low blood glucose (2.4 mmol/L) was on day nine (48 h after commencing co-trimoxazole), and was treated with a bolus of 50 ml of 20% dextrose. This transiently resolved the hypoglycaemia only for it to recur, requiring further boluses of dextrose, and then a continuous infusion of up to 45 ml/h of 10% dextrose. She continued to require supplemental intravenous dextrose for 73 consecutive days, despite multiple attempts to wean the infusion.
Investigations
Potential causes of hypoglycaemia in critical care patients.

Hypoglycaemia may be a feature of sepsis, and the patient suffered several episodes of sepsis. 4 However, the onset of hypoglycaemia was in a sepsis-free period, and the constancy of the hypoglycaemia for 73 days (with minimal blood glucose fluctuation during septic episodes) suggested that the cause was not episodic.
After thorough multidisciplinary review of her medications, the only medication which had hypoglycaemia as a recognised side effect (in the British National Formulary and on literature review) was co-trimoxazole. 2 We therefore changed her Pneumocystis prophylaxis to dapsone after 24 days.
The patient regained normal glucose homeostasis 47 days after stopping co-trimoxazole. The resolution of her glucose control progressed in the context of otherwise general stability in her condition. She currently remains on Neuro ITU (day 145). Since the resolution her hypoglycaemic episodes, she has developed the more familiar requirement for exogenous insulin to treat hyperglycaemia whilst on high-dose glucocorticoid therapy.
Discussion
The sulphamethoxazole component of co-trimoxazole is thought to be responsible for its hypoglycaemic effect. 6 This is because it is structurally similar to sulphonylureas, and it is thus thought to stimulate pancreatic beta-cell insulin secretion. 7 In fact, sulphonylureas were first created from the discovery of sulphonamide-induced hypoglycaemia in 1942. 6 Despite this link, hypoglycaemia is a surprisingly rare side effect of co-trimoxazole. The Medicines and Healthcare products Regulatory Agencyhas only received 23 reports of sulphamethoxazole-associated hypoglycaemia since 1964. 3
The duration of hypoglycaemia (47 days) after stopping co-trimoxazole suggests that its mechanism for hypoglycaemia involves a permanent or semi-permanent process. Although not defined, this could represent irreversible receptor agonism, similar to the antiplatelet effect of aspirin. Another possible pathophysiology could include autoimmune stimulation of insulin processes or inhibition of glucagon pathways.
A PubMed search for literature containing the words ‘hypoglycaemia’ and ‘co-trimoxazole’ found 16 case reports discussing cases of hypoglycaemia associated with co-trimoxazole administration since 1988. Five of these were associated with coinciding sulphonylurea use and are not discussed here.
Case reports documenting co-trimoxazole-induced hypoglycaemia.
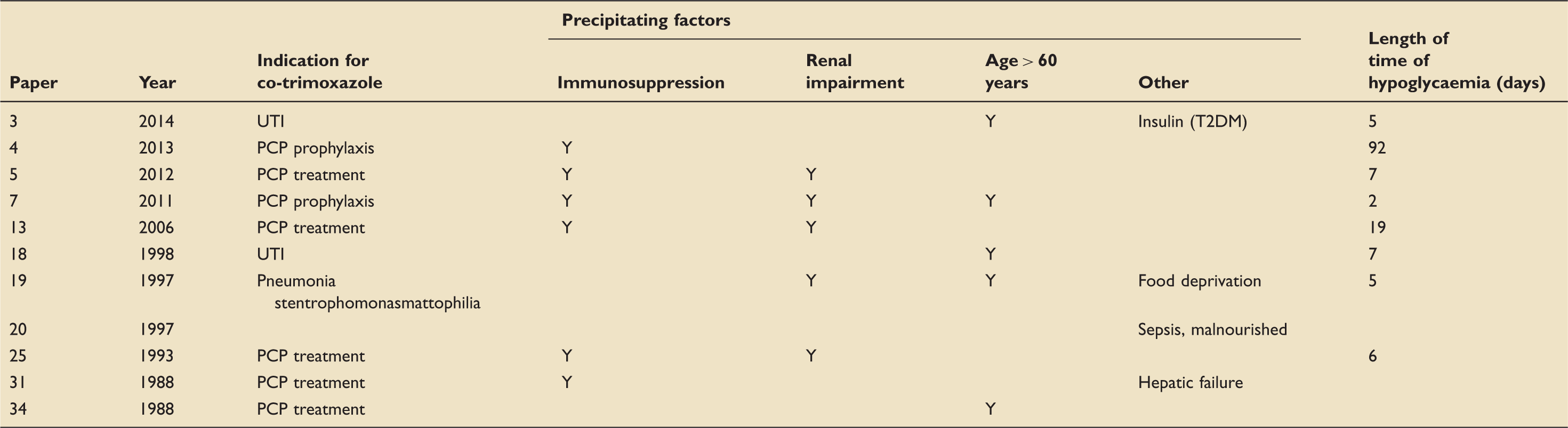
Hypoglycaemia is a life-threatening complication which, when identified, can be safely managed with intravenous dextrose supplementation. This case highlights a poorly recognised, severe side effect of a commonly used medication. However, what makes this case extraordinary is the severity and duration of this complication.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
