Abstract
This editorial examines the epidemiology of nosocomial infection in trauma intensive care. Specifically, ventilator-associated pneumonia, central line-associated blood stream infection, and catheter-associated urinary tract infection rates are described. Two important trends are observed. Firstly, nosocomial infection rates have fallen with time. This trend is evident in all intensive care populations and is thought to be principally due to the adoption of preventative bundle strategies. Secondly, rates remain consistently higher in trauma patients than in other intensive care populations. The reasons for this are likely to be multifactorial. Recognizing the particular vulnerability of this patient group should prompt especially rigorous efforts at prevention, early diagnosis, and management.
Introduction
Health care-associated infection in critically ill patients represents a significant burden of disease.
There is limited information in the medical literature on nosocomial infection in trauma patients admitted to intensive care units (ICUs). This editorial aims to describe infection rates among these patients.
Trauma care in England has become centralized with the advent of regional trauma networks (similar structural change is underway in the rest of the UK). At the heart of each network is a major trauma center. This coordination of care means that higher concentrations of critically ill trauma patients are managed in a smaller number of ICUs. It is therefore more relevant than ever to address the question: are trauma patients different?
Where is the data?
While there are many single-center studies, there is very limited epidemiological data available on nosocomial infection in trauma patients outside the United States.
The National Nosocomial Infection Surveillance System (NNIS), an instrument of the US Centers for Disease Control and Prevention (CDC) that has subsequently been incorporated into the National Healthcare Safety Network (NHSN), is the United States’ most widely used health care-associated infection tracking system, and has produced several reports over the last decade.1–7 The use of nationally accepted definitions, 8 and consistency of data collection and reporting, enables valid comparison of infection rates with time, and between different patient subpopulations.
The International Nosocomial Infection Control Consortium (INICC) is a health care-associated infection control program founded in Argentina in 1998, with a surveillance system based on that of the US NHSN. Using NHSN definitions and protocols, it has similarly produced reports collating data from its largely developing world hospital network.9–11 Currently its practices span 36 countries.
There is less coordination within Europe, partly because methods for national surveillance in different European countries have been too heterogeneous to allow meaningful comparison of results. In the UK there is no coordinated or centralized approach to collecting data on nosocomial infection in ICU patients. Most UK ICUs do supply returns to the Intensive Care National Audit and Research Centre Case Mix Programme, but the scope and breadth of this data are too limited for epidemiological analysis. The Trauma Audit and Research Network collects epidemiological data on trauma patients across England and Wales, but its focus is on overall mortality rates and adherence to key trauma standards of care.
Ventilator-associated pneumonia
Definition
Pneumonia is difficult to define, and variations in definitions have contributed to wide variations in reported ventilator-associated pneumonia (VAP) rates. 12 Operationally, it is widely accepted that VAP is pneumonia developing ≥48 h after endotracheal intubation and/or mechanical ventilation. By US CDC/NHSN definitions, pneumonia is diagnosed using a combination of radiological, clinical, and microbiological criteria, and broadly subdivided into “clinically defined pneumonia,” “pneumonia with specific laboratory findings,” and “pneumonia in immunocompromised patients.” 8 While all three require X-ray changes and combinations of clinical symptoms and signs, “clinically defined pneumonia” does not require any positive microbiology. There is variation in the diagnostic criteria used in the UK.
The available data
Ventilator-associated pneumonia rates in intensive care units.
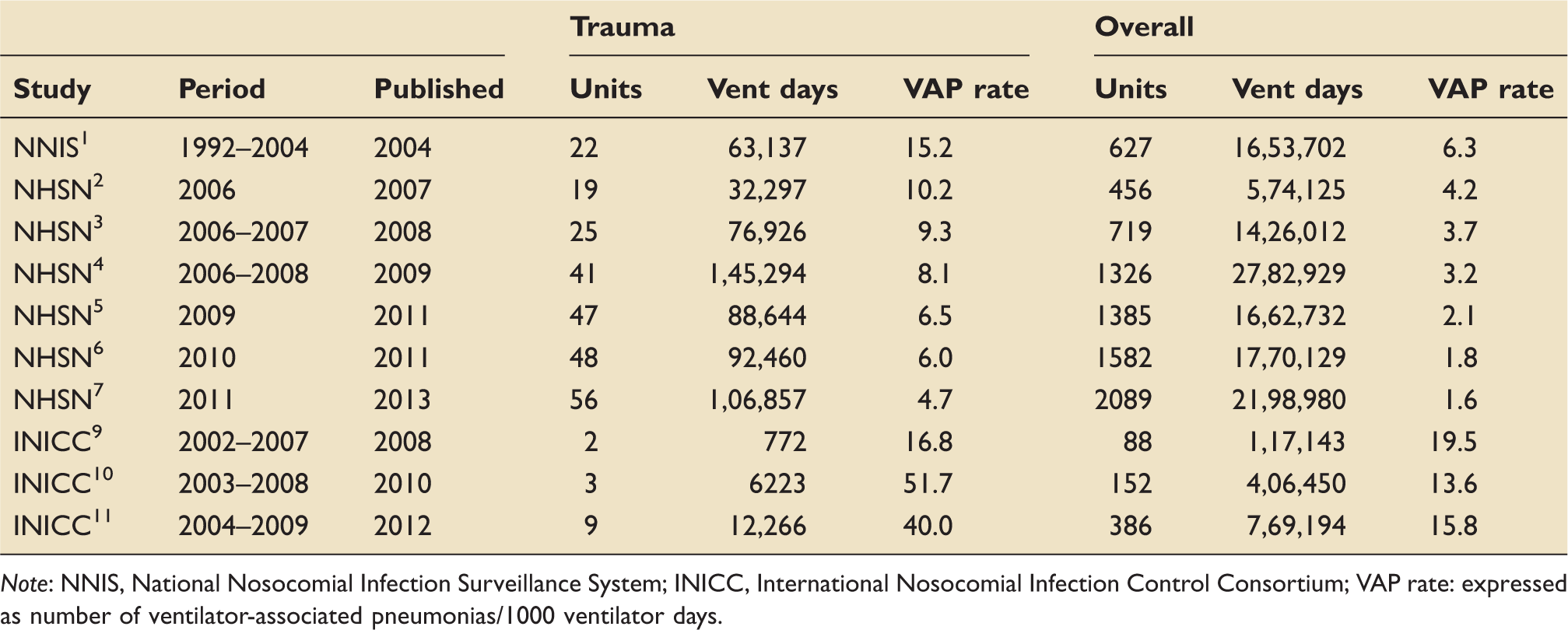
Note: NNIS, National Nosocomial Infection Surveillance System; INICC, International Nosocomial Infection Control Consortium; VAP rate: expressed as number of ventilator-associated pneumonias/1000 ventilator days.
Firstly, the NHSN data show that VAP rates in trauma patients are consistently in the order of two- to threefold higher than in ICU patients overall. The 2011 NHSN data demonstrate a VAP rate of 4.7/1000 ventilator days for trauma patients, the equivalent figure being 1.6 among all ICU patients.
This difference is also evident in the developing world. The most recent INICC report from 2004–2009 demonstrates a VAP rate of 40.0/1000 ventilator days in trauma patients, with a corresponding rate of 15.8 in ICU patients overall. In support of this trend, a study in Shanghai of nine trauma ICUs, using CDC/NHSN definitions and data collection methods, demonstrated a VAP rate of 39.2/1000 ventilator days, compared to 20.8 in the ICU population overall. 13 Similar to the INICC data from developing world countries, these rates are much higher than those in the US, but the relatively higher rate observed in trauma patients is of comparable magnitude (roughly two- to threefold).
Secondly, VAP rates have fallen steadily with time. A decade ago in the US, VAP incidence density was 6.3/1000 ventilator days among all ICU patients; in 2011, the rate was 1.6. The corresponding period saw a reduction among trauma patients from 15.2/1000 ventilator days to 4.7.
Does VAP matter?
From the limited published evidence on VAP in trauma patients, there is general agreement that it increases morbidity, though conflicting evidence exists regarding its impact on mortality.
The EU-VAP (European ventilator associated pneumonia) study was conducted across nine European countries. 14 Trauma itself was associated with a higher risk for developing VAP (odds ratio 2.89, 95% confidence interval [CI] 2.26–3.69), consistent with the NHSN and INICC data examined above. Among those that developed VAP however, mortality was significantly higher in nontrauma than in trauma patients (42.6% vs. 17.2%, p < 0.001). Notably, the trauma patients were significantly younger (mean age 45 vs. 61 years, p < 0.0001) and had fewer comorbidites. A large observational study in Texas of 2591 patients similarly found that trauma patients have a higher incidence of VAP than nontrauma patients (24.3/1000 ventilator days vs. 6.1, p < 0.001), but VAP is of greater consequence in nontrauma patients (mortality 31.4% vs. 11.0%, p = 0.002). 15 The trauma patients who developed VAP remained ventilated (18.2 days vs. 4.9, p < 0.001) and in ICU for longer (28 days vs. 10.2, p < 0.001). However, these patients had no excess mortality (11.0% vs. 9.5% in trauma patients without VAP, p = 0.70).
In patients with severe traumatic brain injury (TBI), VAP was associated with significantly longer duration of mechanical ventilation (15 days vs. 8, p < 0.0001) and longer ICU (17 days vs. 9, p < 0.0001) and hospital stay (60 days vs. 28, p = 0.003), but was not associated with significantly higher mortality (31% vs. 25%, p = 0.6). 16 A case–control study of severe TBI patients with and without VAP similarly reported that VAP was associated with a longer period of ventilation (14 days vs. 10, p = 0.015) and ICU stay (21 days vs. 15.5 days, p = 0.008), but not with increased mortality (20.8% vs. 15.2%, p = 0.54). 17
However, the evidence is not unequivocal. One study did demonstrate an excess mortality for trauma patients with VAP (44% vs. 19% among those without VAP, p = 0.02). 18 It has also been reported that the combination of VAP with associated bacteremia in severely injured trauma patients increases mortality (27.7% vs. 11.2% in VAP alone, p < 0.001). 19
Predicting onset of VAP based on admission characteristics is difficult. The Texas study showed that trauma patients who develop VAP have a significantly lower Glasgow coma score at presentation (7.7 vs. 9.9 in those who did not go on to develop VAP, p < 0.001), 15 and higher injury severity score (24 vs. 17.3, respectively, p < 0.001). A study in a cohort of 322 trauma patients again suggested reduced consciousness as a risk factor for developing pneumonia. 20 However, three independent studies demonstrated no significant correlation between initial Glasgow Coma Score and the subsequent development of VAP.16–18 Elsewhere, chest trauma and pulmonary contusions have been demonstrated to be independent risk factors for developing VAP.15,21
Central line-associated blood stream infection (CLABSI)
Definition
To define a primary central line-associated blood stream infection (CLABSI), the CDC/NHSN require that a central venous catheter (CVC) was in place within the 48-h period before the criteria of a laboratory-confirmed bloodstream infection (BSI) are met: the organism cultured must not be related to infection at another site; if a recognized pathogen is cultured, only one positive blood culture is required; if a common commensal is responsible, it must be cultured in at least two blood samples drawn on separate occasions at separate sites, and the patient must display systemic signs of infection (such as pyrexia >38°C). 8 As such, CLABSI rates may be overestimates, as some BSIs may be secondary to sources other than the central line that are not easily recognized. 22
The available data
Central line-associated blood stream infection rates in intensive care units.
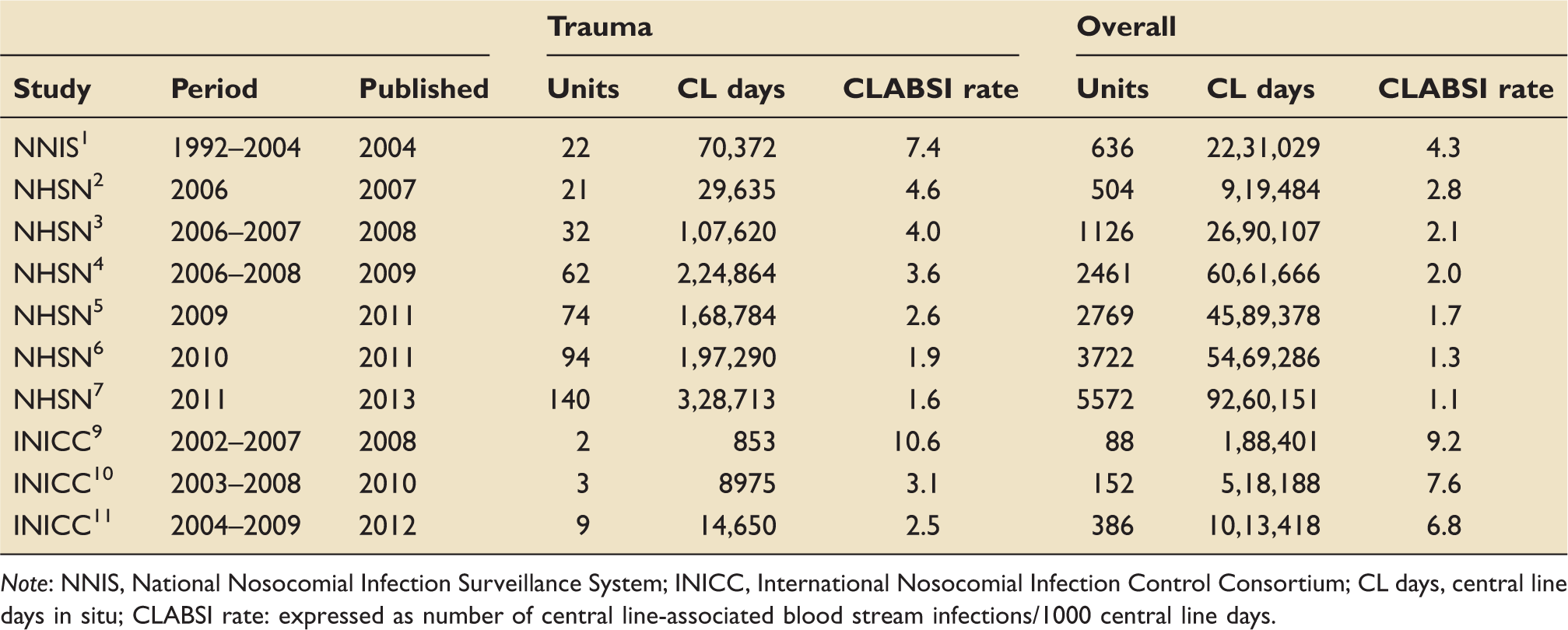
Note: NNIS, National Nosocomial Infection Surveillance System; INICC, International Nosocomial Infection Control Consortium; CL days, central line days in situ; CLABSI rate: expressed as number of central line-associated blood stream infections/1000 central line days.
Rates of CLABSI among trauma patients are consistently in the order of 1.5- to 2-fold higher than in the general ICU population. The most recent NHSN report from 2011 saw a rate of 1.6/1000 central line days in trauma patients, while the equivalent figure for all ICU patients was 1.1.
The most recent INICC data support this observation, though the differences (CLABSI rate of 6.8 among trauma patients, compared to a rate of 1.9 overall) are slightly more pronounced than that seen in the US data. Taken together with the Shanghai study, 13 which demonstrated a CLABSI rate of 3.1 among trauma patients compared to a rate of 1.1 overall, this is further evidence that while infection rates are generally higher in the developing world than in the US, they are also consistently higher in trauma patients.
With time, there has been a steady decrease in the overall rate of CLABSI among ICU patients. From an incidence density of 4.3/1000 central line days detailed in the inaugural NNIS report, the most recent figure for 2011 is 1.1. In the same time frame, the rate among trauma patients has fallen from 7.4/1000 central line days to 1.6 (Table 2).
Central line considerations
The most recent US Institute for Healthcare Improvement (IHI) guidelines clearly recommend avoiding the femoral vein, citing a body of evidence that this access point confers a higher rate of infection. 22 In practice however, the choice of CVC insertion site in trauma patients is often guided by more logistical considerations. The need for cervical spine immobilization adds further risks to cannulation of the internal jugular vein, and lower limb injury may prohibit use of the femoral vein. Meanwhile, the necessity of intercostal drain placement in certain patients with chest trauma may mitigate to some extent against the risk of pneumothorax as a complication of subclavian line insertion.
The US guidelines do not support routine CVC replacement (e.g., after seven days) to prevent catheter-related infections, in the absence of local or systemic evidence of infection. 22 They do recommend the replacement, within 48 h, of CVCs placed when aseptic technique cannot be maintained (e.g., in an emergency). A study in trauma patients, reporting a CLABSI rate of 3.3/1000 central line days showed that in 25 of 35 CLABSI cases (71%), there was a break in sterile technique during placement. 23 That said, 17 of the 25 “dirty” lines had been changed within 24 h, in an effort to reduce the risk of CLABSI, evidently without success.
Does CLABSI matter?
Little is published on the impact of CLABSI in trauma patients, but one study reports an excess length of ICU stay of eight days for trauma patients with CLABSI (24 days vs. 16 days for trauma patients without CLABSI, p = 0.0007). 23 There was no significant difference in mortality between the groups.
Catheter-associated urinary tract infection (CAUTI)
Definition
The CDC/NHSN definition of catheter-associated urinary tract infection (CAUTI) requires that a urinary catheter be in place for ≥48 h. 8 There must be presence of at least one defined symptom–sign, not attributable to any other cause, and a positive urine culture of ≥105 colony-forming units (CFUs)/ml. If there are between 103 and 105 CFU, then an additional criterion must be met (positive dipstick for leucocyte esterase and/or nitrite, pyuria, or microorganisms seen on urine Gram stain). Asymptomatic bacteremic UTI (ABUTI, by CDC/NHSN terminology) may be diagnosed in the absence of symptoms–signs, but the presence of matching uropathogen in blood culture.
The available data
Catheter-associated urinary tract infection rates in intensive care units.
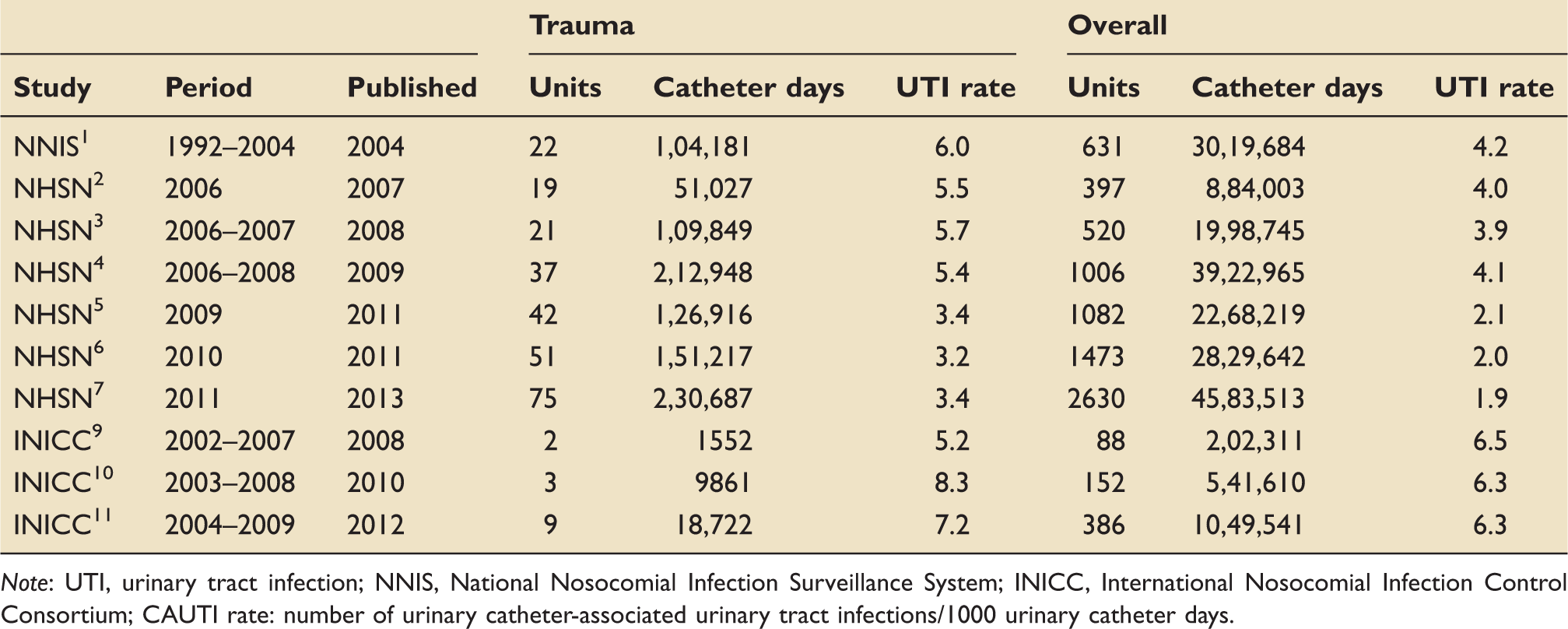
Note: UTI, urinary tract infection; NNIS, National Nosocomial Infection Surveillance System; INICC, International Nosocomial Infection Control Consortium; CAUTI rate: number of urinary catheter-associated urinary tract infections/1000 urinary catheter days.
The US data point toward a consistently higher CAUTI rate among trauma patients than the overall population, of roughly 1.5-fold magnitude. The 2011 data saw a rate of 3.4/1000 urinary catheter days among trauma patients, and a corresponding rate of 1.9 among the entire ICU population. There is some fluctuation in the rates reported in the INICC data, and no consistent difference in orders of magnitude between trauma and overall patient populations. As seen before, the rates are generally higher among the developing world population than in the US.
The reduction in CAUTI rates over time evidenced in the NHSN data is not as pronounced as with VAP and CLABSI, but is nevertheless present. Among both trauma and overall critical care populations, the rate has roughly halved since the inaugural report: in trauma patients, from a CAUTI rate of 6.0/1000 urinary catheter days to a rate of 3.4 in the 2011 data; in all patients from a rate of 4.2 to a rate of 1.9 over the same period.
Does CAUTI matter?
While there is general agreement that CAUTIs, like other nosocomial infections, are detrimental to patients, there is little data clearly delineating the relationship between CAUTI and mortality. A retrospective study in trauma patients (though not exclusively in the ICU setting), did demonstrate a higher mortality in those with UTI than those without (9.6% vs. 3.5%, p < 0.001). 24 Elsewhere, a case–control study in 12 European ICUs, though not limited to trauma patients, demonstrated no association with excess mortality (odds ratio 0.95, 95% CI 0.76–1.18, p = 0.64). 25
Conclusions
Nosocomial infection rates in intensive care have fallen in recent years, which is temporarily associated with the adoption of “bundle” strategies at prevention. The US IHI has championed this approach, and evidence for the efficacy of such strategies, though largely among general ICU populations, is unequivocal. Such strategies have been widely adopted across the world. Epidemiological data from UK ICUs is inadequate to allow comparison of nosocomial infection rates. This could be addressed by broadening the data collection remit of existing networks.
Nosocomial infection rates remain consistently higher in critically unwell trauma patients than in other ICU population (with the exception of burns patients). Whether the remaining infections represent failures of regulatory bundle application, inadequacy of surveillance definition or nonpreventable nosocomial infection is not apparent. The limited evidence available suggests these infections increase morbidity, though their effect on mortality is not clear.
It is beyond the scope of this editorial to speculate at any length as to why these differences exist, but the authors suspect a complex interplay of exogenous mechanisms (such as damage to innate physical barriers and environmental exposure to pathogenic organisms) and endogenous mechanisms (such as the immunomodulatory effects of trauma). The existing literature supports the notion that trauma-related immunosuppression occurs at the cellular level, but further research is warranted. At least, the recognition that trauma patients represent one of the most vulnerable groups for nosocomial infection should stimulate especially rigorous preventative strategies, and prompt focused efforts at early diagnosis and management.
Footnotes
Authors’ note
Institution where the work was performed: Addenbrooke’s Hospital, Cambridge.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
