Abstract

Therapeutic drug monitoring (TDM)-based dose adjustment of piperacillin/tazobactam and meropenem is associated with improved antibiotic exposure in critically ill patients with normal renal function. Level of evidence: 1B (CEBM, RCT of good quality)
Patients: Critical care patients with normal renal function who were receiving piperacillin/tazobactam and/or meropenem.
Intervention: Therapeutic drug monitoring (TDM)-based dose adjustment of piperacillin/tazobactam and meropenem.
Outcomes: The primary outcome was the percentage of time the plasma antibiotic concentration exceeded four times the minimum inhibitory concentration (MIC) of the causative organism (or if unknown, the MIC used was the epidemiological cut-off of wild-type Pseudomonas species of 16 mg/L for piperacillin/tazobactam and 2 mg/L for meropenem) during a dosing interval, over the first 72 h of treatment (% fT>4MIC). The target was 100%.
Secondary outcomes were the percentage of time the plasma antibiotic concentration exceeded the MIC during a dosing interval over the first 72 h of treatment (% fT>MIC) and absolute values for the fT>4MIC and fT>MIC. Clinical response was also evaluated at day 7. ICU, hospital and 28-day mortality were recorded.
Eligible: Patients over 18 years old, receiving piperacillin/tazobactam and/or meropenem on ICU, with an arterial catheter (for blood sampling).
Included: 41 patients from the ICU at Ghent University Hospital, Belgium between April 2011 and February 2012.
Exclusion criteria: Pregnancy, lactation, allergy to administered drugs, impaired renal function (eGFR < 80), haemoglobin <7 g/dL, do not resuscitate order or if the patient was not expected to survive 48 h.
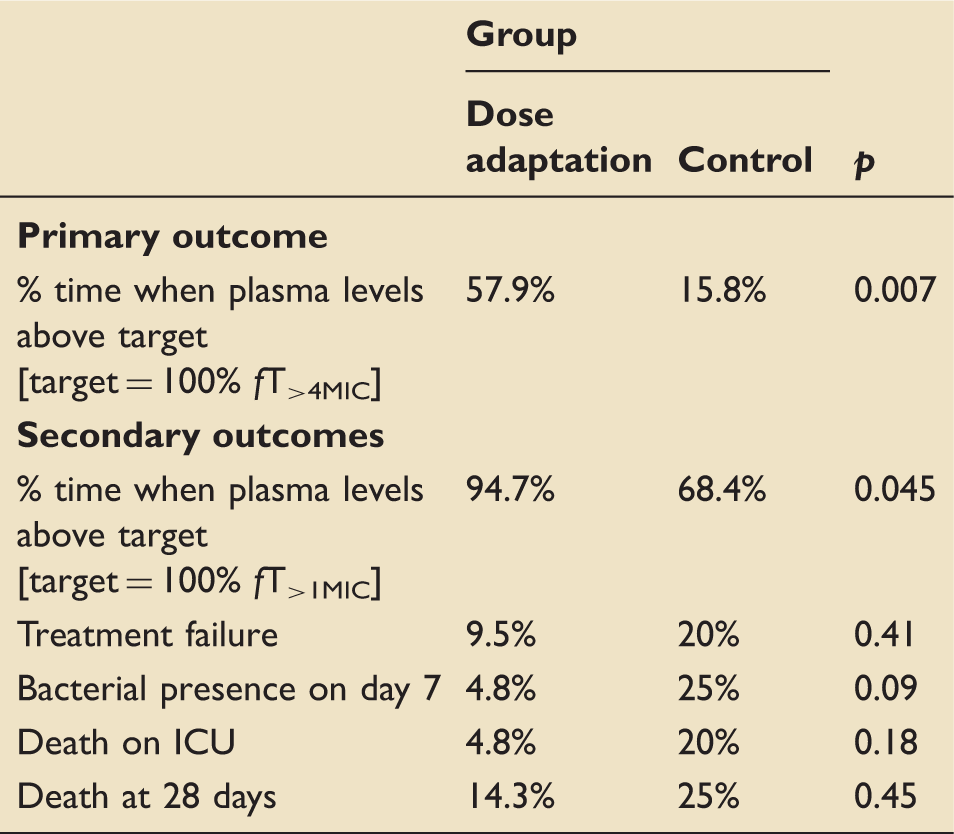
Significantly more patients achieved target antibiotic plasma levels in the intervention group. The intervention significantly increased the median time the antibiotic levels were more than four times MIC (44.5% to 86% on day 2 and 60% to 90% on day 3; p = 0.012). There were no adverse events relating to antibiotics in either group.
Do the methods allow accurate testing of the hypothesis? Do the statistical tests correctly test the results to allow differentiation of statistically significant results? Are the conclusions valid in light of the results? TDM-based dose adaptation of beta-lactam antibiotic therapy improves antibiotic exposure in critically ill patients with normal renal function. Standard dosing does not achieve target concentrations in all patients even with conservative plasma targets. Whether TDM-based dose adaptation leads to improved patient outcomes remains to be determined.
Did results get omitted and why? Three patients did not complete the study protocol and target attainment at day 3 could not be calculated for these patients. Did they suggest areas of future research?
Did they make recommendations based on the results and were they appropriate? Is the study relevant to my clinical practice? What level of evidence does this study represent?
What grade of recommendation can I make on this result alone? What grade of recommendation can I make when this study is considered along with other available evidence? Should I change my practice because of these results? Although it is likely that there are patients with normal renal function on critical care who are receiving insufficient levels of meropenem and piperacillin, TDM is not available in most hospitals and therefore wide-scale implementation is currently not possible. Should I audit my current practice because of these results?
Brendan Spooner, Core Trainee Year 3, ACCS Anaesthetics
Tony Whitehouse, Consultant Intensive Care Medicine, Intensive Care Unit, Queen Elizabeth Hospital, Birmingham, UK
