Abstract
Currently, definitive diagnosis of osteomyelitis involves a combination of clinical signs, symptoms, laboratory tests, imaging modalities and cultures from blood, joint or body fluid. Imaging plays a critical role in the osteomyelitis diagnosis. Each of these tests incurs an additional cost to the patient or healthcare system and their use varies according to the preference of the healthcare professional and the healthcare setup. Imaging plays a critical role in the diagnosis and management of postoperative long bone osteomyelitis, with the aim of reducing long-term complications such as non-union, amputation and pathological fractures. In this review, we discuss the key findings on different radiological modalities and correlate them with disease pathophysiology. Currently, magnetic resonance imaging is the best available imaging modality due to its sensitivity in detecting early signs of long bone osteomyelitis and high soft tissue resolution. Other modalities such as radio-nuclear medicine, computed tomography and ultrasound have been proved to be useful in different clinical scenarios as described in this narrative review.
Keywords
Introduction
With the increase in the number of traumatic accidents and orthopaedic surgeries performed, it is not surprising that there is a rise in the incidence of long bone infection (Calhoun et al 2009). The annual incidence of long bone osteomyelitis was 5.45 cases per 100,000 person years, age and sex-adjusted, between 1969 and 2009 (Kremers et al 2015). In elective trauma surgeries, treatment refractory acute osteomyelitis occurred 1–5% after a closed fracture and 3–50% after an open fracture, depending on the severity (Walter et al 2012). Most healthcare professionals find bone infection intimidating to treat, particularly in the postoperative setting when involving non-united fractures or metal work in situ. There are numerous complications which arise due to osteomyelitis of long bones such as non-union, amputation, pathological fracture and further spread of infection (McKee et al 2010, McNally et al 2016). Diagnosing osteomyelitis is multifaceted, where a combination of clinical examination and diagnostic assessment is required for a diagnosis. A reasonable diagnosis of osteomyelitis can be made if at least one of the four clinical signs are present (fever >38°C and local inflammation, suspicion of osteoarticular infection, reduction in mobility and joint swelling) and one of the four investigations are positive (blood culture, purulent joint fluid, culture from joint aspiration and imaging consistent with bone or joint infection) (Mitha et al 2012). Imaging plays a critical role in the diagnosis of osteomyelitis but incurs an additional cost to the patient or healthcare system and their use varies according to local/professional preference (Lee et al 2016). Some imaging modalities are not completely risk free, involving high doses of radiation and allergenic contrasts (Lin 2010). In this narrative review, we have summarised the important radiological tools which may best evaluate postoperative infection in long bones (Figure 1).
Plain radiographs
Plain radiographs should be the initial imaging for suspected postoperative long bone osteomyelitis (Lee et al 2016) as they may exclude differentials such as fractures or bone tumours. Plain radiographs can be used to look for evidence of osteomyelitis including osteopenia, bone destruction and cortical breaches followed by periosteal reactions (Lee et al 2016). However, 80% of patients who present in the first two weeks of infection will have a relatively normal radiograph (Jaramillo 2011). In chronic osteomyelitis, a focal sclerotic lesion with a lucent rim, sequestrum, is visible on plain radiographs (Lee et al 2016), potentially followed by an involucrum (Pineda et al 2009). These abnormalities are not specific to long bone osteomyelitis and can be found in stress fracture, bone tumours and soft tissue infections (Lee et al 2016). Metalwork in situ makes plain radiographs less useful for the differential diagnosis of osteomyelitis, although the state of the metalwork can be assessed. Despite the lack of specificity, plain radiographs rule out other diagnoses and are therefore the primary initial imaging modality for postoperative long bone infections.
Ultrasound scans
Ultrasound scans (USS) are useful to diagnose acute osteomyelitis, identifying associated subperiosteal collections and adjacent joint effusions or an unspecific fluid collection in soft tissue suggesting an abscess (Lee et al 2016). In a colour doppler, soft tissue oedema is seen in areas around the affected bone (Jaramillo 2011). Another common use is guiding aspiration or needle biopsy for microbiological diagnosis (Lee et al 2016). Furthermore, USS are low cost, at £39 to operate, rapidly completed and highly accessible without known side effects (NHS Improvement n.d.). Although USS are limited in diagnosing osteomyelitis itself, they may identify secondary signs of osteomyelitis and guide aspirations/biopsies (Lee et al 2016).
Computed tomography
Computed tomography (CT) tends to have a limited role in assessment of postoperative long bone osteomyelitis. CT more reliably identifies cortical destruction, periosteal reactions and sequestrum formation, which appears as sclerotic lesions with a lucent rim. CT also identifies blurring of fat planes, increased density of subcutaneous fat and swelling of affected muscles which indicate soft tissue infection surrounding the postoperative long bone osteomyelitis (Palestro et al 2007). It has a higher resolution compared to magnetic resonance imaging (MRI) in imaging the osseous changes from the osteomyelitis and therefore is useful to assess postoperative bone stock in the healing phase. Similarly, to US, CT can also detect periosteal abscesses. CT therefore plays a similar role to US in the diagnosis of osteomyelitis as well as guiding aspirations and biopsies, particularly for deeply seated abscesses in obscure locations (Lee et al 2016) where its high resolution allows differentiation between areas of infection and soft tissue structures such as major blood vessels and nerves (Lee et al 2016).
Intravenous contrasts significantly enhance CT capabilities of distinguishing in-between different tissues. For example, the centre of an abscess will not enhance with contrast due to insufficient blood supply. However, a demarcating increased brightness around the wall will be noted due to hyperaemia and increased capillary permeability within the abscess wall (Palestro et al 2007).
Although signs of long bone osteomyelitis can be detected, CT has a much lower resolution in comparison to MRI for soft tissues. CT is not able to detect bone marrow oedema, hence is not suitable in the detection of early postoperative long bone osteomyelitis (Lee et al 2016). Furthermore, the CT image is degraded by streak artefacts when metalwork is present (Pineda et al 2009); however, metal artefact reduction applications in CT are making the metal impact on imaging quality less significant. Overall, CT is a cheaper alternative for patients with contraindications to MRI, costing £69 per scan (NHS Improvement n.d.), and can be used in surgical planning of treatments of osteomyelitis. However, it has a very limited role in initial diagnosis of osteomyelitis.
Magnetic resonance imaging
(MRI) is known as the gold standard imaging modality for osteomyelitis, providing good cross-sectional views of the bone and surrounding soft tissue (Jaramillo 2011). MRI can identify osteomyelitis as early as one to two days after onset of infection (Jaramillo 2011). Normal bone cortex exhibits low-signal intensities and bone marrow has a higher T1 signal intensity (T1 images highlight fat tissues within the body, whilst T2 images highlight fat and water within the body) (Palestro et al 2007). The earliest sign of osteomyelitis on an MRI is bone marrow oedema which presents as a higher signal intensity in the medullary cavity in comparison to adjacent or contralateral healthy bones (Pugmire et al 2014).
Other common signs picked up by MRI include intraosseous, subperiosteal abscesses, phlegmon, sinus tracts and periostitis (Donovan & Schweitzer 2010, Lee et al 2016). Intraosseous and subperiosteal abscess will have low signal T1 intensities with a thin rim of intermediate intensity around the abscess after intravenous contrast administration, which is indicative of hypervascular granulation tissue (Lee et al 2016). Furthermore, if there is an intramedullary abscess, it presents very similarly to bone marrow as they both have higher signal intensity in the medullary cavity. A phlegmon is a solid inflammatory mass and can be differentiated from abscesses through increased enhancement variability across the mass and a negative penumbra sign (Lee et al 2016). This distinction has significance as abscesses require urgent intervention through aspiration or decompression (Collins et al 2005). Sinus tracts, another complication of osteomyelitis, are fluid filled extensions from the bone to the skin surface and are commonly identified through increased peripheral enhancement after intravenous contrast (Lee et al 2016). Periostitis is observed through low-signal periosteum elevated away from the bone, correlating to the periosteal reaction observed on plain radiographs (Lee et al 2016).
Intravenous contrast agents are administered to increase the enhancement of inflammation due to increased hyperaemia and capillary permeability. T1-weighted images can be used to detect these images because of the short T1 relaxation time of paramagnetic contrasts such as gadolinium-containing compounds (Palestro et al 2007). In uncomplicated cases, gadolinium is not required as the fat-suppressed T2 imaging obtains the same signal abnormalities as post-contrast T1 (Miller et al 1997). Studies have shown, gadolinium should be reserved for refractory cases due to abscess formation and suspected infection revolving around a joint (Miller et al 1997).
Unfortunately, MRI is not useful in identifying sequestra because they appear black just like normal cortical bone. Thus, they are inferior to CT when observing osseous changes of osteomyelitis. Although MRI is a sensitive test for detecting long bone osteomyelitis, it tends to overestimate the extent of infection in the acute phase when bone oedema is presented with a high-signal intensity in the medullary canal (Pugmire et al 2014). The specificity of MRI is much less than the sensitivity, due to the pathology of osteomyelitis overlapping with malignancies and trauma (Lee et al 2016, Pugmire et al 2014). Thus, the interpretation of MRI scan in osteomyelitis will be challenging and requires a radiologist with a specialist interest in musculoskeletal imaging. Furthermore, MRI imaging in postoperative patients is easily misinterpreted because postoperative changes can persist for months or years and can be misinterpreted for infection. Hence, MRI findings should always be correlated to the clinical history and examination to prevent unnecessary further investigations and treatments.
Similar to CT, MRI imaging also will be degraded by metallic implants due to the presence of metallic artefacts (Vaz et al 2017).
There are other factors which limit the usefulness of MRI such as permanent pacemakers and younger patients requiring sedation or general anaesthesia in order to undergo the scan. Also, each MRI scan costs approximately £130 and there is still a lack of accessibility in various regions (Lee et al 2016, NHS Improvement n.d.). Unfortunately, the presence of metalwork is common in postoperative long bone infection thus reducing the usefulness of MRI in the diagnosis of postoperative long bone infections, thus it is pertinent to understand other modalities of imaging which are not limited by metal artefacts such as nuclear imaging (Vaz et al 2017).
However, we are now able to use metal artefact reduction sequence (MARS) MRIs which can minimise the impact from metalwork. MARS consists of a variety of techniques to reduce metal artefacts on MRI and can be categorised into in-plane artefact reduction and through-plane artefact reduction. In-plane artefact reduction aims to reduce the effects of artefact in the image plane through simple modifications of the scan protocol such as 1.5 T magnet strength rather than 3 T, increasing bandwidth during slice selection, maintaining a good signal to noise ratio through increasing the number of excitations (Hargreaves et al 2011). Through-plane reduction involves reducing the effects of metal artefacts in an adjacent plane. The MAVRIC-SL, a combination of multiacquisition variable-resonance image combination technique (MARVIC) and slice-encoding for metal artefact correction (SEMAC), is used for through-plane artefact reduction (Choi et al 2014). Overall, MRI is the most useful diagnostic modality due to its sensitivity in detecting early signs of long bone osteomyelitis and high soft tissue resolution.
Triple-phase bone scan
Technetium-99m-labelled MDP (Tc99m-MDP) is administered intravenously, and a three-phase image is acquired consisting of the flow/perfusion, static tissue and osseous phases (Palestro et al 2007). A positive osteomyelitis sign involves presence of increased focal perfusion, hyperaemia and bone activity (Schauwecker 1992). Because Tc99m-MDP uptake is dependent on perfusion and bone formation (Galasko 1975), in osteomyelitis, increased uptake correlates to increased perfusion and invasion of microorganisms/leukocytes which form the inflammatory reaction, consequentially resulting in bone destruction (Mader et al n.d.). Although triple phase bone scans are easily performed and sensitive for postoperative long bone osteomyelitis, the positive signs are not unique to long bone osteomyelitis (Vaz et al 2017). Hyperperfusion, hyperaemia and increased bone formation are all characteristic of bony malignancies, trauma, aspect inflammation, heterotopic ossification and any other cause of accelerated new bone formation (Vaz et al 2017). Furthermore, Triple phase bone scans are the most expensive scans costing £198 per scan (NHS Improvement n.d.). Additionally, triple-phase scans are difficult to interpret and can be ambiguous, thus further imaging may be necessary to obtain a definitive answer.
Gallium scintigraphy
Gallium scans can be used in conjunction with a triple-phase bone scan to detect postoperative long bone infection. Studies have demonstrated that triple-phase bone scan accuracy ranged from 50 to 70% in infected long bones, whilst Gallium used in conjunction with triple-phase bone scans only marginally increased the accuracy to 65–80% (Love et al 2009). The majority of the circulating gallium is bound to transferrin, thus increased blood perfusion results in increased gallium at the inflammation site (Palestro et al 2007). Bacteria uptake the gallium directly and through siderophores (Palestro et al 2007). The gallium is seen on leukocytes through phagocytosis of bacteria from macrophages and gallium directly binding to the leukocytes (Palestro et al 2007). Gallium scintigraphy is useful in patients with a low white cell count as it can detect osteomyelitis in immunocompromised patients (Palestro 1994). The current literature suggests that gallium scintigraphy may be more useful in spinal osteomyelitis and may not be indicated in postoperative long bone infections (Love et al 2009, Termaat et al 2005). Limitations of gallium scintigraphy include the large radiation exposure and requirement of multiple imaging sessions, potentially taking up to 48–72 h (Christian et al 2007). Similar to triple-phase bone scans, uptake is dependent on inflammation which is not exclusive to long bone osteomyelitis.
Radio-labelled leukocytes (RLL)
RLL is an in vitro labelling technique utilising 111In‐oxine and 99mTc‐exametazime to label the patient’s leukocytes, most commonly neutrophils (Palestro et al 2007). RLL measures radiotracer accumulation in the reticuloendothelial system. In order to differentiate between bone marrow and leukocytes, the white cell scan is combined with Tc99m-labelled colloid scan, which maps out the bone marrow (Lee et al 2016). Any disparity between the white cell scan and the bone marrow scan is indicative of infection. RLL scans combined with bone marrow scans are the most effective in detecting infection in long bones with an accuracy of approximately 90% across various studies (Magnuson et al 1988, Pring et al 1986, Rand & Brown 1990). However, as the RLL are most commonly neutrophils, the infections diagnosed may be limited to bacterial. For opportunistic infection such as mycobacterium or yeast, RLL may not be sensitive enough; therefore, gallium scintigraphy may be preferred in this scenario. A minimum total white count of 2000mL − 1 is necessary to achieve adequate sensitivity (Palestro et al 2007). Furthermore, there have been reports of two fatalities from cardiopulmonary failure shortly after administration of the RLL; however, the precise reasoning behind the fatalities is yet to be clarified (Magnuson et al 1988). Another drawback associated with the RLL scan is that it requires skilled personnel to perform the procedure and it is not readily available. Although RLL scans have the highest accuracy, it seems that there are quite a few limitations which have yet to be resolved.
Positron emission tomography (PET)
Fluorine-18-fluorodeoxyglucose positron emission tomography (FDG-PET) is used to localise inflammation. In inflammation and infection, FDG uptake is thought to be raised due to a combination of increased expression of glucose transporters on activated inflammatory cells and a cytokine-mediated increase in affinity for deoxyglucose from glucose transporters (Mochizuki et al 2001, Paik et al 2004). Although malignancies and infection present similarly on FDG-PET, it is particularly useful in localising the abnormality when it is difficult to determine on other imaging such as MRI (Wang et al 2011). Indeed, Van Vielt et al demonstrated that FDG-PET is a promising tool in discriminating between aseptic and septic delayed union in the lower extremities (van Vliet et al 2018). Furthermore, FDG-PET was proven to be useful in postoperative patients in distinguishing postoperative infection from postoperative bone healing (Koort et al 2004), whilst Lankinen et al showed that FDG may be effective in confirming low-grade bone infections in culture negative patients (Lankinen et al 2017). Overall, FDG-PET proves to be a promising prospect in the diagnosis of long bone infections in postoperative patients.
SPECT-CT
SPECT-CT is useful in imaging osteomyelitis through combining anatomical and functional aspects which allows for more precise localisation of infection foci (Thang et al 2014). In some cases of bone infection with adjacent soft tissue involvement, planar images are not able to distinguish soft tissue from bone infection and SPECT-CT is able to more precisely define the extent of infection. A study by Filippi and Schillaci found that SPECT-CT correctly characterised and localised site uptake in all suspected osteomyelitis patients and discriminated between adjacent soft tissue involvement and bone involvement (Filippi & Schillaci 2006). SPECT-CT was particularly useful in patients with structural bone abnormalities after traumatic injury (Filippi & Schillaci 2006). After trauma, the structural abnormalities persist and this makes it difficult for CT or plain radiograph to detect infection because morphological imaging may not detect infection in structurally abnormal bone (Seabold et al 1989, Tumeh et al 1987). Holger et al found that SPECT-CT has improved specificity in comparison to SPECT alone when diagnosis chronic osteomyelitis (89% vs. 78%, respectively) (Horger et al 2003). Van den Wyngaert et al found that SPECT-CT could be a highly cost-saving imaging modalities in comparison to MRI for postoperative patients with recurrent and persistent lower limb pain (Van den Wyngaert et al 2018). Furthermore, each SPECT-CT is £118 which is £12 cheaper than MRI (NHS Improvement n.d.). SPECT-CT seems to be useful in diagnosing post-traumatic and postoperative long bone osteomyelitis patients with structurally abnormal bone, thus could be considered as an alternative to MRI.
Conclusion
Radiological evaluation plays a critical role in the diagnosis and management of postoperative long bone osteomyelitis. Plain radiographs should be first line to exclude other differentials such as fractures. CT and USS can allow detect the osseous changes which can aid with aspiration and biopsy. MRI should be next and is the most useful imaging to establish postoperative long bone osteomyelitis as it can detect early signs such as bone marrow oedema. With the addition of MARS for CT and MRI, their relative utility will be increased in patients with metalwork. If MRI is contraindicated, CT is an alternative. If the CT and MRI imaging is degraded by metal artefacts, nuclear medicine scans such as triple phase bone scans, SPECT-CT, PET and gallium scintigraphy can be used to localise the inflammation. SPECT-CT shows promise through its cost-effectiveness and should be used in post-traumatic patients with structurally abnormal bone. PET can be indicated to distinguish between postoperative infections and postoperative healing. Gallium scintigraphy may be indicated in immunosuppressed patients with low leukocytes counts which are sufficient for radio-labelled leukocytes scans. Finally, it is up to the treating healthcare professional to choose what may best suit depending on the healthcare system.
Key phrases
Incidence of long bone infections is rising. Definitive diagnosis of long bone osteomyelitis involves a combination of clinical signs, laboratory tests and imaging. MRI is currently the best available imaging modality. Alternative imaging modalities may be preferable in specific clinical scenarios. Imaging modalities are not completely risk free for the patient.
No competing interests declared
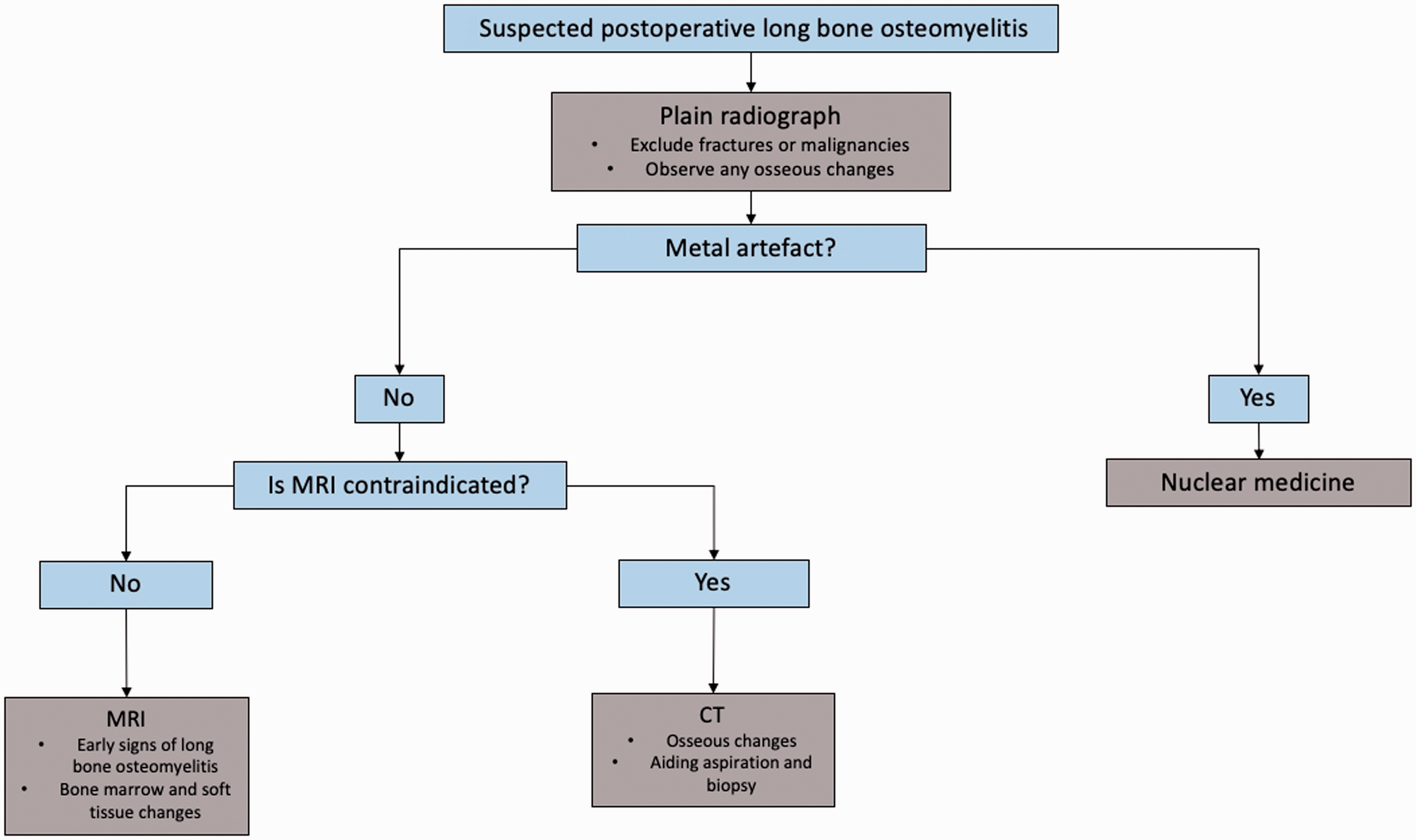
A flowchart guiding the choice of imaging in long bone osteomyelitis
Footnotes
ORCID iDs
Andrew Kailin Zhou https://orcid.org/0000-0001-6656-8123 Milind Girish https://orcid.org/0000-0003-0599-5265 Jiang An Lim https://orcid.org/0000-0003-1610-7956 Xiaoyu Chen ![]()
