Abstract
Retinal image sharpness assessment is one of the critical requirements of automatic quality evaluation in telemedicine screening for diabetic retinopathy. In this paper, a new sharpness metric measuring the spread of edges is presented to quantify fundus image clarity. After edge detection on the region of interest of retinal image, the width of each edge is calculated and the histogram of region of interest generated. Based on the histogram, a distance-based factor is introduced to gain the weighted edge width, which is defined as the sharpness metric for the fundus image. The method was tested on Messidor dataset and a proprietary dataset. The results show that the proposed metric performs well over different image distortion levels and resolutions and is of low computational complexity. The weighted edge width value of gradable retinal image, which is irrelevant to resolution, is always within the range of 3–7 pixels.
Introduction
According to the latest report from the International Diabetes Federation, there are about 415 million adults with diabetes worldwide. 1 Over half of these individuals will develop diabetic retinopathy (DR), which is one of the most common, serious complications of diabetes. 2 During the early phases of disease, there may be no obvious obstacle on the vision, so patients always realize too late to get timely examination or treatment for preventing blindness. As the number of patients with diabetes increasing, DR has become a leading cause of blindness in adults globally. 1
Studies have showed that arteries, veins, and capillaries of human body can be directly observed through fundus noninvasively. 3 Hence, taking DR screening regularly is necessary to achieve early diagnosis and get the effective curing time. 4 Digital fundus photography is used, which enables a noninvasive examination and allows image preservation and transmission for later use. 5
However, due to the imbalance of the professional medical resources, the patients’ compliance of the screening is not satisfied. For example, no more than 50% patients with diabetes participate in screening in time in the developed countries, 6 while as high as 82.71% of patients in China lack awareness about regular fundus examination. 6 In order to provide quality care to patients at any location, many domestic and foreign experts are committed to telemedicine programs addressing DR.7,8 The available achievements have proved that telemedicine is more effective to identify and determine DR severity than conventional clinic-based ophthalmoscopy does and will help preventing severe vision loss. 7
Unfortunately, it is a common phenomenon that images with insufficient quality are collected. Jian-jun et al. 9 pointed out that in the case of nonmydriatic retinal images, 3–30% were unsuitable for retinopathy grading. In other studies, the proportion of single field nonmydriatic (no pupil dilation) images with unsatisfied quality has been reported at 1110 and 20.8%, 11 respectively. In telemedicine, receiving an ungradable fundus image may lead to escaping diagnosis or being misdiagnosed. It is too much trouble for patients to return to the clinic for retaking images. 12 Besides, every examination and image is critically priceless record of that individual in longitudinal studies. Losing information for a given period may reduce the statistical power of the results. Hence, assessing fundus image quality automatically becomes the first key step before reliable lesion detection, especially in the telemedicine systems of delivering eye care. 10
According to Fleming et al.,
11
retinal image quality should include two aspects: clarity and field definition. In this paper, we focus on the former problem. Lesions may be disguised in blurred images, therefore a diseased eye could not be timely point out. Defocus blurring of fundus image may be caused by poor camera focus, cataract, or macular edema. And poor patient fixation or saccadic eye motion during acquisition may lead to motion blurring. Some retinal images of different qualities are shown in Figure 1.
(a) Qualified fundus image, (b) motion-blurred fundus image, and (c) defocus-blurred fundus image.
The clarity of the image can be seen as a function of edge strength. 13 Strength of edges in blurred or low-contrast images is always weak. Edge information can be used to evaluate fundus image sharpness. Thus, in this paper, we develop a sharpness metric for automatic quality evaluation of retinal image based on the edge width.
The rest parts of paper are structured as follows: In the next section, an overview of previous work on sharpness evaluation for fundus images is presented. “Proposed sharpness metric for fundus images” section describes the proposed sharpness metric based on histogram of edge width. Experiment results are shown in the subsequent section, and conclusion of our work comes in the final section.
Previous work on fundus image sharpness assessment
The automatic assessment of retinal image sharpness has gained more and more attention. 10 There exist two groups of methods for solving the task 12 : (i) approaches based on structure segmentation and (ii) sharpness metric.
In methods falling in category (i), image clarity evaluation requires the identification of anatomical landmarks, such as detecting vessels and calculating their total length or destiny. In 2003, the first method for clarity assessment based on eye structure criteria was proposed by Usher et al. 14 The area of segmented vessels over the entire image was used. Then, like Lowell et al., 15 Fleming et al. 11 and Hunter et al. 16 judged image sharpness based on the structure of the blood vessels just within an area around the macula. More recently, based on the segmented vessel map, Yin et al. 17 put forward high level image quality measures including blood vessel density and the maximum edge length or edge spread, while in Welikala et al., 18 authors calculated area, fragmentation, and complexity as the global features.
In those cases where segmentation can be reliably done, the eye structure markers will be very helpful for the later use in DR screening. However, it is always complex and computational to obtain the major anatomical structures. Especially, the failure of the segmentation is not trivial when images are of poor quality. 12 Therefore, these methods are not so suitable for telemedicine programs addressing DR.
On the other hand, (ii) sharpness metrics can provide a continuous measure in an unsupervised manner for the clarity prediction. In 1999, edge intensity histograms were first utilized by Lee and Wang 19 to describe image sharpness. But as Lalonde et al. 20 states, “the link between intensity histogram similarity and image quality is not strong,” the results did not satisfied. In 2009, Bartling et al. 21 calculated the degree of sharpness from the spatial frequencies of the image. In the following time, Davis et al. 22 provided a real-time retinal image quality assessment procedure, where reblurred methods were introduced as a no-reference image sharpness metric. The same metric was employed in study of Pires Dias et al. 12 and of Yin et al. 17
Commonly, blurring will lead to edge diffusion, so evaluation of retinal image sharpness can be made by the spread of the edge. Average edge width has been used to measure image blurriness by Marziliano et al. 23 Based on it, the cumulative probability of blur detection (CPBD), 24 a new local sharpness metric came. Then, CPBD value was calculated on only the green channel to characterize the strength of edges for retinal images in Yu et al. 25
Using sharpness metrics for fundus image quality assessment is simplicity. 12 Reduced computational complexity makes them amenable to use in DR screening. However, metrics mentioned above may be too sensitive to determine whether the retinal image is gradable in DR screening. Retinal images with slight blur are always acceptable for manual grade. For example, there must be a large range between CPBD values of blurred versions of a retinal image and the value of the normal one. But all of them may be acceptable during screening. Therefore, those sharpness metrics are not so suitable for sharpness assessment in DR screening.
As has been said, for more practical, we will pose a fundus image sharpness metric by getting weighted edge width (WEW) based on the histogram.
Proposed sharpness metric for fundus images
For evaluating blur of fundus images, we attempt to measure the spread of the edges. The computation of the proposed sharpness metric is summarized in Figure 2.
Flow chart of the proposed sharpness metric.
Preprocessing
In retinal images, major edges arise from vasculature, optic disk, and lesions. 13 According to statistics, vasculature and red lesions are always invisible in red planes of retinal photographs while there are little useful information but noise can be obtained from the blue planes. 26 Since each part of fundus and types of lesions can be distinguished in green planes well, only the green plane is used in our study.
Like images in Figure 1, the field including all fundus information we concerned is a circle in the fundus image. The black area beyond the circle does not contain any information. Besides, boundary of the field of view will have an adverse effect upon the following analysis. Hence, the inscribed quadrilateral of the fundus field containing major information is select as the region of interest (ROI). The next steps are conducted on the ROI of retinal image.
Edge width histogram computation
Firstly, Sobel operator is applied to achieve both horizontal and vertical edge detection. By applying a threshold to the gradient image, noises are removed.
Based on the edge map, width of each edge location is calculated. According to Lowell et al., 15 the number of pixels with increasing grayscale values in one direction as well as the number of pixels with decreasing values in the other side should be counted. The width around the edge is obtained by computing the sum of both counts except the edge point itself.
Figure 3 showing a row of the image illustrates an example of width measurement. For a detected vertical edge pixel P, local maximum Ps is defined as the start position, while local minimum Pe corresponds to the end location. Edge width of P is |Pe − Ps | = 5. When it comes to width calculation of horizontal edges, the process can easily be extended by scanning each column.
One row of a fundus image.
Edges may have different widths. The probability
Figure 4 shows an example of the edge width histogram computation of ROI.
Edge width histogram computation. (a) Retinal image green plane, (b) ROI, (c) edge detection and (d) edge width histogram. ROI: region of interest.
Sharpness metric WEW
The average of the edge widths
Histogram of edge width with different blurriness is shown in Figure 5. It demonstrates that, with the increasing blur, the histogram becomes diffusional, and the peak moves right.
Histograms of edge width for retinal images with different blurriness. (a) Original image, (b) image filtered by a circular averaging filter with radius = 3, (c) image filtered by a circular averaging filter with radius = 9, (d) image filtered by a circular averaging filter with radius = 15, (e)–(h) are edge width histograms of (a)–(d).
Considering that blurring of invalid edges or noise may lead to too large or too small edge width, a distance-based weight is introduced in order to modify equation (2). The weight
Therefore, weights from equation (3) are introduced into equation (2) resulting in a WEW, defined as our sharpness metric
The sharper the fundus image is, the lower the metric value takes.
Experiments and results
In this section, the performance of proposed sharpness metric for fundus images is presented and discussed. A MATLAB algorithm for WEW calculation was implemented on a laptop with 2 GHZ Intel Core I7 and 4 GB of RAM. To evaluate the proposed method, both motion- and defocus-blurred retinal images were used. The test sets taken from publicly available databases and proprietary dataset are first described. Then, experimental results are shown and analyzed.
Test sets
In order to develop and test the retinal image sharpness metric, publicly available retinal image dataset Messidor 27 was used. It takes 1200 gradable retinal images, all of which have a 45° FOV and resolutions of 2304 by 1536 pixels, 2240 by 1488 pixels, or 1444 by 960 pixels.
Besides, a proprietary dataset consisting of 30 retinal images from the first affiliated Hospital of Fujian Medical University were also adopted. Images from the dataset have a resolution of 3072 by 2048 pixels. With the help of three ophthalmologists from the first affiliated Hospital of Fujian Medical University, we defined 26 retinal images as satisfied images while the other four are unqualified.
Experiments on Messidor database
We chose some fundus images of size The image was filtered with a motion filter with five different lengths The image was filtered with a circular averaging filter with different radius
Sharpness metric was calculated for each test image. It is found that a strong linear relationship is consistent for all the test images. Figure 6 shows the behavior of our sharpness metric Behavior of sharpness metric W EW for a fundus image (a) sharpness measurement versus length of the motion filter and (b) sharpness measurement versus radius of the circle averaging filter. WEW: weighted edge width.
We also calculated CPBD metric on each test image. The CPBD values of test images used in Figure 6(a) are presented in Figure 7. For a more intuitive observation, CPBD is used instead of 1-CPBD in the graph.
Behavior of CPBD metric of blurred versions with different lengths of the motion filter. CPBD: cumulative probability of blur detection.
It is obvious that CPBD value changes sharply when a retinal image is disturbed. However, all of these motion-blurred images are acceptable in DR screening. They are qualified enough for ophthalmologists to grade. Hence,
To corroborate our metrics,
Figure 8 illustrates the result of (a) WEW metric for the 2240 × 1488 fundus images in Base11 subset, (b) WEW metric for the 1440 × 960 fundus images in Base22 subset, and (c) WEW metric for the 2304 × 1536 fundus images in Base33 subset. WEW: weighted edge width.
Performance on proprietary dataset
Here we extended our experiments to the proprietary dataset. WEW metric was calculated for each fundus image which was marked by manual in advance. The results are shown in Figure 9. Metric values of most images are around 5 pixels. However, one blurred image takes low WEW metric value while the value of another acceptable image is beyond 7 pixels. The total AUC is 93.3% when images with value range in 3–7 were marked as “qualified.”
WEW metric for the 3072 × 2048 fundus images from proprietary dataset. WEW: weighted edge width.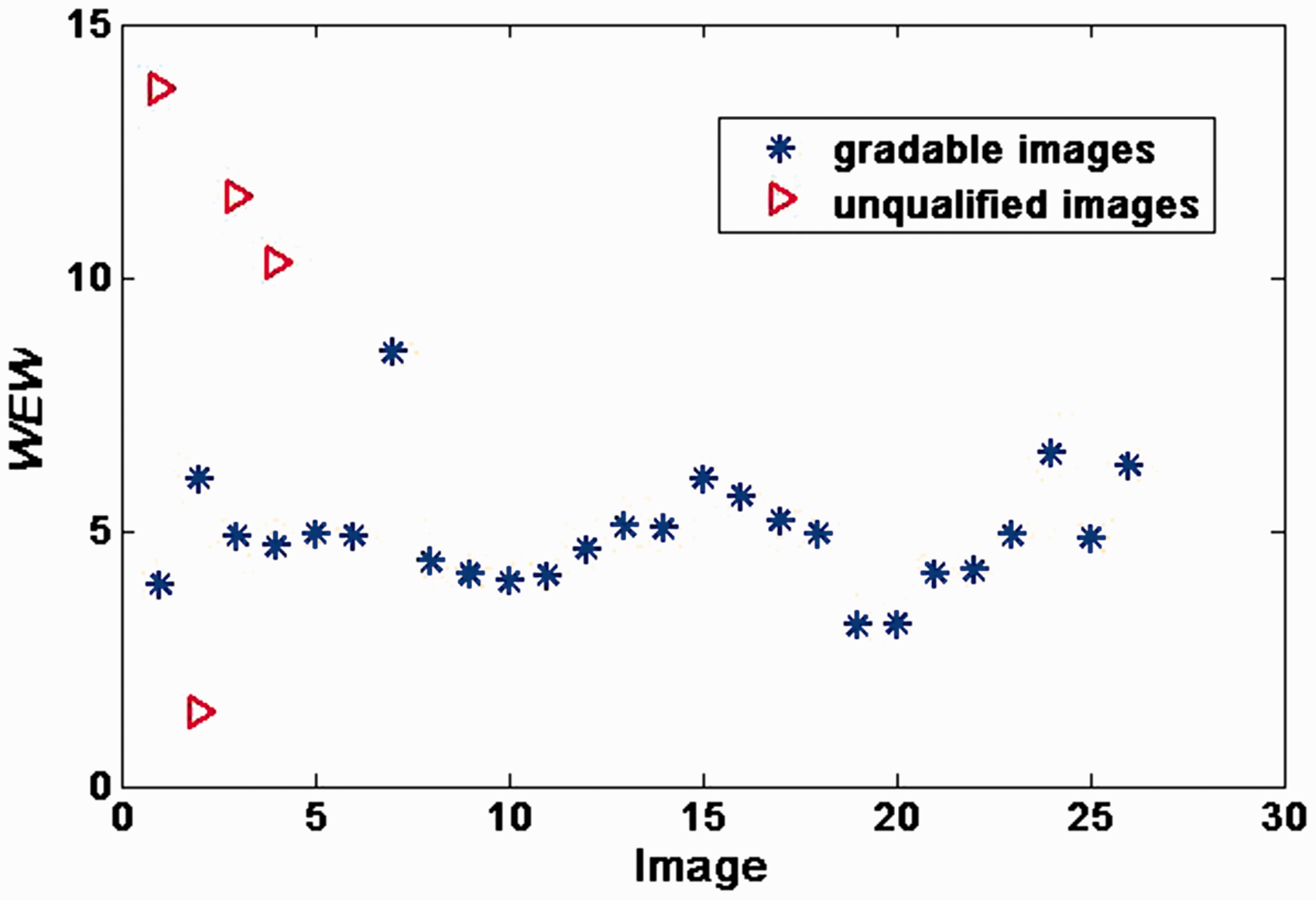
As shown in the Figures 8 and 9, there are still some gradable or blurred images with unexpected values. The main cause of error is mainly due to the problem of reliable determination of edge. When blur is increasing, the total number of edges extracted by Sobel operator goes down, which reduces the probability of edge with various widths. In that case, a serious blurred image with very low WEW value will be mistaken for the clear picture. With the help of an advanced edge operator, the performance will be improved.
Besides, some pictures should have been acceptable are defined useless. Low contrast, uneven illumination or artifacts affect the metric. A proper preprocessing would certainly make it more robust.
Although the method has several disadvantages as mentioned above, low complexity was one of our prime objectives in applying this metric.
Computation time comparison
Comparison of average computation time in different scales.

CPBD: cumulative probability of blur detection; WEW: weighted edge width.
Conclusion
In this paper, we develop a retinal image sharpness metric based on edge width histogram. The WEW metric is of low computational complexity and in good agreement with ophthalmologists’ sharpness assessment. The metric for a gradable retinal image is in the range of 3–7 pixels. According to the satisfied AUC and low complexity, it has the potential to be an efficient feature in quality evaluation for DR screening application.
Future research will focus on the measurement of field definition of the fundus image and the image quality evaluation process for DR screening.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Natural Science Foundation of China (No. 61671152, No. 60827002), by the Natural Science Foundation of Fujian Province, China (No.2016J0129), by the Educational Commission of Fujian Province of China (No. JAT160070).




