Abstract
Microfungi have appeared as one of the most effective and eco-friendly catalysers for derivatizing bio-active molecules in green chemistry. The use of Eurotium cristatum has been established as a method of post-harvest handling and value addition for tea products, especially in enhancing their health-protective properties. From our study, a consortium of E. cristatum was selected and introduced as inoculum on Camellia chrysantha leaves substrate, yielding transformed products with chemo-preventive and antidiabetic potential. Biological activity-guided fractionation incorporated with chromatographic separation of the fermented products gave out compound
Inoculation of Camellia chrysantha leaves with characterized Eurotium cristatum consortium led to the formation of fermented product that exhibited inhibition against cancerous cells and α-glucosidase. Triterpene compound 22-O-tigloyloxycamelliagenin A was isolated from the fermented leaves with enhanced cytotoxic, lipase inhibitory and α-glucosidase inhibitory properties.
Introduction
Microbial fermentation is one of the oldest procedures for preserving agricultural products and producing beverages from organic materials. With enzyme activities to modify natural substrates and further release novel molecules, it has recently been considered a cost-effective catalysing method in environmentally friendly material transformation. The use of specific microorganisms, that is, lactic acid bacteria and filamentous fungi, was found as key factors in yielding characteristic aroma and taste of well-known fermented tea products, such as Yunnan pu’er, Toyama kurocha and Tteokcha dark teas. 1 Particular microbials of Aspergillus, Bacillus, and Penicillium were proved to play a role in supporting valuable medicinal properties for tea products, including reducing blood lipids and providing antioxidative effects. 2 – 4 One of the most significant fungus for processing teas is Eurotium cristatum (Eurotiales, Asperillaceae, syn. Aspergillus cristatus) [https://www.mycobank.org/]. 5 – 7 To date, E. cristatum has extensively been utilized in herbal fermentation of miscellaneous plant taxa, such as Ginkgo biloba, Docynia indica, Eucommia ulmoides and Acanthopanax senticosus, to facilitate value-added products. 8 A comparison of chemical compositions between non-fermented and fermented Cyclocarya paliurus leaves revealed 67 differential metabolites, including 31 terpene compounds. 9 Interestingly, while the flavonoids amount increased by 25.66%, the total contents of polyphenols and triterpenoids decreased by 53.04% and 27.91%, respectively, after the fermentation. 9 The components of herbal leaves fermented by E. cristatum were supposed to undergo structural modifications, correlating with changes in the taste, aroma, and bioactivities of the products. 8
Section Chrysantha (Ericales, Theaceae) of Camellia comprises more than 24 species with unique yellow flowers and was popularly regarded as ‘golden camellias’. 10 Plants of this section attracted attention due to their unique importance in either floriculture or the production of valuable bio-active compounds. 11 In the last decade, the number of publications on the first discovered golden camellia plant Camellia chrysantha has significantly increased, mostly regarding the scientific validity, the medicinal and commercial importance of its flowers. Recently, vitexin, aromadendrin, quercetin, kaempferol, and phenolic acid were reported as components of C. chrysantha’s leaves. 12 Impressively, they were claimed to play a supportive role in controlling cellular activities and protecting cells from harmful free radicals. 10 Another phytochemical investigation showed four triterpenoids of β-amyrin hexanoate, friedelin, friedelanol, and β-amyrin and a steroid chondrillasterol as components of C. chrysantha’s leaves. 13 In addition, 17 types of amino acids from its leaves with averaged amounts ranging from 3.73 to 5.93 g/100 g were recorded. 14 In general, the chemical composition and pharmaceutical potential of C. chrysantha’s leaves have been thoroughly provided, laying the basis for further studies of their development as beneficial bio-transformed products.
In geographical areas with natural distribution of C. chrysantha, systematic studies to preserve and sustainably exploit the plant treasure are recommended. As parts of commercial activities, C. chrysantha is presently cultivated for limited seasonal harvest of the flowers, while the leaves are abundant in all seasons but generally unfavoured for utilization. Methods for improving the tea leaves’ quality to take advantage of this resource are thus required. In these circumstances, microbial fermentation for manufacturing tea beverage is among the most conceivable approaches to be considered. Our study aimed to employ E. cristatum strains in the transformation of golden C. chrysantha’ leaves to yield fermented tea with enhanced biological properties, as well as to characterize the most biologically active component from the tea product. For this purpose, extracts and fractions from the fermented tea leaves were evaluated for their in vitro chemo-preventive and antidiabetic potential on cancerous cell growth and α-glucosidase, respectively. Isolated component from the fermented tea leaves was additionally assessed for its inhibitory activity against pancreatic lipase, the key lipid-digesting enzyme in the intestine.
Materials and methods
Materials
Fungal strains
Six fungal strains of Eurotium spp. were used in this study, inherited from the laboratory of experimental biology – Institute of Natural Products Chemistry. Three of these were isolated from commercial Anhua dark tea (Hunan Provincial Baishaxi Tea Industry Co., China), while other strains were isolated from plants (Sonneratia sp., Zingiber officinale).
Camellia leaves collection and pre-processing
Fresh leaves of C. chrysantha were collected from tea plantations in Ba Che, Quang Ninh (21°19′34.2″N 107°06′07.7″E), taxonomical considered, 15 washed and air dried. The leaf samples are stored at the laboratory of experimental biology – Institute of Chemistry.
Chemicals, reagents and apparatus
Ellipticine, Acarbose, Orlistat, 4-Nitrophenyl α-D-glucopyranoside (pNPG), Sulforhodamine B (SRB) and α-glucosidase were purchased from Sigma-Aldrich Co. (St Louis, MO, USA). Methanol and dimethyl sulfoxide (DMSO) with a purity of over 98% were purchased from Xilong Scientific Co., Ltd (Guangdong, China).
Cell lines
Two human cancer cell lines and a normal cell line were provided by the Department of Experimental Biology–Institute of Natural Products Chemistry, including MCF7 (Human breast adenocarcinoma, ATCC no. HTB-22), LU-1 (Human lung adenocarcinoma, ATCC no. HTB-57) and Vero cells (Chlorocebus aethiops kidney epithelial cells, ATCC no. CCL-81.4).
Methods
Identification of fungal strains
Morphological identification
For macromorphology observations, including colonies’ pigmentation, size and texture, fungal strains were grown on different growth media, including Dichloran Glycerol Agar (DG-18), Czapek’s Agar (CZA) and Sabouraud Dextrose Agar (SDA) plates, at 25°C in 7 days. The fungal specimens were examined by microscope for determination of hyphal and reproductive structures as previously described by Klich. 16
Molecular identification
Fungal genomic DNAs were extracted from biomass using CTAB method. 17 Primer pair ITS1 (5′-TCC GTA GGT GAA CCT GCG G-3′) and ITS4 (5′-TCC TCC GCT TAT TGA TAT GC-3′) was employed for amplification of the internal transcribed spacer (ITS) region from isolated fungal DNAs. 18 PCR was performed with a 25-μL reaction mixture consisting of the following (per reaction): 4 µL of template, 1 µL of each primer, 2 µL of 10 mM dNTPs, 0.25 µL of Taq polymerase, 5 µL of 10X Taq polymerase buffer, 1.5 µL of 50 mM MgCl2 and 10.25 µL of deionized water. After an initial denaturation at 94°C for 3 mins, thirty cycles of amplification were performed. Each cycle composed of a denaturation step at 94°C for 1 min, an annealing step at 55°C for 1 min, and an extension at 72°C for 2 min. PCR products were purified and subsequently sequenced by Perkin Elmer ABI Prism 377 DNA Sequencer (USA). By Basic Local Alignment Search Tool (BLAST) in the National Centre for Biotechnology Information (NCBI) database, identified regions of high similarity to ITS fungal sequences were determined. The phylogenetic tree was constructed using a neighbour-joining method with 1000 bootstrap values available in MEGA 7.0 software. 19
Physiological and biochemical identification
Certain characteristics of isolated strains, including optimal temperatures, pHs and salinities for growth, were identified through experimental procedures. The production of enzymes by fungal strains was determined by measurement of halo size on agar plates containing specific substrates. 20
Preparation of fermented C. chrysantha leaves
Air-dried tea leaves were subjected to an enzyme-inactivation process (121°C, 20 min) by which microbial spores and infusion factors on the surface of tea leaves were eliminated.
Selected strains of Eurotium spp. were activated by cultivation in Sabouraud Dextrose broth (25°C, 3 days) before inoculating into C. chrysantha leaves.
The fermentation process was performed in 500 mL Erlenmeyer flasks for both non-inoculated (NF) and microbial inoculated fermentation (IF). In brief, 100 grammes of pre-processed (121°C for 20 min) raw tea leaves were adjusted to moisture level of 60% to generate C. chrysantha tea infusion. The infusion was inoculated with either 10 mL sterile water or the same volume of fungal suspension (5 × 106 CFU/mL) to produce portions of NF and IF, respectively. The whole fermentation process was taken for 21 days at 25°C and protected from light. Each fermentation mode (NF/IF) was repeated three times for consistency and accuracy determination.
Extraction and chemical isolation
Fermented leaves of C. chrysantha (3 kg) were extracted thrice (each 10 L, 3 days) with ethanol at 50°C using a conventional ultrasound-assisted technique. The solvent was then in vacuo evaporated (EYELA, Japan) to yield ethanol extract (600 g). The ethanol extract residue was suspended in water (2.0 L) and successively partitioned with n-hexane and ethyl acetate (EtOAc) to obtain n-hexane (
Ethyl acetate extract
Structure elucidation
The nuclear magnetic resonance (NMR) spectroscopy, including 1H NMR (600 MHz) and 13C NMR (150 MHz), COSY, HMQC and HMBC of isolated compound were measured on Bruker spectrometer (Germany) at the Institute of Chemistry. Thin layer chromatography (TLC) was performed on pre-coated silica gel DC-Alufolien 60 F254 (Merck), combined with an ultraviolet lamp (UV) at 254 nm and 365 nm, 10% aqueous H2SO4 solution and heating.
Assessment of in vitro cytotoxicity
The cytotoxicity of extracts and compounds was determined by SRB colorimetric method as described by Skehan et al. 21 Briefly, dissolved test samples in DMSO were diluted and incubated with cells (seeded at 0.8 × 104 cells per well) in 96-well microtiter plates (37°C, 5% CO2, 48 h). Cultures were then fixed with trichloroacetic acid and stained by 0.4% (w/v) SRB. The protein-bound dye was extracted with 10 mM Tris-base for the determination of optical density (OD) at a single wavelength of 540 nm in a 96-well plate reader (Tecan F150 microplate reader, Switzerland).
Assessment of inhibitory effects against lipase and alpha-glucosidase
The lipase and α-glucosidase inhibitory activities of extracts were evaluated as mentioned in our previous publication 22 following Lewis and Liu 23 and Tadera et al. 24 with minor modifications.
The absorbance of released p-nitrophenol was measured at 405 nm using a spectrometer system (Tecan F150 microplate reader, Switzerland). Acarbose was included as a standard in α-glucosidase inhibition assay, while orlistat was employed as a positive reference in lipase inhibitory assay. Each experiment was performed in triplicates. IC50 values of test samples were calculated by interpolation from linear regression analysis.
Results
Fungal characterization
In order to select E. cristatum candidates for effective fermenting C. chrysantha leaves, six strains from our microbial collection (E1, E2, E3, E6, E7, and E8) were characterized. These strains were found to exhibit great identities to ITS sequences of published Aspergillus and Eurotium species and typical morphological characteristics of E. cristatum on agar plates. The characterization of fungal candidates in detail was presented in Supplementary file 1.
A screening process of in vitro antioxidative and antidiabetic activities, as well as safety test in Balb/c mice (data not shown) suggested E. cristatum strains E1, E3 and E8 as potential bio-active inoculums. These characterized fungi were then chosen to inoculate in C. chrysantha leaves. Crude extracts yielding from the fermentation of C. chrysantha tea leaves by E. cristatum strains E1, E3 and E8 either as monocultures or associated as a consortium of three strains, were symbolized as IF-E1, IF-E3, IF-E8 and IF-LM, respectively. The biological activities of these extracts were assayed and compared to their non-fermented counterpart (NF) to determine fermenting efficiency.
Biological activities of crude extracts from E. cristatum fermented tea leaves
Cytotoxicity
The effect of tea leaves extracts on the viability of cells was in vitro assessed both on adenocarcinoma cell lines LU-1 and MCF-7 and normal cells (Vero cells). Results depicted in Table 1 indicated cytotoxic effects on cancer cells of fermented extracts (IF-E1, IF-E3, IF-E8 and IF-LM) in comparison to unfermented NF. Remarkably, while NF exhibited no obvious change in survival rates of tested cells, the application of E. cristatum fungal strains, especially as a fungal consortium (IF-LM) seemed to yield extracts with elevated cytotoxic effects against both cancer cell lines.
Cytotoxicity of tea leaves’ extracts in cancer cell lines LU-1 and MCF-7.
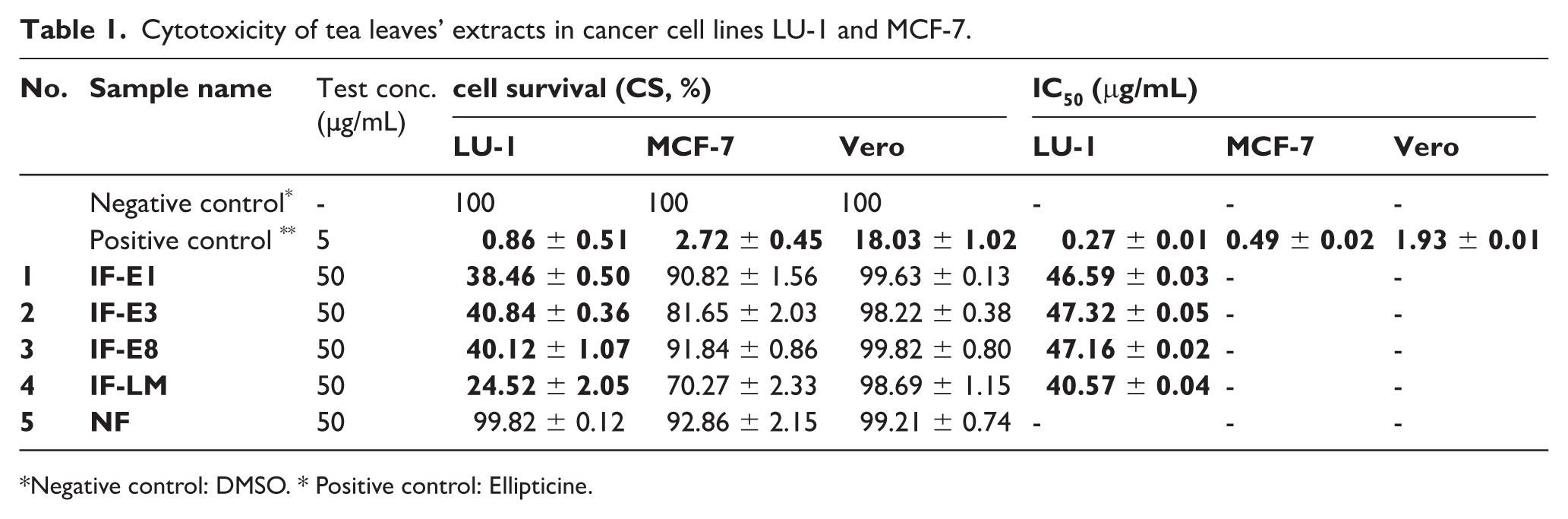
Negative control: DMSO. * Positive control: Ellipticine.
Alpha-glucosidase inhibitory activity
The inhibitory activity of ethanolic crude extracts from unfermented (NF) and fermented tea leaves by E. cristatum strains (IF-E1, IF-E3, IF-E8 and IF-LM) against α-glucosidase was investigated and compared. The results were shown in Table 2.
Inhibitory activities of tea leaves’ extracts against α-glucosidase.
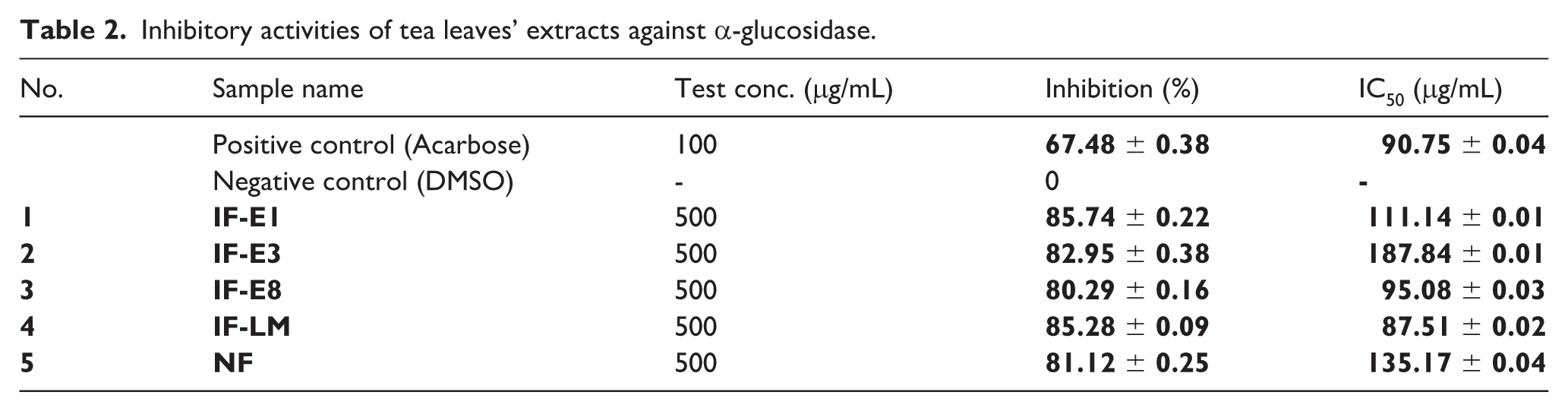
It was observed that not only unfermented tea leaves’ extract (NF) but also microbial fermented leaves’ extracts exhibited inhibition against α-glucosidase, with inhibitory rates ranging from 80.29% ± 0.16% to 85.74% ± 0.22% at the test concentration of 500 μg/mL. Among them, IF-LM extract showed an enhanced inhibitory activity with IC50 at 87.51 ± 0.02 μg/mL, indicating the advantage of using microbial consortium over applying single fungal strain in the fermentation process.
Determined biological activities of extracts (Tables 1 and 2) demonstrated significant differences between non-fermented and fermented leaves and suggested the use of the fungal consortium composed of E. cristatum E1, E3 and E8 in enhancing biological activities. Further steps in our study were undertaken to characterize the most bio-active components from fermented tea leaves when applying the fungal consortium by stepwise bioactivities guided chemical extraction, fractionation and isolation.
Biological activities of extracts and fractions from fermented tea leaves
For selecting effective portion in fermented tea leaves’ extracts, n-hexane (
Biological activities of n-hexane and ethyl acetate extracts from C. chrysantha fermented leaves.

Results indicated ethyl acetate as the appropriated solvent for extraction of active fractions from fermented golden tea leaves. Further chemical fractionation of
Biological activities of fractions from extract THV-LM/E of fermented tea leaves.

Among three investigated fractions,
Chemical elucidation of THV-LM03
Compound
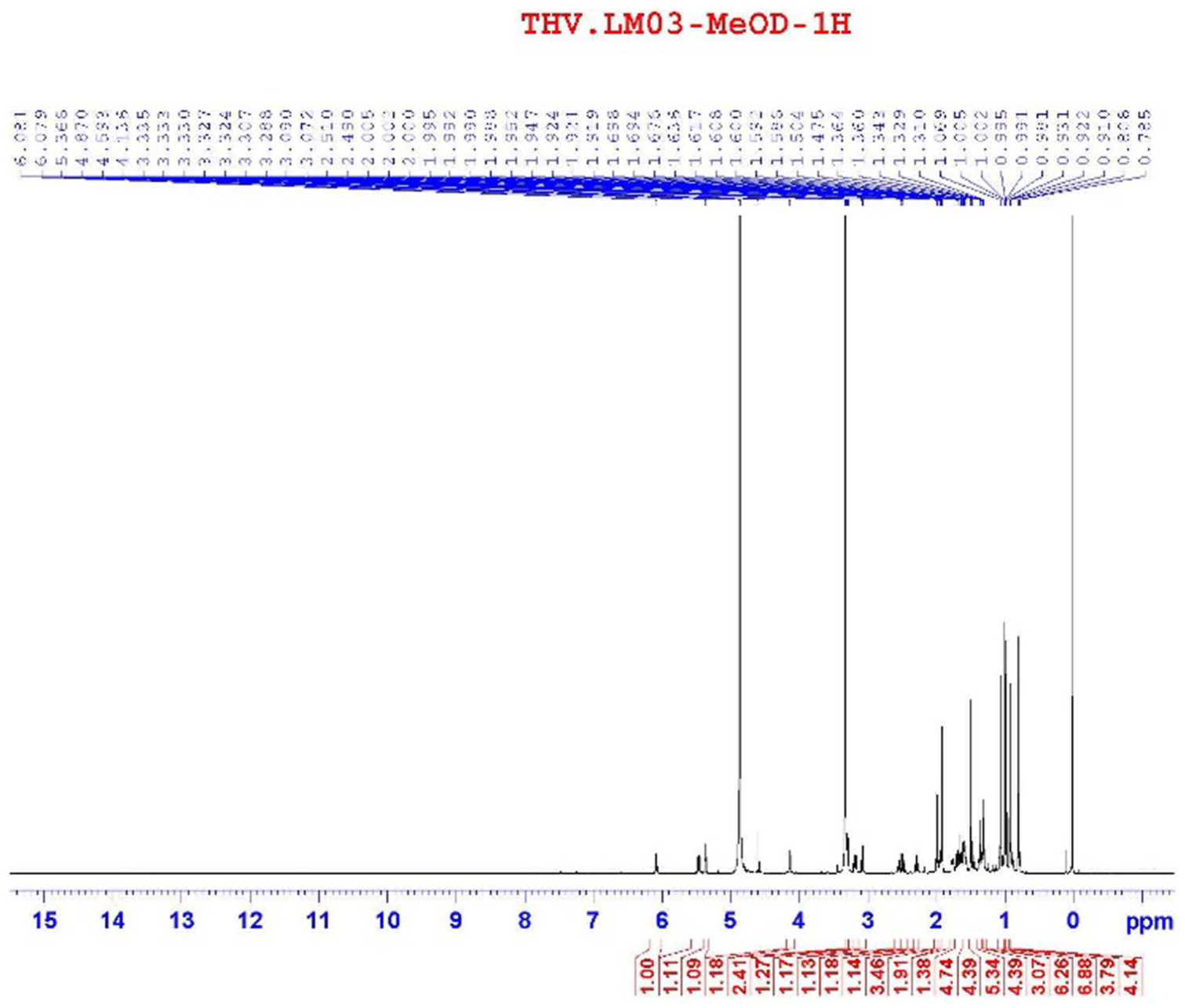
1H-NMR spectrum of compound
The 13C-NMR and HSQC spectroscopic data of compound

13C-NMR spectrum of compound

HSQC spectrum of compound
Correlation signals between δH 5.45 (1H, dd, J = 6.0/12.0 Hz, H-22) and δC 169.75 (C-1′) on HMBC confirmed the attachment of tigloyl group at C-22 position. Similarly, correlation signals between δH 3.08 (1H, m, CH2-28a)/3.29 (1H, m, CH2-28b) and δC 45.32 (C-17) indicated an attachment of hydroxymethyl group at C-17 position (Figure 4).
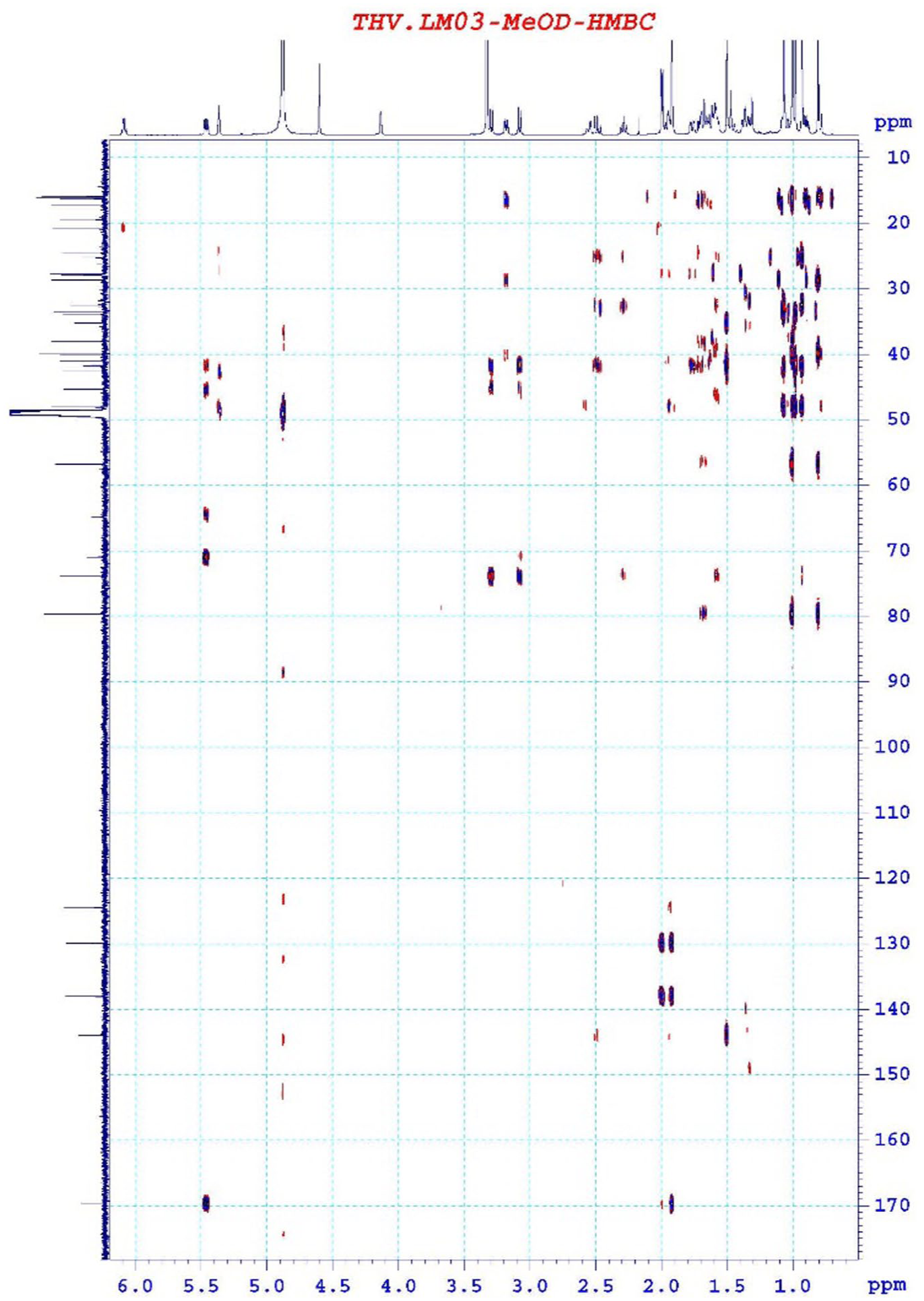
HMBC spectrum of compound
From these spectral data (Table 5) and comparison to those in literature,
25
compound
1H-NMR and 13C-NMR data of 22-O-Tigloyloxycamelliagenin A and compound

Recored in CD3OD, b150 MHz, c600 MHz. δC#: 22-O-Tigloyloxycamelliagenin A (in CDCl3).
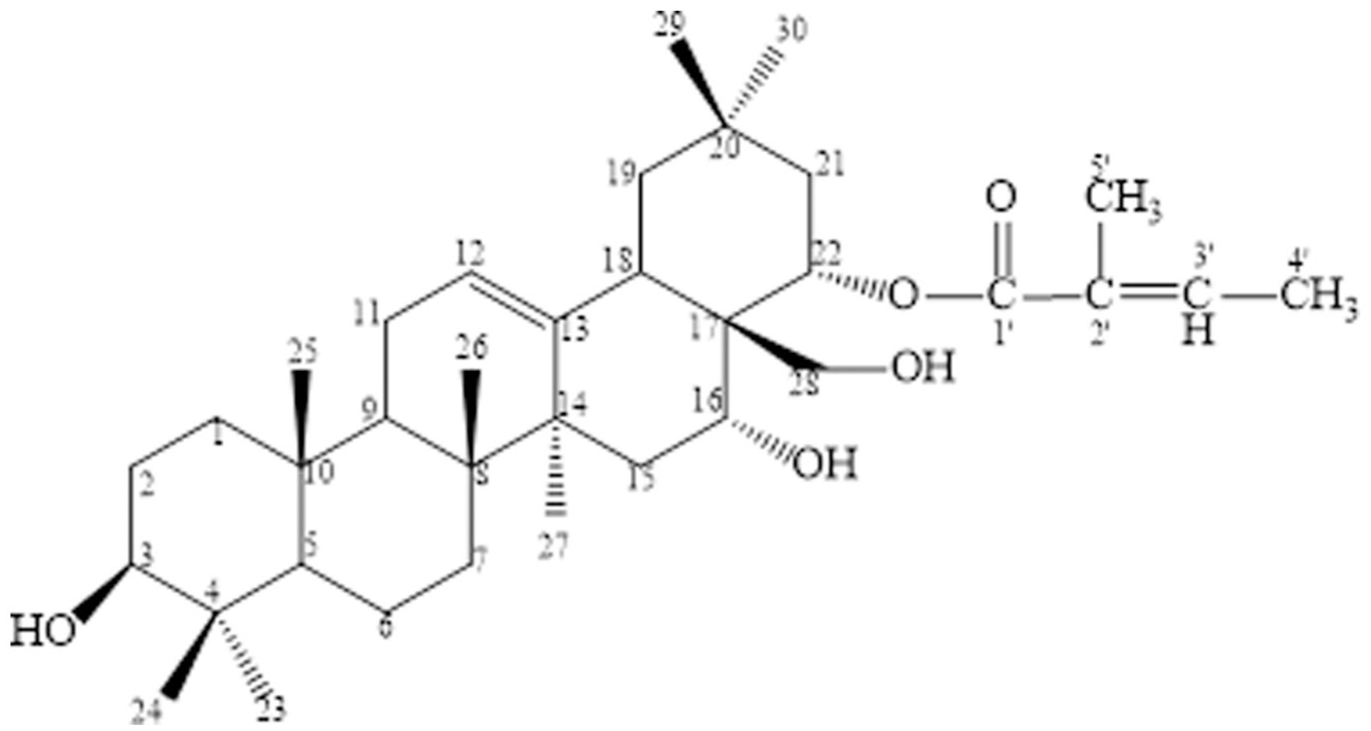
Chemical structure of
Biological activities of compound THV-LM03
Studies on the chemical composition based on biological activity of fermented leaves of C. chrysantha resulted in the isolation of compound 22-O-tigloyloxycamelliagenin A
Biological activities of compound

Notwithstanding previous results concerning cytotoxic property of 22-O-tigloyloxycamelliagenin A,
25
the inhibition against cancer cells of LU-1 and MCF-7 is a new contributing data for its profile. Most importantly, our present study revealed 22-O-tigloyloxycamelliagenin A as a product of fungal biotransformation on tea leaves which exhibited diabetes and obesity associated enzymes inhibition. The IC50 value (~29.33 µM) of
The results indicate the potential of
For the first time, the compound was discovered as a potential pharmaceutical ingredient for treating cancer and weight management. However, further experiments and trials are required to develop biomedical preparations from the transformed terpenoid.
Discussion
Hypothetical novel fungi-driven biotransformation pathway of triterpenoids
Previously, 22-O-tigloyloxycamelliagenin A has been reported as a constituent from the bark of Barringtonia asiatica.
25
Our present result brought out the first isolation of such triterpene as a plant leaves derived compound. Interestingly, this compound has not been isolated from either leaves of C. chrysantha or E. cristatum fungi. Possibly, the fungi transformed triterpenes on tea leaves substrate and led to the production of novel compounds, including 22-O-tigloyloxycamelliagenin A (
In Camellia sinensis, Xiao et al.
26
reported a significant increase of aroma quality and terpene alcohols during E. cristatum driven fermentation, indicating the fungal involvement in the metabolism of terpenoids. Among four previously isolated triterpenoids from C. chrysantha leaves,
13
β-amyrin shared the most similar skeleton to
Possibly, during the process, two major transformations occurred, allowing the insertion of hydroxyl groups and stepwise tigloyl group at β-amyrin’s specific sites to yield the corresponding derivative
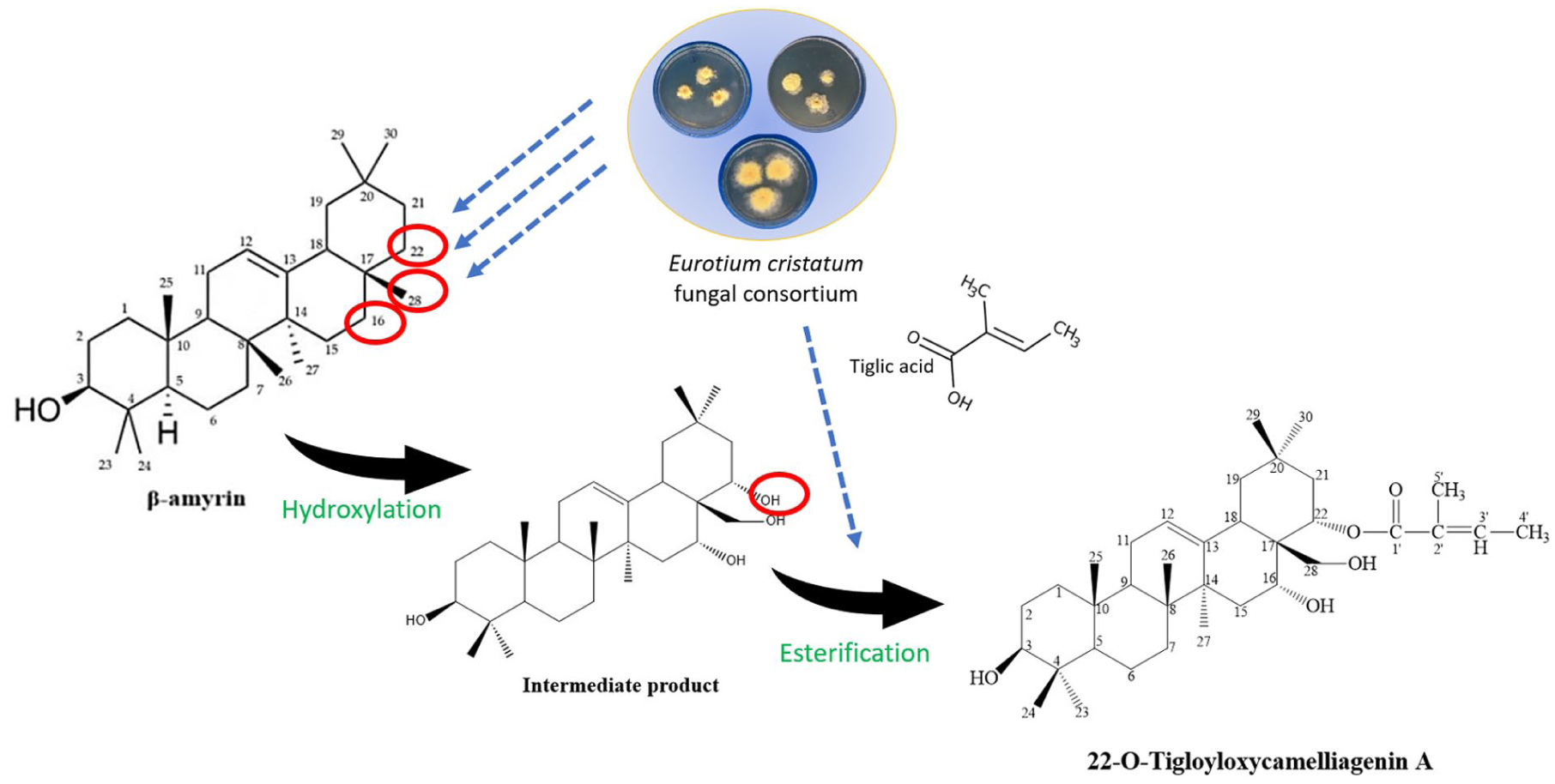
Hypothetical biotransformation of β-amyrin to
The formation of
For determining accurate parameters of novel triterpenoid biosynthesis using the microbial consortium, further studies on optimizing fermentation conditions in herbal substrates, that is, C. chrysantha leaves, are required. As proposed in our hypothetical conversion pathway (Figure 6), β-amyrin and 22-O-tigloyloxycamelliagenin A could serve as chemical signatures for the determination as well as evaluation of fermentation efficiency in extended optimization processes.
An effective microbial consortium for enhancing tea leaves’ value
In the past decades, microbial transformation has been demonstrated to be a versatile tool to enlarge the structural diversity of triterpenoids. For the purpose, the selection of appropriate organisms serving for a desired modification of particular substrates played a key role. 31 Our results shed a light on the use of an E. cristatum fungal consortium for specific hydroxylation and esterification of triterpenes to more bio-active products. In detail, characterized E. cristatum strains E1, E3 and E8 of different origins showed mostly similar bioactivities when inoculated in C. chrysantha leaves’ substrate as monocultures, but even an enhanced effect when fused as a consortium (Table 3, Table 4). As a fungal consortium, the efficiency of catalytic engineering might be more advantageous due to the potential activation of silent metabolic pathways, which are normally not expressed under monoculture condition. 32
Our study lays foundation for the development of fermented tea beverage production using E. cristatum fungi in the form of a consortium. On one hand, the existence of E. cristatum in dark teas might play a role as probiotics for modulation of gut microbiota, 6 on the other hand, the fungal consortium could help improve the beneficial ingredients’ bio-synthesis efficiency in the value-added products.
Conclusion
In our work, three fungal strains of E. cristatum were characterized and successfully inoculated in C. chrysantha leaves. In terms of biological assessments, the fermentation by a fungal consortium including these strains was found with advantages over monocultural and uninoculated counterparts. Further in vitro evaluation results indicated enhanced effects in inhibiting cancer cells and against α-glucosidase of ethyl acetate extract over n-hexane extract from the microbial fermented tea leaves. The biological activities guided chemical isolation of C. chrysantha leaves’ ethyl acetate extract gave out 22-O-tigloyloxycamelliagenin A as the most active ingredient. The compound was found as a potential α-glucosidase inhibitor, α-lipase inhibitor and cytotoxic triterpene against cancer cells and thus worthy for further studies on application aspects. Furthermore, a hypothetical two-step transformation of 22-O-tigloyloxycamelliagenin A from plant triterpenoid component under the catalysis of E. cristatum’s enzymes was proposed, supporting for more comprehensive understandings about the conversion of triterpenoids during microbial fermentation of herbal substrates.
In conclusion, this study brought out the realization of E. cristatum fungal strains as an effective consortium in the fermentation of C. chrysantha leaves and an expanded understanding of us about the biotransformation of triterpenoids in organic materials in general. These might provide a neoteric option for processing golden camellias’ leaves, especially for promoting value-added products with enhanced health benefits. Equally important, our study contributed to establish an environmentally friendly synthesis method of bio-active molecules which is cost-effective with reducing energy use and chemical waste in the future.
Supplemental Material
sj-docx-1-chl-10.1177_17475198261421696 – Supplemental material for The fermentation of Camellia chrysantha leaves with Eurotium cristatum to yield biologically active compound
Supplemental material, sj-docx-1-chl-10.1177_17475198261421696 for The fermentation of Camellia chrysantha leaves with Eurotium cristatum to yield biologically active compound by Hang TN Tran, Quan T Nguyen, Van TH Nguyen, Tuyen T Tran, Bach C Pham, Cuong H Le, Nghi H Do, Ha TH Tran and Chi K Hoang in Journal of Chemical Research
Footnotes
Acknowledgements
We would like to thank Dr Luu Van Chinh and other co-workers for helpful discussion and technical works.
Ethical considerations
The submitted manuscript contains original and new results which are not submitted for publishing to other publications or published elsewhere.
All research was conducted to the highest possible ethical standards.
All authors are involved in the work. Authors and co-authors reviewed and ensured the accuracy and validity of all the results prior to submission.
All authors guarantee that the research findings have not been previously published.
Consent to participate
Not applicable.
Consent for publication
Not applicable.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Vietnam Academy of Science and Technology [grant number THTE00.10/24-25].
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability statement
Data supporting this study are included within the article and supporting materials.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
