Abstract
Gout occurs when high uric acid (UA) levels accumulate in certain joints, primarily in the big toe. When this accumulation surpasses the saturation concentration, reaching a supersaturated state, a UA-based sodium salt precipitates, forming structures of one or more sharp crystals that damage the soft collagen tissue surrounding them, producing intense pain that incapacitates the patient from moving the affected zone. When large amounts of UA salt accumulate in a localized region of the body, they precipitate, forming crystalline structures that cause gout; this process is called crystallization. The opposite process, dissolution, dissolves UA crystals when the local UA concentration decreases below the saturation concentration, reducing the gout’s effects. These processes can be characterized using several techniques, including dynamic light scattering (DLS), X-ray diffraction (XRD), and piezoelectric analysis (PEA). The PEA is sensitive to the molecular structure of piezoelectric materials; then, it can be used to determine the status of gout disease. Then, this technique can provide an early, painless, local, and noninvasive way to detect gout. The diagnosis can be obtained by measuring the intensity of piezoelectric signals in the affected region

Introduction
Gout disease is a type of arthritis characterized by an inflammatory process in the region where uric acid (UA) accumulates at high concentration, leading to the formation of sharp crystals. 1 These crystals produce acute and intense pain as they severely damage nerves, veins, and soft tissue. This disease happens when UA-based sodium salt accumulates, under supersaturated conditions, mainly in the big toe: at this high concentration, this salt precipitates, forming rhomboidal needle-like crystals; 2 these sharp crystals hurst (cut) the surrounding soft tissue, resulting in very intense pain that incapacitates the patient from practically any mechanical movement; even though the affected region is small (big toe) with respect to the whole body, 3 the pain is so intense that prevents any movement of the patient. 4 The affected tissue presents the symptoms: inflammation, arthritis, and redness.3,5,6
Due to these characteristics, the treatment involves the use of non-steroidal anti-inflammatory drugs (NSAIDs) like ibuprofen or naproxen. To reduce swelling, use ice packs, rest, keep legs elevated, lose weight, limit alcohol, and maintain an appropriate diet. These recommendations could be considered “home remedies.” However, it is also worth mentioning other innovative techniques for treating this disease, such as the use of biosensors. These treatments for gout measure uric acid levels in the blood or other body fluids. These devices include a biological recognition element and a transducer. The biological recognition element is an enzyme, uricase, that detects the presence of uric acid, while the transducer converts this information into a measurable signal. This technique enables quick, precise, and portable monitoring, aiding in diagnosing and managing gout. There are various types of biosensors for gout, including electrochemical biosensors, which may include a carbon nanotube matrix with immobilized uricase to detect high concentrations of uric acid with high sensitivity. A portable device consisting of integrated biosensors, developed into mouthguards, measures UA levels in saliva in real time. Nanodevices, which are highly sensitive and designed to analyze blood faster and with smaller sample volumes. Biosensors for gout detection are fast and accurate, enabling quicker and more precise detection of UA levels and helping to diagnose and treat gout early. A recent advancement in imaging for gout is dual-energy computed tomography (DECT). It is a noninvasive method for visualizing, characterizing, and quantifying monosodium urate deposits. This technique helps clinicians with early diagnosis, treatment, and follow-up of this condition.
It has been found that many people with high UA levels do not develop gout;7–9 there are some reasons for this: (a) typical UA measurements are an average over the entire body and not in a specific region of it: UA is distributed throughout the body and may not reach supersaturation concentration in a specific region of the body; (b) the collagen molecules produces a blocking effect, preventing the deposit of UA molecules, and reducing the formation of large crystals structures; and (c) excess water adsorbed in the body can partially dissolve some crystals in the affected region reducing their effects. In any case, it is important that the body properly eliminates UA-based compounds through the appropriate organs, as they accumulate and can cause disease.
Uric acid and urea
UA and urea are different nitrogen-based compounds that produce toxic effects of different intensities when retained and accumulated in high concentrations over long periods in the body.10–13 These compounds are by-products of protein metabolism and must be excreted through feces, urine, or sweat to keep the body healthy. In a healthy person, urea appears in a wide range of concentrations with minimal problems because it is water-soluble, and any excess is excreted in the urine. UA has low solubility in water, so it must be present in the body at lower concentrations, meaning it must be excreted through urine to maintain its level at a minimum. The problem occurs at high UA levels because the patient needs to excrete it quickly to reach safe limits.
UA and urea have different chemical structures, with different water solubilities and toxicities, affecting the human body differently. Toxic compounds are produced during the proteins’ metabolism, and these must be converted into a less toxic product until it is removed from the body; this vital task is carried out by one of the two excretory systems: one is the kidney, urinary, and fecal system, while the second one is the skin, which excretes some toxic substances through sweat. 14 Of the nitrogen-based products excreted by humans, urea is the main one because it appears in large amounts with respect to UA and ammonia; 15 however, small amounts of UA can be excreted in the urine. 16 Birds excreted UA in a solid form as fecal matter.17–19
Urea is widely used for some medical applications, such as dermatological treatments, skin rehydration, nail debridement, earwax removal, psoriasis, xerosis, onychomycosis, ichthyosis, eczema, keratosis, keratoderma, and corns. 20 UA is a weak organic acid formed by two rings (pyrimidine and imidazole), with the formula C5H4N4O3;8,21 UA-based sodium salt is the precursor to monosodium urate, a compound that precipitates into needle-shaped crystals responsible for gout (Figure 1). 22 The body produces UA during the metabolism of some compounds called purines, 8 which are a broad class of nitrogen-based compounds frequently found in nature. This explains the presence of UA in the body: UA is produced in small quantities by the body during the metabolic processes of some proteins, or it is introduced into the body during the intake of certain types of foods and drinks; 23 high content of purines is found in internal organs such as the liver, kidney, heart, and brain, etc. 23

The hydration process of uric acid and the formation of monosodium urate.
Gout
Gout is produced by the formation of UA crystals during the precipitation of supersaturated concentrations of monosodium urate (Figure 1). When UA is in contact with water, three carbonyls (=C=O) are hydrated and transformed into hydroxyls (–OH), and three amines (=NH) are transformed into nitrogen; in a second stage, one hydrogen of the hydroxyl is substituted by Na (–OH → –O–Na) producing the monosodium urate. 22 This salt, in the presence of water, is solvated, giving rise to the ionic form of UA; then, UA will be used to denote uric acid and/or its ionic form. Monosodium urate precipitates within joints and soft tissues, producing needle-like rhomboidal crystals that cut and tear soft tissues, triggering a severe inflammatory response. UA has a low solubility limit concentration in water of CS = 0.06 mg/mL. 24 When this limit is exceeded, the UA concentration reaches supersaturated conditions, and the solute precipitates, forming the crystals alone or in clusters. These crystals grow inside the soft tissue, producing intense pain. 25 The reduced solubility of UA in water causes precipitation at relatively low concentrations and, consequently, the early appearance of crystals. In normal conditions, the kidney eliminates UA from the body in small quantities through the urine; however, at high UA concentrations, the kidney cannot eliminate it, and these high UA concentrations in the body cause diseases such as kidney stones or other types of kidney damage, being the most common disease gout; UA concentration inside the body can be reduced by reducing the intake of certain foods or by medicine. Small amounts of UA can be dissolved in the blood and travel throughout the body, reaching the kidney, where it is excreted in the urine; at a pH less than 5.8, typical of urine, UA is excreted in its nonionic form. A blood or urine test can determine the average concentration of uric acid in the body.
UA crystallization, dynamic light scattering, X-ray diffraction, and piezoelectric analysis
The UA crystallization processes start with a UA solution at a specific concentration and temperature; a solvation process is the first necessary step to obtain this solution. A stable solution with the maximum possible amount of solute is called saturated; if this solution contains less solute, it is unsaturated, and if it contains more solute, it is supersaturated. The solubility of a substance is expressed as the saturated concentration. The typical method for preparing a supersaturated solution is by stirring and/or increasing the temperature, because the solubility of most solids can be increased by raising the temperature; once a saturated solution is prepared at a high temperature, a supersaturated solution can be obtained when the system is cooled down.
Because supersaturated solutions are metastable, the excess solute precipitates, usually very quickly, forming crystals: the UA molecules approach each other, collide, and adhere, forming crystals. As crystals form, the UA concentration decreases, eventually reaching a saturated or unsaturated state; once this condition is reached, the solute remains in equilibrium with the solvent, and precipitation stops. The amount of water relative to UA determines whether crystals form or dissolve. Thus, the amount of water in the body can help control the formation of UA crystals in gout. Water adsorption is important for patients with gout because water can reduce the crystal size through dissolution. Water is transported throughout the body through the interstitial spaces, which are connected. Even though precipitation and crystallization processes describe the separation of a solute from a solution, in crystallization, the solid has a well-defined crystalline structure, whereas in precipitation, the solid forms without a particular structure.
Experimental
Sample preparation
UA crystals (Aldrich), hydrolyzed Collagen (Sense) with active peptides of 150 to 200 nm, and distilled water were used to prepare all samples. The samples were prepared by mixing these powders, because it is not possible to obtain real samples of gout at different stages of UA crystal growth in a soft collagen matrix; however, collagen and HAp are the main constituents of the big toe, the place where gout is most frequently found. Particle size was determined by dynamic light scattering (DLS), and structural changes were measured using piezoelectric analysis (PEA). Each sample contained 0.2 g of collagen with 100 mL of water. To get the crystallization kinetics, five aqueous solutions were prepared at different UA concentrations: three unsaturated (C < CS) C1 = 0.01 mg/mL, C2 = 0.02 mg/mL, C3 = 0.03 mg/mL, one saturated C6 = CS = 0.06 mg/mL, and one blank C0 = 0.00 mg/mL (no UA) with 0.2 g collagen. Collagen in water suffers a swelling, increasing its particle size.
Separately, to obtain the crystallization and dissolution profiles, two different samples were prepared using UA crystals: one below the saturation (C4 = 0.045 mg/mL at 37 °C) and the other supersaturated (0.07 mg/mL at 27 °C). The X-ray diffraction (XRD) samples were prepared by mixing 4.0 g of collagen with different concentrations of UA in 100 mL of distilled water. These samples were then left in a crystallizer for approximately one week until crystals formed. Each sample was ground using an agate mortar. For abrasion, water adsorption, and PEA, all samples were prepared by mixing the collagen and UA powders.
All samples were prepared for these processes by mixing two powders, collagen and UA. However, for abrasion, water adsorption, XRD, and PEA analyses, it was necessary to use a stabilizing agent to provide the samples with the required consistency; for this reason, 20% polyurethane (PU) was used as an agglutinant. The samples for these tests were prepared as follows: UA and collagen powders were mixed in an agata mortar; PU was obtained by mixing poly-isocyanate with a hydroxylated resin (Polyform); this mixture was added to the powders, forming a soft paste which was placed in a stainless steel mold, and subject to a pressure of 100 bar for 1 min; at this time the chemical reaction between the poly-isocyanate and the hydroxylated resin already started providing the samples with some stability; the samples were removed from the mold and placed at rest for a day to finish the chemical reaction that forms PU. The samples were small disks measuring 12.5 mm in diameter and 1.7 mm thick.
Samples characterization
The XRD analysis was performed on a Rigaku, Ultima IV, diffractometer operating at 30 mA and 40 kV with a Cu Kα wavelength of λ = 1.5406 Å; the angle 2Theta was varied from 5° to 80° at 2 °/min. For crystallization and dissolution, the crystallite sizes SC were obtained using Scherrer’s equation, together with the software JADE v8.1, and the broadening of the strongest reflection in the diffraction pattern. The scanning electron microscopy (SEM) images were obtained using a Hitachi SU-8230 microscope operated at an acceleration voltage of 1000 V and a magnification of 300×. The water adsorption was determined according to ASTM-D570-98 standard using distilled water and an analytical balance with a 10-4 g precision. The abrasion results were obtained according to the ASTM-D-1242-95 standard using a rotating steel disk, carbide sandpaper No 600, and an analytical balance with a 10-4 g precision. The PEA was obtained using a digital multimeter USB Steren model MUL605 equipped with data communication software to the PC, with a resolution of 0.01 mV in the 0 to 60 mV range, with an accuracy of ±(0.5%) and an input impedance of 10 MΩ.
Results and discussion
As mentioned, in gout, monosodium urate precipitates in supersaturated conditions, forming crystals. Figure 1 shows how this salt is produced during the hydration process. When an aqueous solution of UA crystalline powder is prepared, some transformations occur: water solvates the aggregates by separating UA crystals, the crystal size is reduced (dissolution process), the molecules are separated, and, finally, an electrolyte is produced where UA remains in ionic form UA− with its counterions Na+, giving rise to monosodium urate. As the crystal dissolves (dissolution process), the concentration of UA− in the medium increases, reaching supersaturation conditions; at this point, precipitation begins, marking the onset of the crystallization process. On the contrary, the crystal formation produces the opposite effect: the concentration of UA ions in water decreases, eventually stopping crystal formation; the system reaches a state of equilibrium that depends on the initial conditions. To observe the complete crystallization and dissolution processes, it is necessary to start well below the saturation concentration to prevent competition between these two processes.
Figure 2(a) shows the crystallization kinetics, where it is possible to observe the UA’s aggregation profiles in water at five different UA concentrations. These profiles represent the onset of crystal formation. Among concentrations lower than CS, C1 produces crystals most rapidly; this is because, at this concentration, there is less blocking by the other ions, so that a UA− ion can be deposited on the crystal for its growth. On the contrary, when the UA− concentration is high, the crystals grow slowly due to the blocking effect. This blocking effect is characteristic of ionic solutions. From these profiles, it is possible to observe that the ions’ precipitation is not homogeneous because the profiles do not follow a linear dependence but exhibit a more complex relationship; this can be noticed by observing the correlation coefficients ranging from [0.93094 to 0.9839], indicating that the fitting is not so good. These profiles were fit with a straight line only for visualization and to estimate the crystallization rate. Figure 2(b) shows the slopes of the profiles as a function of UA concentration; these slopes provide information on the rate of crystal growth. From this figure, it is clear that the system with the lowest UA concentration forms crystals most rapidly as compared to systems at higher concentrations: as the concentration of UA increases from 0.01 to 0.06 mg/mL, the rate of crystal formation decreases from 10.2 to 8.5 nm/min (Figure 2(b)).

(a) Kinetics of UA crystal formation for samples with different UA concentrations; (b) slopes of the crystal size profiles as a function of UA concentration.
This behavior is typical of ionic transport: the ionic transport is modeled as a series of jumps of the ions to neighboring available locations of equal to or larger than the size of the jumping ion; however, this process has a notable characteristic effect, the “obstruction (blocking) effect”: when a high concentration of ions surrounds the jumping ion, these ions inhibit (blocking) the jump, reducing the ionic transport; then, this depends not only on the size and charge of the ions but also on their concentrations: lower UA concentration results in a higher transport velocity.
Knowing crystallization and dissolution kinetics is important because the UA concentrations affect both processes; the size profiles for both processes were obtained using DLS. All aqueous solutions were prepared containing UA at different concentrations and temperatures. For the crystal dissolution (Figure 3(a)), the UA crystals were immersed in water with gentle stirring at a concentration of 0.045 mg/mL and at 37 °C; this concentration is below CS = 0.06 mg/mL, and because water dissolves crystalline aggregates, the crystal size is also reduced (Figure 3(a)). The crystal dissolution kinetics show that this process follows second-order kinetics: the crystal size SC was reduced from 9200 to 4000 nm following a decreasing parabolic law:
with a correlation coefficient of R = 0.99056. Due to the low UA concentration and relatively high temperature (0.045 mg/mL at 37 °C), this process was carried out systematically and homogeneously following a specific parabolic profile. On the contrary, for the crystallization process (Figure 3(b)), the UA samples were immersed in water at a concentration of 0.07 mg/mL and at 27 °C; the precipitation begins forming crystals as soon as the samples are in contact with water because this concentration is higher than CS, and the temperature is reduced. Once the sample is immersed in water, the DLS equipment begins to measure the particle size as a function of time, reporting an increment from 150 to 4200 nm in 60 min; this data was fitted using a power law, resulting in an exponent of 2, that is, the crystallization kinetics also follow a second-order kinetic with a correlation coefficient of R = 0.99279:
In these conditions, UA precipitates quickly and homogeneously, forming large crystals.
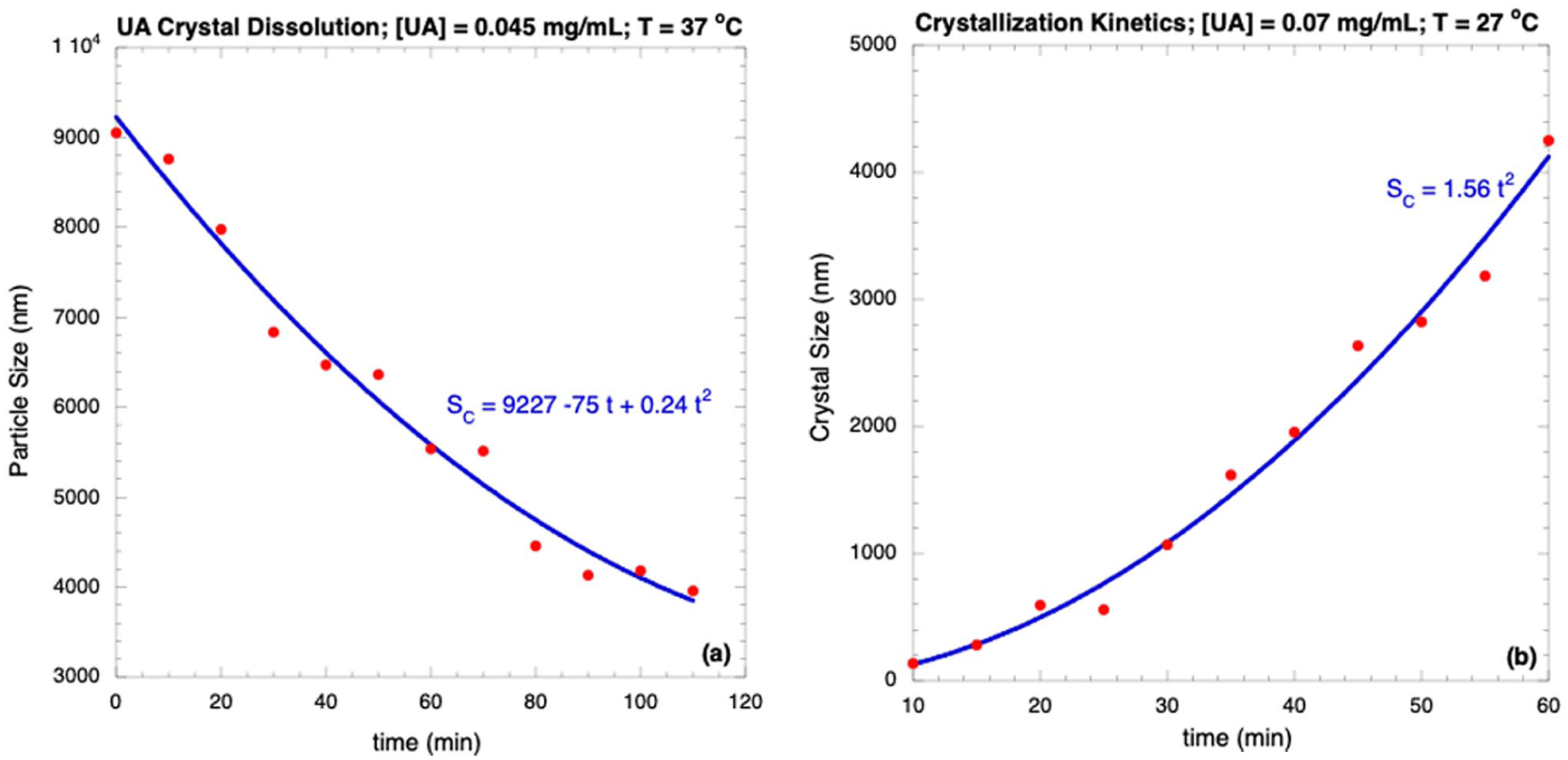
(a) UA crystals dissolution in unsaturation conditions (C4 = 0.045 mg/mL, 37 °C); (b) UA crystals growth at supersaturation conditions (C7 = 0.07 mg/mL, 27 °C).
The XRD technique provides information on the crystallinity of the samples. Figure 4 shows two diffraction patterns: the one in red corresponds to UA crystals (Aldrich), and the other in blue corresponds to crystal growth from the precipitation of UA molecules under supersaturated conditions (0.07 mg/mL at 27 °C). Based on these diffractograms, the crystallite size can be determined from the strongest reflection, at 2Θ = 13.42° for the red (UA crystal, Aldrich), and 2Θ = 19.44° for the blue (crystals grown from supersaturated solutions, 0.07 mg/mL at 27 °C). The crystallite size determination was obtained using Scherrer’s equation, which relates the crystallite size SC with the broadening of the strong reflection in the diffraction pattern:
where SC is the crystallite average size, K is a dimensionless shape factor with a value close to unity (typically 0.94), λ is the X-ray wavelength, β is the line broadening at half the maximum intensity (FWHM) in radians (also denoted as Δ(2Θ)), and Θ is the Bragg angle. Using Equation (2), the crystallite size for the UA crystals (Aldrich) in red is SC = 21.3 nm, while the crystal grown in supersaturated conditions in blue has a crystallite size of SC = 0.82 nm; even though the size of these crystals is small, this was obtained only during the first hour of crystal formation.
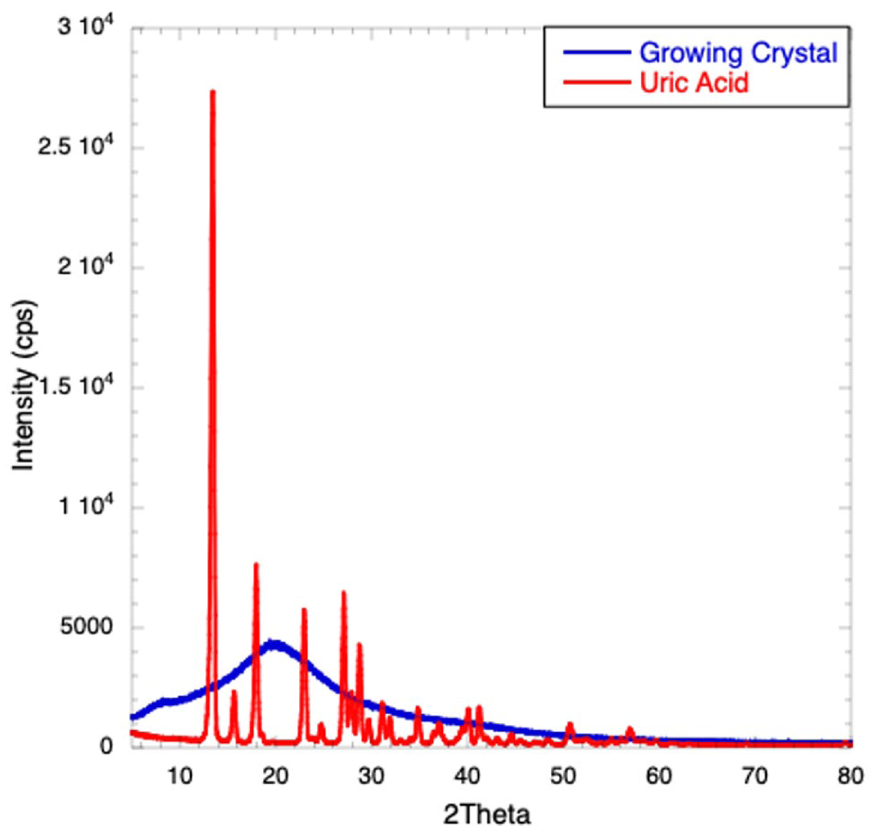
XRD of (a) UA crystals (red), as received from the manufacturer, and (b) crystalline growth of UA obtained by precipitation in saturated conditions (blue).
SEM images of the crystalline aggregates are shown in Figure 5. This image corresponds to the sample obtained from UA analytical reagents (Aldrich) as received; some crystals are sharp with rhomboidal terminations, typical characteristics of the crystals in the big toe in patients with goat. Water adsorption studies are important in this case because the growth and dissolution of the UA crystal occur in an aqueous medium; it is important to clarify that PU does not absorb water significantly. SW is a measure of the amount of water absorbed by the samples; it is defined as SW = (mf − mo)/mo, where mo is the dry initial weight, and mf is the final weight of the sample after an immersion in water for 2 h. Water adsorption has two contributions: the water absorbed by UA, and the water absorbed by collagen. For collagen, this molecule has a length in the range [220, 300] nm; it has been reported changes in the collagen swelling degree from 0 to 2 depending on pH; SW-C = 2 means that the average weight of collage molecules is increased by a factor of 3: mt = 3mo; however, in a rod-like molecule like collagen, an increase in weight by a factor of 3 does not mean that the length L was increased for this factor; because m = πρr2L, a small increment in the thickness r of the molecule can lead to a significant increment in its weight. In Figure 6, the water adsorption results are reported. As mentioned, all samples contain collagen and different UA concentrations except C0, which contains only collagen. UA and collagen exhibit very different water absorption behavior. Collagen is poorly soluble in cold water and slightly more soluble in warm and hot water; however, at 30 °C (near room temperature), it is more soluble, increasing its weight. On the contrary, water dissolves the UA crystals, thus reducing their size and weight (crystal dissolution). From Figure 6, it is possible to see that sample C0 (containing only collagen) produces considerably high water adsorption: Δm ≡ mf − mo = 7.4 mg; this is the reason for the high slope in the SW-C water absorption profile. In samples containing UA and collagen, there is competition for weight increment: collagen adsorbs small amounts of water, increasing its weight, while UA crystals are dissolved in water, reducing their weight. This means that, in a small region, the weight only changes from 0 to 1.5 mg, because these two effects are similar in magnitude but opposite in contribution, significantly reducing the combined effect of water absorption; this is why the slope is significantly reduced in the SW-UA water absorption profile. Additional increments in the amount of UA produce a net but slight weight reduction; in this case, the crystal dissolution dominates over the collagen water absorption. It is also evident that weight loss decreases linearly with increasing UA concentration. Figure 6 clearly shows that in collagen-UA systems, by increasing the water concentration, it is possible to reduce the size of the crystals and, consequently, their effect. This figure is interesting and important because, on one hand, it shows how UA crystals form and on the other hand, how they dissolve. The amount of water absorbed in this process is 5.88 mg, while the amount of water desorbed is 5.82 mg; these water masses are practically the same. The only difference is that during crystallization, the change in UA amount is 0.01 mg, whereas during desorption, it is 0.06 mg.

SEM image of UA crystals obtained from the manufacturer.

Adsorbed water profiles for different samples after 2 h of immersion.
The abrasion results are reported in Figures 7(a) and (b). Figure 7(a) shows the weight loss profiles for all samples; they are straight lines with different slopes, indicating that the samples are homogeneous. The slopes of these profiles represent the rate at which the mass is removed from the sample. In Figure 7(b), the slopes for all samples are plotted as a function of UA concentration; all these samples are in the unsaturated-saturated regime, that is, below CS. Because the collagen content is constant across all samples, its contribution to weight loss is constant and small, as these fibers are mechanically very resistant to abrasion; they can also form entanglements, further increasing their resistance. When the UA concentration is increased, the samples lose weight more quickly (Figure 7(b)). The slope for a CS concentration is 0.226 mg/s, which is the highest value for the slope corresponding to rapid abrasion resistance; at this concentration, many more UA molecules are not chemically linked to collagen molecules, and they are easily removed. For supersaturated conditions (C7 = 0.07 mg/mL), rapid crystal growth increases abrasion resistance because the crystals are highly abrasion-resistant; thus, for C7, the slope is small (0.144 mg/s). This data is not shown in the figure.
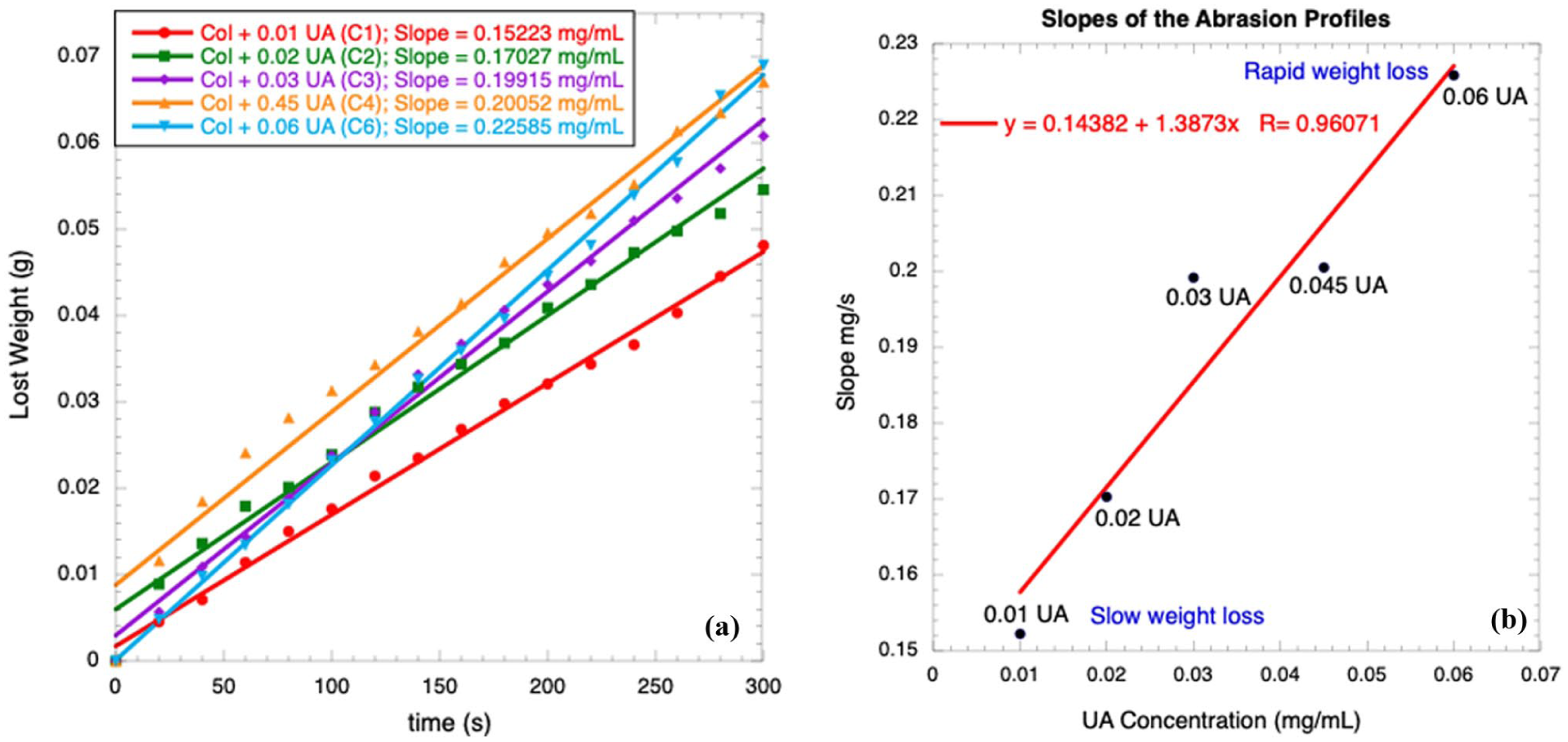
(a) Weight loss profiles in abrasion experiments, (b) slopes of the abrasion.
The piezoelectric technique is sensitive to changes in the molecular structure of the piezoelectric material. Collagen is known to be a piezoelectric material, so if the collagen concentration is kept constant but the UA concentration is increased, the UA crystals growing inside the collagen matrix modify the spatial distribution of collagen molecules near the UA crystals; then, piezoelectric analysis is sensitive for determining changes in collagen morphology when it is in the presence of UA crystals. Due to this, the PEA can be used to follow the kinetics of crystal growth and, consequently, the evolution of gout disease; the piezoelectric output voltages of UA-Collagen samples, as a function of UA concentration, are shown in Figure 8. However, these samples do not include HAp, an essential component in the samples affected by gout. HAp will be considered constant in the same way that collagen, and as a filler; however, HAp is piezoelectric, contributing to the output signal for gout.
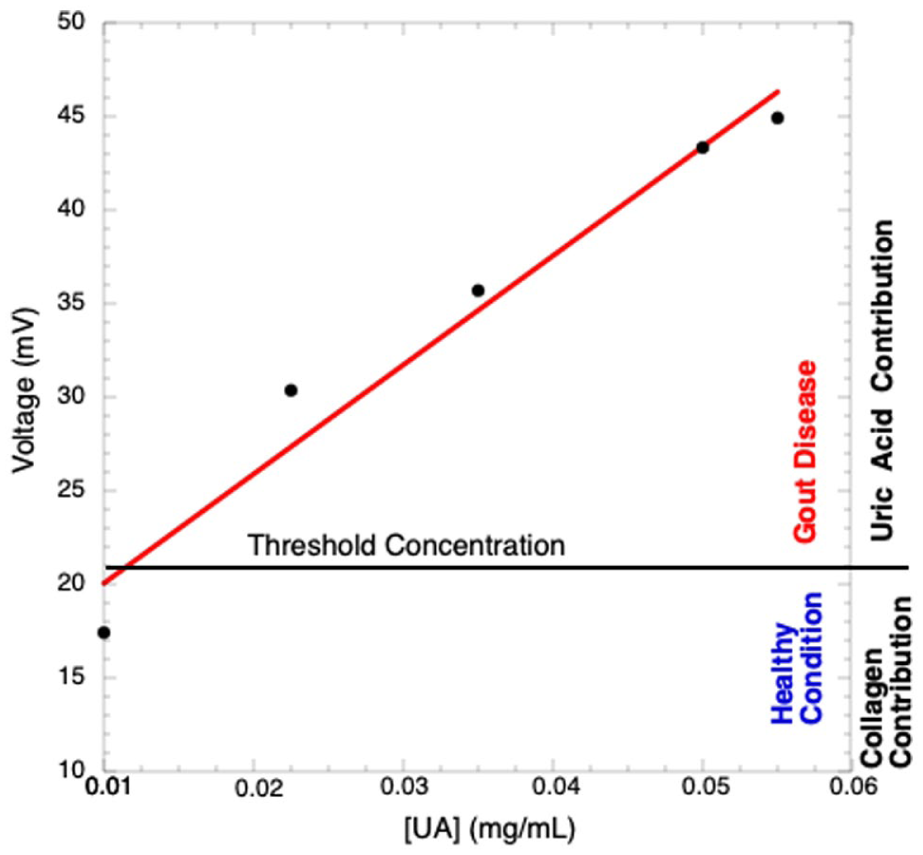
Piezoelectric voltage as a function of UA concentration.
Figure 8 was modified by including a DC voltage due to HAp of 89 mV, as reported elsewhere reference; 26 then, the piezoelectric voltage obtained for HAp-Collagen-UA samples was reported in Figure 9. From this figure, it is possible to see that at 110 mV, the accumulation of UA is incipient 0.01 mg/mL, while at 140 mV, the UA concentration is 0.06 mg/mL, which is the saturation concentration CS; at this concentration of UA, its precipitation is imminent, giving rise to the beginning of crystal growth; in these conditions, gout can be declared, although it is just the beginning, which means that UA crystals can still be small. For voltages below 140 mV, the UA concentration remains unsaturated, and the risk of gout is small, depending on how far the output voltage is from 140 mV. The voltage of 110 mV corresponds to the threshold voltage between gout and no gout. As shown, a simple voltage measurement provides essential information about the status of gout. The procedure for a rapid diagnosis of gout is as follows: place the big toe between electrodes, a small but known pressure has to be applied, and the piezoelectric voltage measured; for voltages below 140 mV, the risk of gout is small; however, for voltages higher than 140 mV, the risk of gout is high, and the patient needs to be referred to a specialist.

Piezoelectric voltage including HAp as a function of UA concentration.
Then, it is possible to use the piezoelectric technique to diagnose “local” accumulations of UA in the big toe; when the piezoelectric voltage increases, so does the UA concentration, accelerating the crystal formation and, consequently, the gout. One of the essential features of the piezoelectric technique is that it determines local properties; in this particular case, this technique provides a voltage that depends directly on the local UA concentration; additionally, it allows an early, painless, and noninvasive gout diagnosis. This result is important from both clinical and patient perspectives. The possibility of obtaining an accurate, rapid, and noninvasive diagnosis is a great advance in gout treatment because it allows anticipating the disease and taking the necessary measures to eliminate or at least reduce its effects. It would be great to have actual gout samples at different stages of crystal growth, but that isn’t possible. However, the results obtained here, using samples prepared by mixing collagen and UA powders, are pretty similar to those from real samples.
Conclusion
Gout disease is caused by monosodium urate precipitation, which forms needle-sharp crystals that injure soft tissue and result in intense pain that incapacitates the patient from practically any movement; this precipitation occurs when its concentration reaches supersaturation and affects the big toe. In this work, the kinetics of UA-based crystal formation and dissolution were determined under aqueous conditions. The kinetics of water adsorption were determined by measuring particle size over time using DLS. Several studies of the collagen-UA systems were also included, using techniques such as abrasion, XRD, DLS, SEM, and PEA. The PEA enables a correlation between the piezoelectric voltage and the UA concentration. It is this correlation that allows for a diagnosis of gout: The PEA allows for determining increments in UA concentration in the analyzed region, and when UA rises to a supersaturated concentration, it precipitates, forming crystals that damage soft collagen tissue; this is the process of gout formation. It is important to note that, typically, UA content in blood is reported as an average over the whole body, not just in the region of interest. However, PEA provides a local measurement of UA concentration. In principle, to make a diagnosis, it is sufficient to place the sample between electrodes, apply low pressure, obtain the piezoelectric voltage, and see if this voltage is in the gout or healthy region. According to Figure 9, for voltages equal or lower than 110 mV, the accumulation of UA at the big toe is incipient and presents no risk that affects health; however, for voltages higher 110 mV corresponds to a saturated UA concentration CS = 0.06 mg/mL; at this concentration, the precipitation is imminent, giving rise to the beginning of crystal growth; this situation corresponds to the onset of the got disease. The diagnosis of gout using the PEA is straightforward, noninvasive, and enables quick results. The piezoelectric signal provides information about local UA concentration and how it changes over time, depending on diet, medications, exercise, and so on. A voltage measurement near the swollen region of the big toe provides essential information about the gout. It is possible to use the piezoelectric technique to diagnose gout early, painlessly, and noninvasively.
Footnotes
Acknowledgements
The authors thank M. in S. Manuel Aguilar Franco for his valuable assistance in acquiring the SEM image. In addition, they want to mention M. in S. Guillermo Vazquez Sanchez for valuable support in computing.
Ethical considerations
This article does not contain any studies with human or animal participants.
Consent to participate
This article does not contain any studies with human or animal participants.
Consent for publication
Not applicable.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability statement
The research data used to support the findings of this study are included in the article.



