Abstract
This study focused on evaluating the effectiveness of different filter media including activated carbon, Pyrolox and Greensand in improving water quality based on key parameters such as color, turbidity, dissolved oxygen, total dissolved solids, pH, hardness, and sludge density index. Preliminary results showed that each filter material had its own advantages. Specifically, activated carbon showed superior performance in removing color (8.3 TCU–9.233 TCU) and turbidity (0.5 NTU–0.767 NTU), while Pyrolox showed the highest efficiency in reducing dissolved oxygen in the range of 4.657–6.85 mg/L. Notably, Greensand was recorded as the best filter material in reducing sludge density index (3.52 ± 0.252), an extremely important factor for membrane filtration systems. The study also found that these filter media had no significant impact on pH, total dissolved solids and hardness of water. In addition, another important finding is that the use of filters with smaller pore sizes significantly improves color removal efficiency. These findings provide valuable data for the selection and optimization of water treatment systems to meet diverse water quality requirements in preparation for reverse osmosis desalination.

Introduction
Saltwater intrusion is the phenomenon of saltwater from the sea moving deep into the land through the river and canal system, especially in the dry season when the freshwater flow from the upper Mekong River decreases sharply. This is a serious and increasing problem in the Mekong Delta (Vietnam), the largest rice and aquaculture granary in the country. According to statistics, in recent dry seasons, saltwater has penetrated 50–110 km inland, seriously affecting daily life, agricultural production and aquaculture.1–4 Many areas are in a state of severe shortage of fresh water for daily life, forcing people to buy water at high prices, or use saline water sources, affecting their health. Faced with this situation, the reverse osmosis (RO) water filtration system has been applied as a feasible solution to treat saline water, especially for the purpose of providing drinking water and daily life. 5 RO technology operates on the principle of using high pressure to push water through the RO membrane, which can remove up to 99% of dissolved minerals and impurities, helping to convert brackish water and slightly salty water into standard fresh water.6,7 Traditional pretreatment technology (coagulation combined with a two-stage filter column using sand and anthracite) and microfiltration membrane technology (polyvinylidene fluoride (PVDF), pore size 0.1µm) have also been studied and evaluated to improve water quality from seawater sources in the San Diego area, California. 8 Different pretreatment technologies including traditional coagulation combined with a three-stage filter column (sand, anthracite, and garnet), slow filtration (gravel and sand), and ultrafiltration membranes (<0.02 µm) have been studied and evaluated for their effectiveness in treating seawater in the Monterey Bay area. 9 In addition. In Saudi Arabia, fine and thick sand media were used before NF and significantly improved the quality of the feed water. In addition, the use of NF as a pretreatment in a seawater reverse osmosis (SWRO) desalination pilot plant increased water production by more than 60%, resulting in a 30% cost reduction. 10 A pilot model using a two-stage filter column tested for pretreatment of seawater in Thermaikos Gulf, Northern Greece was published by Mitrouli et al. 11 The two-stage filter column used a layer of sand (50 cm) and a layer of gravel or anthracite (70 cm) media, both of which were capable of reducing sludge density index (SDI) (<5) and turbidity by nearly 0.2 NTU. Another study by Remize et al. 12 compared the efficiency of a two-stage filter column (sand and anthracite) combined with FeCl3 coagulant (6 ppm) and a microfiltration method using a PVDF membrane with a pore size of 0.1 µm, for the pretreatment of seawater in the Mediterranean region.
Pretreatment is a fundamental step in RO systems, providing many essential benefits to ensure the efficiency and durability of the entire system. This process removes large impurities, microorganisms, and harmful ions and helps prevent fouling, thereby prolonging the life of the RO membrane. At the same time, pretreatment improves filtration performance by increasing water flow, stabilizing pressure and optimizing the osmosis process. In particular, the quality of the output water is ensured by removing harmful impurities, bacteria, viruses, as well as improving the taste and odor of water by treating organic compounds. In terms of cost, pretreatment not only reduces energy consumption but also limits membrane damage, thereby optimizing maintenance and operating costs. With the ability to flexibly adapt to many water sources, pretreatment helps prevent precipitation and membrane fouling, while protecting the system from microbial contamination with effective disinfection methods. Thanks to the integration of many outstanding benefits, pretreatment not only improves system performance but also ensures that the output water meets high safety and quality standards. Therefore, the pretreatment process, including removing impurities before the water enters the RO system, plays a key role in improving filtration efficiency and prolonging the life of the RO membrane. This study aims to evaluate and optimize pretreatment methods for saltwater sources, in order to not only enhance the operating efficiency of RO technology but also contribute to reducing costs, protecting the filtration system and maintaining a sustainable source of fresh water for the community—especially in areas that are heavily affected by saltwater intrusion.
Materials and methods
Materials
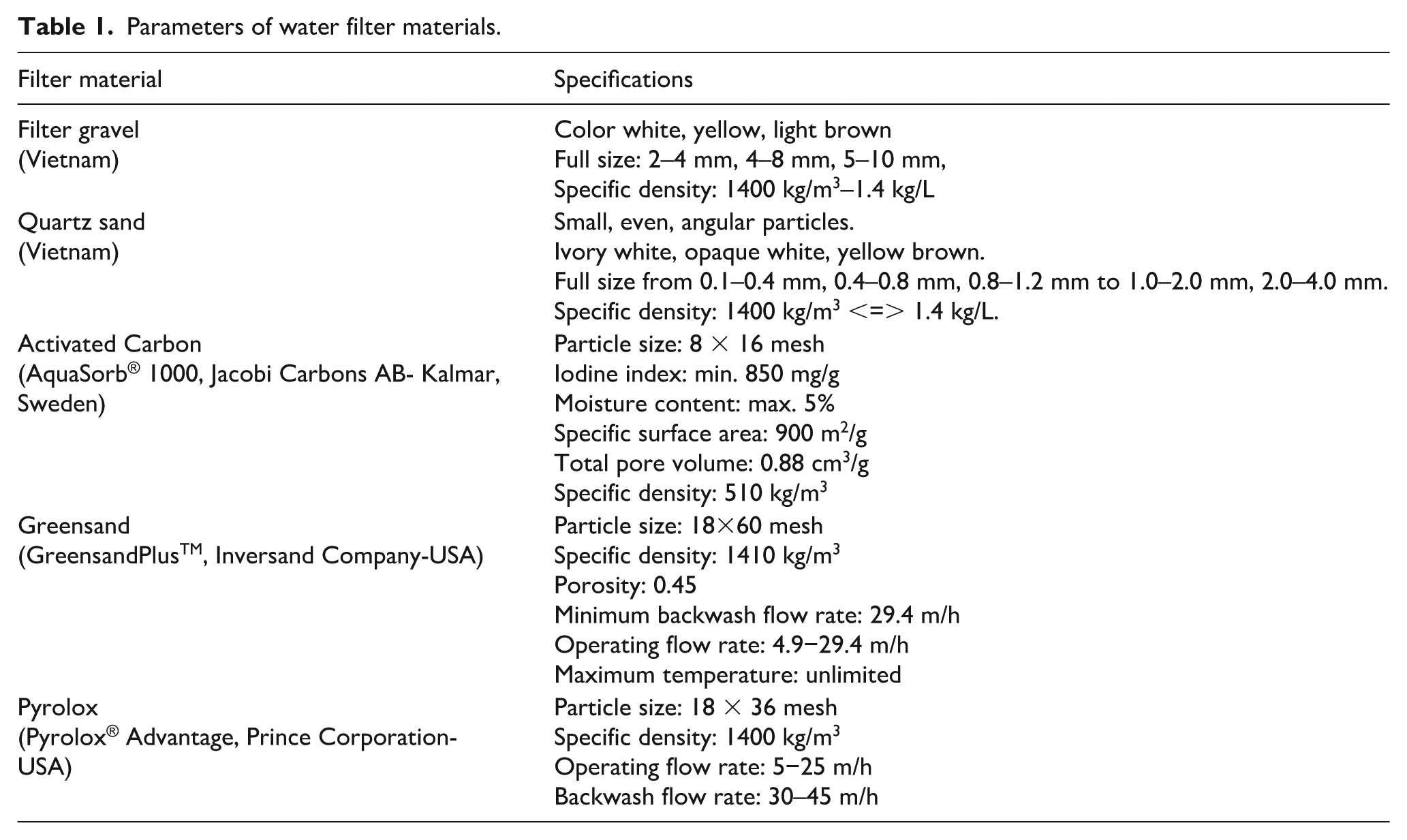
Water Source from the Luong Quoi River, Luong Quoi Commune, Giong Trom District, Ben Tre Province (Vietnam). Filter Materials: Activated carbon, Greensand, and Pyrolox (Table 1). Filter Cartridges: Polypropylene material with pore sizes of 0.2, 1, and 5 µm (Table 2).
Parameters of water filter materials.
Water filter core specifications.

Pretreatment process for experimental system
The water source for the survey will be taken directly from the Luong Quoi River and supplied to two corresponding processes, namely directly supplied to a 10-m3 tank before being fed into the pretreatment system (Process 1, Figure 1) and through the coagulation-flocculation process at the water plant, then continued to be supplied to a 10-m3 tank before being fed into the pretreatment system (Process 2, Figure 2). Water from the 10-m3 tank is pumped through a rough filter cluster including a two-layer filter column containing supporting gravel and quartz sand and a multi-layer filter column containing supporting gravel, quartz sand and the filter material being surveyed. After going through the rough filter, the water continues to go through a cartridge filter column containing filter cores of different sizes being surveyed. Water samples after the cartridge filter column are taken to measure indicators to check water quality parameters after the pretreatment process.

Experimental system operation according to procedure 1.

Experimental system operation according to procedure 2.
Analytical methods
Parameters such as pH, total dissolved solids (TDS), and dissolved oxygen (DO) were measured by P-520 and P-512, PEAK Instruments, China. Sludge density parameter (SDI15) was measured according to ASTM-D4189-07 method by SDI-2000, Applied Membranes, USA.
The turbidity of the water sample is determined by measuring the absorbance at a wavelength of 450 nm and comparing it with the calibration curve using a standard turbidity solution, which is a mixture of hydrazine sulfate and hexamethylene tetramine (SMEWW-2130B).
The color of the water sample is determined by measuring the absorbance at a wavelength of 455 nm (SMEWW-2120C) and comparing it with the calibration curve using a standard color solution, which is a mixture of potassium chloroplatinate and cobaltous chloride.
The hardness of the water sample is determined by the ethylenediaminetetraacetic acid (EDTA) titration method (TCVN 6224:1996). The hard buffer solution is prepared by dissolving 1.18 g of EDTA, 0.644 g of MgCl2, and 16.9 g of NH4Cl in 143 mL of concentrated NH4OH and diluting to 250 mL. 1 mL of the buffer solution is added to a conical flask containing 50 mL of the sample to achieve a pH of 10. Next, a small amount of the indicator Eriochrome Black T (EBT) is added to the solution, and the solution will turn wine-red. The sample is titrated with 0.01 M EDTA solution until a blue color appears, and the volume of EDTA used (1 mL) is recorded.
Results and discussion
Selection of the process for the experimental system
The experimental RO system showed a notable difference between the filter media and operating processes in DO removal capacity, as shown in Figure 3(a) Specifically, the DO concentration in Process 1 was measured at 6.712 mg/L, which decreased to 5.553 mg/L in Process 2 when activated carbon was used as the filter media. Although this decrease was relatively small, it still showed a certain improvement trend in DO reduction when switching to Process 2. With Greensand filter media, the DO concentration in Process 1 was 5.490 mg/L but increased to 6.108 mg/L in Process 2. This shows that Process 2 was not effective in reducing DO with this material, and even increased the DO concentration, so Process 1 was considered a more suitable choice when the goal was to control DO. Most notably, the results obtained with Pyrolox media were obtained. While DO in Process 1 reached 6.672 mg/L, in Process 2, this figure dropped sharply to 4.543 mg/L. This is a significant reduction and shows the superior efficiency of Process 2 when combined with Pyrolox in treating DO. In summary, among the three tested materials, Pyrolox combined with Process 2 gave the best DO reduction efficiency, achieving the lowest DO level in the surveyed cases. Therefore, this choice is considered the optimal solution to control DO in the RO system.
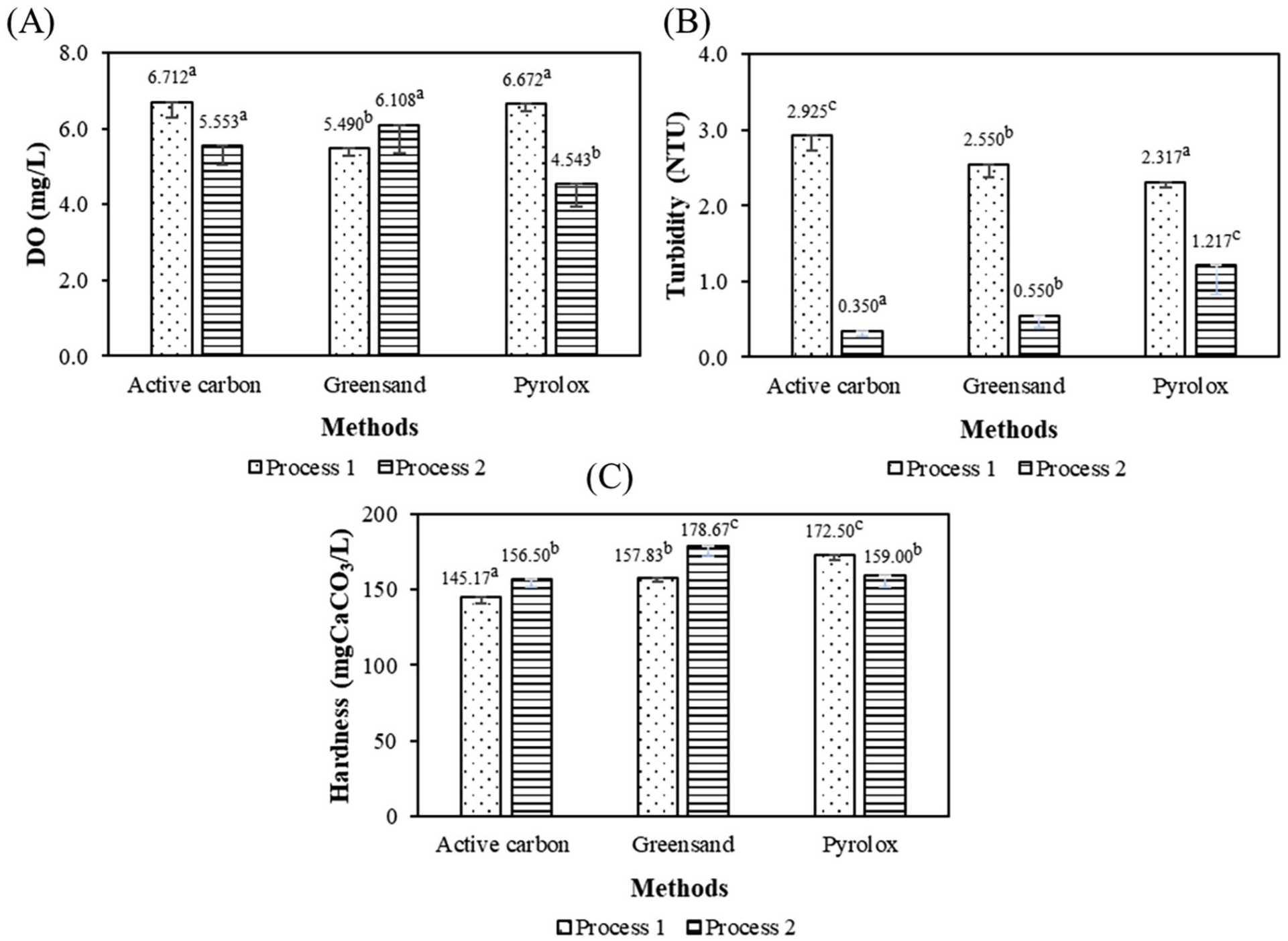
Effect of system operation procedures on dissolved oxygen (a), turbidity (b), and hardness (c) of water.
Figure 3(b) emphasizes the importance of controlling the influent water turbidity. In Process 1, filter materials such as activated carbon (2.925 NTU), Greensand (2.55 NTU), and Pyrolox (2.317 NTU) all failed to meet the turbidity standard, indicating that they did not meet the requirements for water entering the RO system. The filtration efficiency decreased and the power consumption increased when the turbidity was high, leading to membrane clogging. In Process 2, activated carbon and greensand materials contributed to reducing the turbidity to 0.35 NTU and 0.550 NTU, the turbidity value within the allowable range, demonstrating the effective water treatment capability before contacting the RO membrane. In addition, the turbidity decreased to 1.217 NTU when using Pyrolox material in Process 2, the turbidity exceeded the allowable threshold, so the optimal efficiency in membrane protection could not be guaranteed. Overall, in Process 2, the two activated carbon and greensand materials demonstrated excellent turbidity control capability, contributing to reducing the risk of membrane clogging and improving operational efficiency.
In addition, hardness is a factor that negatively affects the treatment efficiency, Mg2+ and Ca2+ ions and treatment chemicals can react with each other, leading to a decrease in the ability to disinfect, creating conditions for microorganisms to grow in the system. At the same time, biological adhesion is a factor that reduces the ability to remove impurities and the quality of water after treatment. The results shown in Figure 3(c) show that both test treatment processes did not bring about significant results in reducing hardness, and in some cases, they even tended to increase the concentration of hardness ions. Specifically, when using activated carbon, the hardness increased from 145.17 mgCaCO₃/L in Process 1 to 156.50 mgCaCO₃/L in Process 2. The water hardness was not improved when using greensand material, on the contrary, in the middle of the two processes, the hardness increased from 157.83 to 178.67 mgCaCO₃/L. Greensand is commonly used to remove manganese and iron and has a negligible effect on improving water hardness. 13 In addition, the ion release of the material is the factor leading to this increase. Meanwhile, the water hardness decreased from 172.50 to 159.0 mgCaCO₃/L in Process 2 for Pyrolox material. However, this reduction still does not meet the required input threshold for the RO system. However, Pyrolox still plays an important role when used as a pretreatment filter, helping to remove heavy metals and impurities that often come with hard water. This helps to prolong the life and improve the efficiency of downstream water softening equipment, such as ion exchange systems or RO membranes. In summary, activated carbon and greensand materials are effective in improving turbidity in Process 2, the DO level is stable and higher than in Process 1, proving that Process 2 is the preferred choice when the priority is to improve the quality of the inlet water, meeting the criteria for DO and clarity.
The effect of filter materials on the quality of water after coarse filtration
The interactions between water components and the ability to remove impurities are directly affected by the pH index during water treatment, especially during the rough filtration stage. It is necessary to check the pH before rough filtration, because when the pH is low, impurities are easily dissolved and heavy metals are difficult to remove during filtration. On the contrary, the filtration system can be clogged with magnesium and calcium compounds that precipitate when the pH is too high. Table 3 shows the pH values of water after filtration with three different materials such as active carbon, greensand, and pyrolox. The water quality is maintained after filtration, the pH value is stabilized in a neutral environment. Specifically, when using activated carbon, greensand and pyrolox materials, the pH values fluctuated at 7.017 ± 0.006, 6.993 ± 0.006 and 7.023 ± 0.012, respectively, proving that the pH was still within the allowable range. After the rough filtration process, pyrolox material was the most effective, being the material with the lowest DO concentration of 4.243 ± 0.235 mg/L. Meanwhile, greensand was the most effective in reducing the DO concentration with a DO value of 5.477 ± 0.021 mg/L, the highest compared to the remaining materials. In addition, activated carbon also demonstrated the ability to reduce DO concentration with a DO value of 5.833 ± 0.103 mg/L, still lower than pyrolox material. The pollution level and filtration capacity of the water treatment system are reflected by the color index during the rough filtration process. The analysis results in Table 3 show that activated carbon is the material with the best color removal ability, the value after filtration is 13.1 ± 0.346 TCU, much lower than the two materials greensand (34.433 ± 1.963 TCU) and pyrolox (38.9 ± 6.966 TCU). In summary, activated carbon shows superior color treatment ability and stability after re-treatment, making it an ideal material for water color treatment. In terms of turbidity index, greensand and pyrolox materials have low treatment efficiency with values of 2.067 ± 0.551 NTU and 2.3 ± 0.889 NTU, respectively. In contrast, the low turbidity value of 1 ± 0.1 NTU of activated carbon showed effective water treatment and stability of the water treatment process with a standard deviation of 0.1. Similarly, activated carbon showed the lowest TDS results, averaging 225 ± 9.539 mg/L, indicating superior treatment efficiency in reducing TDS. This value is far below the maximum allowable threshold (500 mg/L), reflecting the excellent treatment ability of this material. Pyrolox also showed high efficiency with an average TDS value of 250.333 ± 9.292 mg/L, ensuring that TDS concentrations were maintained at low and stable levels within the standard limits. However, the TDS value of greensand material at 468.333 ± 19.858 mg/L indicates that the TDS reduction performance of greensnad is worse than that of pyrolox and activated carbon. The hardness of water treated through three materials of active carbon, greensand and pyrolox, the analyzed hardness values are 164.667 ± 1.528 mg/L, 176.667 ± 4.619 mg/L and 163.333 ± 3.786 mg/L, respectively. Activated carbon is very effective in removing organic pollutants and improving the taste of water. However, it is not effective in removing heavy metals or dissolved iron/manganese. Therefore, it is often combined with other filter materials to increase the overall efficiency. Pyrolox is a natural filter material containing approximately 85% pure MnO2, pyrolox has superior iron, manganese and H₂S removal capabilities, even without the need for chemical regeneration like greensand. 14 However, the material is heavy, requires high flow and sufficient pressure to operate effectively, and requires regular backwashing to avoid clogging. In addition, greensand is effective in removing iron, manganese, H₂S, but requires periodic regeneration with KMnO₄ solution to maintain the activated layer. 15 The operation depends on pH (optimum from 6.2 to 8.5) and DO concentration.
Some indicators of water after rough filtration by different filter materials.

Note. The values a, b, c represent the differences of the filter materials at (p < 0.05).
The effect of filter materials and filter core on post-cartridge filtration water quality
Table 4 shows the pH value of water after rough filtration through three different types of filter materials. The results show that the pH of the water after filtration remains stable at a near-neutral level, and the water quality is maintained after the filtration period. Specifically, the pH value of the three types of filter materials (active carbon, greensand and pyrolox) and filter cores (5 µm, 1 µm, and 0.2 µm) fluctuates slightly in the range of 6.9–7, within the appropriate range, to avoid the dissolution or precipitation of unwanted compounds, improving the efficiency of water treatment.
Water quality parameters when changing filter materials and filter cores by cartridge filtration.

Note. The values a, b, c represent the differences of the filter materials at (p < 0.05).
Activated carbon tends to adsorb organic substances, helping to improve DO when using a 1-µm filter core, achieving the highest DO (6.29 mg/L). However, when using a 0.2-µm filter core, there may be a decrease in flow and gas exchange, leading to a decrease in DO. In addition, the ability to retain aerobic bacteria or the accumulation of organic residues at too small a filtration level can reduce DO.
Greensand works by oxidizing Fe²⁺ and Mn²⁺, which is often accompanied by the consumption of some of the DO in the water. The highest DO values are due to the favorable flow and gas exchange when using a 5-µm filter. The DO concentration tends to decrease slightly when reducing the filter size to 2 and 0.2 µm, probably due to the reduction in flow rate and increased resistance to oxygen diffusion into the water.
Pyrolox was only tested with a 0.2-µm filter, but the results varied greatly between samples (4.657–6.85 mg/L). This shows that DO depends not only on the type of material but also on the specific operating conditions (flow rate, contact time, degree of fouling). The lowest DO sample may be related to oxygen being consumed strongly by the Fe²⁺/Mn²⁺ oxidation reaction or microbial activity in the filter bed. The highest DO sample (6.85 mg/L) indicates effective oxidation conditions without excessive DO reduction. The filter media significantly affects DO, especially when the pore size is reduced too small (0.2 µm), which can cause DO reduction due to reduced gas exchange and retention of aerobic microorganisms. Activated carbon and greensand materials show moderate DO improvement with filter media (1–5 µm), while Pyrolox is highly dependent on system conditions.
In terms of color removal ability of activated carbon, with low and fairly stable TCU color indexes in all three filter media types: 5 µm (8.3 TCU), 1 µm (9.23 TCU), and 0.2 µm (8.5 TCU). This shows that the color removal efficiency of activated carbon is not significantly affected by the filter size, demonstrating the strong adsorption of this material for water-soluble color compounds. Greensand showed a clear increase in color removal efficiency with decreasing filter size. The TCU decreased from 37.633 (5 µm) to 9.467 (1 µm) and then decreased slightly to 9.5 (0.2 µm). The clear improvement when moving from 5 to 1 µm suggests that greensand is more effective in color removal when combined with finer filter sizes, possibly due to the retention of finer particles containing colorants. Similar to greensand, Pyrolox also showed a decreasing trend in TCU with smaller filter sizes: 26.433 (5 µm), 11.333 (1 µm), and 9.9 (0.2 µm). However, the color removal efficiency of Pyrolox is still lower than that of activated carbon and greensand in the 0.2-µm filter core. This shows that Pyrolox is not specialized in adsorbing colorants but mainly operates according to the oxidation-precipitation mechanism, so the efficiency depends more on the physical properties of the filter core. Of the three materials, activated carbon has the highest and most stable color removal efficiency, less affected by the filter core size. Greensand and Pyrolox significantly improve the efficiency when used with small filter cores, especially greensand gives results almost equivalent to activated carbon in the 0.2-µm core. Therefore, to optimize color treatment in water, activated carbon or greensand should be selected in combination with a ⩽1-µm filter core. Turbidity increased with decreasing filter size when using activated carbon material 0.5 ± 0.1 NTU (5 µm), 0.633 ± 0.058 NTU (1 µm), and 0.767 ± 0.0.058 NTU (0.2 µm). This is an opposite trend to the expectation, indicating that finer filter media did not reduce turbidity when used with activated carbon. This may be due to the fact that activated carbon is a porous powder material, which is susceptible to washing or leakage of fine particles through small filter media, increasing turbidity. This suggests that when using activated carbon, a better cleaning or particle immobilization step is needed to avoid increasing turbidity at small filter levels. Greensand showed a very significant reduction in turbidity when using smaller filter media. The turbidity decreased most when moving from the 5-µm core (2.633 ± 0.153 NTU) to the 1-µm core (0.467 ± 0.115 NTU). Although the turbidity increased slightly at the 0.2 µm core (0.6 ± 0 NTU), it remained low. This shows that greensand has good suspended particle removal ability when supported by a fine filter core, suitable for treating turbidity in raw water containing a lot of suspended solids. The improvement in turbidity was obvious when using the 1-µm core and continued to decrease at 0.2 µm, showing that Pyrolox combined with fine filter cores effectively removed turbidity, thanks to the oxidation-precipitation mechanism of metal particles (such as Fe, Mn), which helps increase the ability to remove sediment. 16
Activated carbon slightly reduces hardness, with a decreasing trend as the core size gets smaller, specifically water hardness is 154 mgCaCO3/L (5 µm), 151 mgCaCO3/L (1 µm), and 146.333 (0.2 µm). However, the reduction is not significant. Because activated carbon mainly functions to adsorb organic compounds, color, odor and does not have the ability to exchange ions, the water softening effect (hardness reduction) is low. Greensand shows a clear hardness reduction ability, especially when combined with a finer filter core. At the 0.2-µm core, the hardness drops sharply to 142 mg/L. This may be due to the ability of greensand to oxidize and precipitate metal ions such as Fe²⁺, Mn²⁺—indirectly reducing hardness if these ions contribute to the total hardness, while fine filter cores retain better the precipitates or fine particles generated from the filtration process. Pyrolox shows a relatively stable ability to maintain hardness, without a clear downward trend. In addition, the TDS value after filtration depends significantly on both the type of material and the cartridge size. At 5 µm, greensand gives the highest TDS (456.667 mg/L) due to the leaching of oxide or ion particles from the MnO₂ coating, while activated carbon (285.667 mg/L) and Pyrolox (234 mg/L) maintain lower levels due to their adsorption capacity and more stable structure. When reduced to 1 µm, activated carbon and greensand both reduce TDS (264.333 and 427 mg/L, respectively) due to a combination of mechanical filtration and adsorption, but Pyrolox increases to 265 mg/L due to the ultrafine MnO₂ particles generated during oxidation passing through the medium cartridge. At 0.2 µm, all three materials strongly reduce TDS, with the order from low to high being activated carbon (230.333 mg/L) approximately equal to pyrolox (236.667 mg/L) < greensand (244.333 mg/L), reflecting the ability to retain almost all colloidal particles, fine sediments and minimize leaching. In terms of mechanism, activated carbon mainly reduces TDS through strong surface adsorption and is more effective when the contact time is longer in the small core, while greensand removes iron and manganese by oxidation-precipitation but has the risk of releasing sediment when the core is coarse, for Pyrolox material, it will strongly oxidize but can generate MnO₂ colloidal particles in the intermediate core. Thus, choosing a small core size not only increases the efficiency of particle retention and TDS reduction, but also helps stabilize water quality, especially when combined with materials that tend to leach such as greensand or pyrolox. The SDI index is an important indicator in assessing the ability to cause blockage of the RO filter membrane. The lower the SDI index, the lower the ability to cause membrane blockage, which means the higher the quality of the input water for the RO system. An SDI value of <3 is ideal, <5 is acceptable, and >5 is at risk of membrane blockage. Activated carbon has a high SDI (>5) with slight variation between filter cartridges. Although smaller cartridges are usually expected to reduce SDI, here SDI increases when using 0.2-µm cartridges. This may be due to fine dust leakage from activated carbon into the water, especially when combined with ultra-small filter cartridges. Greensand shows quite good efficiency in reducing SDI, especially when using 0.2-µm cartridges. Although there is a slight increase when using 1-µm cartridges, at the smallest cartridge (0.2 µm), SDI reaches 3.52, approaching the safe threshold for feeding into the RO system. Greensand combined with fine filter cartridges helps reduce fine sediment and flocculation, reducing the risk of RO membrane fouling. Pyrolox has the highest SDI of all three materials, although it decreases slightly when using smaller cartridges, it still remains above the threshold of 5, not good enough for RO systems. The cause may be due to the generation of metal precipitates (Fe, Mn) from the oxidation reaction but not completely retained in the current filter cores. 17 Pyrolox itself has a rough structure, making it easy for fine particles to be washed out if not well fixed.
The effect of salinity on water quality after cartridge filtration
Table 5 shows that the concentration of the assumed salt significantly affects the water quality as shown by the indicators such as sediment density index (SDI), turbidity, and TDS. The significant increase in the dissolved ion content when increasing the concentration from 2 to 10 ppt leads to an increase in the TDS index. Specifically, at 2 ppt the TDS index is 3.002 g/L and increases to 9.981 g/L at a concentration of 10 ppt, indicating that the water environment is highly saline. This is a completely reasonable trend and consistent with the theory. Salt (mainly NaCl) is a dissolved solid. When the concentration of salt in water increases, the total amount of dissolved solids in the solution will also increase accordingly. Regarding turbidity, the fluctuation is not clear. Turbidity was high at concentrations of 6 ppt (0.725 NTU) and 8 ppt (0.541 NTU) and lowest at 10 ppt (0.354 NTU). The non-linear variation in turbidity suggests that salt concentration was not the sole or most direct factor influencing turbidity in this experiment. The SDI increased gradually with increasing salt concentration. Specifically, the SDI increased from 6.343 (2 ppt) to 5.373 (10 ppt). Salt can alter the ionic strength of a solution, affecting the stability of colloidal particles and suspensions. This can lead to the aggregation of small particles into larger clusters or, conversely, fragmentation of larger particles, thereby affecting the possibility of filter clogging. If the salinity is in the range of 2–10 ppt, it creates favorable conditions for the growth of microorganisms (algae, bacteria), these organisms can form biofilm on the filter membrane surface or contribute to the amount of suspended particles, increasing SDI and causing membrane fouling.
Water quality index at different salt concentration levels.

Note. The values a, b, c represent the differences of the filter materials at (p < 0.05).
Hermawan et al. treated brackish water by combining local filter materials such as activated carbon, sand, coconut shell charcoal, pumice, kaolin, red brick, zeolite, and seashell with RO technology. The special feature is the application of Internet of things (IoT) technology to monitor and control the treatment process, helping to optimize efficiency and operation. 18 The effectiveness of two brackish water treatment methods using locally available filter materials such as activated carbon, sand, gravel, kaolin, and RO membranes was compared. The study was able to evaluate factors such as cost and efficiency of removing impurities (salinity, turbidity, ions), thereby determining whether the water treatment process using RO equipment is more effective than locally available materials. 19 Another study in 2024, Idrissi et al, focused on selecting effective pretreatment methods for brackish water before proceeding with the main desalination processes (such as RO). The study thoroughly analyzed the physical and chemical composition of brackish water from rural areas in Morocco, using pretreatment methods such as sedimentation, coagulation, activated carbon filtration, sand filtration, or even micro/ultrafiltration, to remove impurities that could clog membranes or reduce the efficiency of subsequent treatment steps. 20
Conclusion
The research process has shown the distinct role of each type of filter material in improving water quality. Specifically, activated carbon filter material stands out with its ability to remove color at a filter error of 5 µm (8.3 TCU) and turbidity (0.5 NTU), providing high efficiency in removing colored compounds and suspended particles, contributing to making water clearer and eliminating unwanted aesthetic factors. Meanwhile, Pyrolox filter material is proven to have the strongest impact on reducing DO index with 4.657 mg/L, which is especially important in applications that require oxygen concentration control to prevent oxidation. For membrane filtration system protection, Greensand filter material with a SDI of 3.52 (0.2 µm) is lower than the remaining materials. However, the study also showed that the studied filter media did not have a significant impact on basic chemical parameters such as pH, TDS, and hardness. In summary, each type of filter media has its own advantages for specific water treatment objectives. The selection of the appropriate filter media should be based on the requirements for the output water quality and the parameters to be improved.
Footnotes
Authors’ Note
The authors declare that this manuscript has been read and approved by all authors, and that the content represents honest and original work.
Author contributions
Van Phuoc Nguyen: Writing—original draft. Quoc Khuong Anh Nguyen and Thi Kim Ngan Tran: Writing—original draft and Writing—review & editing. Chi Sy Phung: Data curation. Xuan Thanh Bui: Conceptualization.
Ethical considerations
Not applicable.
Consent to participate
Not applicable.
Consent for publication
Not applicable.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research is funded by Nguyen Tat Thanh University, Ho Chi Minh City, Vietnam under grant number 2024.01.128.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability statement
The data supporting the findings of this study are available from the corresponding authors upon reasonable request.
