Abstract
Three efficient, general approaches were investigated for the total synthesis of cis-(3,3-dimethylcyclopropane-1,2-diyl)dimethanol (cis-DMCP) (

Introduction
Dimethylcyclopropane is an important and interesting organic moiety that has unexpectedly been found in many natural and non-natural bioactive molecules and exhibits robust biological activity. 1 3-Carene, a natural bicyclic monoterpene, occurs coniferous oleoresins and aromatic essential oils.2,3 This starting material has been reported to have pluripotent biological effects, notably as an antimicrobial,4–6 antioxidant,5–8 anticancer,9,10 semiochemical,11,12 and fumigant properties.13,14
(+)-3-Carene is particularly interesting because of reactivity originates from two electron-rich sites: the olefinic C=C linkage and the strained gem-dimethylcyclopropane scaffold. Therefore, (+)-3-Carene has been used a dimethylcyclopropane source for the synthesis of many drugs,15–18 such as (+)-ingenol,
19
2,2-dimethyl-1,3-disubstitutedcyclopropane,
20
Δ-9-tetrahydrocannabinol,
21
a 5-lipoxygenase-activating protein (FLAP) modulator,
22
the first seco-casbane diterpene (EBC-329) (Figure 1),
23
boceprevir (Figure 2),
24
the sex pheromone of the Madeira mealybug (Figure 3),
25
and tefluthrin (Figure 4).
26
Therefore, in this work, we develop the synthesis of cis-DMCP(

Structure of EBC-329.

Structure of boceprevir.

Structure of the madeira mealybug sex pheromone.

The structure of tefluthrin.
Results and discussion
In this study, three efficient, general approaches were investigated for the total synthesis of cis-DMCP (

Influence of the reaction temperature.
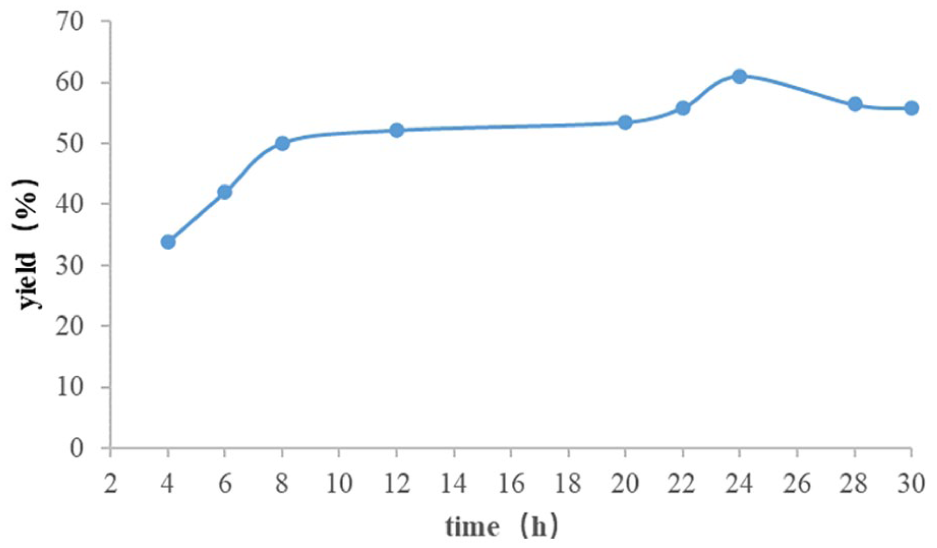
Influence of reaction time.

Catalyst loading-dependent reaction yield.

Influence of the amount of TBHP product yield.
The synthesis of compound
Influence of KMnO4 and KMnO4/SiO2..

In this study, three approaches to synthesize the target product
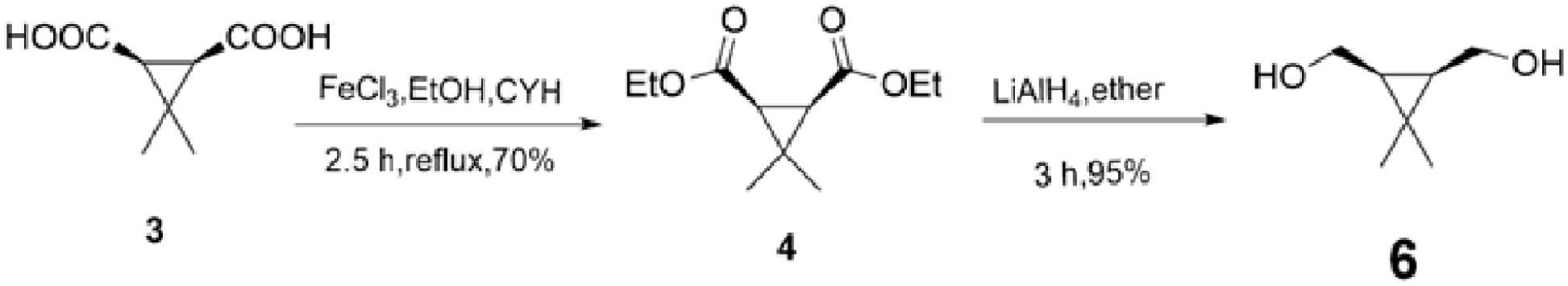
In the synthetic route, cis-3,3-dimethylcyclopropane-1,2-dicarboxylic acid (

Synthesis of (3,3-dimethylcyclopropane-1,2-diyl) dimethanol.
Synthesis of cis-DMCP.
In this synthetic route, compound
In the second synthetic route: cis-6,6-dimethyl-3-oxabicyclo[3.1.0]hexane-2,4-dione (

Synthesis of cis-DMCP.
We used acetic anhydride as the dehydrating agent and compound
In the synthetic route, compound

Synthesis of cis- DMCP.
The direct reduction of diacid
Experimental section
Unless explicitly purified, commercial starting materials and commercial reagents were employed without further purification. Powdered activated carbon (AR), (+)-3-carene (1) (RG), tetrahydrofuran (THF), and diethyl ether (Et₂O) were performed via Na/benzophenone. Flash chromatography used 200–300 mesh silica gel (FCC (SiO₂, 200–300 mesh). The 1H and 13C nuclear magnetic resonance (NMR) spectra were acquired on a Bruker Avance 500 MHz spectrometer. NMR data (δ, multiplicity): s, d, t, q, m. FT-IR spectra were acquired on an Shimadzu Japan Affinity-1 spectrometer. High-resolution mass spectra (HRMS) were acquired spectrometer (Bruker solari X 70 FT-MS) using ESI ionization.
CoPc/Al2O3 was prepared according to previous methods. 28 CoCl2·6H2O (7.14 g, 0.03 mol) was delivered to a stirred suspension of Al2O3 (50 g, 0.49 mol) in ethanol. The mixture was macerated for 24 h at room temperature (rt), filtered and dried before being placed in a muffle furnace at 300°C for 4 h and then cooled to rt. Maceration was then repeated as described above CoCl2·6H2O, o-phthalic anhydride, and urea were mixed at a molar ratio of 0.25:1:25, with a final mass percentage of ammonium molybdate of 1%. The samples were then ground, mixed well and melted to yield a thick, purple–black, which was placed in a high-temperature muffle furnace set to 240°C for 4 h. The crude CoPc/Al2O3 was washed repeatedly with water, ethanol, and acetone.
Synthesis of compound 2
CoPc/Al2O3 (0.15 g) and TBTP (3.00 g) were added to (+)-3-carene (
Synthesis of compound 3
(−)-3-Carene-2,5-dione (
Synthesis of compound 4
Ferric chloride (0.12 g, 0.70 mmol) and ethanol (1.00 g, 21.71 mmol) were charged to a solution of compound 3 (1.00 g, 6.32 mmol) in cyclohexane(CYH) (30 mL) and the suspension was heated to reflux at 65 °C for 2.5 h. Then the solution was washed successively with water (10 mL), 15 wt% aqueous sodium hydrogen carbonate (NaHCO3) (15 mL) and sodium chloride brine (10 mL). The organic phase was then dried over anhydrous Na2SO4, followed by solvent evaporation under vacuum to afford a faint-yellow mobile oil (0.95 g, 4.43 mmol, 70%), which was pure enough to be used in the next step. IR (KBr): v 1739 (C=O), 1159(C-O-C) cm-1; 1HNMR (500 MHz, CDCl3): δ 4.12 (q, 4H, J=7.0), 1.84 (s, 2H), 1.40 (s, 3H), 1.23 (t, 6H, J=7.0), 1.20 (s, 3H, CH3); 13CNMR (125 MHz, CDCl3): δ 169.00, 60.4, 32.0, 28.0, 25.7, 15.4, 14.1; HRMS (ESI) for C11H18O4Na [M+Na] calcd 237.1103; found: 237.1099.
Synthesis of compound 5
In a 50 mL reaction vessel assembled with a reflux apparatus and desiccant tube, compound 3 (0.50 g, 3.16 mmol) and redistilled acetic anhydride (0.53 g, 5.19 mmol) were added. The mixture was mixed gently in an oil bath with a magnetic stirrer until a clear solution was obtained, after which the reaction was refluxed at 140 °C for 3 h. The complete assembly was removed from the oil bath, followed by solvent evaporation under vacuum, giving the compound
Synthesis of compound 6 (一)
Compound 4 (0.50 g, 2.33 mmol) dissolved in anhydrous ether (10 mL) was dripped slowly into an ice-cold stirred suspension of LiAlH4 (0.53 g, 13.99 mmol) in anhydrous ether (20 mL) under N₂. The reaction liquid was heated to reflux at 35°C for 3 h. With the reaction complete, the reaction mixture was cooled to 0°C, quenched dropwise with water (1 × 0.53 mL) and 15 wt% aqueous NaOH (3 × 0.53 mL) after which water (1 × 0.53 mL) was added to minimize exotherm. The gelatinous precipitate was filtered off, and washed with dichloromethane (DCM) (50 mL). The filtrate was dehydrated with anhydrous Na2SO4, followed by solvent evaporation under vacuum to obtain the compound 6. Purification by FCC(SiO2, EtOAc/pet. ether = 4: 1) afforded compound
Synthesis of compound 6 (二)
Compound 5 (1.00 g, 7.12 mml) dissolved in anhydrous THF (20 mL) was dripped slowly into a ice-cold vigorously stirred LiAlH4 (0.60 g, 15.70 mmol) suspension in anhydrous THF (20 mL) under N₂. The reaction liquid was warmed to rt and agitated for 30 min. When the reaction had finished, the liquid was cooled to 0°C and cautiously quenched dropwise with water(1 × 0.6 mL) and 15 wt% aqueous NaOH (3 × 0.6 mL), after which water (1 × 0.6 mL) was introduced cautiously. The gelatinous precipitate was filtered off, and washed with DCM (20 mL). Water adsorption by anhydrous Na2SO4, followed by solvent evaporation under vacuum to provide the crude product. Purification by FCC(SiO2, EtOAc/pet. ether = 4: 1) afforded compound
Synthesis of compound 6 (三)
Compound 3 (2.00 g, 12.64 mmol) dissolved in anhydrous THF (30 mL) was dripped slowly to a ice-cold s vigorously stirred LiAlH4 (2.80 g, 73.77 mmol) suspension in anhydrous THF (30 mL) over 4 h under N₂. The mixture was maintained at reflux (68°C) for 4 h. After full conversion, the reaction was cooled to 0°C and cautiously quenched dropwise of water (1 × 2.8 mL), 15% aqueous NaOH (3 × 2.8 mL) and then a single aliquot of water (1 × 2.8 mL) was carefully introduced. Gelatinous precipitate was filtered off, and DCM-washed (40 mL). The filtrate was desiccated over anhydrous Na2SO4, and followed by solvent evaporation Purification by FCC(SiO2, EtOAc/pet.ether=4:1) afforded compound
Conclusion
In summary, we presented efficient synthetic route for the synthesis of cis-(3,3-dimethylcyclopropane-1,2-diyl)dimethanol from 3-carene. The key synthetic step is the selective allylic oxidation of (+)-3-carene using CoPc/Al2O3 as the catalyst. In addition, three approaches to synthesize target product
Footnotes
Ethical considerations
This study required no ethical approval as it involved neither human participants nor animal subjects.
Consent to participate
This study did not involve human or animal subjects, and no consent to participate was required.
Consent for publication
All participants provided consent for the publication of their data in this journal.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability statement
The data used to support the findings of this study are included within the article.
