Abstract
The purpose of the research was to synthesize hybrid molecules based on the imide group by preparing diazonium salts, perform coupling reactions for these salts, and study the biological activities of the synthesized compounds. Therefore, three innovative heterocyclic imide compounds (

Keywords
Introduction
There has been a steady increase in the synthesis of hybrid molecules and their evaluation as powerful medications and a variety of pharmacological agents.1,2 In the fields of chemistry and medicine, the most common chemical work to develop modified scaffolds with several enhanced and remarkable features is hybrid molecules, which are produced by combining the structural characteristics of two differently active segments.3,4 In general, these compounds are antibacterial against a variety of Gram-negative pathogenic bacteria.5,6 It has been reported that several compounds with anticancer, antimalarial, antibacterial, antidiabetic, anti-inflammatory, and antioxidant effects are based on heterocyclic sulfonamide derivatives.7,8
Sulfonamides are chemical compounds with a benzene ring S-linked to a distinctive nitrogen and sulfur dioxide group. The biological and pharmacological actions of sulfonamide and its derivatives have drawn a lot of attention. 9 Sulfonamides are widely used substances with a wide range of applications in the literature. 10
The promising antibacterial potential of sulfonamide derivatives against both Gram-positive and Gram-negative bacteria has been underlined by recent investigations (2022–2025).11,12 Strong action against Klebsiella pneumoniae was demonstrated by sulfonamide-based indole derivatives, while azithromycin-sulfonamide conjugates improved efficacy against Streptococcus pyogenes-resistant bacteria. 13 Against a variety of diseases, novel 7-methoxyquinoline sulfonamide compounds showed strong antibacterial and antibiofilm properties. Furthermore, broad-spectrum antibacterial properties were demonstrated by sulfonamides with oxyacetal and pyrimidine moieties. In addition, recent quinoline–sulfonamide hybrids demonstrated potent inhibitory activity against a variety of bacterial strains, highlighting the significance of structural alterations to enhance antibacterial efficiency. These results imply that sulfonamide derivatives continue to be a useful platform for creating novel antimicrobial medicines, particularly those that target resistant bacterial species. 14
In the current treatment plan for sulfonamide therapy, prodrug sulfonamides are crucial. 15 Sulfonyl succinyl, for example, acts as a prodrug of sulfathiazole. Because it ionized in the alkaline environment of the colon and was gradually hydrolyzed by gut enzymes, it was used to treat gut infections. 16 The amide group increases the sulfonamide’s hydrophilic properties and decreases its polarity. It makes it easier for the medication to pass through the intestinal wall and be broken down by enzymes. 17
In addition to their use in medicine, sulfonamide derivatives are widely used as dyes, plasticizers, corrosion inhibitors, and synthetic fibers. 18 Similarly, antitumor and antiulcer medicines are made from benzene sulfonamide and its derivatives. 19 Therefore, this study aimed to the preparation and characterization of hybrid functionality molecular-based varieties of heterocyclic rings and investigated their correlation between compound structures and their biological activities.
Material and methods
All the chemicals applied in our study are obtainable from Fluka. And for Sigma Aldrich, the melting points have been specified by Electro thermal capillary apparatus. Infrared spectra were obtained using the ATR technique Shimadzu 8400S, and Fourier Transform Infrared spectroscopy SHIMADZU in the range (400–4000) cm−1. The 1H-NMR spectra were obtained on a Bruker model ultra-shield 400 MHz in the laboratories of the University of Science and Technology (Tehran). Using tetra methyl silane (TMS) as an internal reference and DMSO-d6 as a solvent, mass spectrum analyses were performed by the Agilent Technology MS 5973 device.
Results and discussion
New compounds containing isoxazoles and benzothiazoles of heterocyclic rings have been synthesized successfully. Phthalic anhydride, maleic anhydride, and 1,8-naphthalic anhydride were used with 2-aminobenzothiazole as a starting material to prepare the target compounds.
To synthesize diazonium salts, two solutions were prepared: the first one containing of HCl dissolved in 2-aminobenzothiazole, and the second solution was sodium nitrite. The two solutions were mixed and stirred at 5 °C for 3 h. The end of the reactions was monitored by thin-layer chromatography (TLC). Then, the substituted benzaldehyde was added to obtain the target compounds (Scheme 1). The biological efficiency of the products was studied on four types of bacteria. They showed good biological activities toward Gram-positive and Gram-negative bacteria.

Synthesis route of cyclic imide compounds (
The prepared compounds were confirmed by Fourier transform infrared (FTIR) and 1H NMR through the appearance of the amide carbonyl stretching vibration and the absence of carbonyl group which clearly supported the formation of the target compounds (Tables 1 and 2).
20
However, the structure of the compounds (
FTIR spectrum data for cyclic imide derivatives (

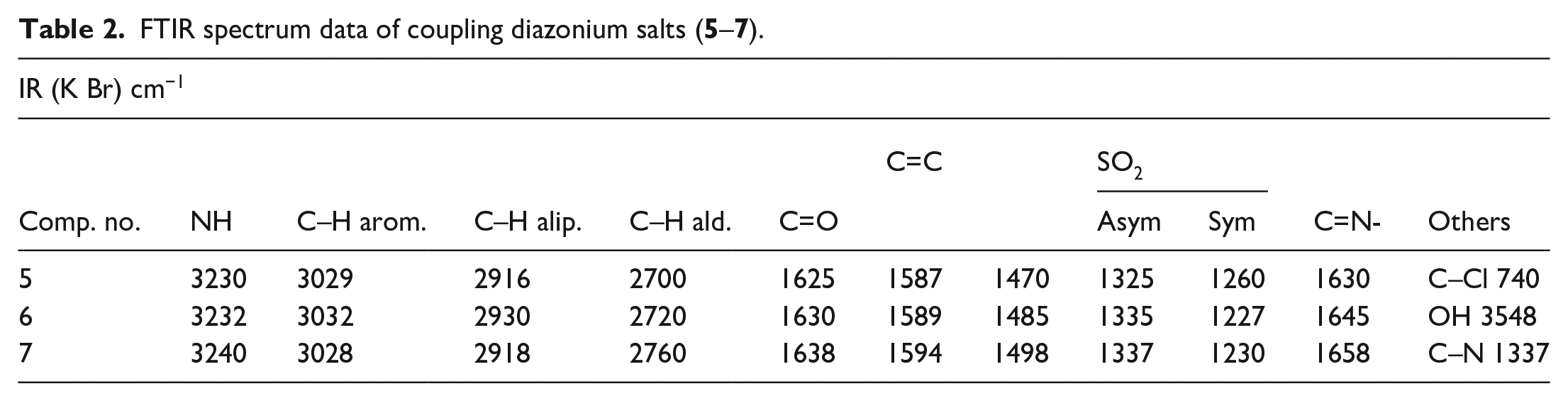
FTIR spectrum data of coupling diazonium salts (
Biological activity
The biological activity of the synthesized compounds was investigated by evaluating their effects on four bacterial species: two Gram-negative bacteria (E. coli and K. pneumoniae) and two Gram-positive bacteria (Staphylococcus aureus and S. pyogenes). Mueller-Hinton Agar (MHA) was used as the culture medium, which is commonly employed in determining the biological activities of antibiotics and chemical substances for medical use during the measurement of minimum inhibitory concentrations (MIC). The antibacterial activity data presented in Table 3 for the prepared compounds (
Antibacterial activity data for compounds (

The antibacterial activity data reveal that among the synthesized compounds (

Antibacterial activity of the prepared compounds at 10% concentration.
Therefore, compound
Besides, compound
Moreover, some of these compounds outperformed conventional drugs in combating certain bacterial species, as shown in Table 3 and Figure 2. In the study of biological activity, a comparison was made between various drugs and the synthesized compounds. Sulfamethoxazole demonstrated good efficacy at a 10% concentration against all bacterial species, with a particularly strong effect against K. pneumoniae (21 mm), surpassing most of the synthesized compounds. Naproxen exhibited moderate activity against S. aureus and S. pyogenes at the same concentration, but its impact on E. coli and K. pneumoniae was lower compared to the synthesized compounds. On the contrary, ibuprofen generally showed weak efficacy in comparison to the other drugs and synthesized compounds. Penicillin displayed strong activity against S. aureus at a 10% concentration (30 mm), although its effect on K. pneumoniae was less pronounced compared to the other drugs.

Biological activity of the prepared compounds: (a) compound
Experimental section
General procedure for synthesis of compound (1 –3 )
A mixture of compound
2-(2,5-dioxo-2,5-dihydro-1H-pyrrol-1-yl)-N-(5-methylisoxazol-3-yl)benzo[d]thiazole-6-sulfonamide (1 )
Bright yellow solid; yield: 1.53 g (75%); m.p. = 155–157 °C. 1H NMR (400 MHz, CDCl3) spectrum signal appeared at δ 8.3–7.8 (m, 3H, benzothiazole), δ -6.50 (s, 2H, pyrrole -2,5-dion (C=O), 6.10 (s, H, isoxazol). FTIR (KBr): 1636 cm−1 (C=O), 1736 cm−1 (C=N), 3066 cm−1 (C–H, ArH), 2992 cm−1 (C–H, alip.H), 1566–1460 cm-1 (C=C).
2-(1,3-dioxoisoindolin-2-yl)-N-(5-methylisoxazol-3-yl)benzo[d]thiazole-6-sulfonamide (2 )
Dark-yellow solid; yield: 78%, m.p. = 162–164 °C. FTIR (KBr): 1700 cm−1 (C=N), 1665 cm−1 (C=O), 3066 cm−1 (C–H, ArH), 2992 cm−1 (C–H, alip.H), 1585–1461cm-1 (C=C).
2-(1,3-dioxo-1H-benzo[de]isoquinolin-2(3H)-yl)-N-(5-methylisoxazol-3-yl)benzo[d]thiazole-6-sulfonamide (3 )
Orange solid; yield: 80%, m.p. = 174–176 °C. (KBr): 1636 cm−1 (C=O), 1736 cm−1 (C=N), 3066 cm−1 (C–H, ArH), 2992 cm−1 (C–H, alip.H), 1566–1460 cm−1 (C=C).
General procedure for synthesis of compounds (5 –7 )
Synthesis 2-(chlorodiazenyl)-N-(5-methylisoxazol-3-yl)benzo[d]thiazole-6-sulfonamide (4a )
2-((2-chloro-5-formylphenyl)diazenyl)-N-(5-methylisoxazol-3-yl)benzo[d]thiazole-6-sulfonamide (5 )
Light-yellow solid, yield: 70%; m.p. = 166–168 °C . FTIR (KBr): 1630 cm−1 (C=N), 1626 cm−1 (C=O) 3029 cm−1 (C–H, ArH), 2916 cm−1 (C–H, alip.H), 1587–1470 cm-1 (C=C) 2700 cm−1 (C–H, ald.H), 740 cm−1 (C–Cl).
2-((5-formyl-2-hydroxyphenyl)diazenyl)-N-(5-methylisoxazol-3-yl)benzo[d]thiazole-6-sulfonamide (6 )
Yellow solid; yield: 80%; m.p. = 170–172 °C. FTIR (KBr): 1630 cm−1 (C=N), 1626 cm−1 (C=O), 3032 cm−1 (C–H, ArH), 2930 cm−1 (C–H, alip.H), 1589–1485 cm−1 (C=C), 2720 cm−1 (C–H ald.H), 3548 cm−1 (C–OH).
((2-(dimethylamino)-5-formylphenyl)diazenyl)-N-(5-methylisoxazol-3-yl)benzo[d]thiazole-6-sulfonamide (7 )
Dark-orange solid; yield: 83%; m.p. = 178–180 °C. 1H NMR(400 MHz, CDCl3) spectrum signal appeared as follows: δ 9.66 (s, 1H, C H=O), δ 7.72–7.66 (m, 3H, ArH), δ 2.96–2.5 (s, 6H, C H3-N-CH3), δ 2. (s, 3H, C H3-C-O). FTIR (KBr): 1630 cm−1 (C=N), 1626 cm−1 (C=O), 3032 cm−1 (C–H, ArH), 2930 cm−1 (C–H, alip.H), 1589–1485 cm-1 (C=C), 2720 cm−1 (C–H ald.H), 3548 cm−1 (C–OH).
Conclusion
The current study focuses on the synthesis and characterization of heterocyclic imide compounds, as well as the synthesis and characterization of dual diazonium salts based on 2-aminobenzothiazole. This was achieved in two stages. The first stage involves the synthesis of cyclic imide compounds through the reaction of 2-aminobenzothiazole with various cyclic anhydrides. The second stage includes the reaction of 2-aminobenzothiazole with an HCl solution diluted with distilled water at a 1:1 ratio and a sodium nitrite solution in an ice bath to obtain diazonium salt as an intermediate stage. This salt was then converted into a pair or conjugation by reacting with benzaldehyde derivatives. The prepared compounds were confirmed using spectroscopic methods, and their efficiency was demonstrated in inhibiting four types of bacteria, two of which are Gram-positive and two are Gram-negative. The compounds
Supplemental Material
sj-docx-1-chl-10.1177_17475198251362960 – Supplemental material for Synthesis, characterization, and biological activity of some novel sulfonamide derivatives
Supplemental material, sj-docx-1-chl-10.1177_17475198251362960 for Synthesis, characterization, and biological activity of some novel sulfonamide derivatives by Bushra T Mahdi, Huda Turki Mahdi, Mohammed Hadi Ali Al-Jumaili and Malath Khalaf Rasheed in Journal of Chemical Research
Footnotes
Acknowledgements
The completion of this research paper would not have been possible without the support and guidance of the University of Anbar and the University of Fallujah. I would like to clarify that the H NMR spectra for compounds
Consent to Participate
The authors confirm that all authors participated in the research work of this manuscript.
Consent for Publication
The authors confirm that we give our full consent for publishing this manuscript.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
The data that support this study are accompanying as a supplementary material.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
