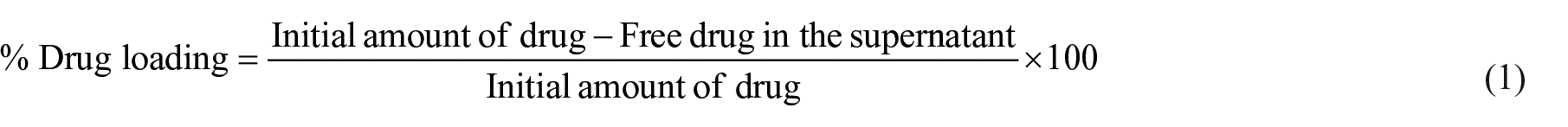Abstract
The present research highlights the preparation of multifaceted zinc oxide nanoparticles (ZnO NPs) using an eco-friendly, cost-effective, efficient, and clean approach, that is, quince seed-derived gel (QG). The characterization of the as-synthesized NPs was performed using UV-Vis spectroscopy, Fourier transform infrared (FTIR) spectroscopy, scanning electron microscopy (SEM), energy-dispersive X-ray spectroscopy (EDX), photoluminescence (PL) spectroscopy, and zeta potential analysis. The sulphadimidine drug was loaded on QG-mediated ZnO NPs to design drug-loaded NPs (DL-NPs). The cumulative drug release profile of DL-NPs and the antimicrobial efficacies of pure drug, ZnO NPs, and DL-NPs were evaluated. UV-Vis analysis established the surface plasmon resonance (SPR) peak at 370 nm, whereas PL study revealed the emission nature of the NPs. FTIR spectrum of ZnO NPs demonstrated distinct peaks at 885.24 and 457.49 cm–1, confirming tetrahedral coordination of Zn and ZnO bond formation, respectively. At the same time, DL-NPs exhibited a specific FTIR peak at 635.58 cm–1, indicating a shift resulting from interactions between ZnO NPs and the drug. The zeta potential exhibited the presence of a surface charge on the NPs. SEM revealed that the NPs have a spherical shape, and EDX analysis confirmed the elemental composition of the synthesized NPs. ZnO NPs were loaded with the drug with an entrapment efficiency of 72% ± 3.5%. In addition, the cumulative sulphadimidine release from DL-NPs was maximum after 2.0 h at pH 6.8 (54%) compared to pH 7.4 (35%), and no significant changes were observed thereafter. The antimicrobial assay revealed that the NPs, pure drug, and DL-NPs were potent against both Gram-positive (Bacillus subtilis and Bacillus licheniformis) and Gram-negative (Escherichia coli and S. shigella) bacteria. The MIC and MBC results showed that ZnO NPs and the drug were more effective in preventing the growth of Gram-negative bacteria, while DL-NPs were more impactful against Gram-positive bacteria.

Keywords
Introduction
Nanotechnology, a multidisciplinary field, has attracted attention due to its widespread implementation in medication delivery, agriculture, electronics, civil engineering, environmental management, and the food industry.1,2 The term “nanomaterial” denotes those having a size less than 100 nm, unlike their bulk materials. 3 Their structure, size, and physicochemical properties are the parameters that help in their categorization. Recently, the production of metal and metal oxide NPs, such as Ag, Au, CuO, Fe2O3, and ZnO, has been undertaken in nanotechnology.4 –7 Compared to other metal oxides, ZnO NPs are favorable due to their cost-effectiveness, safety, and UV-blocking properties. 8 They show far-reaching and promising expectations for bioimaging, gene delivery, cancer therapy, energy storage, photocatalytic, and packaging applications.9 –13
ZnO NPs are being studied extensively for drug delivery applications thanks to their easy fabrication, non-toxicity, extraordinary drug loading capability, controlled drug release, and targeted delivery.14,15 For instance, the encapsulation efficiency and cisplatin release from ZnO NPs were found to be 50.52% and 8.64%, respectively. 16 In another study, Paulownia tomentosa leaf extract-mediated and polyethylene glycol (PEG)-modified ZnO NPs demonstrated in vitro doxorubicin release profiles of approximately 25% (48 h) and 85% (24 h) at pH 7.4 and 5.5, respectively. 17 In addition, the quercetin release rates from phenyl boronic acid-conjugated ZnO NPs were observed to be approximately 13%, 28%, and 47% at pH levels of 5, 6, and 7.4, respectively, establishing their suitability as effective drug delivery vehicles. 18 ZnO NPs have also become a focal point of research as robust antimicrobial agents, thereby treating infectious diseases and addressing concerns related to multidrug resistance.19,20 These materials inhibit bacterial cells by generating reactive oxygen species and simultaneously inducing oxidative stress, which disrupts enzyme and protein synthesis pathways, distorts plasma membrane structure, and causes the leakage of extracellular content. 21
Various techniques, including mechanochemical, solvothermal, hydrothermal, precipitation, sol-gel, microemulsion, and vapor transport, have been explored for fabricating ZnO NPs. 8 However, these methods are problem-oriented as they produce hazardous by-products, demand high energy, and cause unintended health problems. 22 In this regard, green synthesis has come forward as a promising alternative to conventional approaches. It presents numerous merits, including reduced waste production, minimized pollution, renewable feedstock, and improved environmental and human safety.22,23
Quince (Cydonia oblonga), a small to medium-sized deciduous tree, is indigenous to the Hyrcanian forest region, 24 which can withstand a wide range of environmental conditions and grows at latitudes as far north as Scotland. The plant contains several phytochemicals and secondary metabolites, including terpenoids, tannins, sugars, organic acids, steroids, phenolics, flavonoids, and volatiles. 25 Plant seeds comprise mucilage, fixed oil, a small amount of cyanogenetic glycoside, and an enzyme that affects the hydrolysis of glycosides.26,27 Pharmacological activities, including cardioprotective, renoprotective, antibacterial, antioxidant, hypocholesterolemic, and anti-inflammatory effects, have been ascribed to various quince parts.25,28
Plant materials, including fruits, seeds, leaves, and roots, can serve as nanofactories for reducing metal ions and synthesizing NPs. For example, Solanum nigrum, Laurus nobilis, Arthrospira platensis, Myristica fragrans, and Spirogyra hyalina have been utilized in the preparation of ZnO NPs with multifaceted biological properties, including antibacterial, anticancer, antidiabetic, and free-radical scavenging effects.29 –33 However, additional substances might be necessary to lower the interfacial energy between the particles and control their growth. Natural polymers, such as latex, hydrogels, and gums, are key sources of structurally diverse bioactive compounds, which can be employed as natural reductants and surface protectors. Therefore, the current study represents a detailed assessment of QG as a facile reducing and capping agent. QG stands out for being environmentally friendly, cost-effective, widely available, and biocompatible, thus rendering the NPs well-suited for therapeutic applications. To date, no investigation has been documented regarding the antibacterial, drug-encapsulation, and controlled-release capabilities of quince seed gel-mediated ZnO NPs. The as-synthesized NPs were evaluated for their physicochemical characteristics using UV-vis, PL, SEM, EDX, FTIR, and a zeta size analyzer techniques. Furthermore, the efficiency of ZnO NPs as a carrier for Sulphadimidine sodium was examined. Sulphadimidine is an antibacterial drug used to treat animal and human infections, such as toxoplasmosis, coccidiosis, and chlamydiosis. 34 Afterward, the in vitro stimulus-responsive release of sulphadimidine sodium-loaded ZnO NPs was inspected to analyze their effectiveness for controlled therapy. Finally, the microbicidal potencies of the pure drug, bare NPs, and drug-loaded NPs were evaluated against both Gram-positive (B. subtilis and B. licheniformis) and Gram-negative (E. coli and S. shigella) bacteria.
Experimental
Materials
Quince seeds were obtained from a local authorised vendor, while zinc acetate, sodium hydroxide, potassium dihydrogen phosphate, and sulphadimidine sodium were procured from Sigma-Aldrich and used as received. The microbiology laboratory at Government College University, Lahore, Pakistan, provided the bacterial strains used in this study. Distilled water was utilized as a solvent for the preparation of buffer and other solutions.
Isolation of QG
Quince seeds (200 g) were washed, dried, and drenched in distilled water (300 mL) for 30 min. The mixture was subjected to heating (Simmerstat CH2090-001 PTFE plate) at 60-70 °C for 2.0 h until a homogeneous gel was formed. The formed gel was isolated from the mixture using nylon fabric and subsequently dried in an electric oven (IRMECO GmbH incubator model B-53) at 55 °C for 48 h. Thereafter, it was ground and kept in a vacuum desiccator.
Preparation of ZnO NPs
To proceed with the biogenic fabrication of ZnO NPs, a QG suspension (1.0%) was mixed with an aqueous solution of zinc acetate (50 mM) and heated at 60 °C for approximately 2 h. In the next step, the reaction mixture was dehydrated in the oven at 70 °C for 8.0 h. The created NPs were finally calcined (500 °C for 2.0 h) and stored for subsequent characterization.
Analytical characterization
The Cary-60 UV-spectrophotometer was utilized to validate the formation of ZnO NPs, while the emission nature of the NPs was detected through a Cary Eclipse fluorescence spectrophotometer. The surface functionalities of QG, ZnO NPs, drug, and DL-NPs were determined via an FTIR spectrophotometer (Nicolet iS5 Spectrophotometer, Thermo Scientific, UK) in the 4000-400 cm–1 range. A Zeta size analyzer (Anton Paar, Litesizer 500), equipped with special conducting cells, was employed to evaluate the stability and zeta potential of the NPs. The structure of the NPs was assessed using a Nova Nano-SEM (SEM-450), and the elemental composition was analyzed using an energy-dispersive X-ray (EDX) analyzer.
Applications of ZnO NPs
Various activities, including drug delivery and antibacterial effectiveness, were explored to understand the release profile of ZnO NPs-based delivery systems and assess their inhibitory effects against both Gram-positive and Gram-negative bacteria, ultimately paving the way for advanced biomedical strategies.
Drug delivery
In this study, in vitro drug loading and release assays were conducted under various pH environments to investigate the controlled and sustained delivery pattern of QG-mediated ZnO NPs. The underlying hypothesis is that ZnO NPs, upon exposure to the acidic conditions thriving in the small intestine, will undergo decomposition to liberate the encapsulated ingredient without premature leakage and compromising the therapeutic efficacy.
Drug loading
Sulphadimidine, an antibacterial drug, 35 was selected as a model. The drug was loaded by stirring ZnO NPs (100 mg) in an aqueous solution of the drug (50 mL) for 24 h at 25 °C. DL-NPs were centrifuged at 3500 rpm for 30 min and rinsed with distilled water several times. The DL-NPs were characterized through the FTIR technique and subsequently desiccated in the oven at 70 °C. The loading percentage was calculated spectrophotometrically by determining the initial drug concentrations and the remaining unbound drug in the supernatant after centrifugation. The encapsulation content (%) was determined using the following formula:
Drug release study
The in vitro drug release was studied via the dialysis bag method. 36 The DL-NPs (10 mg) were enclosed in a dialysis bag with a 12-14 kDa molecular weight cut-off, and the release profile was evaluated in a phosphate buffer (100 mL, pH 6.8) at 37 °C with continuous magnetic stirring. At specific time intervals (every hour), a sample of 2.0 mL was taken, and a fresh dissolution medium of 2.0 mL was added to maintain the volume and avoid sink conditions. The collected dialysates were then investigated for the free active ingredient using a UV-Vis spectrophotometer at 241 nm. A similar drug delivery procedure was performed at pH 7.4.
Antibacterial activity
The antibacterial activities of the pure drug, ZnO NPs, and DL-NPs were examined using an agar well diffusion assay against the aforementioned bacterial strains. The experimental procedure commenced with the creation of a non-selective/non-differential solid layer on petri plates, using Mueller-Hinton agar. After solidification, the bacterial culture with an adjusted concentration (McFarland turbidity standard 0.5) was swabbed onto the top of the solidified agar. Dimethyl sulphoxide was employed as a negative control, while the sulphadimidine sodium was taken as a positive control. A total of five wells were created in each plate, out of which three were filled with samples (100 µL) such as ZnO NPs and DL-NPs in their respective petri dishes, and the remaining two were filled with the negative control and the positive control (pure drug). The inhibition zone around each well was analyzed to assess the bactericidal competence of all samples after incubating the plates at 37 °C for 24 h. Each experiment was triplicated.
Determination of MIC and MBC
The MIC and MBC of three different samples, that is, ZnO NPs, pure drug, and DL-NPs, were estimated against 24 h old bacteria at different concentrations such as 10, 9.5, 9, 8.5, 8, 7.5, 7, 6.5, 6, 5.5, 5, 4.5, 4, 3.5, 3, 2.5, 2, 1.5, 1, and 0.5 mg/mL. The nutrient broth (3.0 mL), bacterial culture (30 µL), and various sample concentrations (ZnO NPs, drug, and DL-NPs) were added to their respective sterile test tubes, except for those used as controls. The cultures were kept at 37 °C for 24 h to calculate the optical density at 523 nm. MBC values were determined by dispersing the MIC samples (100 µL) onto an agar plate and then incubating them at 37 °C for 24 h. Each test was executed thrice.
Results and discussion
Synthesis and characterization
In this experimental work, ZnO NPs were prepared using QG as a natural and environmentally friendly reducing and capping agent. QG was treated with an aqueous solution of zinc acetate (50 mM) in subsequent heating, drying, and calcination steps. The formation of ZnO NPs was verified by the appearance of white-colored precipitates. 33 The bioconstituents (polysaccharides, proteins, and minor compounds such as flavonoids, alkaloids, phenols, steroids, tannins, and terpenoids) present in QG are hypothesized to act as electron donors, reducing Zn2+ into Zn0 atoms. After undergoing nucleation and oxygenation, metal atoms were simultaneously capped and stabilized by biological entities, leading to the formation of ZnO NPs. A recent investigation reported a similar mechanism for synthesizing ZnO NPs utilizing Abelmoschus esculentus mucilage. 37 The plausible mechanism involved in synthesizing ZnO NPs using QG is shown in Figure 1.

Schematic illustration of the synthesis of ZnO NPs using QG.
UV-visible analysis
UV-Vis spectroscopy is one of the most accessible methods for determining the reduction of Zn salt into ZnO NPs. The absorption spectrum revealed a prominent peak at 370 nm (Figure 2), which is believed to arise from the nucleation of ZnO NPs.38,39 This peak corresponds to the collective oscillation of the conduction electrons undergoing excitation on the surface of the analyzed NPs by absorbing light at a specific wavelength. 40 In addition, the band gap energy of ZnO NPs at 370 nm was found to be 3.357 eV, as calculated using the given equation:

UV-visible spectrum of ZnO NPs fabricated using QG.
Herein, the determined value aligns well with prior research.41,42 It is known that the optical response of the NPs largely depends on the synthesis method. ZnO NPs synthesized using biofabrication generally exhibit higher UV-Vis absorbance and a blue shift in the absorption maxima, which can be attributed to the smaller particle size and quantum confinement property. Conversely, ZnO NPs prepared via chemical reduction typically exhibit excitonic absorption peaks above the band gap wavelength of their counterparts (red shift), implying that the particle size increases, thereby causing a decrease in the absorption.43,44
Photoluminescence study
PL spectroscopy is a non-destructive and contactless technique for determining the luminescence properties and surface defects of nanostructured semiconductors. The emission behavior of ZnO NPs was assessed as a function of the excited photon energy over a wavelength range of 200-600 nm (Figure 3). An intense broad peak was observed at approximately 270 nm, resulting from the near-band-gap excitonic emission. 45 Herein, the results align with the existing literature, which indicates that nanocrystalline ZnO exhibits an excitation wavelength in the UV region (220-270 nm). 46 Biogenic ZnO NPs display higher PL intensity than those obtained through chemical synthesis. 47 In addition, weak-to-moderate peaks centered around the 420-550 nm wavelength range, including 423, 460, 485, and 537 nm, were located, representing greater availability of singly ionized oxygen vacancies. 48 The occurrence of sub-peaks in the visible region is typical for ZnO NPs. This is likely due to the recombination of a deeply trapped hole with a delocalized electron near the conduction band,49,50 suggesting that the prepared sample contains impurity phases. Overall, the disparity in the optical properties of ZnO NPs might be associated with surface defects, variations in particle size/surface morphology, or the presence of impurities (biological molecules from QG), ultimately leading to shifts in the absorption edges (UV-Vis spectrum)/inducing additional emission peaks (PL spectrum).

The PL spectrum showing the emission behavior of ZnO NPs.
Surface chemistry of QG, ZnO NPs, drug, and DL-NPs
The active functional groups of QG, QG-capped ZnO NPs, pure drug, and DL-NPs were identified using FTIR spectroscopy. QG displayed significant absorption peaks at 3320, 2926, 2855, 1736, 1593, 1417, 1244, and 1038 cm–1. The broad peak at 3320 cm–1 was attributed to the –OH stretching, 51 while two stretching frequencies at 2926 and 2855 cm–1 were assigned to –CH bonds, 52 thereby revealing the occurrence of polysaccharide molecules in the gel. 53 The vibrations at 1736, 1593, and 1417 cm–1 illustrated the C=O, anti-symmetric COO, and symmetric COO stretching modes, respectively. The band at 1244 cm–1 corresponded to the pectin’s ether and ring (C-C) bonds. 54 A prominent peak at around 1038 cm–1 was consistent with the oligosaccharide linkage of sapogenins (C-O-C). 55 The FTIR spectrum profile of QG-ZnO NPs was also analyzed to establish that the NPs were reduced, capped, and stabilized by the biomolecules of QG. The synthesis of ZnO NPs was confirmed by the distinguishing bands at 885 and 457 cm–1, which agreed well with the Zn tetrahedral coordination and the stretching vibration of ZnO, respectively.56,57 The peaks at 3388 cm–1 (-OH stretching), 2930 cm–1 (C-H stretching), 1422 cm–1 (COO symmetric stretching), and 1031 cm–1 (oligosaccharide linkage) demonstrated shifts due to interactions occurring during the NPs formation. Hence, ZnO NPs showed various characteristic peaks of QG with some nominal shifts, suggesting the strong binding ability of the gel’s functional groups with the NPs via hydrogen bonding, van der Waals forces, or electrostatic interactions. The FTIR spectra of QG and biosynthesized ZnO NPs are presented in Figure 4.
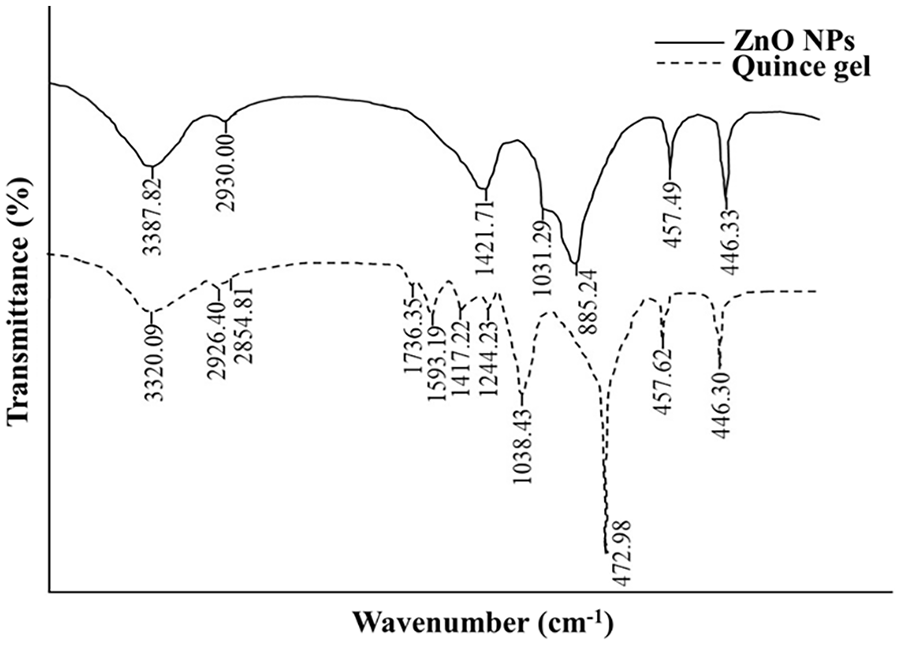
FTIR spectra showing functional groups of QG and QG-ZnO NPs.
The FTIR spectrum of sulphadimidine sodium revealed a series of significant bands at 3564, 3335, 1597, 1571, 1372, 1298, and 822 cm–1 (Figure 5). The presence of the NH group in the drug was confirmed by stretching vibrations at 3564 and 3335 cm–1. 58 Absorptions corresponding to the stretching frequency of the phenyl ring (C=C) were centered at 1597 and 1571 cm–1. 59 At the same time, the peaks at 1372 and 1298 cm–1 established the symmetric and asymmetric S=O stretchings in the sulphonamide group, respectively.59,60 A medium band at 822 cm–1 appeared in response to two adjacent aromatic hydrogen atoms. 61 Similarly, in the case of ZnO NPs loaded with sulphadimidine, distinct peaks were observed at 3490, 3363, 3154, 1639, 1583, and 1559, 1377, 1243, and 833 cm–1 corresponding to OH, NH, CH, C=O, C=C, S=O symmetric, and S=O asymmetric vibrations and adjacent aromatic hydrogen atoms, respectively. The encapsulated product also exhibited a band of ZnO at 635.58 cm–1, 62 indicating the presence of ZnO NPs within the sample (Figure 5). However, encapsulation induced changes in the positions and intensities of all vibration bands, confirming the interactions between the functional groups of the drug and DL-ZnO NPs.
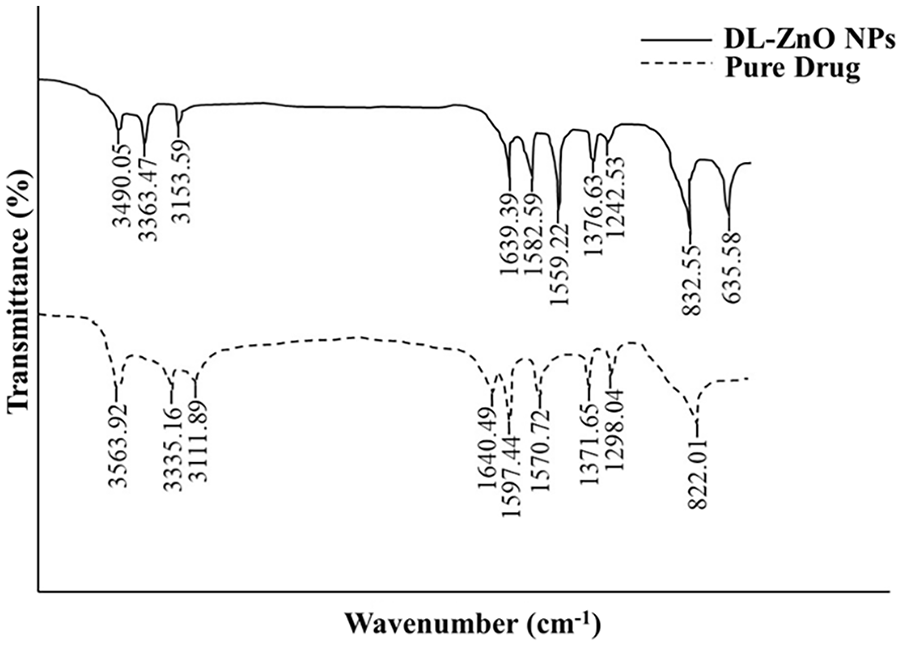
FTIR spectra of sulphadimidine drug and DL-NPs.
Zeta potential of ZnO NPs
Zeta potential provides insight into the physical stability of biosynthesized NPs based on their surface charge and corroborates their dispersion capacity. 33 A zeta potential value of at least 15 mV is a practical guideline for determining the stability of colloids. The present study calculated an average zeta potential value of −21.85 mV, as shown in Figure 6, representing that as-synthesized ZnO NPs are stable and uniformly dispersed in an aqueous colloidal phase. The negative charge may be due to the potential capping of the NPs with negatively charged polyhydroxy compounds in the gel. 63 In a relevant study, a similar zeta potential value of ZnO NPs was measured at −22.1 mV, ensuring consistency with the literature. 33

Zeta potential analysis demonstrating the stability of ZnO NPs.
Morphological nature of ZnO NPs
SEM is the most commonly employed electron microscopy technique for characterizing the surface morphology of the NPs at the micro- and nano-levels. 64 The size distribution and structure of ZnO NPs were analyzed at different magnifications (Figure 7(a) and (b)). As per the results, the biosynthesized NPs were spherical and uniformly distributed. The average particle size, determined using ImageJ software, was found to be 231 nm (Figure 7(c)). Herein, the outcomes closely align with the previous literature reports, where spherical ZnO NPs were produced in the size range of 50–200 nm.65,66 In another investigation, ZnO NPs were reported to have an average diameter of 228 nm. 67 These studies suggest resonance behavior and validate our findings regarding the successful synthesis of ZnO NPs.

SEM micrographs of ZnO NPs at different magnifications (a, b), size distribution curve (c), and EDX pattern (d) of ZnO NPs synthesized using QG.
The qualitative and quantitative analyses of as-prepared NPs were performed using the EDX method. The detector revealed prominent peaks corresponding to Zinc and oxygen elements, with weight percentages of 58.59% and 21.95%, respectively (Figure 7(d)). Owing to the presence of bioconstituents of QG and impurities in ZnO NPs, additional elements, including calcium, carbon, magnesium, potassium, chlorine, and aluminium, also appeared in the EDX spectrum. The results are consistent with those of previous studies.32,42
Applications of ZnO NPs
Drug loading and drug delivery
The loaded drug content on ZnO NPs was determined using an analytical method authenticated by UV-visible spectroscopy, after creating a calibration curve ranging from 50 to 350 ppm (r² = 0.9798). Herein, the loading efficiency was 72 ± 3.5% after 24 h. The outcomes validate earlier research, which found that green (Rhodococcus pyridinivorans) and chemically synthesized ZnO NPs showed relatively similar loading percentages for quercetin (72.4%) and anthraquinone (79%).68,69 The study proposes biogenic ZnO NPs as a suitable platform for encapsulating drugs or active ingredients.
The in vitro release of sulphadimidine from DL-NPs was quantified in phosphate buffer at pH 6.8 (an acidic environment similar to the small intestine) and pH 7.4 (normal physiological conditions), with a temperature of 37 °C to ensure experimental conditions were identical to those in the body fluid. Initially, the drug diffused out of the ZnO NPs and passed through the sealed dialysis membrane, while the NPs, being larger, were retained inside. The dialysis membrane functions similarly to ultrafiltration, allowing selective passage of specific materials through size exclusion based on the pore size. 70 The present investigation demonstrated a rapid discharge of sulphadimidine until 2.0 h, which remained nearly unchanged afterward. The release rates corresponding to 2, 6, 9, 12, 15, 18, 21, and 24 h were 54%, 60%, 64%, 68%, 72%, 78%, 81%, and 85%, respectively at pH 6.8, while at pH 7.4, approximately 35%, 42%, 47%, 52%, 58%, 63%, 67%, and 73% of the encapsulated drug was discharged, respectively (Figure 8).

Cumulative drug release at pH 6.8 and 7.4.
In vitro delivery of doxorubicin from ZnO NPs resulted in a release rate of 65% within the initial 5 h, followed by sustained release for the subsequent 24 h. 71 In another study, ZnO NPs loaded with curcumin exhibited a biphasic release profile, characterized by a fast unloading phase within the initial 12 h and a slow, constant diffusion over a prolonged period. 72 The release profile is strongly influenced by the temperature and pH of the medium, which can induce its disruption and result in the burst discharge of the drug molecule.73,74 At pH 6.8 (an acidic environment), sulphadimidine was liberated due to the dissociation of metal-ligand bonds, 75 ensuring high therapeutic efficacy. Meanwhile, the drug carrier might remain stable under normal physiological conditions, exhibiting minimum release. Therefore, ZnO NPs may be effective drug delivery systems for treating bacterial infections.
Antibacterial assay
Antibiotic resistance and the resulting risk of disease spread are major public health concerns in both developing and developed countries. The number of effective antimicrobial therapies is limited due to the rise of new and multidrug-resistant infections. Addressing the global challenge of antibiotic resistance requires a new supply of eco-friendly and biocompatible materials, such as plant-mediated NPs.76 –78 In the present investigation, the antibacterial efficacy of QG-mediated ZnO NPs, DL-NPs, a positive control (pure drug), and a negative control was evaluated against Gram-positive bacteria (B. subtilis and B. licheniformis) and Gram-negative bacteria (E. coli and S. shigella). According to the results, notable growth delay and bactericidal effects were witnessed with the application of ZnO NPs, DL-NPs, and the positive control (Figure 9). In contrast, the bacterial growth was not disturbed by the negative control.
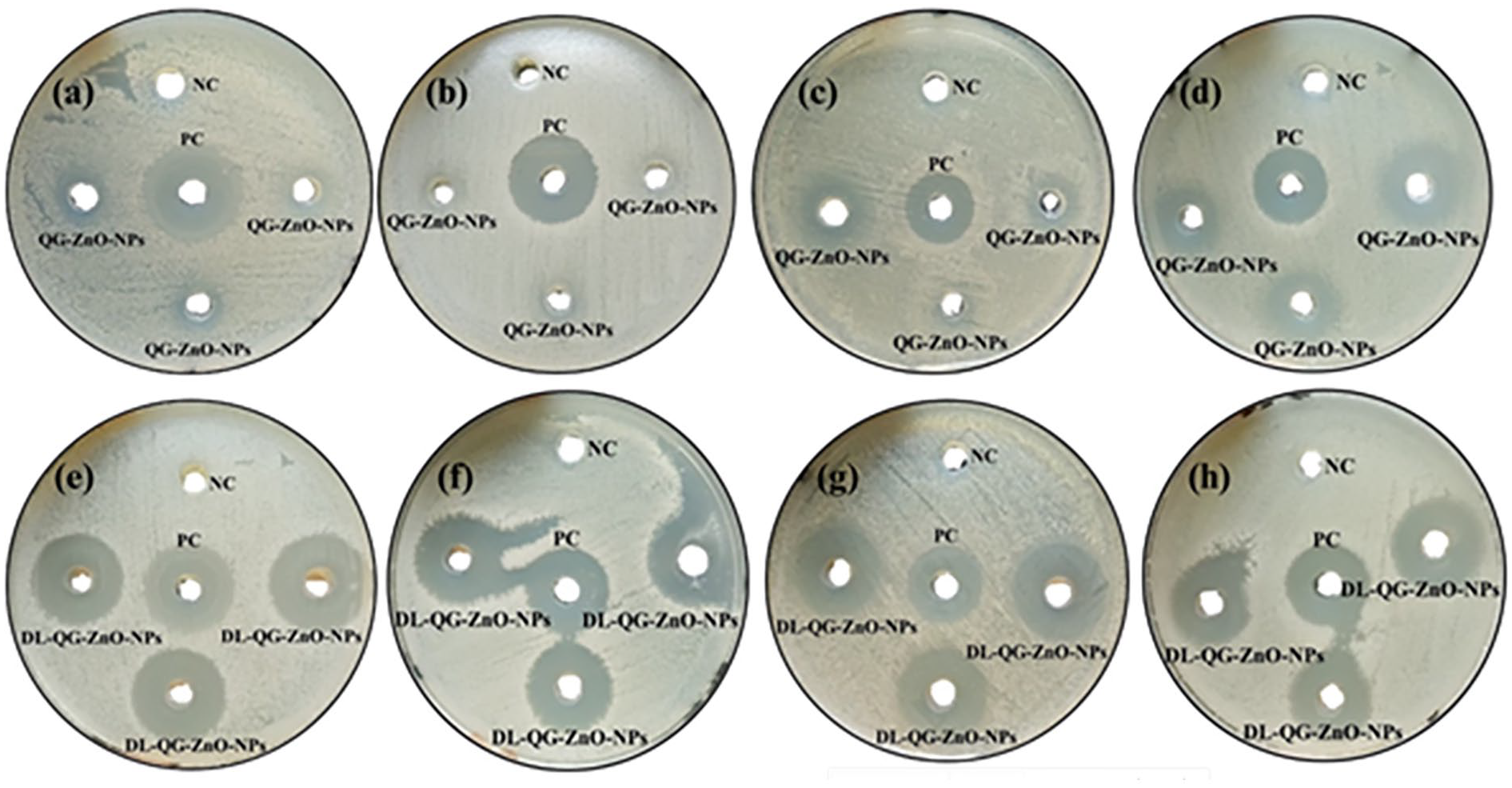
Bactericidal potential of ZnO NPs (a), (b), (c), and (d) and DL-NPs (e), (f), (g), and (h), evaluated using agar wall diffusion method.
In particular, pure drug and DL-NPs exhibited a pronounced antibacterial potential, in contrast to ZnO NPs. DL-NPs were potent against both Gram-positive and Gram-negative bacteria, with negligible differences in ZOI values. The ZnO NPs showed significant ZOI against all bacteria except B. licheniformis. Both the drug and ZnO NPs were observed to control the growth of Gram-negative bacteria more effectively than that of Gram-positive bacteria. One possible explanation is that the Gram-negative bacteria have a thinner peptidoglycan layer, making them more vulnerable to membrane damage. 79 Gram-positive bacteria, on the other hand, possess a thick layer of peptidoglycan, which provides robust structural support and additional stability to the cell wall.80,81 The antimicrobial capacities of pure ZnO NPs and NPs developed as a neodymium delivery system were validated against two bacterial isolates, namely E. coli and Klebsiella pneumoniae. The results suggested that neodymium-coated ZnO NPs unveiled superior bactericidal effects compared to pure NPs. The impacts of the tested samples on bacterial species were assumed to depend on several factors, including the generation of reactive oxygen species and the release of Zn2+. 82 Quercetin-capped ZnO nanorods appeared to inhibit E. coli substantially than their bare counterparts, including ZnO and pure quercetin. 68 Another study demonstrated the significant bactericidal potential of ZnO NPs and multiple-drug-conjugated ZnO NPs against MDR bacteria, 83 highlighting their usefulness in reducing pathogenic infections. The antibacterial activities of ZnO NPs, DL-NPs, and the pure drug, as determined using the agar well diffusion method, are presented in Table 1.
Antibacterial Activities of QG-ZnO NPs, DL-NPs, and the Pure Drug, Particularly on ZOI (mm).

ZnO NPs combat bacterial invasion via different mechanisms such as the disruption of the cell wall, protein, lipid as well as DNA, impairment of cell membrane permeability, leakage of cellular contents, reactive oxygen species (H2O2, O22–, OH–) generation, and the release of Zn2+ ions, ultimately leading to damage and cell death.82,84 Figure 10 illustrates a potential mechanism underlying the antibacterial activity of ZnO NPs.

Mechanism underlying the antibacterial action of QG-mediated ZnO NPs.
Next, the MIC and MBC values were investigated for ZnO NPs, DL-NPs, and the pure drug against all types of bacteria, as disclosed in Table 2. It has been found that DL-NPs revealed enhanced inhibition and bactericidal ability against the tested bacteria. ZnO NPs more effectively targeted B. subtilis, although typically to a lesser extent than DL-NPs, but more significantly than that of the pure drug. The Gram-positive bacteria were more susceptible to DL-NPs, while ZnO NPs and the drug were more influential against Gram-negative bacterial strains. MBC values were higher than MIC, demonstrating that a lower concentration is required to inhibit growth than to kill bacteria. The results suggest that encapsulating ZnO NPs with an antibacterial drug enhances their ability to eradicate bacterial populations. Hence, DL-ZnO NPs may offer an attractive solution for combating difficult-to-treat infections.
MIC and MBC Values of QG-ZnO NPs, DL-NPs, and the Pure Drug.

Conclusion
The present research focuses on the green, straightforward, and cost-effective production and characterization of ZnO NPs using UV-vis, FTIR, PL, zeta potential analyzer, SEM, and EDX analyses. In the UV-Visible absorption spectrum, a prominent peak at 370 nm was observed for the NPs. FTIR analysis indicated that the surface chemistry of QG played a crucial role in the functionalization of the NPs. The emission behavior and an average zeta potential (−21.85 mV) of as-prepared NPs were confirmed through PL and zeta size analyzer techniques, respectively. The SEM pattern revealed a spherical shape, while the EDX method detected zinc (58.59%) and oxygen (21.95%) as the core elements in the obtained sample. The drug loading efficiency was approximately 72%, with a cumulative release rate of 54% and 35% at pH 6.8 and 7.4, respectively, after 2 h. Thereby, ZnO NPs are potent nanocarriers for the controlled delivery of antibiotics with maximum efficiency under acidic conditions. Besides the drug release study, ZnO NPs, pure drug, and DL-NPs presented profound antibacterial potential. DL-NPs were potent against both Gram-positive and Gram-negative bacteria, while the drug and ZnO NPs inhibited Gram-negative bacterial strains more significantly. Lower concentrations of ZnO NPs, pure drug, and DL-NPs were required to compare MIC against MBC, exhibiting vigorous bacteriostatic activity. Collectively, these findings indicate that ZnO NPs functionalized with specific moieties could pave the way for developing antimicrobial delivery systems with enhanced therapeutic outcomes.
Footnotes
Ethical considerations
This article does not contain any studies with human or animal participants.
Consent to participate
All authors have agreed to participate.
Consent for publication
All authors have read the final manuscript and agreed to its publication.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability statement
All data generated or analyzed during this study are included in this manuscript and will be available upon request from the authors.