Abstract
A novel thermoregulated phase-separable Pt nanocatalyst stabilized by ionic liquid [CH3(OCH2CH2)22N+Et3][PF6−] (ILPEG1000-PF6) was prepared and first explored for the selective hydrogenation of quinoline. The conversion of quinoline and selectivity of 1,2,3,4-tetrahydroquinoline were both >99% at 130 °C under 3 MPa for 1.5 h, and the ILPEG1000-PF6 stabilized Pt nanocatalyst can be reused four times efficiently. In addition, the ILPEG1000-PF6 stabilized Pt nanocatalyst was also extended to the selective hydrogenation of 3-methylquinoline, 6-methylquinoline, 8-methylquinoline, and 6-chloroquinoline with >99% conversion and selectivity, respectively.
Keywords

Introduction
Soluble transition-metal nanocatalyst (TMNC) has attracted much attention as a result of high activity and selectivity. However, it also suffers from the problem of separation from the reaction system, which is quite similar to the traditional homogeneous catalysts.
1
To solve this problem, a series of biphasic system were developed.2
–4 Ionic liquids were increasingly investigated in the catalysis over the last few years5
–7 and used to establish biphasic catalytic system involving TMNC, for examples, supported ionic liquid phase system8,9 and ionic liquid biphasic system.
10
In our previous studies, a thermoregulated phase-separable (TPS) catalysis system composed of ionic liquid [CH3(OCH2CH2)nN+Et3][CH3SO3−] (
Tetrahydroquinolines have a wide range of synthesis applications in medicines, pesticides and fine chemicals.15,16 Selective hydrogenation of quinolines has been recognized as a feasible method for the preparation of tetrahydroquinolines owing to atom efficiency (Scheme 1).17,18 Various TMNC were reported for selective hydrogenation of quinolines, such as Pd, 19 Au, 20 Ru, 21 Rh, 22 and Pt23 –29 (Table 1). Among these TMNC, the Pt nanocatalyst drew much attention due to its prominent capability for H2 activation.30,31 In our previous work, [CH3(OCH2CH2)22N+Et3][CH3SO3−] (ILPEG1000) stabilized Pt nanocatalyst was explored as catalyst for selective hydrogenation of quinolines, but the catalytic activity needs further improvement. 32 Herein, a novel ionic liquid [CH3(OCH2CH2)22N+Et3][PF6−] stabilized Pt nanocatalyst (abbreviated as ILPEG1000-PF6-Ptnano), which possess the TPS property, was studied in the selective hydrogenation of quinoline as well as its derivatives with the aim to further improve the catalytic activity of Pt nanocatalyst.

Selective hydrogenation of quinolines.
The catalyst reported for the hydrogenation of quinoline in literatures.

Experimental section
Materials and reagents
The ionic liquid ILPEG1000 was synthesized as reported method.
33
Toluene,
Characterizations
The 1 H NMR and 31 P NMR spectra were obtained from Bruker Avance II 400, the 13 C NMR was recorded on Bruker Avance Ⅲ 500. Transmission electron microscopy (TEM) was measured by JEM-2000EX instrument (120 kV). GC was conducted on Tianmei 7900 GC equipped with OV-101 column and flame ionization detector (N2 as carrier gas). GC-MS was conducted on Agilent 7000B instrument equipped with DB-1701 column (He as carrier gas). Inductively Coupled Plasma–Atomic Emission Spectrometry (ICP-AES) analysis was recorded on PerkinElmer Avio 220 instrument. The UV-Vis spectra were carried out by Flash SP-Max 2300A.
Preparation of ILPEG1000-PF6
Typically, ILPEG1000 (2.36 g, 2 mmol) and KPF6 (0.37 g) were mixed with 30 mL deionized water in a flask (50 mL capacity). The mixture was extracted by 3 × 30 mL of CH2Cl2 after stirring for 12 h under RT and dried with MgSO4. Then, CH2Cl2 was removed and the obtained ILPEG1000-PF6 was dried under vacuum.
1
H NMR (400 MHz, CDCl3): δ 1.32 (t,
Preparation of ILPEG1000-PF6-Ptnano
In a typical procedure, an aqueous solution of H2PtCl6·6H2O (1 mL, containing 7.4 × 10-4 mmol of Pt) and 0.90 g ILPEG1000-PF6 were added to a 75 mL Teflon-lined stainless-steel autoclave. The mixture was stirred at 70 °C for 2 h to evaporate water. After that, the autoclave was flushed and pressurized up to 4.0 MPa with H2, and heated along with stirring at 120 °C for 2.5 h. After cooling and depressurization, a brown ILPEG1000-PF6-Ptnano was obtained.
Hydrogenation of quinoline and its derivatives
Take quinoline hydrogenation as an example, 0.115 g quinoline, 0.200 g
Hg poisoning experiment
Hg (0.15 g, Hg/Pt molar ratio = 1000) was added to the autoclave involving the as-prepared ILPEG1000-PF6-Ptnano and stirred under RT for 2 h, subsequently the hydrogenation was performed at 130 °C under 3 MPa for 1.5 h.
Results and discussion
The studies started with the preparation and characterization of the ILPEG1000-PF6-Ptnano. The ILPEG1000-PF6-Ptnano was prepared by the method of hydrogen reduction and analyzed firstly by TEM. As depicted in Figure 1, the newly obtained Pt nanocatalyst displayed a mean diameter of 2.4 ± 0.3 nm, indicating good stabilization of the ILPEG1000-PF6. Furthermore, the UV-Vis absorption spectra of the ILPEG1000-PF6-Ptnano (Figure 2) showed that the characteristic peak disappeared completely after reduction, which also indicated the formation of Pt0. 34
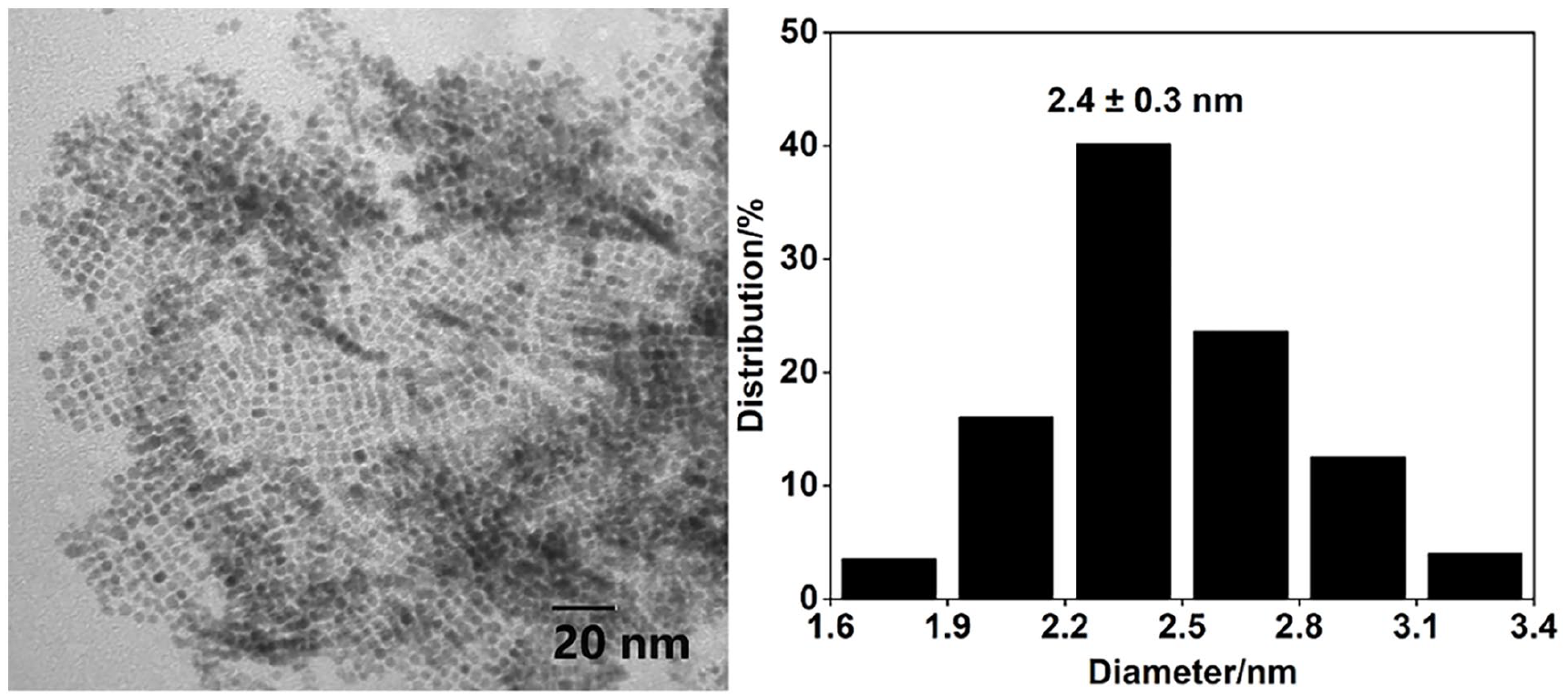
TEM image and particle size distribution of the newly prepared Pt nanocatalyst (the molar ratio of ILPEG1000-PF6/Pt = 1000).
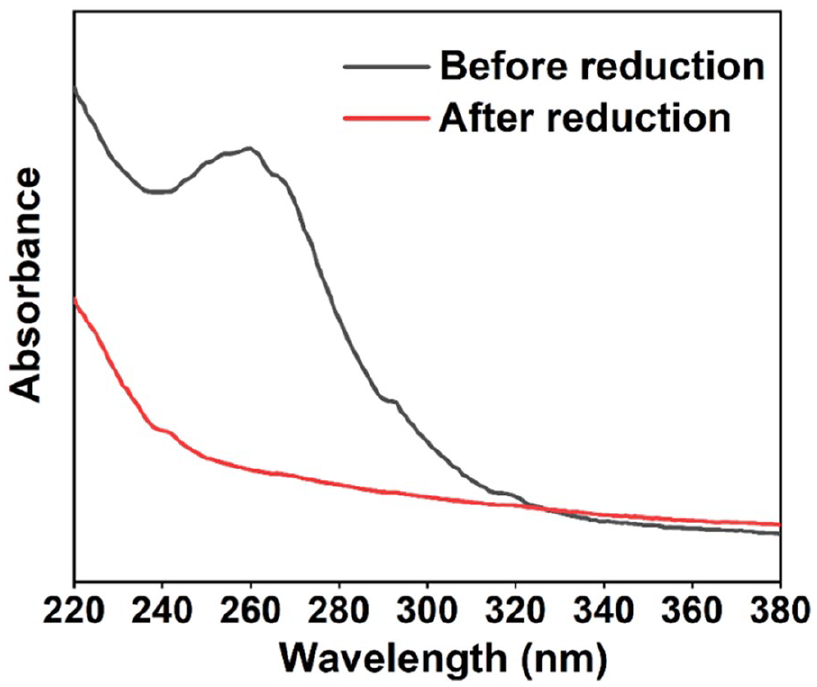
UV-Vis absorption spectra of the ILPEG1000-PF6-Ptnano.
The schematic depiction of the structure of Pt with ILPEG1000-PF6 was displayed in Figure 3. The PF6− anions interact with Pt nanocatalyst preferentially by electrostatic force and form a protective layer around the Pt surface, and the cations of ILPEG1000-PF6 form the second protective layer to protect them from aggregation.5,6

Schematic depiction of the structure of Pt with ILPEG1000-PF6.
After preparation and characterization of the ILPEG1000-PF6-Ptnano, the following experiments were carried out to certify that the ILPEG1000-PF6-Ptnano possessed the TPS property in the mixed solvent (toluene/

TPS property of ILPEG1000-PF6-Ptnano in the mixed solvent (toluene/
With the above results in hand, the reactivity of ILPEG1000-PF6-Ptnano in the selective hydrogenation of quinoline was explored, and the results are displayed in Table 2. The influence of reaction temperature was first explored from 80 ℃ to 130 ℃. The >99% conversion was achieved as the temperature increased to 130 ℃ and the selectivity of 1,2,3,4-tetrahydroquinoline was >99% (Table 2, entry 4). Then the influence of time was studied. As the reaction time changed from 0.5 to 1.5 h (Table 2, entries 4–6), the conversion gradually increased to >99% and the selectivity of 1,2,3,4-tetrahydroquinoline remained >99%. Subsequently, the influence of H2 pressure on the reaction was examined (Table 2, entries 4, 7, 8), >99% conversion of quinoline was achieved when the H2 pressure was 3 MPa. This may be due to the fact that high pressure results in high hydrogen solubility in the organic phase, thus benefits the increasing of catalytic activity. Next, the influence of quinoline/Pt molar ratio was investigated (Table 2, entries 4, 9, 10). The conversion of quinoline decreased to 83% as the molar ratio of quinoline/Pt increased to 2000. The high molar ratio of quinoline/Pt may lead to a decreasing of H2 at the Pt catalyst surface and therefore becomes unfavorable to the hydrogenation of quinoline. The influence of ILPEG1000-PF6/Pt molar ratio was finally investigated. When the molar ratio of ILPEG1000-PF6/Pt was 500, although the >99% conversion and selectivity could be achieved (Table 2, entry 11), the Pt nanocatalyst was agglomerated after the reaction because of the poor stability. Moreover, the conversion decreased to 90% when the ILPEG1000-PF6/PF molar ratio was 1500 (Table 2, entry 12). The possible reason for this could be the diffusion barrier of quinoline. Therefore, the turnover frequency (TOF) value was 784 h−1 under optimal reaction conditions (Table 2, entry 4). Considering that the anion of ionic liquids may influence reaction activity,35,36 the reactivity of ILPEG1000 stabilized Pt nanocatalyst was also investigated and showed a much lower catalytic activity (3% conversion of quinoline) compared with the ILPEG1000-PF6-Ptnano (Table 2, entries 4, 13). This may be due to the weakly coordinating effect of anion PF6−, 37 which favors the absorption of H2 and quinoline, therefore benefits the increasing of catalytic activity.
Selective hydrogenation of quinoline catalyzed by ILPEG1000-PF6-Ptnano a .

Reaction conditions: Pt 7.4 × 10-4 mmol,
Conversion of quinoline, determined by GC and GC-MS.
Selectivity of 1,2,3,4-tetrahydroquinoline, determined by GC and GC-MS.
TOF: moles of 1,2,3,4-tetrahydroquinoline formed per mole of Pt per hour.
The ILPEG1000 was used as stabilizer.
With the above experimental results in hand, the reusability of ILPEG1000-PF6-Ptnano was evaluated. Due to TPS property, the ILPEG1000-PF6-Ptnano can be simply separated from the product after reaction and reused in the next cycle. As shown in Figure 5, the ILPEG1000-PF6-Ptnano could be reused four cycles with >99% conversion and selectivity. The conversion was decreased at the fifth cycle. The possible reason for the decrease in conversion were further investigated. The TEM image indicated that the mean diameter of Pt nanocatalyst increased to 3.1 ± 0.6 nm after four cycles (Figure 6). Besides, the ICP-AES results indicated the Pt leaching in the organic phase gradually decreased in each cycle, the mean leaching of Pt was 0.68 wt% (Table 3). Therefore, the increasing of Pt nanocatalyst particle size and the leaching of Pt may lead to the decreased catalytic performance at the fifth cycle.
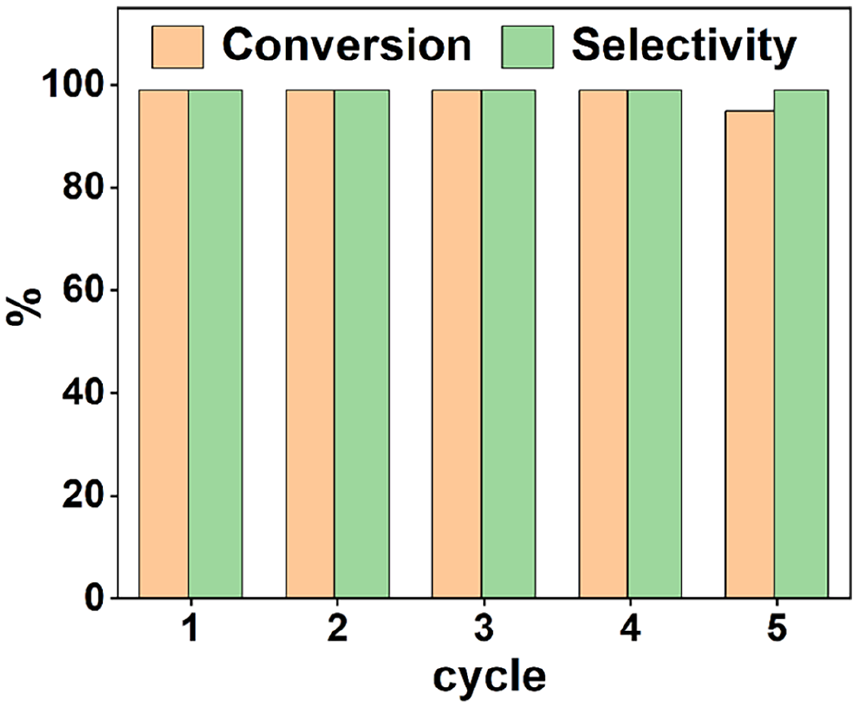
The reusability of ILPEG1000-PF6-Ptnano in the selective hydrogenation of quinoline. Reaction conditions: Pt 7.4×10-4 mmol, ILPEG1000-PF6/Pt = 1000 (molar ratio), quinoline (0.115 g),

TEM image and particle size distribution of Pt nanocatalyst after four cycles (the molar ratio of ILPEG1000-PF6/Pt = 1000).
The Pt leaching in the organic phase.

In order to get further insight in this catalytic reaction, Hg poisoning experiment was carried out to determine whether the ILPEG1000-PF6-Ptnano is homogeneous or heterogeneous. As shown in Table 4, the ILPEG1000-PF6-Ptnano was deactivated by adding Hg before the reaction, the conversion of quinoline was reduced from >99% to 2%. Therefore, the ILPEG1000-PF6-Ptnano might be heterogeneous in this catalytic process. 38
Hg poisoning experiment a .

Reaction conditions: Pt 7.4×10-4 mmol, ILPEG1000-PF6/Pt = 1000 (molar ratio), quinoline (0.115 g),
Conversion of quinoline, determined by GC.
Reaction without adding Hg.
To explore the generality of ILPEG1000-PF6-Ptnano, 3-methylquinoline, 6-methylquinoline, 8-methylquinoline, and 6-chloroquinoline were employed as substrates. As shown in Table 5, the above-mentioned substrates afforded the corresponding tetrahydroquinolines with >99% conversion and selectivity under the optimized reaction conditions.
Selective hydrogenation of quinoline derivatives catalyzed by ILPEG1000-PF6-Ptnano a .

Reaction conditions: Pt 7.4 × 10-4 mmol, ILPEG1000-PF6 (0.60 g),
Conversion of substrate, determined by GC and GC-MS.
Selectivity of product, determined by GC and GC-MS.
Conclusion
A novel ILPEG1000-PF6-Ptnano with TPS property was synthesized and applied to catalyze the selective hydrogenation of quinoline and its derivatives. The ILPEG1000-PF6-Ptnano was easily reused four cycles that still maintained catalytic activity and selectivity. Further catalytic applications of ILPEG1000-PF6-Ptnano in hydrogenation of other substrates will be carried out in the future.
Footnotes
Author contributions
Mingyang Liu: Investigation, Writing—original draft, Writing—review & editing.
Yanhua Wang: Supervision, Writing—review & editing.
Consent to participate
Not applicable.
Consent for publication
Not applicable.
Data availability
The data generated and analyzed during the current study are available from the corresponding author on reasonable request.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Natural Science Foundation of China (21173031).
Ethical consideration
Not applicable.

