Abstract
Seven metabolites (

Introduction
The Scutellaria genus, commonly known as Skullcap, belongs to the Lamiaceae family and includes numerous species found in temperate and tropical regions worldwide. These regions encompass North China, Japan, Russia, Pakistan, Taiwan, the Philippines, Thailand, Vietnam, and New Guinea.1,2 Within this diverse genus, some species are cultivated for ornamental purposes, while others have recognized medicinal properties. All of them are categorized as herbaceous plants.1,3 Skullcap species have contributed to a wide array of natural compounds, including steroids, flavonoids, phenolic acids, iridoids, alkaloids, phenylpropanoids, clerodane diterpenoids, triterpenes, lignans, phytosterols, tannins, polysaccharides, and essential oils.4,5 These plants have been utilized in traditional medicine practices for millennia. 1 For example, in traditional Siberian medicine, the aerial parts of the plant are used to address various ailments such as nausea, dyspepsia, ascites, high blood pressure, gastrointestinal issues, bleeding, malaria, and acute respiratory infections. 6 Similarly, Tibetan traditional medicine incorporates skullcap to treat conditions like tachycardia, myocarditis, pneumonia, gastrointestinal diseases, polyarthritis, and kidney and liver pain. 7 In addition, skullcap-based remedies are known to alleviate headaches and insomnia, dilate blood vessels, and reduce arterial pressure, potentially reducing the risk of heart disease.8,9
The Scutellaria genus encompasses 300 to 350 species, among which Scutellaria indica stands out due to its rich phytochemical profile, with approximately 61 compounds isolated from the plant. 6 Research has highlighted various health benefits of S. indica, including its potential anti-cancer, 10 anti-arginase, 11 anti-inflammatory, 12 antitumor, 13 and antiviral properties. 14 However, within the existing literature, only one study has investigated the inhibitory effects of flavonoids from this plant on nitric oxide (NO) production. 12 There is a noticeable research gap in the study of phenolic acid glycosides, particularly phenylethanoid glycosides isolated from S. indica, and their NO production inhibitory activity. Based on the existing research void, the chemical constituents of S. indica and their inhibition of NO production were investigated.
Results and discussion
Chemical structure elucidation
The S. indica methanolic extract was partitioned into n-hexane, ethyl acetate, and butanol-soluble fractions. Chromatographic purification of the ethyl acetate and butanol-soluble fraction led to the isolation of seven compounds (

Structure of the isolates (1–
The 1H-NMR spectrum of compound
Compound
Compounds 3–
NO production inhibition
In the first experiment, to determine the non-toxicity of the isolates (1–

Compounds
In the next experiment, the anti-inflammatory activity of the isolates (
In vitro inhibitory activity of the isolates (

L-NMMA: N(G)-monomethyl L-arginine.
Results are represented as IC50 value (µM).
Positive control; The data are expressed as the mean ± SD of three replicates (n = 3).
P < 0.05 compared with the control group.
It is important to note that the control group in this experiment was not exposed to either Lipopolysaccharide (LPS) or the samples. Therefore, any observed inhibitory effects of the isolated compounds on NO production were not influenced by cytotoxic impact. When the control group was stimulated with LPS (1 µg/mL), NO production increased approximately 11-fold after 24 hours. In contrast, compound

Inhibition of LPS-induced NO production in RAW 264.7 cells by compound
In our study, scrocaffeside A (
Molecular docking
Scrocaffeside A (
Interaction and binding affinity between compounds

Compounds and iNOS interactions.
Molecular docking results of the iNOS.

The binding affinity and interactions of compounds

Compounds and COX-2 interactions.
Molecular docking results of the COX-2.

The binding affinity and interactions between studied compounds (

Compounds and IL-8 interactions.
Molecular docking results of the IL-8.

Scrocaffeside A (
In this experiment, Lipinski’s 27 rule of five was employed to evaluate a compound’s druggability. According to this rule, a compound with good drug-likeness should have a molecular weight (MW) below 500 Daltons, fewer than 5 hydrogen bond donors (HBD), fewer than 10 hydrogen bond acceptors (HBA), and a log P value less than 5 (Table 5).
Analysis results of physicochemical property.

MW: molecular weight; log P: log of octanol/water partition coefficient; nHBD: number of hydrogen bond donor(s); nHBA: number of hydrogen bond acceptor(s); TPSA: total polar surface area; MR: molar refractivity; log S: log of solubility; nRotB: number of rotatable bond(s).
In addition, aqueous solubility (log S), the number of rotatable bonds, molar refractivity (MR), and total polar surface area (TPSA) were also assessed. For optimal oral bioavailability and intestinal absorption, the TPSA value should not exceed 140 Å, the MR value should range between 40 to 130, and the number of rotatable bonds should be below 10 (Table 5).
27
Scrocaffeside A (
Table 6 presents the in silico predictions of the absorption, distribution, metabolism, and excretion (ADME) properties of the studied compounds. Both scrocaffeside A (
Absorption, distribution, metabolism, and excretion (ADME) predictions results.

GI Abs: gastrointestinal absorption; BBB per: blood-brain barrier permeability; P-gp: P-glycoprotein; CYP: cytochrome P.
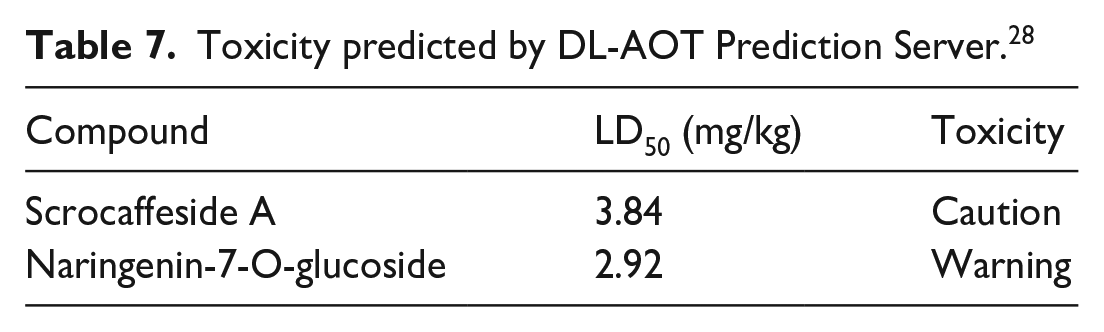
Table 7 results indicate that the LD50 values of (
Toxicity predicted by DL-AOT Prediction Server. 28
Limitations of the study
Limitations of the study include the exclusive focus on isolating compounds (
Conclusion
In conclusion, the phytochemical investigation of the entire S. indica from Vietnam has resulted in the isolation of seven metabolites, namely scrocaffeside A (
Experimental
Chemistry
General experimental procedure, plant material, extraction and isolation, and physiochemical and NMR data of isolated compounds (
Biology
Cell culture, cell viability assay, and determination of NO production, please see Supplemental materials file.
Molecular docking study
Preparation of proteins and ligands
The crystal structures of iNOS, COX-2, and IL-8 are available in the Research Collaboratory for Structural Bioinformatics (RCSB) Protein Data Bank (PDB ID: 3E7G, 5IKQ, and 5D14, respectively) and were downloaded. UCSF Chimera 1.17.3 was used to prepare the proteins and ligands. The “Dock Prep” tool removed all non-standard residues, added polar hydrogen atoms, and assigned Gasteiger charges to the proteins. The prepared proteins and ligands were saved in PDB format.
Molecular docking
AutoDock Vina 1.2.5, integrated into UCSF Chimera, conducted the molecular docking. The CASTp 3.0 server identified the active sites of the proteins. Grids were centered to encompass all residues identified by CASTp. 29 The docking process used a maximum of 10 conformers based on the Broyden-Fletcher-Goldfarb-Shanno algorithm. Default parameters of AutoDock Vina were employed, and conformers were ranked by binding free energy. Conformers with the lowest binding free energy were selected. AutoDock Vina was executed on a Windows 10 Pro operating system with a 2.53 GHz Intel Core i5 processor.
Absorption, distribution, metabolism, excretion, and toxicity predictions
All compounds underwent screening based on Lipinski’s rule of five for drug-likeness. The SwissADME web tool calculated pharmacokinetic parameters for the compounds. In addition, acute oral toxicity predictions were performed using the DL-AOT prediction server.27,28,30
Supplemental Material
sj-docx-1-chl-10.1177_17475198241272457 – Supplemental material for Nitric oxide production inhibitors from Vietnamese Scutellaria indica: An in vitro and in silico study
Supplemental material, sj-docx-1-chl-10.1177_17475198241272457 for Nitric oxide production inhibitors from Vietnamese Scutellaria indica: An in vitro and in silico study by Dao Cuong To, Phi-Hung Nguyen, Le Minh Hoang, Hoa Thi Nguyen, Truong Thi Viet Hoa, Truong Thi Thuy Nhung, Phuong Dai Nguyen Nguyen, Ngu Truong Nhan, Hong Khuyen Thi Pham and Phu Chi Hieu Truong in Journal of Chemical Research
Footnotes
Acknowledgements
The authors thank Dr Nguyen Quoc Binh for his help in the identification of the plant materials.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was financially supported by the Vietnam Academy of Science and Technology under the grant number: UDPTCN 03/21-23.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
