Abstract
Tachysterol is an intermediate product in the synthesis of vitamin D2 from ergosterol and vitamin D3 from 7-dehydrocholesterol under ultraviolet irradiation. Recent studies have shown that tachysterol is also a crucial intermediate in the synthesis of the natural anti-tumor product Chaxine C. Although there have been numerous studies on the synthesis of vitamin D2 and other photoisomers from ergosterol, research on the synthesis of tachysterol is relatively limited. In addition, the yield of tachysterol is not high (57.0%), and the mechanism is unclear. In this study, a method was developed for the qualitative and quantitative analysis of tachysterol. Meanwhile, an efficient synthesis process for tachysterol was reported. The qualitative and quantitative analysis of ergosterol isomers, including tachysterol, a precursor of vitamin D2, and photosterol, was conducted using high-performance liquid chromatography, and the quantitative method for tachysterol was confirmed using quantitative nuclear magnetic resonance. There were strong linear relationships between tachysterol content and the testing parameters, with the correlation coefficients of 0.9997 and 0.9971, respectively. The synthesis process of tachysterol from ergosterol through our self-designed photoreactors was optimized, and the effects of reaction temperature, reaction time, photoprotectants, reaction substrate concentration, and the power of ultraviolet lamps were investigated. The yield of the reaction was 85.7%, which is 28.7% higher than the previously reported results. The mechanism and by-products of the reaction were further studied using ultraviolet spectrophotometry and gas chromatography mass spectrometry. This study explored the potential for the large-scale preparation of tachysterol, particularly for the total synthesis of the anti-tumor natural product Chaxine C. It also offered insights into the synthesis of structural similar compounds and the analysis of photocatalytic reaction products.

Introduction
Photochemistry studies the changes caused by the interaction between matter and light. It refers to the phenomenon where light promotes molecules from the ground state to the excited state.1,2 Photochemical synthesis unlocks a unique reaction pathway that cannot be achieved through traditional synthesis methods. Photochemical synthesis has demonstrated favorable reaction outcomes in the synthesis of specific molecular isomers.3,4
It is well known that vitamin D is essential for human health, and its synthesis is induced by solar UV (ultraviolet) radiation on the skin’s epidermis.5–7 Tachysterol is an intermediate product in the synthesis of vitamin D2 or vitamin D3 from ergosterol or 7-dehydrocholesterol under UV irradiation. Since Havinga and his colleagues have made significant efforts in studying the photoreaction mechanism of previtamin D photosynthesis, we now have a better understanding of vitamin D research (see Figure 1).8,9 The conversion of ergosterol to vitamin D2 occurs in two stages. First, ergosterol undergoes cleavage and opens the B-ring under UV irradiation, producing previtamin D2. It is heat-labile and is converted into vitamin D2 through heat induction. 10 Previtamin D2 triggers the formation of the other two isomers under continuous UV irradiation. One possibility is that the B-ring of previtamin D2 closes to form lumisterol, which has a structure similar to ergosterol. Another possibility is that the A-ring of previtamin D2 rotates 180° to form tachysterol, which has a structure similar to vitamin D2. When exposed to UV light for an extended period, compounds such as supersterols, 11 toxisterols,12,13 and other by-products are also formed.

Schematics of ultraviolet photolysis of ergosterol.
According to the study, it was found that the proportion of photoreaction products in the stable state was influenced by the UV wavelength of irradiation. The short wavelength (248–254 nm) primarily produces tachysterol, the 296.5 nm wavelength contributes to the formation of previtamin D2, and the longer wavelength (> 305 nm) is conducive to the production of lumisterol.14–16 These studies mentioned the synthesis wavelength of tachysterol, but did not delve into the synthesis process, particularly due to the lack of suitable synthesis equipment and low yields.
Due to their structural similarity, photoisomers such as tachysterol also exhibit specific activities, as seen in dihydrotachysterol, which is used to treat hypocalcemia.
17
Recent studies have shown that tachysterol is also a crucial intermediate in the synthesis of the natural anti-tumor product Chaxine C (see Figure 2).18,19 Chaxine C and its analogs are unique oxidative steroid compounds. Choi and Xiao isolated and identified several Chaxine C and its analogs from an edible mushroom

Biomimetic synthesis of Chaxine C and its related compounds.
During the study of the photocatalytic conversion of ergosterol and the synthesis of Chaxine C, it was discovered that the synthesis of tachysterol was challenging. The qualitative and quantitative analysis of ergosterol isomers was performed using high-performance liquid chromatography (HPLC) and quantitative nuclear magnetic resonance (qNMR).22,23 The synthesis process of tachysterol from ergosterol was optimized. The energy conversion mechanism between the photoisomers of ergosterol was analyzed using UV spectroscopy, and some irreversible toxisterols produced by excessive irradiation were analyzed using gas chromatography mass spectrometry (GC-MS).
In summary, previous studies on the synthesis of tachysterol were not comprehensive. The product yield is relatively low (the highest product yield is only 57.0%), and there is very little research on the reaction mechanism and by-products. In this paper, using a self-designed photoreactors (see Supplementary file, Figure S1). we carefully studied the reaction process conditions, developed qualitative and quantitative analysis methods, and discussed the mechanism. This study provided a reliable method for the synthesis of tachysterol, laid a foundation for the synthesis of the anti-tumor lead compound Chaxine C, and offered a reference for the photodegradation reaction of ergosterol analogs.
Results and discussion
Tachysterol was synthesized, separated, and its structure was confirmed using proton nuclear magnetic resonance and 13C nuclear magnetic resonance (1H NMR and 13C NMR). HPLC and qNMR were used for qualitative and quantitative analysis of ergosterol. The synthesis process of tachysterol from ergosterol was optimized. The mechanism and by-products of the reaction were further studied using UV spectrophotometry and GC-MS.
The qualitative and quantitative analysis of tachysterol
High-performance liquid chromatography
Tachysterol and its photochemical isomers were identified and analyzed by HPLC (see Figure 3). Based on literature and standard samples, the HPLC peaks occur in the following order: previtamin D2 (precursor of vitamin D2), lumisterol, tachsterol, and ergosterol, with a retention time ranging from 10.0 to 14.5 min.
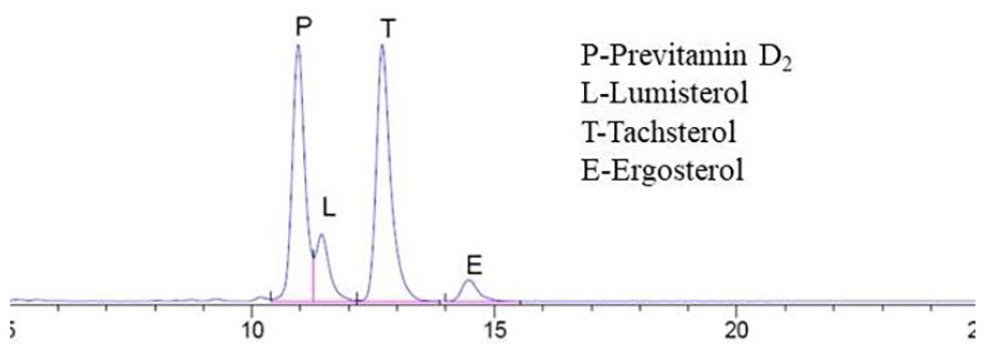
High-performance liquid chromatography of ergosterol reactants.
The tachysterol standard solution was prepared, and the calibration curve was obtained by plotting the peak areas against tachysterol concentrations. The curve was determined using linear regression. The standard concentration range of tachysterol was 20–100 μg mL−1. The regression equation for tachysterol was found to be
Quantitative nuclear magnetic resonance
The internal standard and the quantitative peak for qNMR were determined. Figure 4 displays the 1H NMR spectra of the tachysterol standard solution, while Figure 5 presents the 1H NMR spectra of tachysterol with 1, 2, 4, 5-tetrachlorobenzene internal standard (TeCB), and Figure 6 exhibits the 1H NMR spectra of the tachysterol reaction mixture. The figure shows that TeCB had only one signal peak at 7.56 ppm. The signal peak was chosen as the internal standard peak. The peak signal of tachysterol was intense in the high-field region, while it was less pronounced in the low-field region. The tachysterol emits a doublet signal at 6.74 ppm, which does not overlap with other signals. Therefore, the proton signal at δ 6.74 (d, 1H, H-6) of tachysterol was chosen as the quantitative peak.
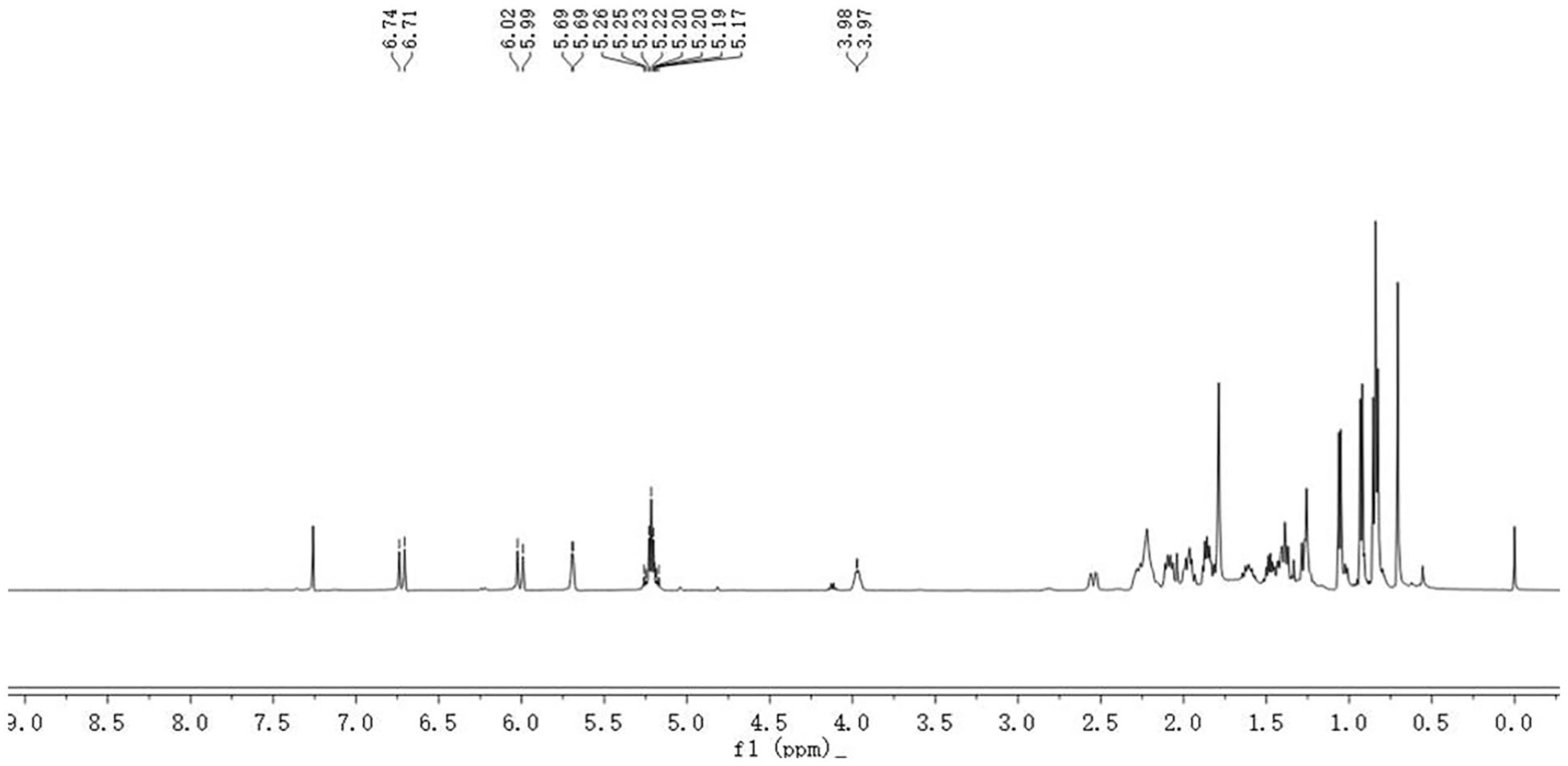
1H NMR spectrum of tachysterol.
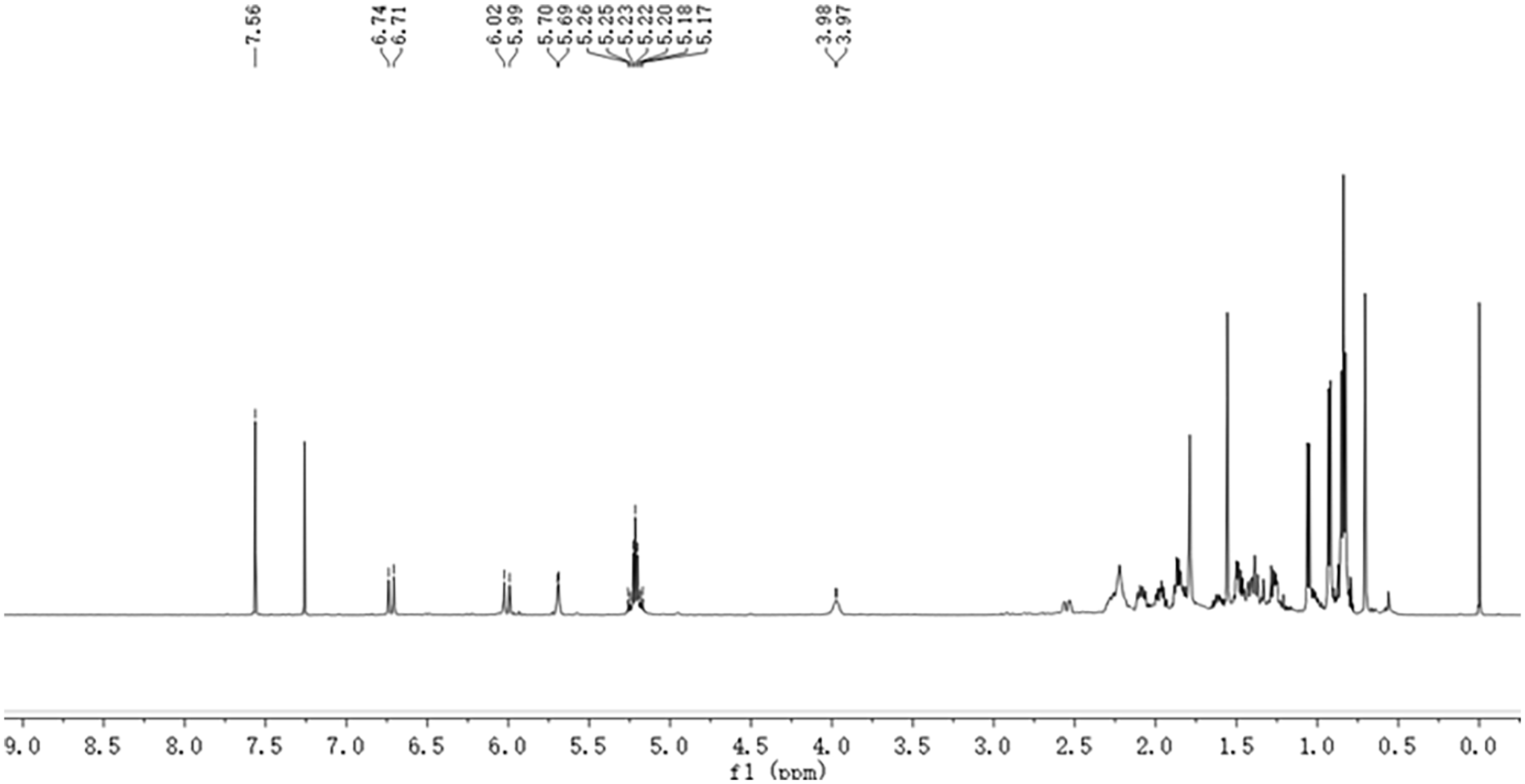
1H NMR spectrum of mixture of tachysterol and TeCB.

1H NMR spectrum of mixture of tachysterol.
The 1H NMR process described was validated by measuring the tachysterol content in tachysterol reactant samples of varying mass. For comparison purposes, the tachysterol content in the reactants was determined using the HPLC method described previously. The tachysterol content obtained using the two methods is presented in Table 1.
The quantitative analysis of tachysterol (qNMR and HPLC).

qNMR: quantitative nuclear magnetic resonance; HPLC: high-performance liquid chromatography.
The results indicated that there was no significant difference between the two methods. In some isomeric compounds that were not clearly distinguishable by liquid chromatography, the compound content could be determined more rapidly using the nuclear magnetic resonance quantitative method (see Supplementary file).
Optimization of synthesis process
Using ergosterol as the raw material, the self-designed photoreactors containing ergosterol (500 mg, 1.26 mmol) and a magnetic stirring bar was placed on a magnetic stirrer.
In order to optimize the synthesis conditions of ergosterol and increase the yield, factors such as reaction time, reaction temperature, photoprotectants, substrate concentration, and UV lamp power on the reaction were discussed.
Reaction time
First, the study examined the effect of reaction time on the synthesis of tachysterol. Other reaction conditions were consistent with those mentioned above. After continuous irradiation at room temperature for 2 h and detection by HPLC every 0.5 h, it was found that the product yield exceeded 50.0% after 0.5 h, and the highest yield was achieved after 1 h. The yield of tachysterol reached as high as 66.2% (refer to Figure 7). As the reaction time increases, the concentration of tachysterol decreases. The experimental results indicate that prolonged irradiation is beneficial for the generation of impurities such as toxisterols and reduces the production of tachysterol.

The effect of reaction time on the yield.
Reaction temperature
Subsequently, the impact of reaction temperature on the synthesis of tachysterol was investigated. The literature reports that the formation of vitamin D2 is the second stage of the photoreaction,
9
during which previtamin D2 is converted into vitamin D2 through a thermochemical process. Considering the time constraint, only the thermal effects of 1 h will be reflected in this study. So, we selected five different temperatures (0, 10, 20, 30, 40 °C) to study their effects on the production of squalene. The reactor device was added with ergosterol (500 mg, 1.26 mmol) and

The effect of reaction temperature on the yield.
Photoprotectants
It is well known that tachysterol and its isomers are sensitive to oxygen.
14
The effects of N2 and antioxidant 2, 6-Di-
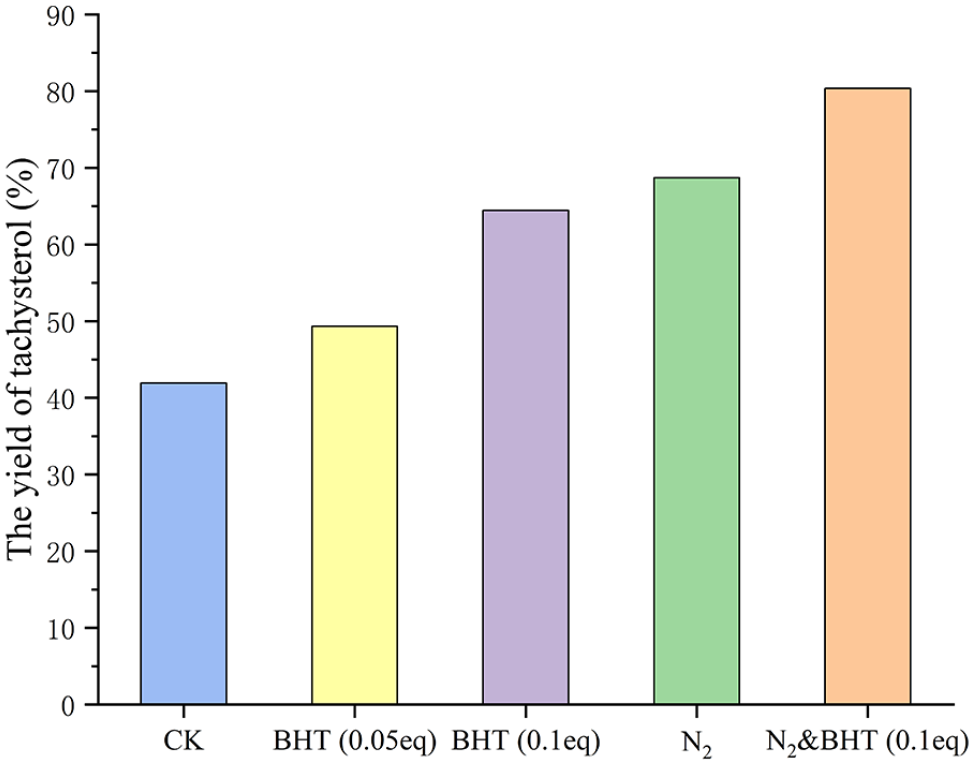
The effect of protectant on the yield.
Reaction substrate concentration
The conversion of ergosterol to tachysterol induced by light was influenced by the intensity of the light. Five different reaction concentrations of 0.5, 1, 2, 4, and 8 mg/mL were selected. The study examined the effect of the concentration of the reaction solution on the yield. The reaction was conducted at a temperature of 20 °C, with 500 mL of

The effect of concentration on the yield.
The power of UV lamps
Under the same conditions, we investigated whether the power of an UV lamp also affects the yield of tachysterol. Three groups of UV lamps with different powers of 10, 15, and 40 W were selected. The concentration of the reaction solution was 2 mg mL−1, the reaction was conducted at 20 °C, and the reaction time was 1 h. N2 and BHT (0.1 equiv.) were employed as protective agents. The study found that the yield of a 40 W UV lamp was significantly higher, reaching up to 85.7% (see Figure 11). With the increase in power, the yield of the product also rises. Given that the product yield has surpassed 85%, further increase in power has not been explored.
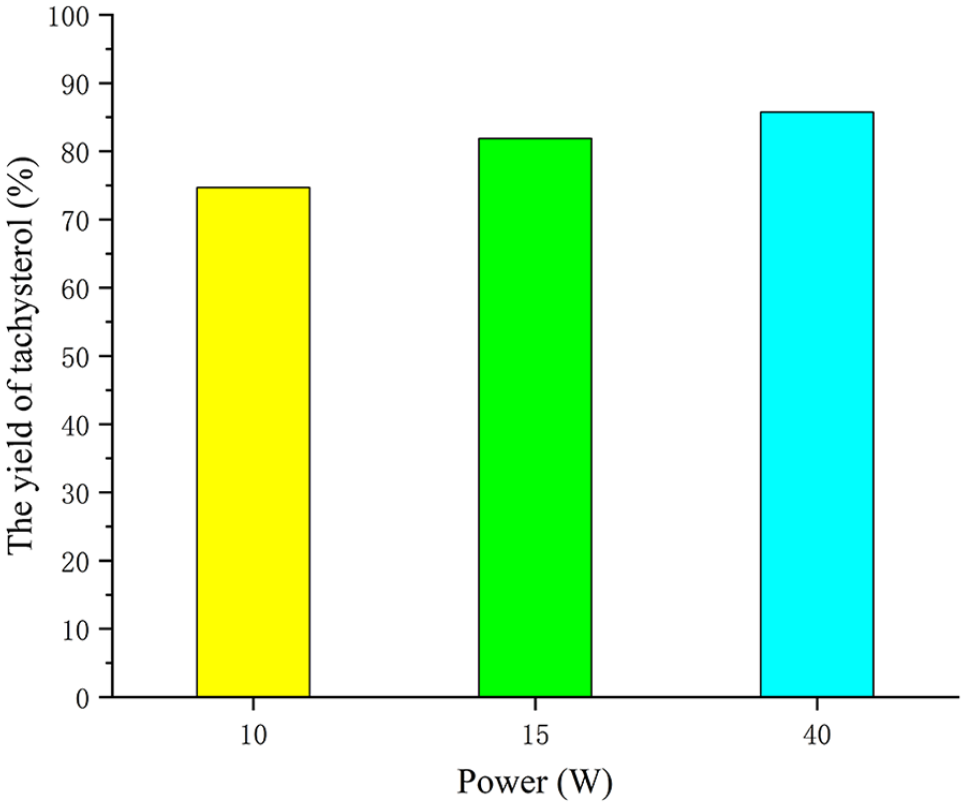
The effect of ultraviolet power on the yield.
In summary, the synthesis process of tachysterol from ergosterol was optimized. The appropriate reaction conditions are as follows: when the reactor device is charged with ergosterol (500 mg, 1.26 mmol), BHT (0.1 equiv., 27.8 mg), and
The mechanism and by-products
UV spectrophotometry
The photochemical stage of converting ergosterol to vitamin D2 is quite complex, involving overlapping absorption spectra. 24 To facilitate the observation of the tachysterol transformation, UV absorption spectroscopy was employed to monitor the changes in tachysterol. The spectral changes were measured using UV spectrophotometry every 15 min over a period of 2 h.
Figure 12 displays the UV spectra of tachysterol, ergosterol, and previtamin D2. It was evident that tachysterol exhibits strong UV absorption at the same concentration, with a maximum UV absorption wavelength of 279 nm. The maximum UV absorption of ergosterol occurs at 281 nm, but the absorption intensity is significantly reduced. The maximum UV absorption wavelength of previtamin D2 was shorter, at 263 nm.

UV absorption spectra of the photoisomers.
We could clearly observe that at the maximum absorption wavelength (280 nm), the absorbance rapidly increased from 0.31 to 0.59 after 15 min of the reaction. Subsequently, the absorbance gradually increases over time. At 1 h, it reached its peak (see Figure 13(a): 0–60 min), which was consistent with the results of liquid chromatography and nuclear magnetic resonance quantification mentioned earlier. Subsequently, the concentration began to decrease as the irradiation time increased after 1 h (see Figure 13(b): 60–120 min). The decrease in absorbance was due to the formation of irreversible byproducts resulting from excessive irradiation.
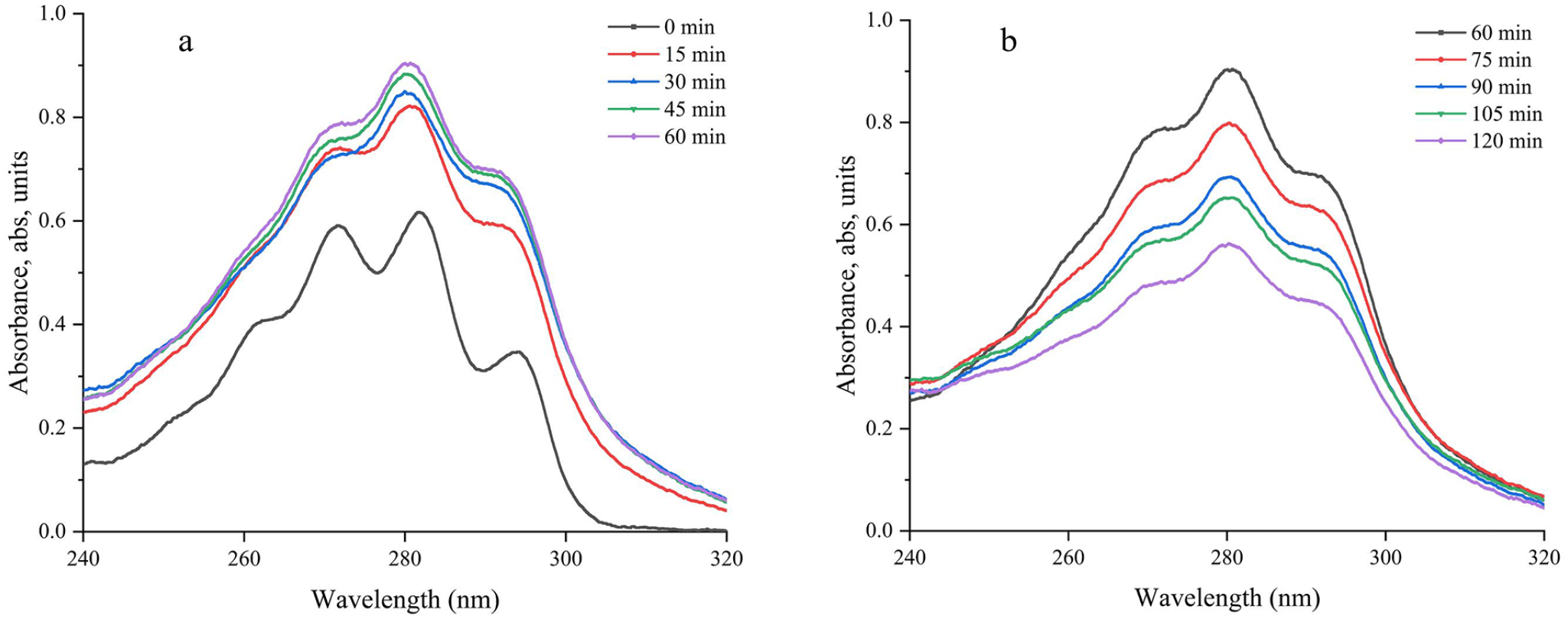
UV absorption spectra of the reaction solution. (a) 0–60 min and (b) 60–120 min.
Gas chromatography mass spectrometry
Extending the reaction time to 6 h, it was observed (using GC-MS, see Figure 14) that the content of tachysterol decreased sharply, and supersterols, toxisterols, and other by-products were also formed. Compounds were formed through cleavage. Samples were obtained every hour and analyzed qualitatively by GC-MS for unknown cleavage compounds.
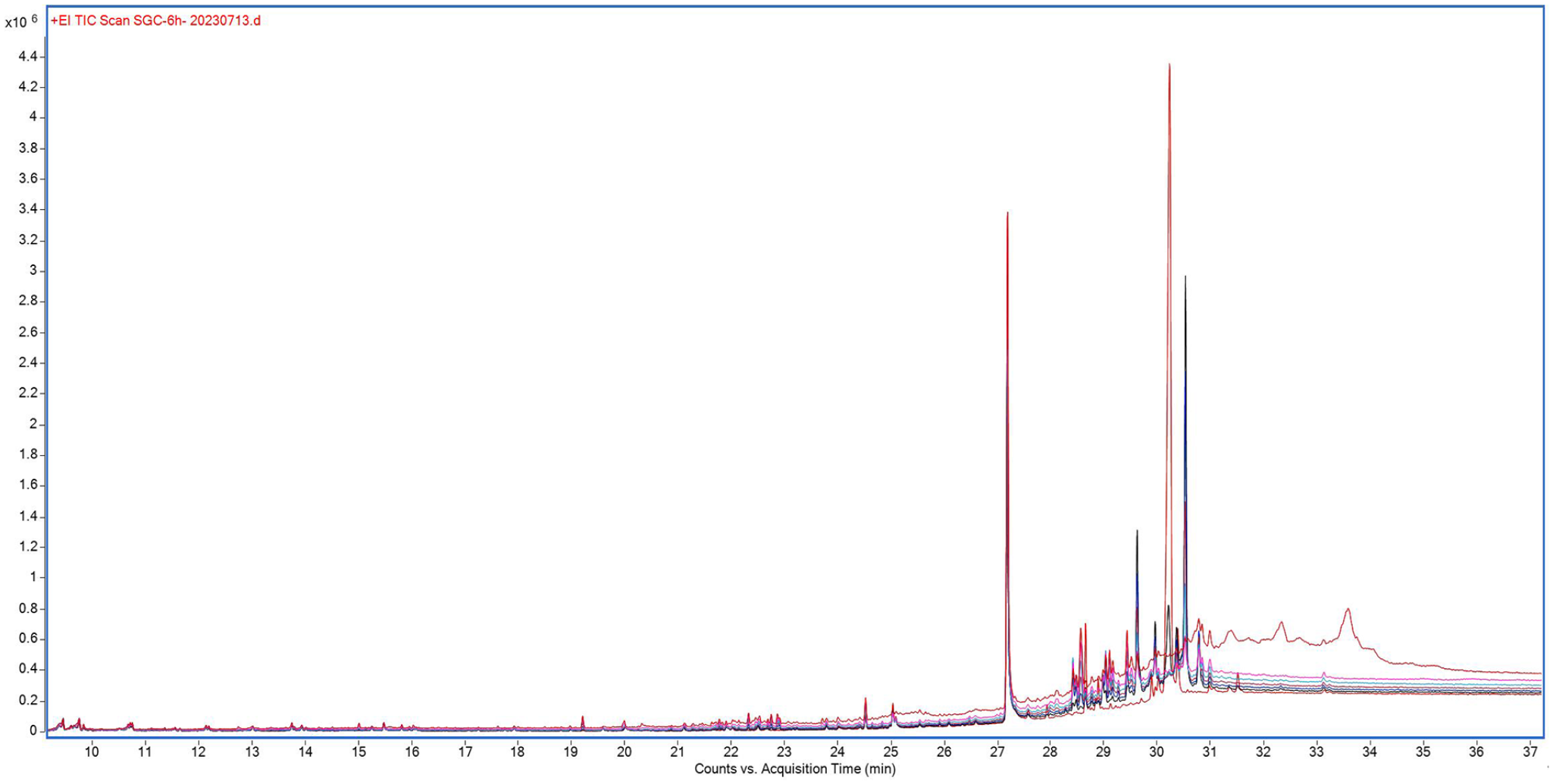
GC-MS of the reaction solution.
By analyzing the GC-MS, particularly the sample after 6 h of reaction, it was observed that the main product, tachysterol, had been mostly broken down, and fragments with mass-to-charge ratios of 266.2, 281.0, and 300.2 were detected (see Supplementary file). It was speculated that at this time, the rapid sterol had already undergone degradation in the double bonds, hydroxyl groups, and other functional groups. Hence, extending the reaction time was unfavorable for the synthesis of tachysterol.
Based on the research findings above, we discovered that ergosterol can rapidly generate previtamin D2 (within 15 min) at 254 nm, and then gradually convert into tachysterol (within 1 h), while also produce by-products such as vitamin D2. Further prolonging the reaction time will result in the degradation of the product under the influence of UV light. It was speculated that the product was a small molecule alkane with branched chains removed (see Figure 15 and Supplementary file, Table S3).

The synthesis mechanism of tachysterol.
Conclusion
In summary, this paper investigated the efficient synthesis of tachysterol under UV irradiation and qualitatively and quantitatively analyzes it. The qualitative and quantitative analysis method for tachysterol based on HPLC and qNMR was established. This method is significantly valuable for analyzing ergosterol derivatives. By optimizing the reaction temperature, time, photoprotectants, reactant concentration, and power of UV lamps, the yield of tachysterol increased to 85.7%. The yield has increased by 28.7% compared to the previously reported results (57.0%). This provides the possibility for the large-scale preparation of tachysterol, especially for the large-scale total synthesis of the anti-tumor natural product Chaxine C.
The synthesis mechanism of ergosterol to produce tachysterol was speculated using UV spectrophotometry, and the by-products of the photolysis reaction were preliminarily identified by GC-MS. With UV spectrophotometry, we can clearly observe the reduction in tachysterol and the subsequent increase of tachysterol over time. The photodegradation products have been preliminarily identified using GC-MS. Excessively prolonging the reaction time is detrimental to the generation and accumulation of products. This study presents a rapid method for synthesizing tachysterol, which lays a foundation for the synthesis of the anti-tumor lead compound Chaxine C. In addition, it enhances and supplements the understanding of the photodegradation mechanism of ergosterol, providing a reference for the photodegradation reaction of its analogs.
Experimental
Chemical reagent
Ergosterol, 2,6-Di-tert-butyl-4-methylphenol, 1,2,4,5-Tetrachlorobenzene, and CDCl3 (+0.03% V/V TMS) were obtained from Aladdin (Jiangsu, China). Methanol (HPLC-grade) and vitamin D2 standards were acquired from Sigma-Aldrich.
Synthesis of tachysterol
The synthesis method utilizes a self-made reactor device. 25 The reaction device is constructed from quartz, with an open upper end where the UV lamp is inserted, and a sealing ring is used for closure. The nitrogen (N2) inlet and outlet are located on the right side, while the injection port is positioned on the left side.
A reactor device containing ergosterol (500 mg, 1.26 mmol) and a magnetic stirrer bar was positioned on a magnetic stirrer. The
The reaction solution was concentrated to dryness under vacuum. The residue was purified by flash column chromatography (PE/EtOAc, 20:1→10:1) to yield tachysterol as a pale yellow oil. The structure of tachysterol was confirmed by 1H NMR and 13C NMR spectra (see Supplementary file).26–28 The structural identification data of the compound are as follows.
Tachysterol, 1H NMR (500 MHz, Chloroform-
High-performance liquid chromatography
Chromatographic conditions
The ergosterol reaction solution was analyzed using the Agilent 1100. The photochemical isomers of ergosterol were separated on an Ecipse XDB-C18 (4.6 mm × 250 mm, 5 µm) column. Reversed-phase chromatography was used. The mobile phase employed an isocratic elution of 90% methanol and 10% water (vol %) with a flow rate of 1.3 mL min−1 and a column temperature of 30 °C. The injection volume was 20 µL, and the UV detection wavelength was set at 270 nm. 29
Preparation of reference solution
Precisely weighing 5 mg of tachysterol, we placed it in a 5 mL volumetric flask and then filled the flask with methanol to create a reference solution with a concentration of 1 mg mL−1. Then 500, 400, 300, 200, and 100 μL of the reference solution were transferred from the reference solution and placed in a 5 mL volumetric flask with methanol. The volume was configured into a reference solution with various concentration gradients.
Calibration curve
The determination of the tachysterol standard solution involved creating the calibration curve by plotting the peak areas against the prepared concentrations and calculating it using linear regression. The standard concentration range of tachysterol is 20-100 μg mL−1. The regression equation for tachysterol was determined as
Quantitative nuclear magnetic resonance
NMR method
The photochemical isomer spectra were obtained using a Bruker DRX500 NMR spectrometer, with 1H and 13C NMR observed at 500 and 125 MHz, respectively (Bruker, Germany). The test conditions are as follows: pulse sequence zg30, pulse width 9.64 μs, spectral width (SWH) 20 ppm, detection temperature (T) 303 K, number of dummy scans (DS) 2, relaxation time (D1) 30 s, and number of scans (NS) 16. 30
Preparation of the reference solution
A total of eight samples of 2–9 mg of tachysterol standard were accurately weighed and then dissolved in 0.8 mL of deuterated chloroform. Then, accurately weigh 50 mg of TeCB and place it in a 5 mL volumetric flask. The flask was filled to a constant volume with CDCl3 to prepare a 10 mg mL−1 internal standard solution. The 0.2 mL internal standard solution was added to the tachysterol standard solution. After complete dissolution, 0.7 mL was drawn into the nuclear magnetic tube.
Calibration curve
The determination of the tachysterol standard solution involved using the mass ratio of tachysterol to the internal standard as the abscissa and the characteristic peak area of both as the ordinate to plot the calibration curve. Linear regression was then employed for calculation purposes. The standard concentration range of tachysterol is 2~9 mg. The regression equation for tachysterol was determined as
Preparation of reactant samples for testing
Prepare the tachysterol solution for a 1-h reaction. The reaction solution was evaporated and concentrated, and samples weighing 6 mg and 7 mg were accurately measured. Then 2 mg of internal standard was added and dissolved in 1 mL of CDCl3. After the dissolution was completed, 0.7 mL of the sample was accurately drawn into the nuclear magnetic tube.
Ultraviolet
The samples were prepared in a 1 mg per 50 mL
The samples were prepared in a 1 mg per 50 mL
Gas chromatography mass spectrometry
The reaction solution of tachysterol was analyzed using an 8860/5977B GC-MS system equipped with a 7693A automatic injection system (1 µL). The carrier gas used was high-purity helium with a purity of 99.999%, flowing at a rate of 1 mL min−1. An HP-5MS column with a length of 30 m, a diameter of 0.25 mm, and a film thickness of 0.25 μm was used. The analysis method was as follows: After heating at 50 °C for 1 min, the temperature was increased from 10 °C min−1 to 320 °C for 10 min. The temperatures of the ion source, quadrupole, injector, and transmission line are 230 °C, 150 °C, 250 °C, and 325 °C, respectively. Data were recorded in full scan mode from m/z 20 to 500 after a 5-min delay in the solvent. 31
Supplemental Material
sj-docx-1-chl-10.1177_17475198241265931 – Supplemental material for Optimization of synthesis process and photocatalytic mechanism of tachysterol under ultraviolet irradiation
Supplemental material, sj-docx-1-chl-10.1177_17475198241265931 for Optimization of synthesis process and photocatalytic mechanism of tachysterol under ultraviolet irradiation by Wuzhang Liu, Yu Shan, Songtao Liu, Ye Zhou, Yan Wang, Linwei Li, Shu Xu, Yu Chen, Xu Feng, Li Lin, Jinyue Luo and Fei Liu in Journal of Chemical Research
Footnotes
Acknowledgements
The author would like to thank Mr. Deng Hua for his contributions during the research process and for providing funding for this study.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this paper.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this paper: This work was supported by the National Natural Science Foundation of China (grant no. 32000258 and 31970375).
Supplemental material
Supplemental material for this paper is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
