Abstract
This study revealed a new inhibitor of Ca2+/calmodulin-dependent protein kinase II (CaMKII), a crucial factor in cardiovascular disease and hypertension. The study focuses on the bioactivity compounds that combine adamantane/1,3,4-oxadiazole, potentially inhibiting CaMKIIδ. Various adamantyl-1,3,4-oxadiazole derivatives were synthesized and tested for their efficiency against CaMKIIδ kinase, with

Introduction
Ca2+/calmodulin-dependent protein kinase II (CaMKII), one of the Ca2+/calmodulin-dependent protein kinase family (comprising 3 subgroups: I, II, and IV), is a versatile serine/threonine kinase pivotal for modulating calcium signaling. It exerts significant influence over a multitude of cellular processes, encompassing excitation-contraction coupling, gene expression, and cell apoptosis. CaMKII phosphorylates a diverse array of substrates that participate in the regulation of calcium-induced adjustments in cellular function. The four distinct CaMKII isoforms (alpha, beta, gamma, and delta) are spread across different tissues: CaMKIIα, β, and γ predominantly inhabit the brain, while CaMKIIδ is primarily situated in the heart.1,2 Within the cardiac context, CaMKIIδ is implicated in the development of heart failure, arrhythmias, and atrial fibrillation. Research has demonstrated heightened CaMKIIδ activity in individuals with heart failure and atrial fibrillation. Inhibiting CaMKIIδ has been observed to ameliorate cardiac function and reduce the occurrence of arrhythmias in animal models of heart failure.3,4 Moreover, inhibition of CaMKIIδ has been shown to protect cardiac mitochondria from Ca2+ overload, loss of inner membrane potential, increased ROS, and reduce the incidence of arrhythmias in models of heart failure.4–6
CaMKII’s activity is regulated by calmodulin (CaM), a calcium-sensing protein. When intracellular calcium levels surge, Ca2+ binds to CaM, initiating the activation of CaMKII. The sensitivity of CaMKII to calcium and CaM is subject to regulation through variable and self-associative domains.4,7 The binding of CaM to CaMKIIδ promotes structural modifications in CaMKIIδ, alleviating autoinhibition and enabling complete enzymatic activity. 8
The potential of CaMKIIδ as a target for clinical intervention in cardiovascular disorders has been established through the emergence of clinical trials involving potent CaMKIIδ inhibitors, such as KN-93, KN-62, and staurosporine the natural CaMKIIδ inhibitor (see Figure 1).4,8 However, therapeutic inhibition of CaMKIIδ is challenged by the diversity of CaMKII isoforms and splice variants and by physiological activities of CaMKII isoforms that contribute to learning and memory. 9

Commercially available and natural used CaMKII inhibitor.
CaMKIIδ inhibitors encounter additional drawbacks encompass poor cellular permeability, a tendency for general toxicity that can impede their clinical applicability, a deficiency in specificity potentially resulting in unintended off-target effects and constraining their therapeutic promise. In addition, certain CaMKIIδ inhibitors may interfere with the interaction between CaMKIIδ and scaffolding proteins, potentially disrupting kinase signaling domains.4,5,10–12
As part of our ongoing interest in the discovery and design of adamantyl-1,3,4-oxadiazole hybrids as bioactive compounds.13–17 As potential CaMKIIδ kinase inhibitors, we had an interest in creating hybrid adducts of adamantane/1,3,4-oxadiazole. The synthesis of target compounds pharmacophore was obtained using a Cu-catalyzed Mannich reaction13,14,18,19 (Scheme 1).

Compounds
In silico profiling against many targets proposed our hybrid compounds block CaMKIIδ kinase, 20 and were, therefore, assessed in vitro as CaMKIIδ kinase blockers. One of them showed micromolar IC50s. It’s interesting to note that no prior research has been done using hybrid molecules of adamantyl-1,3,4-oxadiazole as CaMKIIδ kinase inhibitors.
Results and discussion
Chemistry
As shown in Scheme 1, our synthetic approach started from adamantane carboxylic acid (
Structures of synthesized

High-resolution mass spectrometry (HRMS), 1H, 13C NMR, and IR spectroscopy were used to characterize the synthesize compounds (spectra of all compounds HRMS, IR, 1H and 13C NMR, are available in SI file). The mass spectra were found to be in highly agreement with estimated values and provided the right molecular ion peaks. Aliphatic protons related to the adamantyl group in the region δ = 0.72–2.08 ppm and protons from aliphatic amines were detected in the 1H NMR spectra. The SCH2 protons showed as singlets at around δ = 4.00 ppm.
Resonances were observed for all aromatic rings at δ = 6.70–8.22 ppm. However, all of the produced compounds’ distinctive signals were seen in the 13C NMR spectra, which also included several aliphatic peaks for the aliphatic amine (region δ = 12.0–39.5 ppm) and adamantly (region δ = 41.0–58.0 ppm) moieties. In the aromatic area (region δ = 103.0–152.0 ppm), every aromatic moiety displayed accurate and distinct carbon signals. For majority of the produced compounds, the alkyne carbon atoms peaks (78.5–80.0 ppm) are easily identifiable. At approximately δ = 162.0 and 174.0 ppm, the two quaternary oxadiazole peaks were visible.
Evaluation of biological activities
Profiling against numerous pharmacophores revealed one of the molecules, namely,

Inhibitory profiles of synthesized compounds against CaMKIIδ.

Average of duplicate measurements ± standard deviation.
Standard inhibitor.

Dose-response curves of compound
Docking study
In order to gain a mechanistic understanding of the bioactivity of 6f, we chose to dock 6f into the CaMKIIδ kinase binding site. Using CDOCKER, the docking investigation was completed.25,26
However, prior to docking 6f, the docking parameters had to be validated by comparing the docked pose of the same ligand with the crystallographic pose of the co-crystallized inhibitor within CaMKIIδ kinase (PDB code: 5VLO). Fortunately, with a root mean square difference of 0.27 Å, the docking settings nearly matched the crystallographic position (Figure 4), which gave rise to the decision to dock 6f with the same parameters.

Docked and crystallographic positions of a ligand inside CAMKIIδ (PDB code: 5VLO) (a) The bound ligand in its docked position. The hinge region’s DFG amino acids are represented by green residues. (b) A comparison of the complexed ligand’s crystallographic structure within the CAMKIIδ binding pocket (green) and its docked pose (red).
Figure 5 shows docked
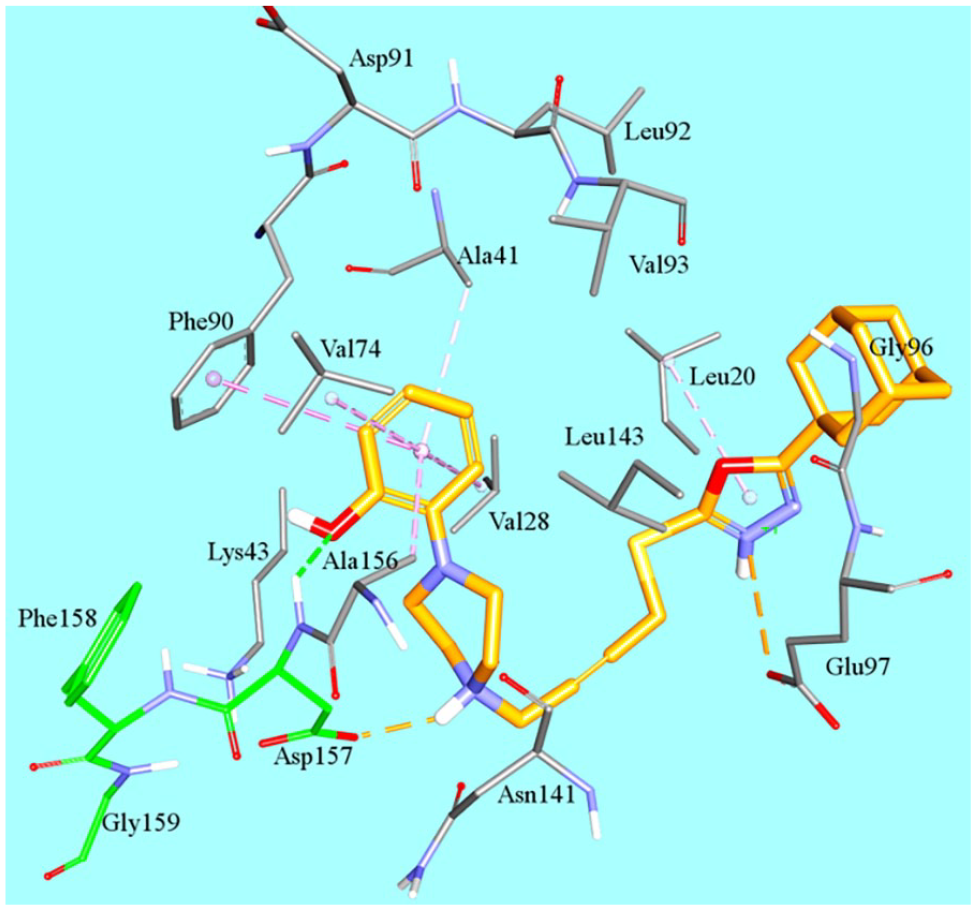
Docked pose of active compounds
Conclusion
Due to CaMKIIδ kinase plays a crucial role in mitosis and cell cycle regulation, targeting it in anticancer research shows potential. This study focuses on using the copper-catalyzed Mannich reaction to generate hybrid adamantine/1,3,4-oxadiazole compounds as potential CaMKIIδ kinase inhibitors. Compound
Experimental
Chemicals and instruments
All compounds were purchased commercially from Across Organics and Sigma-Aldrich Co. and were utilized exactly as supplied, without further purification. The study’s solvents were procured from Scharlau, Fluka, and Aldrich. Thin layer chromatography (TLC) was used to monitor all reactions. Merk aluminum plates pre-coated with silica gel PF254; 20 × 20 × 0.25 mm; these plates were seen under a UV light to identify the reactions. Using TMS as an internal standard, 1H NMR spectra were captured using a Bruker Avance III-500 MHz spectrometer. 13C NMR spectra were collected at 125 MHz on the same equipment. Chemical shifts were expressed as ppm-values for δ-. Spectra were obtained in CDCl3. In addition, 2D (COZY, HMQC, and HMBC) and DEPT spectra were acquired. On a Bruker APEX-IV (7 Tesla) instrument, HR-MS were obtained using the electrospray ion trap (ESI) technique. Using a Thermo-Nicolet Nexus 670 FTIR device, FTIR spectra were captured.
Synthesis and characterization
Methyl adamantane-1-carboxylate (2 )
Following the literature procedure,14,15 compound
1-adamantanecarbohydrazide (3 )
Following the literature procedure,14,15 compound
5-(1-adamantyl)-1,3,4-oxadiazole-2-thiol (4 )
Following the literature procedure, compound
2-(1-adamantyl)-5-(prop-2-yn-1-ylthio)-1,3,4-oxadiazole (5 )
Na2CO3 (44.80 mmol) was added to a stirred solution of
Synthesis of compounds (6a–h)
4-((5-((3r,5r,7r)-adamantan-1-yl)-1,3,4-oxadiazol-2-yl)thio)-N, N-2-yn-1-amine (6a–h )
CuI (25 mg) and aqueous formaldehyde (35%, 1 mL) in 2 mL dimethyl sulfoxide (DMSO) were combined with an alkyne
4-(4-((5-((3r,5r,7r)-adamantan-1-yl)-1,3,4-oxadiazol-2-yl)thio)but-2-yn-1-yl)morpholine (6a )
Pale brown oily product (0.200 g, 69%). IR (cm–1): 3417, 2910, 2852, 2818, 2763, 2644, 2250, 1568, 1485, 1240, 1176, 1116, 1053, 756. 1
2-((3r,5r,7r)-adamantan-1-yl)-5-((4-thiomorpholinobut-2-yn-1-yl)thio)-1,3,4-oxadiazole (
6b
)
Pale brown oily product (0.199 g, 65%). IR (cm–1): 3450, 2908, 2852, 1485, 1454, 1417, 1242, 1174, 1053, 754. 1
2-((3r,5r,7r)-adamantan-1-yl)-5-((4-(4-benzylpiperidin-1-yl)but-2-yn-1-yl)thio)-1,3,4-oxadiazole (6c )
Pale brown oily product (0.273 g, 70%). IR (cm–1): 2910, 2850, 1568, 1485, 1174, 1053, 750. 1
2-((3r,5r,7r)-adamantan-1-yl)-5-((4-(4-(3-methoxyphenyl)piperazin-1-yl)but-2-yn-1-yl)thio)-1,3,4-oxadiazole (6d )
Pale brown oily product (0.281 g, 72%). IR (cm–1): 3377, 2852, 1778, 1668, 1485, 1298, 1236, 1215, 1172, 1043, 768. 1
2-((3r,5r,7r)-adamantan-1-yl)-5-((4-(4-(3-chlorophenyl)piperazin-1-yl)but-2-yn-1-yl)thio)-1,3,4-oxadiazole (6e )
Pale brown oily product (0.276 g, 68%). IR (cm–1): 2910, 2852, 1568, 1485,1236, 1174, 945, 769. 1
2-(4-(4-((5-((3r,5r,7r)-adamantan-1-yl)-1,3,4-oxadiazol-2-yl)thio)but-2-yn-1-yl)piperazin-1-yl)phenol (6f )
Pale brown oily product (0.288 g, 71%). IR (cm–1): 3304, 2914, 2850, 1623, 1568, 1485,1247, 1174, 1053, 927, 763. 1
4-(4-(4-((5-((3r,5r,7r)-adamantan-1-yl)-1,3,4-oxadiazol-2-yl)thio)but-2-yn-1-yl)piperazin-1-yl)phenol (6g )
Pale brown oily product (0.350 g, 79%). IR (cm–1): 3242, 2916, 2852, 2825, 1588, 1490,1217, 827, 759. 1
2-((3r,5r,7r)-adamantan-1-yl)-5-((4-(4-(3-methoxyphenyl)-2-methylpiperazin-1-yl)but-2-yn-1-yl)thio)-1,3,4-oxadiazole (6h )
Pale brown oily product (0.290 g, 73%). IR (cm–1): 3377, 2852, 1778, 1668, 1485, 1298, 1236, 1215, 1172, 1043, 768. 1
In vitro bioassay against CaMKIIδ kinase
The Invitrogen Z’-LYTE Kinase Assay Kit was utilized to evaluate the effectiveness of the generated compounds. The bioassay was carried out using 500 μM of ATP and CaMKIIδ kinase at an ideal concentration range of 2.7–4.7 nM. Molecules were first made into stock solutions with a concentration of 10 millimolar (mM) in DMSO. After that, these stock solutions were gradually diluted in a buffer solution to produce final molecular concentrations that ranged from 0.01 micromolar (µM) to 100 µM. In the final kinase reaction (10 μL), the DMSO content did not go above 1%. Using GraphPad Prism 7.04, nonlinear regression analysis of the log (concentration) versus inhibition % data was performed to estimate the inhibition percentage and IC50 value.
Docking study
Compounds
The current project has implemented the following CDOCKER parameters: A binding site sphere with a radius of 9.0 Å, centered around the co-crystallized ligand (PDB code: 5VLO), was incorporated into the crystallographic structures of CaMKIIδ kinase. The initial conformations of the ligands were subjected to energy minimization and subsequently heated to a temperature of 1000 K using 1000 molecular dynamics steps. For every ligand, this procedure was carried out 10 times, producing 10 randomly generated initial conformations. To modify its energy, each arbitrary conformer rotated 10 times inside the binding pocket. After determining the Van der Waals energies of the resultant conformers/poses, any energies that were 300 kcal/mol or higher were disregarded. A simulated annealing process including 2000 heating steps up to a target temperature of 700 K and 5000 cooling steps down to a target temperature of 300 K was applied to the conformers/poses that survived. Energy minimization was applied to the docked postures until a final minimization gradient tolerance of 0 kcal/mol/Å was reached.
Supplemental Material
sj-doc-1-chl-10.1177_17475198241262467 – Supplemental material for Synthesis, evaluation, and docking study of adamantyl-1,3,4-oxadiazol hybrid compounds as CaMKIIδ kinase inhibitor
Supplemental material, sj-doc-1-chl-10.1177_17475198241262467 for Synthesis, evaluation, and docking study of adamantyl-1,3,4-oxadiazol hybrid compounds as CaMKIIδ kinase inhibitor by Mohammed M Al-Mahadeen, Areej M Jaber, Raed A Al-Qawasmeh and Mutasem O Taha in Journal of Chemical Research
Footnotes
Author contributions
M.M.A.: Methodology, Investigation, Formal analysis, Resources, Review & Editing. A.M.J.: Investigation, Formal analysis, Writing, Review & Editing. M.O.T.: Conceptualization, Investigation, Writing, Review & Editing. R.A.A.: Formal analysis, Review & Editing.
Data availability
The corresponding author can provide the data sets created and/or analyzed during the current work upon reasonable request.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
