Abstract
In this work, 20 curcumin derivatives containing halogen atoms or nitrogen atoms were synthesized for suppression of castration-resistant prostate cancer. These curcumin derivatives were prepared by aldol condensation between the substituted benzaldehydes and acetylacetone or ethyl 3-acetyl-4-oxopentanoate. All synthesized compounds were characterized by 1H nuclear magnetic resonance, 13C nuclear magnetic resonance, high resolution mass, ultraviolet–visible spectroscopy, fluorescence spectroscopy, and high-performance liquid chromatography. The in vitro cytotoxicity of the prepared compounds against 22Rv1 cells was evaluated by the standard MTT assay. The compounds with good inhibitory activity against 22Rv1 cells were further tested for androgen receptor suppression effect. The results show that all compounds were successfully prepared and the purities are all over 90%. Among all synthesized compounds, compound
Keywords

Introduction
Prostate cancer is the most frequently diagnosed cancer in males in the western countries. 1 Androgen deprivation therapy (ADT) targeting androgens via surgical or medical castration is used as the primary therapeutic strategy for patients with advanced prostate cancer.2,3 However, the efficacy of ADT is limited because prostate cancer cells usually progress to a castration-resistant phenotype over 1–2 years of therapy.4,5 The newly developed anti-androgen therapies with either androgen receptor (AR) antagonists or androgen biosynthesis inhibitors are demonstrated to be beneficial.6,7 However, these therapies might also induce drug resistance due to the alterations in AR/androgens axis. 8
Curcumin (Scheme 1) is a well-known compound found in turmeric. Numerous studies have reported the remarkable cancer preventive and therapeutic properties of curcumin. 9 In addition, various curcumin derivatives (Scheme 1) have been designed and synthesized to discover new compounds with better anticancer properties.10,11 Among these, dimethylcurcumin (DMC, ASC-J9) is found to exhibit efficient prostate cancer cell inhibition effect.12–14 DMC is reported to selectively promote AR degradation, resulting in the inhibition of AR transcriptional activity and prostate cancer cell growth.15–17 In addition, DMC is proven to be effective in the control of various AR-associated diseases.18,19 Compared with anti-androgen therapies, anti-AR therapy with DMC might better suppress castration-resistant prostate cancer progression and metastasis. 19
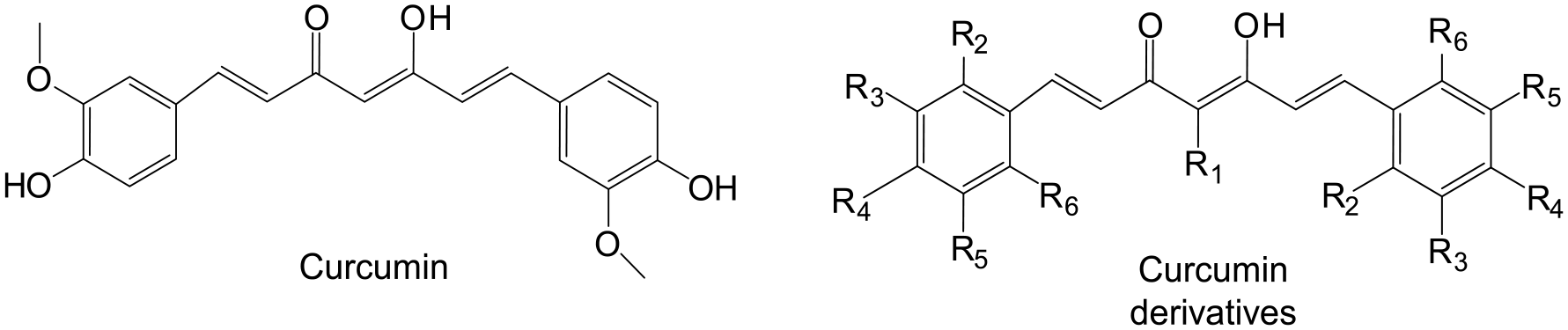
The chemical structure of curcumin and its derivatives.
To discovery novel curcumin derivatives with better castration-resistant prostate cancer cell inhibition effect, 20 curcumin derivatives containing halogen atoms or nitrogen atoms were synthesized by aldol condensation in the present work. The synthesized compounds were fully characterized. The in vitro cytotoxicity and AR protein suppression effect against castration-resistant prostate cancer cells of these compounds were assessed and compared with DMC.
Results and discussion
Curcumin derivatives containing halogen atoms or nitrogen atoms were prepared by an one-pot method,
20
Scheme 2. Acetylacetone or ethyl 3-acetyl-4-oxopentanoate was first reacted with boric acid in N-dimethylformamide (DMF) and then reacted with substituted benzaldehydes in the presence of tributyl borate and n-butylamine. After reaction, acetic acid aqueous solution (20%) was added to afford curcumin derivatives

Synthetic scheme of curcumin derivatives.

Chemical structures of the synthesized curcumin derivatives
The in vitro cytotoxicity of compounds
Average half maximum inhibitory concentration (IC50) of DMC and

As for curcumin derivatives containing halogen atoms, compounds
Among the prepared curcumin derivatives
Compound
Taken together, these results demonstrate that compounds
DMC is reported to suppress AR-related signal pathways by degradation of AR, resulting in suppression effect on castration-resistant prostate cancer (CRPC) cell proliferation. Accordingly, compounds

AR protein suppression effect of compounds
Conclusion
In summary, curcumin derivatives containing halogen atoms or nitrogen atoms were prepared for castration-resistant prostate cancer cell inhibition. Among compounds
Materials and methods
Materials
Acetylacetone (99%), boric acid (99%), tributyl borate (98%), n-butylamine (98%), substituted benzaldehydes (95%-99%), 3-(4,5-dimethyl-2-thiazolyl)-2,5-diphenyl-2-H-tetrazolium bromide (MTT, 98%) were purchased from Aladdin Reagent Co., Ltd. (China). Roswell Park Memorial Institute 1640 (RPMI1640) medium, fetal bovine serum (FBS), penicillin-streptomycin solution, trypsin-EDTA (0.25%) were purchased from Fisher Scientific International, Inc. (USA). All other chemicals were of HPLC grade or analytical grade and used as received.
Synthesis of 2,4-diacetylaminobenzaldehyde
2,4-Diacetylaminobenzaldehyde was synthesized by the reaction between 2,4-diaminobenzaldehyde and acetic anhydride in the presence of triethylamine, Scheme S1. Briefly, 2,4-diaminobenzaldehyde (544 mg, 4 mmol) was dissolved in 30 mL of dichloromethane. To the solution, acetic anhydride (2.04 g, 20 mmol) and triethylamine (1.2 g, 12 mmol) were added and the mixture was refluxed for 48 h. After that, the reaction mixture was evaporated to remove the solvent, and ethanol was added to precipitate the product. The precipitate was isolated by filtration, washed with ethanol for three times and dried under vacuum afford 2,4-diacetylaminobenzaldehyde as a faint yellow solid (yield: 92%). 1H NMR (400 MHz, DMSO-D6): δ (ppm) 10.90 (s, 1H), 10.42 (s, 1H), 9.79 (s, 1H), 8.42 (d, J = 1.8 Hz, 1H), 7.76 (d, J = 8.5 Hz, 1H), 7.66 (dd, J = 8.5 Hz, 1.8 Hz, 1H), 2.13 (s, 3H), 2.06 (s, 3H). 13C NMR (100 MHz, DMSO-D6): δ (ppm) 193.8, 169.8, 169.7, 146.1, 141.6, 136.3, 119.1, 113.8, 109.8, 25.1, 24.8. HRMS: m/z of [M+Na]+ calculated for C11H12N2O3: 243.0746, found: 243.0746. m.p.: 232.8–234.4 °C. HPLC (CH3CN/H2O = 80/20, 1 mL/min, 254 nm): Retention time 2.997 min, estimated purity 98.1%.
Synthesis of ethyl 3-acetyl-4-oxopentanoate
Ethyl 3-acetyl-4-oxopentanoate was synthesized by the reaction between ethyl chloroacetate and acetylacetone in the presence of potassium carbonate,21,22 Scheme S2. In brief, potassium carbonate (13.8 g, 100 mmol) was dispersed in 60 mL of ethanol. To the mixture, acetylacetone (10.0 g, 100 mmol) was added. Then, ethyl chloroacetate (13.5 g, 110 mmol) dissolved in 20 mL of ethanol was dropwise added. The resulting mixture was heated to 60 °C and stirred for 24 h. The solvent was removed by evaporation under vacuum and deionized water was added to the residues to form two phases. The organic phase was separated, and the aqueous phase was extracted with ethyl acetate for three times. The obtained organic phases were combined and dried overnight with anhydrous manganese sulfate. The crude product was obtained by removing the solvent by evaporation under vacuum, which was further purified by column chromatography (eluent: ethyl acetate/petroleum ether = 1:8, v/v) to afford ethyl 3-acetyl-4-oxopentanoate as a pale yellow oil (yield: 71%). HRMS: m/z of [M+Na]+ calculated for C9H14O4: 209.0790, found: 209.0789.
Synthesis of curcumin derivatives
Briefly, 10 mmol of acetylacetone (or ethyl 3-acetyl-4-oxopentanoate) and 5 mmol of boric acid were added to 10 mL of DMF and the resulting mixture was heated to 80 °C and stirred for 20 min. Then, 20 mmol of tributyl borate and 20 mmol of substituted benzaldehyde were added and stirred at 80° for 30 min. After that, 10 mmol of n-butylamine was added and the resulting mixture was stirred at 80° for 1–3 h. After reaction, 50 mL of acetic acid aqueous solution (20%) was added to the reaction mixture and stirred overnight. The formed precipitate was isolated by filtration and purified by recrystallization in methanol.
Characterization
The NMR spectra were recorded on a on a NMR spectrometer (AVANCE III, Bruker) using dimethyl sulfoxide-D6 (DMSO-D6) as the solvent and tetramethylsilane (TMS) as an internal reference. The HRMS spectra were recorded on an Agilent Technologies 6200 series TOF/6500 series Q-TOF B.06.01 (B6157) instrument (CA, USA). The ultraviolet-visible (UV-Vis) absorbance spectra and fluorescence spectra were recorded on a microplate reader (Spark, Tecan). The HPLC measurement was performed on an Agilent 1260 Infinity HPLC system. The melting point was recorded on a B-545 melting point apparatus (Buchi, Switzerland).
Cell culture
Human prostate cell line 22Rv1 was purchased from Procell Life Science & Technology Co., Ltd. (Wuhan, China). 22Rv1 cells were cultured in RPMI1640 medium supplemented with 10% FBS, 100 units/mL penicillin and 100 g/mL streptomycin at 37 °C under 5% carbon dioxide atmosphere. 22Rv1 cells are androgen-responsive and androgen-independent human prostate cell line, which express endogenous AR.
In vitro cytotoxicity against 22Rv1 cells
The in vitro cytotoxicity of the curcumin derivatives against 22Rv1 cells was assessed by MTT assay. In briefly, 22Rv1 cells were seeded on 96-well plates in quadruplicates at a cell density of 5 × 103 cells per well. The curcumin derivatives were dissolved in DMSO and diluted with RPMI1640 medium to the desired concentrations. After incubation with curcumin derivatives with different concentrations for 48 h or 72 h, the cell viability of each group was determined by standard MTT assay. Three separated experiments were performed.
AR protein suppression against 22Rv1 cells
22Rv1 cells were seeded in six-well plates in triplicates at a density of 2 × 105 cells per well. The cells were treated with different compounds (2 μg/mL) for 24 h. Then, the culture medium was removed, and the cells were washed three times with phosphate-buffered saline (PBS; pH 7.4, 6.7 mM). After that, 150 μL RIPA Pyrolysis liquid with 1% protease inhibitor was used to lyse 22Rv1 cells. The lysates we collected into 1.5 mL centrifuge tube and bathed in ice for 30 min and then centrifuged at 12,000 r/min for 5 min. The supernatant was collected as the total protein solution. After quantification, 25% loading buffer was added into protein solution. The total protein solutions were stored at −80 °C for further use.
Equal protein amounts with denaturation treatment were loaded and separated in 5–15% SDS-PAGE acrylamide gel electrophoresis and transferred onto polyvinylidene difluoride membranes. These membranes were blocked with 5% defatted milk powder in tris-buffered saline containing 0.1% Tween-20 for 1.5 h. The membranes were then incubated with 5 mL primary antibodies to AR and β-actin at 4 °C overnight. After that, the membranes were washed three times and probed with horseradish peroxidase-conjugated goat anti-mouse secondary antibody at room temperature for 1.5 h. The protein bands were detected and imaged with the immobilon western chemiluminescent horseradish peroxidase (HRP) substrate on a chemiluminescence imaging analysis system (Tanon-5200Multl, China).
Supplemental Material
sj-docx-1-chl-10.1177_17475198241230042 – Supplemental material for Synthesis of curcumin derivatives for suppression of castration-resistant prostate cancer
Supplemental material, sj-docx-1-chl-10.1177_17475198241230042 for Synthesis of curcumin derivatives for suppression of castration-resistant prostate cancer by Shunze Gong, Chixiang Yuan, Hang Hu and Defeng Xu in Journal of Chemical Research
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the grant from the Key Research and Development Program of Changzhou, China (Applied Basic Research, grant no. CJ20210030).
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
