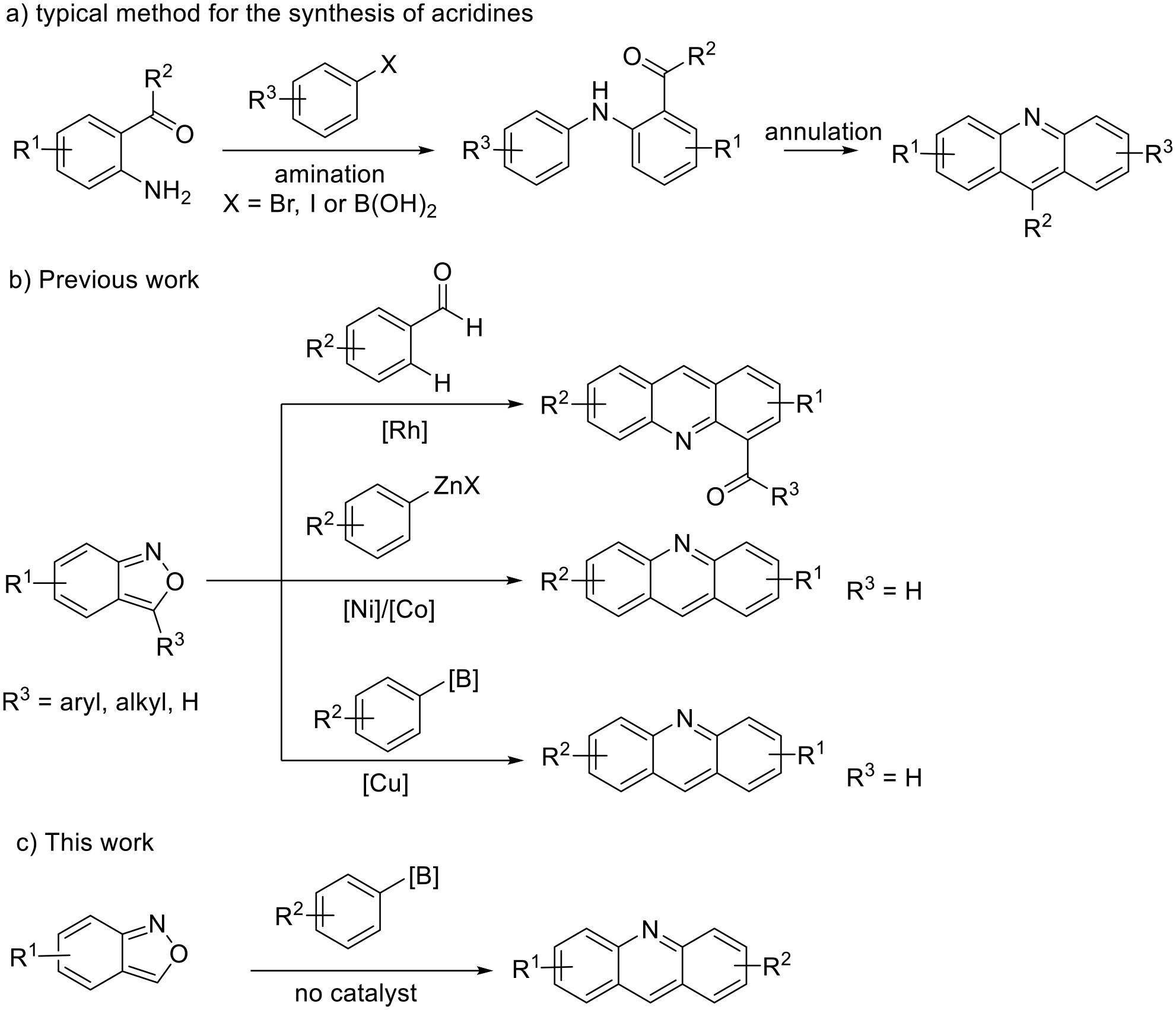
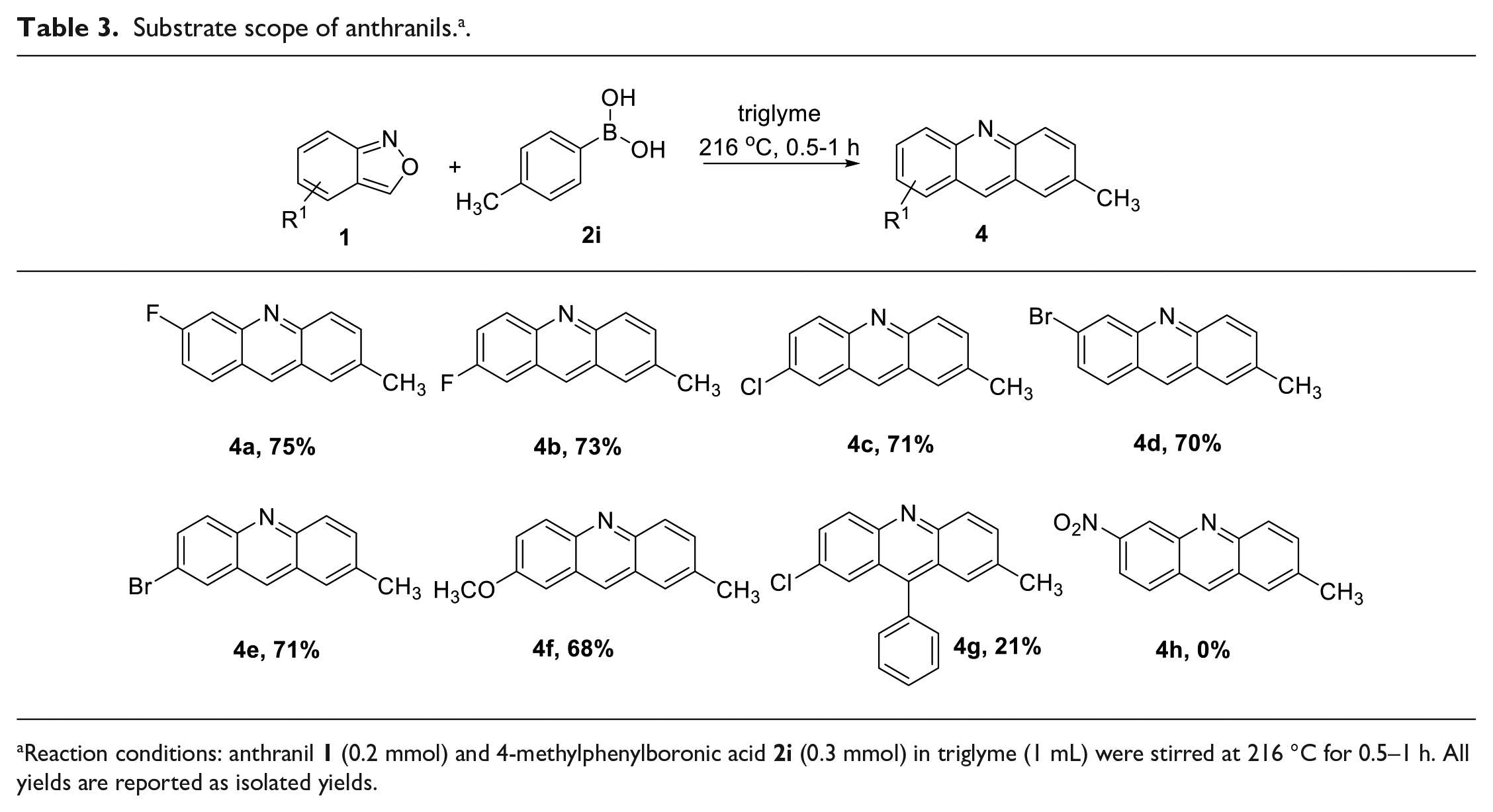
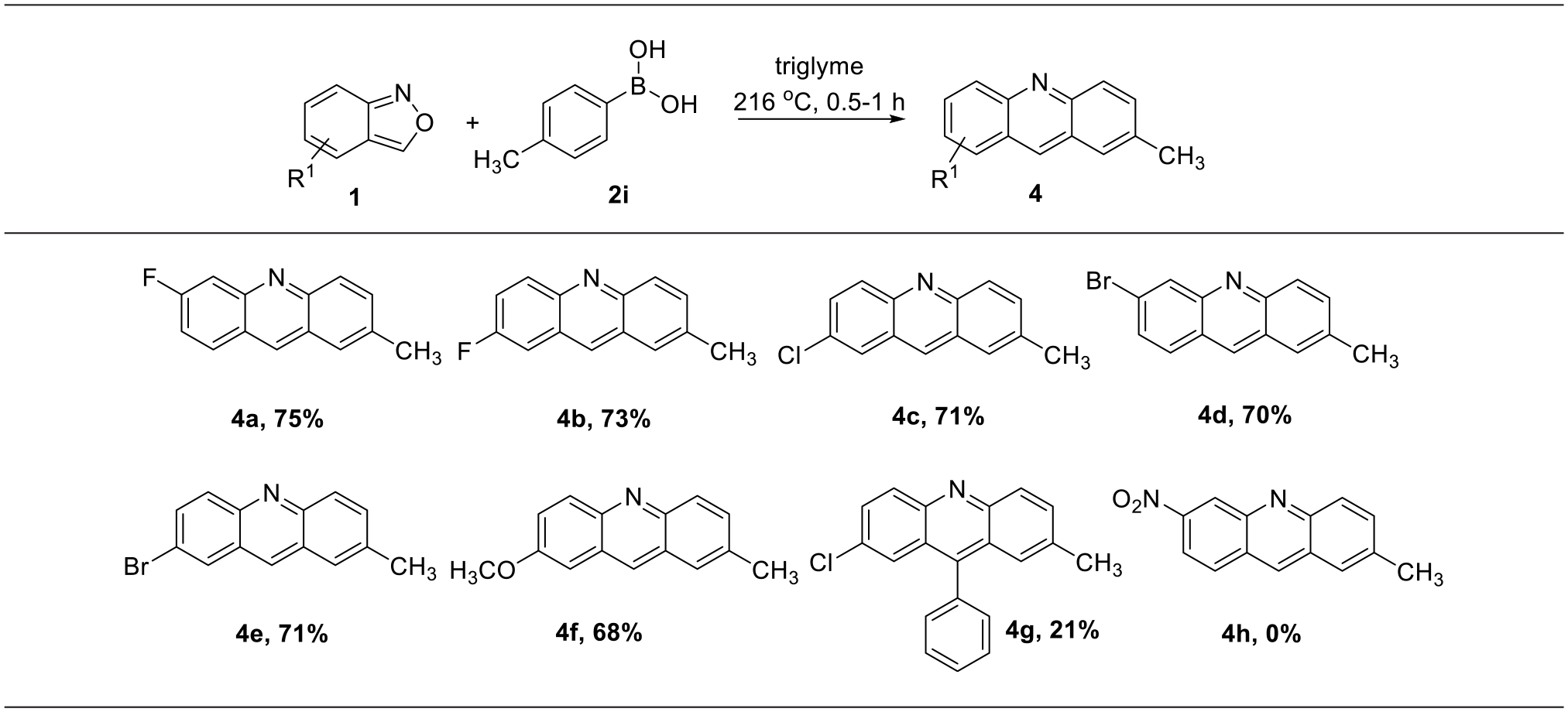
General procedure for synthesis of acridines
Anthranil 1 (1.0 mmol) and arylboronic acid 2 (1.5 mmol) were dissolved in triglyme (5.0 mL). The reaction mixture was stirred at 216 °C using an oil bath for 0.5–1 h and monitored via TLC. Upon completion, the reaction mixture was cooled to room temperature and poured into water. The product was extracted with ethyl acetate (EtOAc) twice, the combined organic phases were washed with water, brine solution, dried over Na2SO4, and filtered. The solution was removed under vacuum and the residue was purified by column chromatography. The crude reaction mixture was purified on silica gel using hexanes/EtOAc as the eluent to afford the desired product 3.
The substituted anthranils were prepared according to the literature methods with minor modifications.
32
All arylboronic acids were all commercially purchased without further purification.
Acridine (3a). The title compound was prepared according to the general procedure.
33
The crude compound was purified by flash column chromatography over silica gel (EtOAc/n-hexane = 1/20) to provide the product as an orange powder (114 mg, yield 64%); m.p. 110 °C–112 °C (literature
33
112 °C–113 °C); Rf 0.50 (10:1 hexane/EtOAc). 1H NMR (400 MHz, CDCl3) δ 8.74 (s, 1H), 8.24 (d, J = 8.8 Hz, 2H), 7.98 (d, J = 8.4 Hz, 2H), 7.78 (t, J = 8.0 Hz, 2H), 7.52 (t, J = 7.6 Hz, 2H). 13C NMR (101 MHz, CDCl3) δ 149.1, 136.1 × 2, 130.3 × 2, 129.4 × 2, 128.2 × 2, 126.6 × 2, 125.7 × 2. HRMS (ESI) m/z [M+H]+ calcd for C13H10N 180.0813, found 180.0835.
4-Chloroacridine (3b). The title compound was prepared according to the general procedure.
10
The crude compound was purified by flash column chromatography over silica gel (EtOAc/n-hexane = 1/20) to provide the product as a yellow powder (123 mg, yield 58%); m.p. 83 °C–85 °C; Rf 0.50 (20:1 hexane/EtOAc). 1H NMR (500 MHz, CDCl3) δ 8.79 (s, 1H), 8.38 (d, J = 8.5 Hz, 1H), 8.01 (d, J = 8.5 Hz, 1H), 7.93 (d, J = 4.0 Hz, 1H), 7.91 (d, J = 3.0 Hz, 1H), 7.84-7.81 (m, 1H), 7.58 (t, J = 7.5 Hz, 1H), 7.44 (t, J = 8.0 Hz, 1H). 13C NMR (125 MHz, CDCl3) δ 149.5, 145.2, 136.9, 133.5, 130.9, 130.3, 130.0, 128.1, 127.6, 127.6, 127.0, 126.7, 125.3. HRMS (ESI) m/z [M+H]+calcd for C13H9ClN 214.0424, found 214.0462.
3-Chloroacridine (3c). The title compound was prepared according to the general procedure.
33
The crude compound was purified by flash column chromatography over silica gel (EtOAc/n-hexane = 1/20) to provide the product as a yellow powder (106 mg, yield 50%); m.p. 119 °C–121 °C (literature
33
119 °C–120 °C); Rf 0.60 (10:1 hexane/EtOAc). 1H NMR (400 MHz, CDCl3) δ 8.76 (s, 1H), 8.24 (s, 1H), 8.21 (d, J = 8.8 Hz, 1H), 7.99 (d, J = 8.4 Hz, 1H), 7.94 (d, J = 8.8 Hz, 1H), 7.81 (t, J = 7.2 Hz, 1H), 7.56 (t, J = 7.2 Hz, 1H), 7.48 (d, J = 8.8 Hz, 1H). 13C NMR (101 MHz, CDCl3) δ 149.5, 148.9, 136.3, 136.2, 130.9, 129.5, 129.3, 129.2, 128.2, 127.9, 126.5, 126.0, 124.8. HRMS (ESI) m/z [M+H]+calcd for C13H9ClN 214.0424, found 214.0458.
2-Chloroacridine (3d). The title compound was prepared according to the general procedure.
33
The crude compound was purified by flash column chromatography over silica gel (EtOAc/n-hexane = 1/20) to provide the product as a yellow powder (149 mg, yield 73%); m.p. 173 °C–175 °C (literature
33
172 °C–173 °C); Rf 0.50 (10:1 hexane/EtOAc). 1H NMR (400 MHz, CDCl3) δ 8.69 (s, 1H), 8.22 (d, J = 8.8 Hz, 1H), 8.18 (d, J = 9.6 Hz, 1H), 8.01–7.99 (m, 2H), 7.81 (t, J = 8.0 Hz, 1H), 7.71 (d, J = 9.0 Hz, 1H), 7.57 (t, J = 7.6 Hz, 1H). 13C NMR (101 MHz, CDCl3) δ 149.1, 147.2, 135.1, 131.6, 131.5, 131.1, 130.7, 129.4, 128.1, 126.9, 126.7, 126.4, 126.3. HRMS (ESI) m/z [M+H]+ calcd for C13H9ClN 214.0424, found 214.0447.
2-Bromoacridine (3e). The title compound was prepared according to the general procedure.
33
The crude compound was purified by flash column chromatography over silica gel (EtOAc/n-hexane = 1/20) to provide the product as a brown powder (179 mg, yield 70%); m.p. 148 °C–150 °C (literature
33
147 °C–148 °C); Rf 0.50 (10:1 hexane/EtOAc). 1H NMR (400 MHz, CDCl3) δ 8.67 (s, 1H), 8.22 (d, J = 8.4 Hz, 1H), 8.17 (s, 1H), 8.11 (d, J = 8.8 Hz, 1H), 7.99 (d, J = 8.0 Hz, 1H), 7.82 (d, J = 8.0 Hz, 2H), 7.57 (t, J = 6.8 Hz, 1H). 13C NMR (101 MHz, CDCl3) δ 149.2, 147.3, 135.0, 133.9, 131.1, 130.8, 129.8, 129.5, 128.2, 127.3, 126.8, 126.5, 119.7. HRMS (ESI) m/z [M+H]+calcd for C13H9BrN 257.9918, found 257.9935.
2-Fluoroacridine (3f). The title compound was prepared according to the general procedure.
33
The crude compound was purified by flash column chromatography over silica gel (EtOAc/n-hexane = 1/20) to provide the product as an orange powder (149 mg, yield 76%); m.p. 119 °C–121 °C (literature
33
120 °C–121 °C); Rf 0.50 (10:1 hexane/EtOAc). 1H NMR (500 MHz, CDCl3) δ 8.65 (s, 1H), 8.24–8.19 (m, 2H), 7.93 (d, J = 8.0 Hz, 1H), 7.76 (t, J = 7.5 Hz, 1H), 7.59–7.51 (m, 3H). 13C NMR (125 MHz, CDCl3) δ 159.8 (d, J = 250 Hz), 148.7, 146.6, 135.2, 132.3 (d, J = 5.0 Hz), 130.2, 129.7, 127.9, 126.8, 126.6, 126.4, 122.2 (d, J = 28.75 Hz), 109.8 (d, J = 22.5 Hz). 19F NMR (470 MHz, CDCl3) δ −113.28. HRMS (ESI) m/z [M+H]+calcd for C13H9FN 198.0719, found 198.0759.
2-(Trifluoromethyl)acridine (3g). The title compound was prepared according to the general procedure.
23
The crude compound was purified by flash column chromatography over silica gel (EtOAc/n-hexane = 1/20) to provide the product as a brown powder (121 mg, yield 49%); m.p. 129 °C–131 °C (literature
23
130 °C–133 °C); Rf 0.40 (10:1 hexane/EtOAc). 1H NMR (500 MHz, CDCl3) δ 8.88 (s, 1H), 8.35–8.37 (m, 2H), 8.27 (d, J = 9.0 Hz, 1H), 8.04 (d, J = 8.5 Hz, 1H), 7.91 (dd, J = 9.0, 1.5 Hz, 1H), 7.88–7.85 (m, 1H), 7.62–7.59 (m, 1H). 13C NMR (125 MHz, CDCl3) δ 150.5, 149.3, 137.8, 131.7, 131.1, 129.8, 128.5, 127.8, 126.8 (q, J = 5.0 Hz), 126.7, 126.1 (q, J = 258.5 Hz), 125.5 (q, J = 2.5 Hz), 125.1, 123.1. 19F NMR (470 MHz, CDCl3) δ −62.75. HRMS (ESI) m/z [M+H]+calcd for C14H9F3N 248.0687, found 248.0696.
Acridine-2-carbonitrile (3h). The title compound was prepared according to the general procedure.
23
The crude compound was purified by flash column chromatography over silica gel (EtOAc/n-hexane = 1/20) to provide the product as a yellow powder (104 mg, yield 51%); m.p. 198 °C–201 °C (literature
23
200 °C–202 °C); Rf 0.30 (5:1 hexane/EtOAc). 1H NMR (500 MHz, CDCl3) δ 8.82 (s, 1H), 8.42 (s, 1H), 8.28 (d, J = 9.0 Hz, 1H), 8.24 (d, J = 8.5 Hz, 1H), 8.03 (d, J = 8.5 Hz, 1H), 7.89–7.86 (m, 1H), 7.82 (dd, J = 9.0, 1.5 Hz, 1H), 7.62 (t, J = 8.0, 1H). 13C NMR (125 MHz, CDCl3) δ 150.8, 148.8, 137.4, 135.7, 132.3, 129.8, 129.6, 128.6, 127.2, 127.1, 125.3, 125.3, 118.8, 109.6. HRMS (ESI) m/z [M+H]+calcd for C14H9N2 205.0766, found 205.0745.
2-Methylacridine (3i). The title compound was prepared according to the general procedure.
33
The crude compound was purified by flash column chromatography over silica gel (EtOAc/n-hexane = 1/20) to provide the product as an off-white powder (142 mg, yield 74%); m.p. 127 °C–129 °C (literature
33
126 °C–128 °C); Rf 0.50 (20:1 hexane/EtOAc). 1H NMR (500 MHz, CDCl3) δ 8.62 (s, 1H), 8.21 (d, J = 8.5 Hz, 1H), 8.13 (d, J = 9.0 Hz, 1H), 7.95 (d, J = 8.5 Hz, 1H), 7.74 (t, J = 7.0 Hz, 1H), 7.71 (s, 1H), 7.61 (d, J = 9.0 Hz, 1H), 7.50 (t, J = 7.0 Hz, 1H), 2.56 (s, 3H). 13C NMR (125 MHz, CDCl3) δ 148.7, 148.2, 135.6, 135.0, 133.4, 129.9, 129.5, 129.2, 128.2, 126.9, 126.9, 126.3, 125.7, 21.9. HRMS (ESI) m/z [M+H]+calcd for C14H12N 194.0970, found 194.0943.
2-Methoxyacridine (3j). The title compound was prepared according to the general procedure.
23
The crude compound was purified by flash column chromatography over silica gel (EtOAc/n-hexane = 1/20) to provide the product as a yellow powder (146 mg, yield 70%); m.p. 92 °C–95 °C; Rf 0.50 (20:1 hexane/EtOAc). 1H NMR (400 MHz, CDCl3) δ 8.59 (s, 1H), 8.20 (d, J = 8.0 Hz, 1H), 8.13 (d, J = 8.8 Hz, 1H), 7.93 (d, J = 7.2 Hz, 1H), 7.71 (s, 1H), 7.51–7.46 (m, 2H), 7.14 (s, 1H), 3.97 (s, 3H). 13C NMR (101 MHz, CDCl3) δ 157.0, 147.3, 146.1, 133.7, 130.8, 129.2, 129.1, 127.6, 127.4, 126.9, 125.8, 125.6, 103.0, 55.5. HRMS (ESI) m/z [M+H]+calcd for C14H12NO 210.0919, found 210.0934.
2-(Methylthio)acridine (3k). The title compound was prepared according to the general procedure. The crude compound was purified by flash column chromatography over silica gel (EtOAc/n-hexane = 1/20) to provide the product as a yellow powder (146 mg, yield 65%); m.p. 94 °C–97 °C; Rf 0.50 (20:1 hexane/EtOAc). 1H NMR (400 MHz, CDCl3) δ 8.59 (s, 1H), 8.19 (d, J = 8.8 Hz, 1H), 8.10 (d, J = 8.8 Hz, 1H), 7.95 (d, J = 8.0 Hz, 1H), 7.74 (t, J = 6.8 Hz, 1H), 7.63–7.61 (m, 2H), 7.52 (t, J = 7.2 Hz, 1H), 2.63 (s, 3H). 13C NMR (101 MHz, CDCl3) δ 148.4, 147.6, 136.7, 133.9, 130.4, 129.8, 129.4, 129.3, 128.0 × 2, 127.0, 126.0, 121.1, 15.4. HRMS (ESI) m/z [M+H]+calcd for C14H12NS 226.0690, found 226.0652.
Acridine-2-carbaldehyde (3l). The title compound was prepared according to the general procedure. The crude compound was purified by flash column chromatography over silica gel (EtOAc/n-hexane = 1/20) to provide the product as a yellow powder (107 mg, yield 52%); m.p. 150 °C–152 °C; Rf 0.40 (10:1 hexane/EtOAc). 1H NMR (400 MHz, CDCl3) δ 10.22 (s, 1H), 8.96 (s, 1H), 8.52 (s, 1H), 8.31–8.22 (m, 3H), 8.07 (d, J = 7.6 Hz, 1H), 7.89 (t, J = 7.6 Hz, 1H), 7.62 (t, J = 6.4 Hz, 1H). 13C NMR (101 MHz, CDCl3) δ 191.3, 150.6, 150.5, 138.6, 136.2, 133.8, 132.1, 130.7, 129.5, 128.6, 126.9, 126.7, 126.3, 125.5. HRMS (ESI) m/z [M+H]+calcd for C14H10NO 208.0762, found 208.0738.
Acridine-2-carboxamide (3m). The title compound was prepared according to the general procedure. The crude compound was purified by flash column chromatography over silica gel (EtOAc/n-hexane = 1/20) to provide the product as a yellow powder (117 mg, yield 53%); m.p. 167 °C–169 °C; Rf 0.40 (10:1 hexane/EtOAc). 1H NMR (400 MHz, CDCl3) δ 9.47 (s, 1H), 8.63 (d, J = 6.8 Hz, 2H), 8.09 (d, J = 8.4 Hz, 1H), 7.93–7.89 (m, 2H), 7.61 (t, J = 7.6 Hz, 1H), 7.59–7.53 (m, 3H). 13C NMR (125 MHz, CDCl3) δ 161.1, 160.5, 150.8, 138.1, 134.1, 133.9, 130.6, 128.7 × 2, 128.6, 128.6, 127.3, 127.2, 123.6. HRMS (ESI) m/z [M+H]+calcd for C14H11NO2 223.0871, found 223.0917.
Benzo[c]acridine (3n). The title compound was prepared according to the general procedure. The crude compound was purified by flash column chromatography over silica gel (EtOAc/n-hexane = 1/20) to provide the product as a yellow powder (141 mg, yield 62%); m.p. 110 °C–112 °C; Rf 0.40 (10:1 hexane/EtOAc). 1H NMR (500 MHz, CDCl3) δ 9.53 (d, J = 7.5 Hz, 1H), 8.64 (s, 1H), 8.39 (d, J = 8.5 Hz, 1H), 8.03 (d, J = 8.0 Hz, 1H), 7.87 (d, J = 7.5 Hz, 1H), 7.84–7.81 (m, 1H), 7.79–7.76 (m, 1H), 7.76–7.72 (m, 2H), 7.71–7.69 (m, 1H), 7.60 (t, J = 7.0 Hz, 1H). 13C NMR (125 MHz, CDCl3) δ 148.0, 147.9, 135.1, 134.1, 131.8, 129.9, 129.8, 129.2, 128.0, 127.9, 127.8, 127.4, 127.2, 126.1, 125.9, 125.4, 125.3. HRMS (ESI) m/z [M+H]+calcd for C17H12N 230.0970, found 230.0934.
Benzo[b]acridine (3o). The title compound was prepared according to the general procedure. The crude compound was purified by flash column chromatography over silica gel (EtOAc/n-hexane = 1/20) to provide the product as a yellow powder (160 mg, yield 71%); m.p. 298 °C–300 °C. Rf 0.40 (10:1 hexane/EtOAc). 1H NMR (500 MHz, CDCl3) δ 9.36 (s, 1H), 8.71 (d, J = 8.0 Hz, 1H), 8.27 (d, J = 9.0 Hz, 1H), 8.06 (d, J = 9.5 Hz, 1H), 8.01 (d, J = 9.5 Hz, 1H), 7.93 (d, J = 9.5 Hz, 1H), 7.87 (dd, J = 8.0, 1.0 Hz, 1H), 7.83–7.79 (m, 1H), 7.71–7.68 (m, 1H), 7.64 (td, J = 7.5, 1.0 Hz, 1H), 7.61–7.57 (m, 1H). 13C NMR (125 MHz, CDCl3) δ 149.5, 148.3, 132.6, 131.4, 130.6, 130.2, 130.1, 129.2, 129.0, 128.6, 128.4, 127.8, 127.6, 126.6, 126.2, 124.3, 123.0. HRMS (ESI) m/z [M+H]+calcd for C17H12N 230.0970, found 230.0946.
[1,3]Dioxolo[4,5-b]acridine (3p). The title compound was prepared according to the general procedure.
23
The crude compound was purified by flash column chromatography over silica gel (EtOAc/n-hexane = 1/40) to provide the product as a yellow powder (118 mg, yield 53%); m.p. 158 °C–161 °C (literature
23
159 °C–162 °C); Rf 0.40 (20:1 hexane/EtOAc). 1H NMR (400 MHz, CDCl3) δ 8.78 (s, 1H), 8.03 (d, J = 8.4 Hz, 2H), 7.74 (t, J = 8.0 Hz, 1H), 7.53 (t, J = 7.2 Hz, 1H), 7.45 (s, 1H), 7.41 (s, 1H), 6.26 (s, 2H). 13C NMR (101 MHz, CDCl3) δ 152.8, 148.2, 148.1, 147.2, 134.3, 129.8, 128.6, 128.3, 125.7, 125.6, 124.5, 103.6, 102.7, 101.9. HRMS (ESI) m/z [M+H]+calcd for C14H10NO2 224.0712, found 224.0735.
1,3-Dimethoxyacridine (3q). The title compound was prepared according to the general procedure. The crude compound was purified by flash column chromatography over silica gel (EtOAc/n-hexane = 1/40) to provide the product as a yellow powder (117 mg, yield 49%); m.p. 116 °C–118 °C; Rf 0.40 (20:1 hexane/EtOAc). 1H NMR (400 MHz, CDCl3) δ 9.02 (s, 1H), 8.13 (d, J = 8.4 Hz, 1H), 7.96 (d, J = 8.0 Hz, 1H), 7.75 (d, J = 6.8 Hz, 1H), 7.45 (t, J = 7.2 Hz, 1H), 7.09 (s, 1H), 6.44 (s, 1H), 4.03 (s, 3H), 3.98 (s, 3H). 13C NMR (101 MHz, CDCl3) δ 162.5, 156.2, 150.8, 149.1, 131.7, 130.7, 128.8, 128.1, 124.8, 124.5, 117.4, 97.7, 97.6, 55.9, 55.8. HRMS (ESI) m/z [M+H]+calcd for C15H14NO2 240.1025, found 240.1058.
Benzo[b][1,8]phenanthroline (3r). The title compound was prepared according to the general procedure. The crude compound was purified by flash column chromatography over silica gel (EtOAc/n-hexane = 1/2) to provide the product as a yellow powder (156 mg, yield 68%); m.p. 236 °C–239 °C; Rf 0.30 (2:1 hexane/EtOAc). 1H NMR (400 MHz, CDCl3) δ 9.07 (d, J = 8.0 Hz, 1H), 8.64 (d, J = 7.6 Hz, 1H), 8.46 (d, J = 7.6 Hz, 1H), 7.89 (t, J = 7.6 Hz, 1H), 7.87-7.83 (m, 1H), 7.72 (td, J = 7.6, 1.2 Hz, 1H), 7.67–7.62 (m, 2H), 7.53–7.49 (m, 1H), 7.02 (d, J = 7.6 Hz, 1H). 13C NMR (101 MHz, CDCl3) δ 159.5, 147.6, 146.2, 134.8, 132.9, 132.2, 128.5, 127.5, 127.5, 127.3, 127.3, 126.5, 125.8, 121.9, 117.8, 113.2. HRMS (ESI) m/z [M+H]+calcd for C16H11N2 231.0922, found 231.0946.
6-Fluoro-2-methylacridine (4a). The title compound was prepared according to the general procedure. The crude compound was purified by flash column chromatography over silica gel (EtOAc/n-hexane = 1/10) to provide the product as a yellow powder (158 mg, yield 75%); m.p. 124 °C–127 °C; Rf 0.50 (10:1 hexane/EtOAc). 1H NMR (500 MHz, CDCl3) δ 8.59 (s, 1H), 8.08 (d, J = 9.0 Hz, 1H), 7.92 (t, J = 9.0 Hz, 1H), 7.78 (dd, J = 11.5, 2.0 Hz, 1H), 7.68 (s, 1H), 7.61 (dd, J = 9.0, 1.0 Hz, 1H), 7.33–7.29 (m, 1H), 2.55 (s, 3H).13C NMR (125 MHz, CDCl3) δ 163.4 (d, J = 250 Hz), 149.2 (d, J = 12.5 Hz), 148.7, 135.6, 135.3, 133.9, 130.5 (d, J = 11.25 Hz), 128.8, 126.5, 126.3, 124.1, 117.7 (d, J = 27.5 Hz), 117.7 (d, J = 20.0 Hz), 21.8. 19F NMR (470 MHz, CDCl3) δ −107.76. HRMS (ESI) m/z [M+H]+calcd for C14H11FN 212.0876, found 212.0889.
2-Fluoro-7-methylacridine (4b). The title compound was prepared according to the general procedure. The crude compound was purified by flash column chromatography over silica gel (EtOAc/n-hexane = 1/20) to provide the product as a yellow powder (154 mg, yield 73%); m.p. 156 °C–159 °C; Rf 0.50 (10:1 hexane/EtOAc). 1H NMR (500 MHz, CDCl3) δ 8.56 (s, 1H), 8.21 (dd, J = 10.0, 5.5 Hz, 1H), 8.11 (d, J = 9.0 Hz, 1H), 7.69 (s, 1H), 7.60 (d, J = 9.0 Hz, 1H), 7.56–7.52 (m, 2H), 2.57 (s, 3H). 13C NMR (125 MHz, CDCl3) δ 169.8 (d, J = 250 Hz), 147.8, 146.1, 136.4, 134.1 (d, J = 7.5 Hz), 133.3, 132.3 (d, J = 10.0 Hz), 129.3, 127.1, 126.9 (d, J = 10.0 Hz), 125.9, 121.6 (d, J = 28.75 Hz), 109.7 (d, J = 21.25 Hz), 22.0. 19F NMR (470 MHz, CDCl3) δ −113.64. HRMS (ESI) m/z [M+H]+calcd for C14H11FN 212.0876, found 212.0897.
2-Chloro-2-methylacridine (4c). The title compound was prepared according to the general procedure. The crude compound was purified by flash column chromatography over silica gel (EtOAc/n-hexane = 1/30) to provide the product as a yellow powder (161 mg, yield 71%); m.p. 216 °C–218 °C; Rf 0.60 (10:1 hexane/EtOAc).1H NMR (500 MHz, CDCl3) δ 8.49 (s, 1H), 8.13 (d, J = 9.5 Hz, 1H), 8.09 (d, J = 9.0 Hz, 1H), 7.91 (d, J = 2.0 Hz, 1H), 7.68 (s, 1H), 7.64 (dd, J = 9.5, 2.5 Hz, 1H), 7.61 (dd, J = 9.0, 2.0 Hz, 1H), 2.56 (s, 3H). 13C NMR (125 MHz, CDCl3) δ 148.3, 146.9, 136.4, 133.9, 133.7, 131.4, 131.3, 131.1, 129.3, 127.1, 127.0, 126.3, 126.2, 21.9. HRMS (ESI) m/z [M+H]+calcd for C14H11ClN 228.0580, found 228.0572.
6-Bromo-2-methylacridine (4d).
33
The title compound was prepared according to the general procedure. The crude compound was purified by flash column chromatography over silica gel (EtOAc/n-hexane = 1/30) to provide the product as a yellow powder (189 mg, yield 70%); m.p. 187 °C–189 °C (literature
33
188 °C–189 °C); Rf 0.60 (10:1 hexane/EtOAc).1H NMR (500 MHz, CDCl3) δ 8.57 (s, 1H), 8.40 (s, 1H), 8.09 (d, J = 9.0 Hz, 1H), 7.80 (d, J = 9.0 Hz, 1H), 7.68 (s, 1H), 7.62 (dd, J = 9.0, 1.5 Hz, 1H), 7.55 (dd, J = 9.0, 2.0 Hz, 1H), 2.56 (s, 3H). 13C NMR (125 MHz, CDCl3) δ 148.8, 148.6, 136.1, 135.2, 134.0, 131.6, 129.4, 129.4, 129.2, 126.9, 126.4, 125.2, 124.3, 21.9. HRMS (ESI) m/z [M+H]+calcd for C14H11BrN 272.0075, found 272.0085.
2-Bromo-2-methylacridine (4e). The title compound was prepared according to the general procedure. The crude compound was purified by flash column chromatography over silica gel (EtOAc/n-hexane = 1/10) to provide the product as a brown powder (189 mg, yield 71%); m.p. 254 °C–258 °C; Rf 0.30 (10:1 hexane/EtOAc). 1H NMR (500 MHz, CDCl3) δ 8.50 (s, 1H), 8.11–8.09 (m, 2H), 8.06 (d, J = 9.5 Hz, 1H), 7.76 (dd, J = 9.0, 2.0 Hz, 1H), 7.69 (s, 1H), 7.62 (dd, J = 9.0, 1.5 Hz, 1H), 2.57 (s, 3H). 13C NMR (125 MHz, CDCl3) δ 148.3, 147.0, 136.5, 133.9, 133.8, 133.4, 131.3, 129.9, 129.3, 127.6, 127.1, 126.3, 119.7, 21.9. HRMS (ESI) m/z [M+H]+calcd for C14H11BrN 272.0075, found 272.0096.
2-Methoxy-7-methylacridine (4f). The title compound was prepared according to the general procedure. The crude compound was purified by flash column chromatography over silica gel (EtOAc/n-hexane = 1/10) to provide the product as a yellow powder (151 mg, yield 68%); m.p. 130 °C–133 °C; Rf 0.30 (10:1 hexane/EtOAc). 1H NMR (500 MHz, CDCl3) δ 8.44 (s, 1H), 8.09 (dd, J = 9.0, 4.0 Hz, 2H), 7.65 (s, 1H), 7.53 (d, J = 9.0 Hz, 1H), 7.42 (d, J = 9.5 Hz, 1H), 7.10 (s, 1H), 3.95 (s, 3H), 2.55 (s, 3H). 13C NMR (125 MHz, CDCl3) δ 157.1, 146.6, 145.8, 135.7, 132.7, 132.2, 131.0, 129.2, 127.7, 127.1 125.9, 125.1, 103.2, 55.6, 21.9. HRMS (ESI) m/z [M+H]+calcd for C15H14NO 224.1075, found 224.1039.
2-Chloro-7-methyl-9-phenylacridine (4g). The title compound was prepared according to the general procedure. The reaction mixture was stirred at 216 °C using an oil bath for 6 h. The crude compound was purified by flash column chromatography over silica gel (EtOAc/n-hexane = 1/10) to provide the product as a yellow powder (63 mg, yield 21%); m.p. 235 °C–237 °C; Rf 0.30 (10:1 hexane/EtOAc). 1H NMR (500 MHz, CDCl3) δ 8.19 (d, J = 9.0 Hz, 1H), 8.15 (d, J = 9.0 Hz, 1H), 7.65–7.60 (m, 6H), 7.41 (dd, J = 7.5, 2.0 Hz, 2H), 7.39 (s, 1H), 2.45 (s, 3H). 13C NMR (125 MHz, CDCl3) δ 147.9, 146.5, 145.2, 136.3, 135.5, 133.2, 131.4, 131.3, 130.7, 130.4 × 2, 129.4, 128.7 × 2, 128.6, 125.7, 125.4, 125.0, 124.7, 22.0. HRMS (ESI) m/z [M+H]+calcd for C20H15ClN 304.0893, found 304.0911.