Abstract
This article explores the application of Fenton oxidation as a sustainable and efficient method for the treatment of glycerol, a major byproduct in biodiesel production. Glycerol, rich in impurities, poses environmental challenges if not properly managed. The research aims to contribute to the understanding of Fenton oxidation mechanisms specific to glycerol transformation into dl-glyceraldehyde and to highlight its potential as a sustainable solution. Glycerol, a trihydroxy sugar alcohol, is a versatile compound derived primarily from the hydrolysis of triglycerides and has found applications in various industries, including pharmaceuticals, cosmetics, and food production. One intriguing aspect of glycerol is its potential conversion to dl-glyceraldehyde, a key intermediate with significant importance in various biological processes. Employing Fenton oxidation emerges as beneficial approach in glycerol processing, considering the tenets of environmental sustainability.
Keywords

Introduction
The Fenton oxidation of glycerol (GLY) to glyceraldehyde is a versatile and environmentally friendly process with applications in both chemical synthesis and wastewater treatment. Understanding the mechanism, optimizing reaction conditions, and exploring recent advancements contribute to the ongoing development and implementation of this oxidation process. These processes leverage the generation of highly reactive species to facilitate the degradation of recalcitrant organic compounds. Fenton oxidation, named after its discoverer Fenton, 1 relies on the catalytic properties of iron ions (Fe2+) and hydrogen peroxide (H2O2) to generate hydroxyl radicals (•OH). In this reaction, as well known, ferrous ions (Fe2+) catalyze the decomposition of hydrogen peroxide (H2O2), producing ferric ions (Fe3+), hydroxyl radicals, and hydroxide ions. The hydroxyl radicals generated in the process are highly reactive and nonselective in their reactivity, making Fenton oxidation effective. Fenton oxidation’s versatility lies in its ability to operate under mild conditions and its adaptability to a wide range of starting materials.
Fenton oxidation involves a series of intricate chemical reactions. The primary steps include the oxidation of ferrous (Fe2+) ions to ferric (Fe3+) ions. The nonselective nature of hydroxyl radicals makes Fenton oxidation a versatile method for degrading complex and persistent organic compounds. The overall efficiency of Fenton oxidation is influenced by factors such as temperature, pH, and the concentrations of Fe2+ and H2O2.
Research studies have consistently demonstrated the efficacy of Fenton oxidation in GLY treatment. Work by Zhang et al. 2 highlighted the successful removal of GLY impurities using Fenton oxidation, emphasizing its ability to efficiently degrade soap molecules and organic byproducts. This literature underscores the potential of Fenton oxidation as a robust solution for GLY treatment in biodiesel production processes. Fenton oxidation’s versatility lies in its ability to operate under mild conditions and its adaptability to a wide range of pH levels. This makes it a practical choice for integration into biodiesel production processes where GLY purification is essential. Furthermore, Fenton oxidation exhibits selectivity in targeting specific GLY impurities, demonstrating its capability to address the intricate mixture of contaminants present in GLY byproducts.
The concentrations of ferrous ions (Fe2+) and hydrogen peroxide (H2O2) are key factors influencing Fenton oxidation efficiency. Higher concentrations generally lead to increased hydroxyl radical production; however, an optimal molar ratio must be maintained to avoid diminishing returns or the formation of byproducts.
The pH of the reaction medium profoundly influences Fenton oxidation. The Fenton reaction is highly pH-dependent, with the optimal pH range typically lying between 2 and 4. At lower pH values, the rate of Fe2+ oxidation increases, leading to more efficient hydroxyl radical generation. However, extremely low pH can result in the precipitation of iron ions, reducing their availability for the Fenton reaction. Conversely, at higher pH values, the precipitation of Fe(OH)3 occurs, limiting the effectiveness of Fenton oxidation. Research by Neyens and Baeyens 3 and Klavarioti et al. 4 has explored the intricate relationship between pH and Fenton oxidation, emphasizing the importance of maintaining an acidic environment for optimal performance.
Fenton oxidation has found diverse applications in the treatment of GLY from various industries, including textile, pharmaceutical, and petrochemical sectors. Moreover, Fenton oxidation can be integrated into the remediation of GLY manufacturing sites, offering a sustainable approach to environmental cleanup.
Haber and Weiss 5 laid the groundwork for understanding Fenton oxidation mechanisms, investigating the effect of reactant concentrations on the overall reaction kinetics. Neyens and Baeyens 3 provided comprehensive insights into the influence of pH on the Fenton process, highlighting the significance of maintaining an acidic environment for optimal hydroxyl radical generation.
Buxton et al. 6 contributed to understanding the temperature dependence of the Fenton reaction, elucidating the impact of temperature on the decomposition of hydrogen peroxide. Wang et al. 7 explored the interplay between Fe2+ and H2O2 concentrations, emphasizing the importance of finding the right balance to enhance Fenton oxidation efficiency. Elevated temperatures generally enhance reaction kinetics, accelerating the generation of hydroxyl radicals.
The synergistic action of Fe2+ and H2O2 in Fenton reactions enhances the degradation of various organic pollutants, making it a versatile and efficient method for environmental remediation. The nonselective nature of hydroxyl radicals allows Fenton oxidation to target a broad spectrum of contaminants, including dyes, pesticides, and pharmaceuticals. Understanding the pivotal roles of iron ions and hydrogen peroxide in Fenton reactions is crucial for optimizing reaction conditions in practical applications. Ongoing research focuses on enhancing Fenton oxidation efficiency, exploring catalyst modifications, and tailoring reaction parameters to address specific environmental challenges. The symbiotic relationship between iron ions and hydrogen peroxide forms the backbone of Fenton oxidation, making it a powerful and widely applicable advanced oxidation process.
Materials and methods
dl-glyceric aldehyde was prepared as described elsewhere 1 with minor modifications. The main difference in procedure was that the hydrogen peroxide was all added in 6 h. All chemicals used were of analytical grade and purchased from Sigma-Aldrich, Germany, and were used without further purification. Solutions were all prepared using doubly distilled water. All reactions were performed using a thermostated and stirred round-bottom flask with 250 cm3 capacity with magnetic stirring. All reactions were performed at or below a temperature of 5°C. The reaction mixture was kept constantly in an ice-water bath.
The reaction was investigated parallel with electron paramagnetic resonance (EPR) spectroscopy. EPR spectroscopy stands as a powerful analytical technique for elucidating the intricacies of chemical reactions, particularly in the context of advanced oxidation processes like the Fenton reaction. EPR spectroscopy is a technique specifically designed for studying materials with unpaired electrons. In the context of the Fenton reaction, EPR allows researchers to detect and quantify paramagnetic species, including the Fe3+ ion. X-band EPR spectroscopy measurements were carried out at the EPR laboratory of the Budapest University of Technology and Economics by using a Bruker ElexSys E500 X-band EPR spectrometer. Solution samples (1 mL each) were measured at 150 K in quartz tube. The modulation amplitude: 1G, the microwave power: 2.074 mW, microwave frequency: f = 9.3 GHz. Quartz tube filled with ca. 1 mL distilled water was measured as baseline. The baseline was subtracted from the spectra before further processing. In addition, the magnetic field axis was rescaled to f = 9.33 GHz microwave frequency in all cases (Figure 1).

EPR measurement samples.
EPR measurement samples
Results
The crude dl-glyceric aldehyde (GLA) syrup was prepared essentially as described by Fenton and Jackson. 12.00 g (0.13 mol, 9.85 mL, d = 1.22 g/cm3) of GLY in 50 mL of water, to which 1.00 g of ferrous sulfate (FeSO4*7H2O) previously dissolved in 15 mL of water and 1 mL cc. H2SO4 have been added and are placed in a 250 mL flask. About 80 mL of 6% hydrogen peroxide (1.1 mols hydrogen peroxide for 1 mol GLY) was added at the rate of about 13 mL in an hour, care being taken to avoid a rise in temperature of more than 5°C with the following rate:
13 mL was added in the first hour,
13 mL in the second hour,
13 mL in the third hour,
13 mL in the fourth hour,
13 mL in the fifth hour,
and the remainder (15 mL) in 1 h more, as shown in Figure 2.
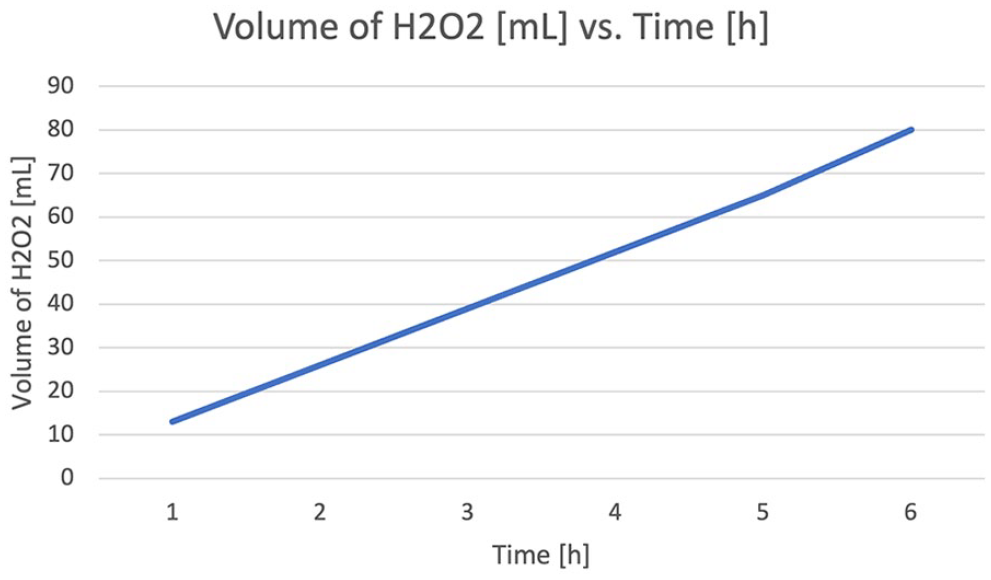
Volume of H2O2 in time.
By titration with Fehling’s solution, shows the sugar derivable from GLY. Fehling’s solution reduced the reaction mixture slowly in the cold, in a minute or 2 at room temperature and instantly at 50°C.
After adding a portion of the hydrogen peroxide, it seems desirable to wait before adding subsequent portions until the red color of the reaction mixture has faded. The mixture was kept in ice water throughout the experiment. The mixture was highly acidic, around pH = 2 during the experiment. The hydrogen peroxide was added in small portions at first (2–3 mL) but in the later stages as much as 5 mL was added at one time.
The pH of reaction was monitored by pH strips. No more H2SO4 have been added to maintain and keep the pH condition at pH = 2. All the hydrogen peroxide was added in 1 working day within 6 h. When all the hydrogen peroxide has been added, the mixture is allowed to stand 24 h to allow the oxidation to continue until the solution becomes clear and pale yellow in color. This mixture, on standing in the refrigerator overnight, had become pale yellow in color and gave a negative test for hydrogen peroxide, means the absence of GLY.
This reaction mixture was then neutralized at room temperature as completely as possible by shaking with excess of powdered calcium carbonate. The sediment (calcium carbonate) was filtered off by means of a suction filter. The filtrate is then concentrated to a syrup by distilling in vacuo (bath temperature: 45°C), during which process a considerable quantity of calcium salts separate in the bottom of the flask (mostly calcium sulfate). The residue from the distillation was then mixed with two volumes of absolute alcohol. This solution was then freed from its precipitate by sedimentation and filtration and again reduced to a thick, clear, pale yellow syrup as a crude product. The pale yellow syrup was purified by distilling in vacuum at 0.8 mmHg. The boiling point of the liquid was between 140–144°C, same as in the literature, 1 data clearly indicated that the product is the dl-GLA. The total yield of 10.05 g (85.6%) pure pale yellow syrup as dl-GLA was produced within 6 h reaction time. All other chemical properties measured are identical as written in literature earlier. The HPLC (high performance liquid cromatography) spectra showed 95% purity of the distilled dl-GLA as a sole product. As a proof, 2 mL portion of our syrup was treated with calculated quantity of hydroxylamine, and there was no sign of the formation of 1,3-dihydroxyacetone oxime crystals, concluded that the product obtained is dl-GLA. All the above-stated facts harmonize well with the supposition that the syrup contains only the dl-GLA product, as discovered by Fenton and Jackson.
The reaction was monitored by the classical quantitative Benedict’s test, based on that only dl-GLA reacts selectively with Benedict’s reagent in this reaction mixture. The amount produced GLA was measured hourly, when 1 mL of sample was used for the titration, the results are shown in Figure 3. The test is based on Benedict’s reagent, which is a mixture of sodium citrate, sodium carbonate, and CuSO4*5H2O. Briefly, when a GLA is subjected to heat in the presence of an alkali, it gets converted into an enediol; therefore, when GLA is present in the analyte, the cupric ions (Cu2+) in Benedict’s reagent are reduced to cuprous ions (Cu+). These cuprous ions form copper(I)oxide with the reaction mixture and precipitate out as a brick-red colored compound. Our standard Benedict’s solution for qualitative measurements was prepared by dissolving 17.3 g of copper sulfate pentahydrate (CuSO4*5H2O), 100 g of sodium carbonate (Na2CO3), and 173 g of sodium citrate in 1000 mL of distilled water. We found that GLA had a reducing power 1.145 times that of glucose, means the corresponding value for 10 mg of GLA is 23.358 mg of copper.

Amount of glycerol in time.
The amount of remained GLY measure was calculated using the initial amount of the GLY and the hourly measured GLA data, as shown in Figure 4.

Amount of glyceric aldehyde in time.
Discussions
The overall reaction for Fenton oxidation of GLY can be represented as follows: 1
The mechanism of Fenton oxidation involves the following steps:
(a) Formation of hydroxyl radicals: H2O2 is decomposed in the presence of Fe2+ to form hydroxyl radicals (•OH): H2O2 + Fe2+ → •OH + OH− + Fe2+
(b) Oxidation of GLY: Hydroxyl radicals react with GLY to initiate the oxidation process: C3H8O3 + •OH → C3H6O3 + H2O
(c) Regeneration of Fe2+: The formed Fe3+ in the reaction is reduced back into Fe2+ ion form: Fe3+ + e− → Fe3+
Iron ions, particularly in the ferrous state (Fe2+), are integral to the Fenton oxidation process. The catalytic role of Fe2+ involves its interaction with hydrogen peroxide, initiating the chain of reactions that lead to the formation of hydroxyl radicals. The iron ions facilitate the decomposition of hydrogen peroxide into hydroxyl radicals and other intermediates, creating a cascade of reactive species that target and break down organic pollutants.
Hydrogen peroxide serves as the oxidizing agent in Fenton oxidation, providing the necessary oxygen source for the generation of hydroxyl radicals. The interaction between Fe2+ and H2O2 leads to the formation of Fe3+, hydroxyl radicals, and hydroxide ions, as outlined in the Fenton reaction. The steady supply of hydrogen peroxide ensures the sustained production of hydroxyl radicals, amplifying the oxidative potential of Fenton oxidation. The temperature of the Fenton oxidation process plays a pivotal role in determining reaction rates and overall efficiency. Therefore, maintaining an optimum temperature is crucial for maximizing the efficiency of Fenton oxidation. Temperature significantly influences reaction kinetics in Fenton oxidation. Elevated temperatures generally accelerate the Fenton reaction, leading to increased hydroxyl radical production. However, excessively high temperatures may lead to undesired side reactions or thermal decomposition of hydrogen peroxide. The temperature of the Fenton oxidation process plays a pivotal role in determining reaction rates and overall efficiency. The pH of the reaction medium plays a critical role in Fenton oxidation. The optimal pH range typically lies around 2, as lower pH values enhance the rate of iron oxidation, while higher pH values lead to the precipitation of iron or iron hydroxides, limiting the effectiveness of the process. The concentrations of ferrous ions (Fe2+) and hydrogen peroxide are critical determinants of Fenton oxidation efficiency.
Nevertheless, there exists an optimal molar ratio of Fe2+ to H2O2 that maximizes efficiency without excessive generation of byproducts. Literature, including studies by Haber and Weiss, emphasizes the importance of carefully controlling the concentrations of Fe2+ and H2O2 to avoid diminishing returns or the formation of byproducts. The measure of GLY and GLA in time is shown in Figure 5. The used conditions driven to the almost pure dl-GLA. Our optimal GLY conversion could reach 85.6% with 95% selectivity to dl-glyceraldehyde. Earlier Fenton and Jackson treated their portion of the syrup with prussic acid according to the method of Fischer and Tafel. Tri-hydroxyisobutyric acid, which should have been formed if dihydroxy-acetone was present, could not be found, indicated the dl-GLA as a sole product.

Glycerol and glyceric aldehyde in time.
The pH of the reaction medium profoundly influences Fenton oxidation. The Fenton reaction is highly pH-dependent, with the optimal pH range typically lying around 2. At lower pH values, the rate of Fe2+ oxidation increases, leading to more efficient hydroxyl radical generation. However, extremely low pH reducing their availability for the Fenton reaction. Conversely, at higher pH values, the precipitation of Fe(OH)3 occurs, limiting the effectiveness of Fenton oxidation. Important relationship between pH and hydroxyl radical generation during Fenton oxidation, emphasizing the importance of maintaining an acidic environment for optimal performance. The used pH conditions driven to the almost pure dl-GLA.
We delve into the role of EPR spectroscopy in unraveling the reactive species involved and providing mechanistic insights into the Fenton reaction. Understanding the intricate mechanisms and reactive species involved in the Fenton reaction is crucial for optimizing its application in environmental remediation and industrial processes. EPR spectroscopy emerges as a key tool for probing these elusive radicals and gaining mechanistic insights into the Fenton reaction. The characteristic EPR signal arising from the Fe3+ provides a quantitative measure of its concentration. This information is invaluable for understanding the kinetics of radical formation and the factors influencing their generation. Beyond hydroxyl radicals, EPR spectroscopy is instrumental in characterizing various iron species involved in the Fenton reaction.
EPR allows the discrimination between ferrous (Fe2+) and ferric (Fe3+) ions, providing insights into the redox transformations occurring during the catalytic cycle. EPR spectroscopy acts as a molecular probe, unveiling the mechanistic intricacies of the Fenton reaction. It assists in elucidating the radical-generating pathways, identifying key intermediates, and discerning the influence of reaction conditions on the overall process. EPR spectroscopy ability to provide real-time, in situ information on radical concentrations and iron speciation positions it as a valuable tool for researchers and practitioners alike. The non-destructive nature of EPR allows for continuous monitoring of Fenton reactions, facilitating dynamic investigations and optimizing reaction conditions for various applications.
Complexation sites in fulvic acid were investigated earlier with Klencsár and Köntös, 8 where manganese (Mn2+) ion was investigated also, which opens us a new way to investigate the Fenton reaction with EPR spectra. Mn2+ sextet to have equal value in the spectra, but allowing the amplitudes of the sextets to change independently during the fit. EPR spectrum of Mn2+ experienced and modeled earlier, with parameters g = 2.00232, D = −0.045 cm−1, λ = 0.33333, A = −23.75 T, and Γf = 0.6 GHz. A sextet of equidistant (Aav = 89.24 G) Lorentzian derivatives superimposed on a broad Voigt derivative provided an acceptable approximation of the theoretical spectrum in the low-field range. 8
The amplitude of the full range theoretical signal is set arbitrarily to match well that of the measured spectrum. Appeared that average hyperfine splitting (the average distance measured in Gauss) between neighboring peaks of the Mn2+ sextet reflects, approximately, the degree of ionicity of the bonds involving the manganese ion. Negative ligands of Mn2+ can establish covalent bonding by donating electrons to states which is responsible for the hyperfine splitting.
In this experiment, a very small, but calculated amount of MnCl2*4H2O was added to the FeSO4*7H2O solution; therefore, the Fenton reaction mixture contained a little amount of manganese (Mn2+) ion. The sextet remains unchanged during the Fenton reaction. The EPR spectra of Fenton reaction samples may be used to follow and characterize changes in reaction conditions, when manganese ion has stable structures in response to be in the future as a base sign of the other variable conditions during Fenton-type reactions in time. By using the newly suggested way, by adding little amount manganese ion (Mn2+) to the Fenton reaction mixture, it is possible to account for the low-field range of X-band EPR spectra of Fe3+ and Mn2+ containing Fenton reaction mixture and frozen solution samples with high accuracy, with the corresponding analysis providing detailed information about the reaction pathway for future scientists. A sharp peek ranging roughly from 2500 to 4500 G with a sextet feature centered at around B = 3300 G. The sextet signal centered at geff = 2 is a fingerprint of paramagnetic Mn2+ ions subject to hyperfine magnetic interaction with their I = 5/2 spin55 Mn nucleus, as described earlier. The applied method in this research may, therefore, open up new ways to study the structure and chemical interactions of Fenton reactions on the basis of their EPR spectra and the manganese sign. The lower amplitude of the low-field Mn2+ sextet signal in the frozen solution sample indicates that with the incorporation of further water molecules into the first coordination sphere of the Mn2+ complexes.
Amount of mononuclear Fe3+ complexes (red magnetic field range) clearly correlates with the amount of H2O2 added. Broad background peaks may indicate the presence of magnetic polynuclear complexes or nanoparticles, as shown in Figures 6 and 7. The samples contained 0.01 g of MnCl2*4H2O also as our new reference signal. Using the S0 sample, the ratio between the Fe2+ and H2O2 was 10:1 in the S1 sample, but 100:1 within the S2 sample.

Amount of mononuclear Fe3+ complexes.
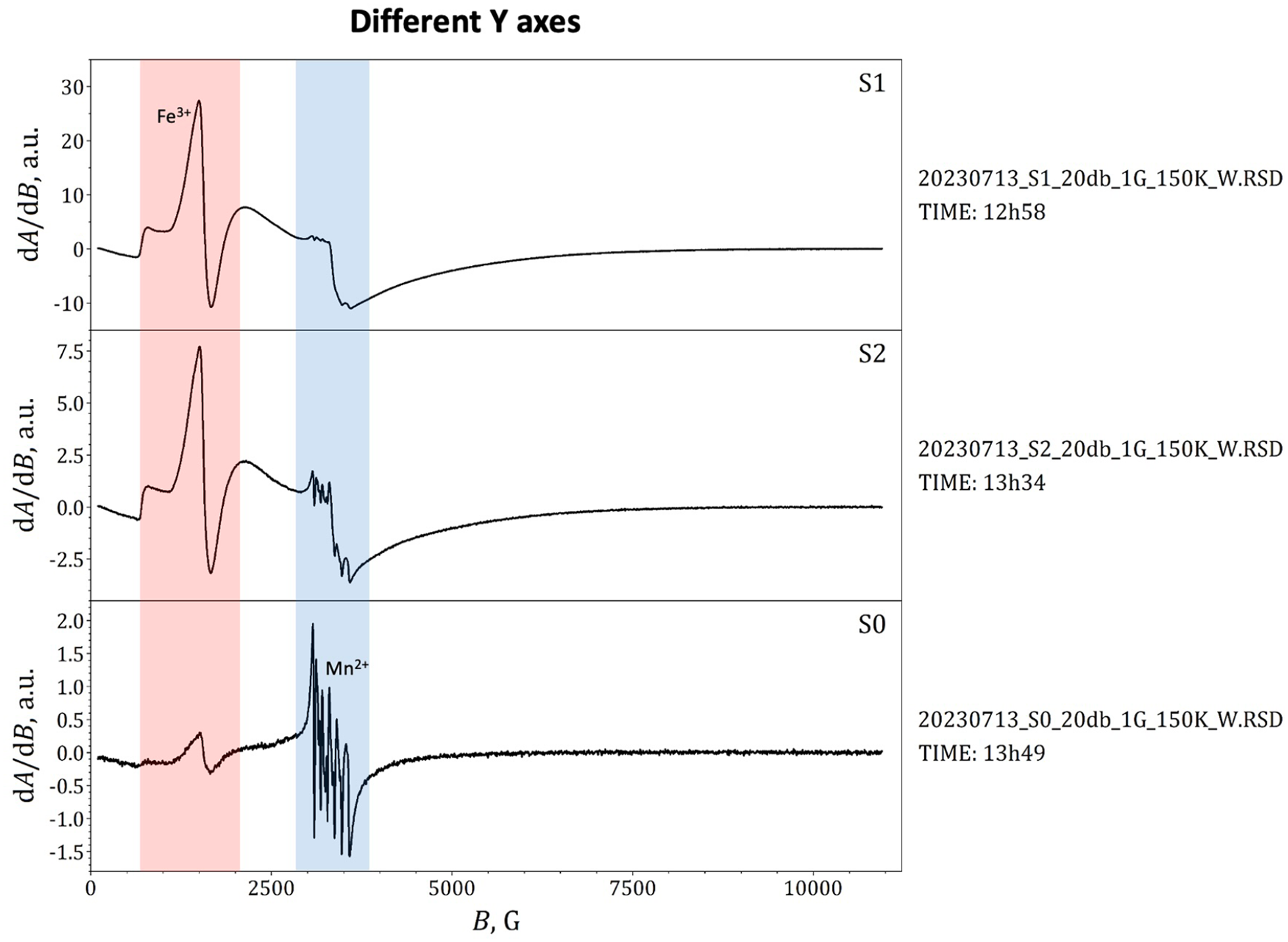
Amount of mononuclear Fe3+ complexes.
In the S1 sample, observed a slight decrease in signal amplitudes with the progress of time, as shown in Figure 8. The ratio between the Fe2+ and H2O2 was 10:1 in the S1 sample.

Fe3+ signal change in time in the S1 sample.
The S2 sample shows an increase first and a slight decrease in signal amplitudes with the progress of time, as shown in Figure 9. The ratio between the Fe2+ and H2O2 was 100:1 in the S2 sample.

Fe3+ signal change in time in the S2 sample.
Different ratios of Fe2+ and H2O2 show different picture of the Fe3+ generation from Fe2+ ion. When the ratio between Fe2+ and H2O2 was 10:1 in the S1 sample, a negative linear gradient was observed, when the highest concentration of Fe3+ was decreased to the minimum value within 300 min. Observed that, when 1.1 mL H2O2 was added to the reaction mixture, Fe2+ ions were oxidized immediately, within 1 s to Fe3+, then produced Fe3+ turned back to Fe2+ form within 300 min, as observed the decreasing trend of the Fe3+ ionic form. EPR spectra shows that amount of mononuclear Fe3+ complexes clearly correlates with the amount of H2O2 added.
When the ratio between Fe2+ and H2O2 was 100:1 in the S2 sample, and only 0.11 mL H2O2 was added to the reaction mixture, we discovered a long 150 min period, when Fe2+ ions were slowly oxidized to Fe3+, then Fe3+ ions turned back into Fe2+ form in 300 min. Starting from 150 min, the same negative linear gradient was observed as we seen above, when the highest concentration of Fe3+ decreased to the minimum value at 300 min. Amount of mononuclear Fe3+ complexes clearly correlates with the amount of H2O2 added.
Recent research has focused on optimizing the Fenton oxidation process to enhance its efficiency and selectivity. During our experience, the hydrogen peroxide was added in small portions and aim to the slow production of Fe3+ ions. Higher initial concentration of Fe3+ may lead to increased hydroxyl radical production, but in most cases, byproducts given. An optimal molar ratio between Fe2+ and H2O2 must be maintain to avoid byproducts, by increasing the time needed to achieve the initial maximum concentration of Fe3+ ions in the reaction mixture.
Conclusion
Fenton oxidation has found diverse applications in the treatment of GLY from various industries, including pharmaceutical, and petrochemical sectors. Moreover, Fenton oxidation can be integrated into the remediation of contaminated sites, offering a sustainable approach to environmental cleanup by converting GLY to useful products like dl-GLA. Fenton oxidation is generally considered environmentally friendly compared with traditional methods. The process produces nontoxic byproducts, and the recovery of iron as a catalyst contributes to resource efficiency. In conclusion, Fenton oxidation offer a powerful and versatile approach to the treatment of GLY. The conversion of GLY to dl-GLA represents a fascinating area of research with significant implications for both the chemical and biological sciences. Various methods, ranging from chemical oxidation to enzymatic processes, have been explored to achieve this conversion. The potential applications of dl-glyceraldehyde in diverse industries underscore the importance of advancing our understanding of Fenton-type conversion processes. The synthesis of dl-glyceraldehyde from GLY may pave the way for sustainable and economically viable pathways in chemical synthesis. The optimal concentration ratio between ferrous ions (Fe2+) and hydrogen peroxide looks as key factors influencing Fenton oxidation efficiency, may lead high yields with high purity of compounds as using in GLY oxidation into dl-GLA. GLY valorization through Fenton oxidation is a good way of obtaining many different molecules with high added value like dl-GLA, which is an important intermediate in the synthesis of various chemicals, including pharmaceuticals and fine chemicals. Fenton oxidation provides a green and efficient route to produce dl-GLA from GLY. Fenton oxidation faces challenges such as the real generation dynamics of Fe3+ ions, one of the needs of controlled, manageable reaction conditions. Future research may explore novel reaction engineering strategies to overcome these challenges and further establish the Fenton oxidation of GLY as a sustainable and economically viable process.
Footnotes
Acknowledgements
The author expresses his thanks to Professor Ferenc Simon (Institute of Physics, Budapest University of Technology and Economics, Budapest, Hungary) for making available the applied spectrometer for recording the EPR spectra. The author expresses his special thanks to Dr. Zoltán Klencsár.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.

