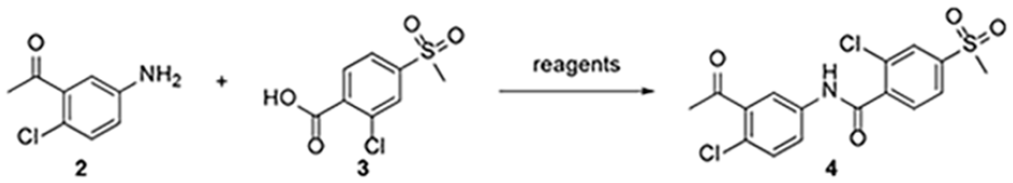
Abstract
Vismodegib is the first hedgehog signaling pathway inhibitor approved by the Food and Drug Administration (FDA) for the treatment of basal-cell carcinoma. The reported methods for the preparation of vismodegib mostly require the employment of precious metal catalysts and phosphine ligands. An alternative low-cost method avoiding the employment of precious metal catalysts and phosphine ligand is developed to prepare vismodegib in a laboratory. The reported method is applied in an undergraduate experimental course to verify its reproducibility and to develop the experimental skills of students. Moreover, the teaching practice experiences are summed up to provide suggestions for the development of exploratory and further experimental courses.
Introduction
Vismodegib is a hedgehog signaling pathway inhibitor developed by the Genentech company of the Roche Group. It was approved by the Food and Drug Administration (FDA) in January 2012 and by the European Medicines Agency (EMA) in July 2013 for the treatment of metastatic and locally advanced basal-cell carcinoma (BCC) that cannot be treated by surgery or radiotherapy. 1 In terms of neoadjuvant therapy, vimodegib is recommended for non-recurrent BCC in functional sensitive sites. 2 In addition to being approved for the treatment of BCC, a number of clinical trials are underway utilizing vismodegib in combination with other drugs. 3
2-(2-Chloro-5-nitrophenyl)pyridine is the key intermediate for the synthesis of vismodegib, which is converted into 4-chloro-3-(pyridin-2-yl)aniline

Representative synthetic routes for the preparation of vismodegib.
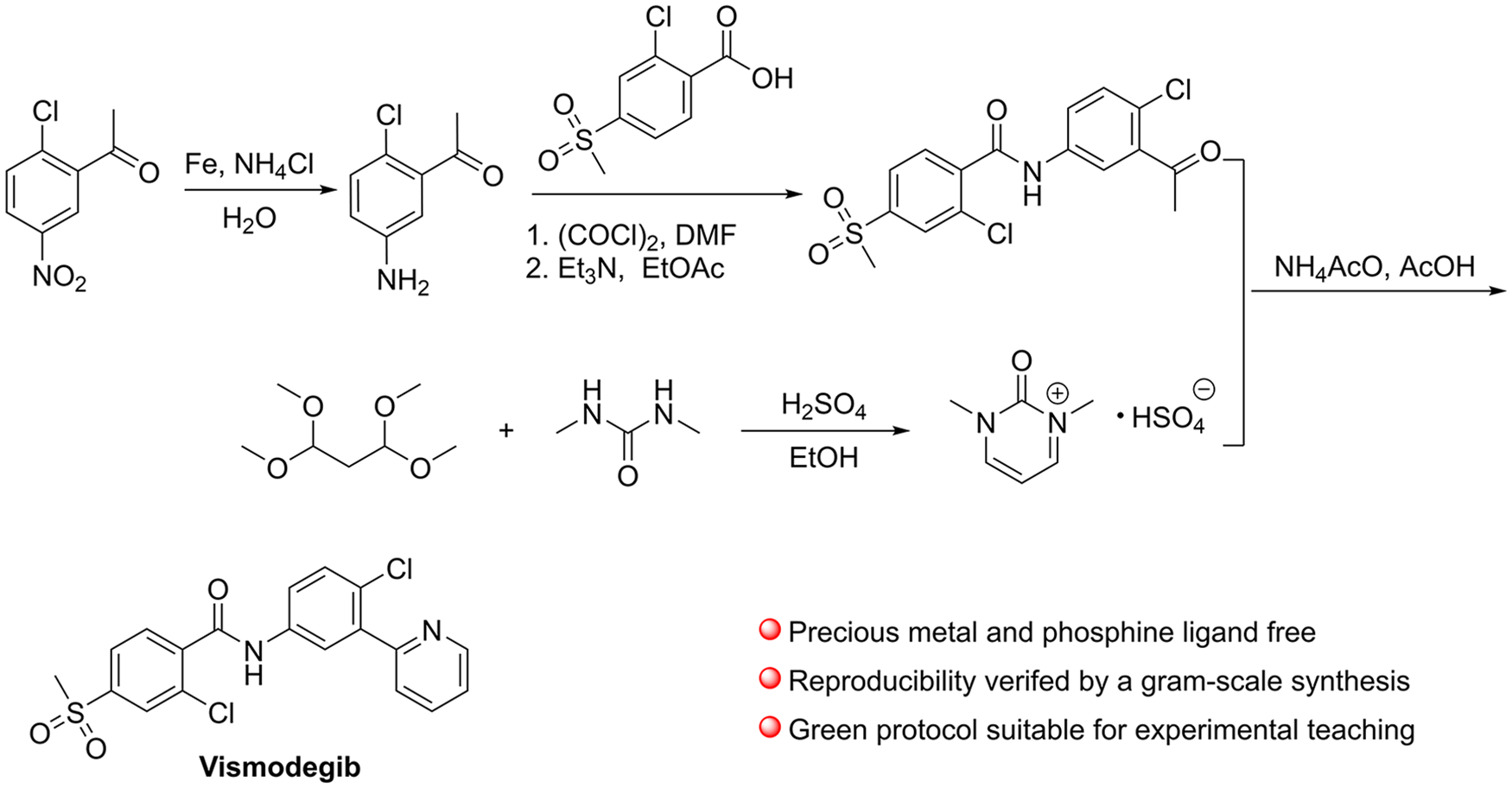
In route F, 2-chloroacrylaldehyde, employed as the starting material, is not commercially available and is highly toxic.12,13 In route G, 2-(2-chloro-5-nitrophenyl)pyridine was synthesized
In terms of the reduction of 2-(2-chloro-5-nitrophenyl)pyridine, catalytic hydrogenation using molecular hydrogen and reduction using Fe under acidic conditions are the most commonly used methods reported in the literature (Scheme 1).4 –11 Catalytic hydrogenation is environmentally friendly and widely used in industry; however, as mentioned earlier, metal residues in drugs will be encountered due to the use of palladium or platinum, and the determination of their levels is necessary. Besides, the use of pressurized equipment is required, but some laboratories cannot use hydrogen gas cylinders due to safety reasons. Although reduction employing low-cost iron can be carried out in almost any chemical laboratory, appropriate disposal of the generated waste is necessary to reduce environmental pollution.
To avoid the use of precious metals, the method for the preparation of 2-(2-chloro-5-nitrophenyl)pyridine reported in route G was adopted, and the catalytic hydrogenation employing platinum/vanadium or palladium was replaced with iron-mediated reduction, which is suitable for preparing vismodegib at low cost under conditions where catalytic hydrogenation cannot be used. Moreover, in order to simply remove the alkaline chemicals such as the remaining reactant-substituted aniline, during the condensation step by washing with acid aqueous, the construction of the phenyl pyridine was conducted after the condensation, and reduction of the substituted nitrobenzene was brought forward. In this alternative method, 2-chloro-5-nitroacetophenone (

Our route to vismodegib (see the tables below for yields and additional details).
In addition, because of the low cost and the implementability in undergraduate laboratories, this method was introduced in undergraduate laboratory courses, the reproducibility of the synthetic protocol was validated, and the experimental skills of undergraduate students were developed. The course included studies on the experimental operations, data analysis, purity, and structure determination, as well as calculation metrics to “green” chemistry, and so on.
Results and discussion
There was no commercial supply of 1,2-dihydro-1,3-dimethyl-2-oxypyridinium salt; however, it could be obtained
According to the nitro reduction methods reported in the literature,16 –18 iron, zinc, and sodium dithionite were employed to prepare 2-chloro-5-aminoacetophenone. Compared with the catalytic hydrogenation using Pd-C/H2, these methods could effectively avoid generation of the dechlorination byproduct and were suitable in situations without hydrogen gas. As shown in Table 1, iron-mediated reduction gave the highest yield, and the amount of iron powder and ammonium chloride influenced the reaction rate and yield. Four equivalence of iron powder and ammonium chloride were suitable, and under these conditions, the reaction was complete within 1.5 h with yields of 86%~93%. When the amount of iron powder and ammonium chloride were reduced, an extended time was required to complete the reaction and the yield decreased. As for the reduction using zinc and acetic acid, not only was the yield the lowest, but also an impurity with a large Rf value was present according to the thin-layer chromatography (TLC). The reduction using sodium dithionite, required tetrahydrofuran (THF) as a co-solvent to improve the yield, which increased the cost. Although the iron sludge produced in the ferric acid reduction may not be conducive to environmental protection, as a secondary resource with a high iron content, it can be used as a raw material for making iron and for producing iron products. 19 Therefore, recycling the iron sludge and selling it to an iron sludge recycling company reduces environmental concerns. Thus, we employed iron and ammonium chloride to reduce 5-amino-2-chloroacetophenone. Besides, the 2-chloro-5-aminoacetophenone obtained in the reduction step was unstable, and a few impurities with high polarities were observed according to TLC after standing at room temperature for 24 hours. Therefore, 2-chloro-5-aminoacetophenone should be stored in a refrigerator and used in the next step as soon as possible.
Optimization of the reduction reaction. a

Reaction conditions:
Yields of crude product without further purification.
The synthesis of amides is a very common reaction in organic synthesis, and there are two main methods. The first uses condensing agents (1-Hydroxybenzotriazole (HOBt), 1-ethyl-3-(3-dimethylaminopropyl)carbodiimide hydrochloride (EDCI), 2-(7-azabenzotriazol-1-yl)-
Optimization of the condensation reaction. a

RME: reaction mass efficiency; DCM: dichloromethane; DCC:
Reaction conditions:
The yields of the crude product were obtained after workup involving washing with 10% hydrochloric acid and 10% sodium carbonate solution.
In the last step of the vismodegib synthesis, we adopted the method of pyridine synthesis in route G. In order to consume the substituted acetophenone completely, the amount of dihydropyrimidine bisulfate required was 1.1 equivalents with respect to the substituted acetophenone. The reaction temperature had a significant impact on the reaction rate and product purity. The reaction mixture appeared dark red when reacting at 110 °C, and red impurities with smaller Rf values were observed on TLC. At 80 °C, the red impurities decreased, but the reaction was not complete within 24 hours. The commonly used methods for the purification of vismodegib are recrystallization and column chromatography. At first, recrystallization was adopted, and multiple single or mixed solvents were investigated, but none of them worked well. Finally, impurities were adsorbed on silica gel and filtered out to afford the product with acceptable purity.
By application of this process to the laboratory courses offered to undergraduates in our school, the reproducibility of the experimental results was verified, and the experimental skills of the undergraduates were developed. New experimental teaching is needed to stimulate the interest of students in drug preparation. Thus, the whole exploration procedure for developing the synthetic method to vismodegib was employed for the college students’ innovative and entrepreneurial training program (an open 1-year experimental course) and five undergraduates participated. In this laboratory course, the students work started with literature retrieval and involved chemical synthesis, data analysis, reaction optimization, purity, and structure determination, as well as environmental influence assessment. For example, by synthesizing
In addition, in the scientific research ability training course (an open 1-week experimental course), three batches of validation experiments at gram level were conducted by six undergraduates, and the results are presented in Table 3. In this laboratory course, the experimental skills of the students, such as the ability to analyze experimental results and make observation notes, were particularly important and formed a key point of the course evaluation. All six undergraduates completed the corresponding operations of any step of the process within the specified time (4 h) and obtained qualified products. Thus, these validation experiments are not only applicable to an open experimental course but also to laboratory courses with fixed class hours, which can serve as a teaching tool involving drug synthesis to train experimental skills and develop the green chemistry consciousness of students.
Results of validation experiments.
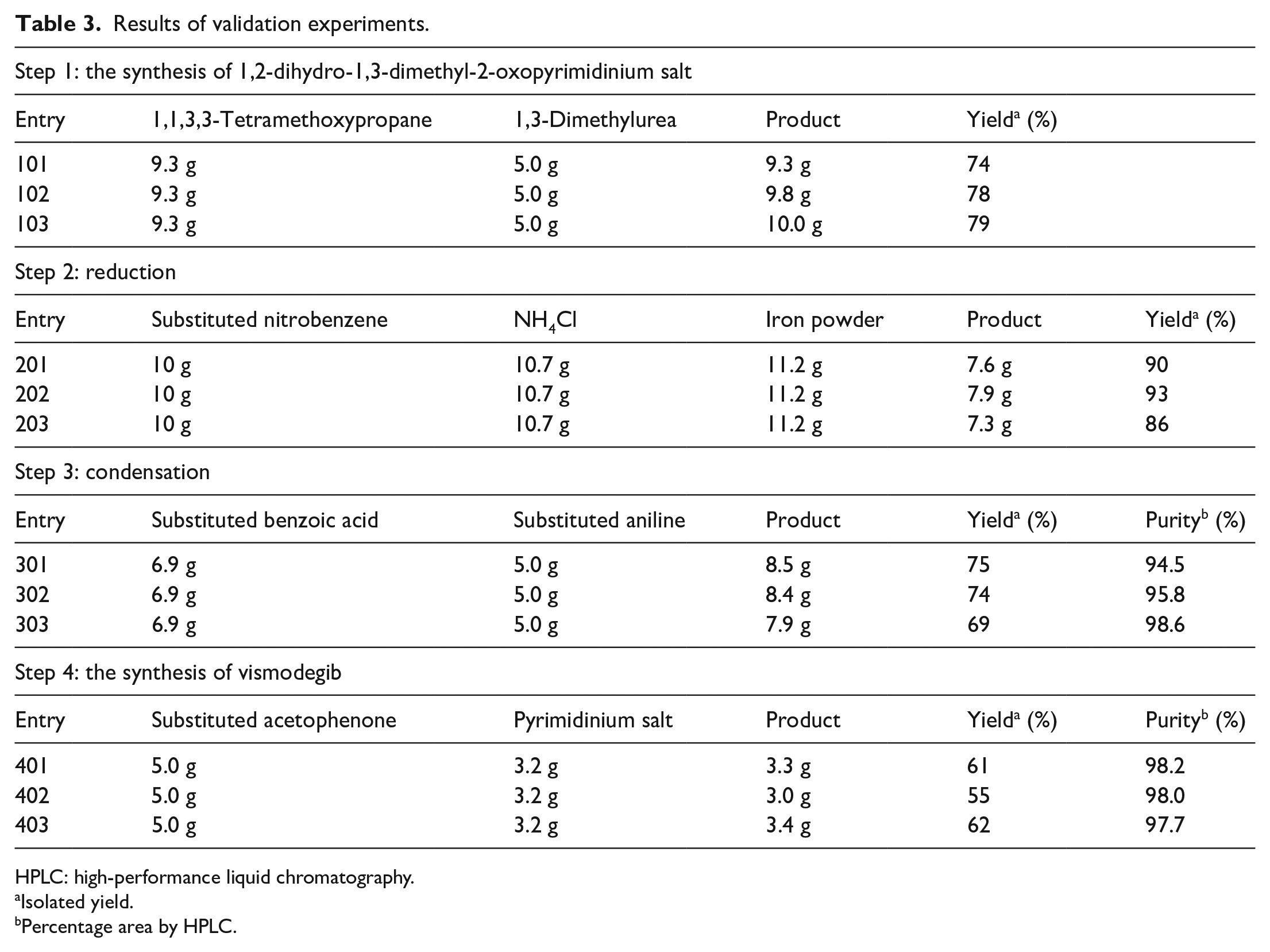
HPLC: high-performance liquid chromatography.
Isolated yield.
Percentage area by HPLC.
After the completion of the aforementioned courses, we invited students to evaluate them. All students indicated that the courses strengthened their understanding of drug synthesis and that they had gained more experimental skills. In addition, the course, as an exploratory experimental process, was more interesting and challenging, which inspired students to investigate related career paths. Three students planned to conduct medicinal chemistry or drug preparation research in the future.
Moreover, four teaching experiences were summed up, which might be useful in exploratory and continuous experimental courses.
The time for students to participate in open experimental courses is scattered because of the many other courses that students also need to participate in. Therefore, the employed experiment must adapt to dispersed time frames to ensure the smooth implementation of the course.
As an exploratory experiment, teachers must lead the whole experiment course and assist students in designing the experiment plan, correct experimental operations, resolve problems, and so on. Increasing discussions between undergraduates and teachers, as well as between undergraduates and postgraduates serving as assistants, is an effective method to improve learning outcomes.
The course assessment and teaching management suitable for specific experiments are important for improving course quality. Compared to “traditional, exactly repeating” experiments, exploratory experiments are more important in making students not settle for the results they obtain but to reflect on how they can do an investigation better the next time.
To ensure safety and cultivate safety awareness, for example, the wearing of goggles, gloves, and laboratory coats as mandatory. Students are required to review the material safety data sheet (MSDS) for materials and reagents prior to conducting experiments. All materials were handled using standard procedures. Chemical waste was disposed of in accordance with local regulations.
Conclusions
An alternative method for the preparation of vismodegib has been developed involving reduction, condensation, and cyclization reactions, in which 2-chloro-5-nitroacetophenone and 2-chloro-4-methylsulfonylbenzoic acid were employed as starting materials. The total yield of the route was about 40%, and the purity of the product was about 98%. The characteristics of the method are low-cost and widely available reagents, no precious metal catalyst or phosphine ligand is required, and reproducible results are obtained. In addition, the method was used in a laboratory course to build the capabilities of undergraduates and to develop a laboratory-scale drug synthesis method. Finally, the teaching practice experiences were summed up, which might be useful in exploratory and continuous experimental courses.
Experimental section
General
All the starting materials, reagents, and solvents are commercially available and were used without further purification. Melting points were determined with an X-4 apparatus and are uncorrected. The nuclear magnetic resonance (NMR) spectra were recorded on a Bruker Ascend 400 (Billerica, MA, USA) using tetramethylsilane (TMS) as an internal standard. Electrospray ionization mass spectrometry (ESI-MS) was performed using an Agilent 1100 Series MSD Trap SL (Santa Clara, CA, USA). The reactions were monitored by TLC (HG/T2354-92, GF254). The purities of vismodeigb and key intermediate
1,2-Dihydro-1,3-dimethyl-2-oxopyrimidinium hydrogen sulfate (7 )
1,3-Dimethylurea (5 g, 60 mmol) in 50 ml of absolute ethanol was added to 1,1,3,3-tetraethoxypropane (9.3 g, 60 mmol), and the resulting mixture was cooled in ice-water bath. Following dropwise addition of 95% sulfuric acid (5 mL) below 10 °C, the stirred mixture became light yellow and deposited precipitates. The mixture was then heated at 50 °C for 1.5 h. After cooling to room temperature, the crystals were filtered, washed with absolute ethanol, and dried to afford 1,2-dihydro-1,3-dimethyl-2-oxopyrimidinium hydrogen sulfate, as a pale white solid, to an amount of 9.3 g with a 74% yield (m.p. 201–203 °C [literature 12 value 204–206 °C]).
5-Amino-2-chloroacetophenone (2 )
A 250-mL three-necked flask with a magnetic stir bar was charged with Fe powder (11.2 g, 0.20 mol), NH4Cl (10.7 g, 0.20 mol), and H2O (100 mL). The mixture was stirred in an electric heating mantle (100 °C) and refluxed for 10 min. Next, 2-chloro-5-nitroacetophenone (10.0 g, 0.05 mol) was added, and the mixture was stirred for another 1.5 h. After cooling to room temperature, the mixture was diluted with 100 mL of EtOAc and filtered through a pad of diatomaceous earth, which was washed with EtOA and recovered as a secondary resource with high iron content. After separating the EtOA layer from the filtrate, the water layer was extracted with EtOA (50 mL × 2). The combined extracts were washed with water (100 mL), dried over anhydrous sodium sulfate, and filtered, and the solvent was evaporated under vacuum. 5-Amino-2-chloroacetophenone was obtained in 90% yield as light-yellow oil (yellow oil
16
) and was used directly in the next step without purification. A sample of purified
N-(3-Acetyl-4-chlorophenyl)-2-chloro-4-methylsulfonylbenzamide (4 )
To a solution of the 2-chloro-4-methylsulfonylbenzoic acid (6.9 g, 30 mol) in DCM (50 mL) was added 1 drop of
To a solution of the 2-chloro-4-methylsulfonylbenzoyl chloride in EtOA (50 mL) was added Et3N (8.2 mL, 0.06 mol) and 5-amino-2-chloroacetophenone (
Vismodegib
Supplemental Material
sj-pdf-1-chl-10.1177_17475198231214490 – Supplemental material for An alternative method for the preparation of vismodegib: A tool for an undergraduate laboratory course
Supplemental material, sj-pdf-1-chl-10.1177_17475198231214490 for An alternative method for the preparation of vismodegib: A tool for an undergraduate laboratory course by Xuefei Bao, Chuqiao Song, Dan Liu and Guoliang Chen in Journal of Chemical Research
Footnotes
Acknowledgements
The authors would like to thank Yuxiang An, Weihao Jiang, Honggao Duan, Yuxuan Zhao, Hui Zhang, and Yuning Wang who participated in the scientific research ability training course in our school and contributed their work to validate the reproducibility of the method.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was financially supported by the Liaoning Province Graduate Education Teaching Reform Project (research and practice on improving the quality of postgraduate theses in medicinal chemistry), Key Research Project of Department of Education of Liaoning Province (LJKZZ20220108), the foundation of Liaoning Provincial Department of Education (LJKQZ20222351), the college students’ innovative and entrepreneurial training program (No. S202210163053).
Supplemental material
Supplemental material for this article can be found via the “Supplementary Content” section of this article’s web page.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.