Abstract
Alkyl-substituted testosterone derivatives are promising platforms for new drug discovery in medicinal chemistry. This approach provides a simple and efficient method for introducing an alkyl substituent to steroids at the C-4 position. In this study, the novel compound 3α-hydroxy-3β-methyl-4,4-dimethyl-5α-21-bromo-19-nor-pregnan-20-one is synthesized from 19-nor-testosterone. The protocol involves methylation of the dienolate, reduction of the alkene, the Grignard reaction of the carbonyl group, a Wittig reaction, hydroboration with BH3, the oxidation and bromination with a 49% overall yield. For the methylation and reduction steps, the effects of the base, solvent, and reactant ratio on the conversion and yield are investigated. The structures of the synthesized compounds are determined by nuclear magnetic resonance (NMR) and high-resolution mass spectrometry (HRMS) (electrospray ionization (ESI)).
Keywords
Introduction
Steroids are natural products with cyclopentane polyhydrophenanthrene structures and are widely employed in the field of medicinal chemistry.1 –3 For example, important steroids such as cholesterol, progesterone, estradiol, and hydrocortisone (Figure 1) have various physiological activities, such as promoting bone growth, 4 anti-inflammatory, 5 and management of levels of water and salt. 6 In addition, with rapid developments in synthetic chemistry, more and more novel steroid compounds are developed, which play increasingly important roles in the prevention and treatment of cardiovascular diseases,7,8 hypoglycemia, 9 malaria, 10 human immunity, 11 and digestive disorders. 12

The chemical structures of several important steroids.
During intensive studies on steroid biological activities, scientists have found that introducing nitrogen atoms or functional groups onto the polyhydrophenanthrene framework can impact unique properties or improve physiological activities over the parent compounds. In particular, the structure of the A-ring plays a crucial role in the study of antirheumatic, paralytic, and antitumor drugs. Apart from ring aromatization and extension, many progesterone and 19-nor-progesterone drugs are also prepared by alkylating the A-ring with new groups and by subsequently introducing nitrogen-containing heterocyclic functional groups such as pyrazoline, thiazole, and oxazole with high physiological activity to the novel steroid C-17 branched chains.13,14 Further screening of their activity, selectivity, and toxicity will be beneficial for the discovery and invention of new antitumor, anti-inflammatory, and antibacterial drugs. For example, it has been found that C-3 alkylation on the steroid A-ring can improve the overall pharmacokinetics (PK) parameters and reduce potential toxicity by eliminating the possibility of partial oxidation of hydroxy groups to ketones, preventing further metabolism and reducing the possibility of secondary elimination pathways. 15 Steroid derivatives with C-4 dimethylation show significant antifungal activity against Fusarium oxysporum in soybean sudden death syndrome. 16 Our previous studies have indicated that 3α-hydroxy-3β-methyl-4,4-dimethyl-5α-21-bromo-19-nor-pregnan-20-one is a key intermediate for the synthesis of novel positive allosteric modulators (PAM) of synaptic and extrasynaptic GABAA.
Alkyl-substituted testosterone derivatives represent promising platforms for new drug discovery in medicinal chemistry. There have been some reports on modification at C-2 or C-3 of the A-ring (Scheme 1);17,18 however, alkylation of C-4 is complicated and reported rarely.

Alkylation of progesterone derivatives at the C-2 and C-3 positions.
It is well-known that alkylation of α,β-unsaturated ketones ortho to the carbonyl with alkylating agents followed by reduction of the alkene affords α-alkyl-substituted ketone derivatives.19,20 In a reverse process, Schmidt et al.
21
disclosed the C-4 monoalkylation of the steroid alkene

Reported method for the C-4 alkylation of steroids.
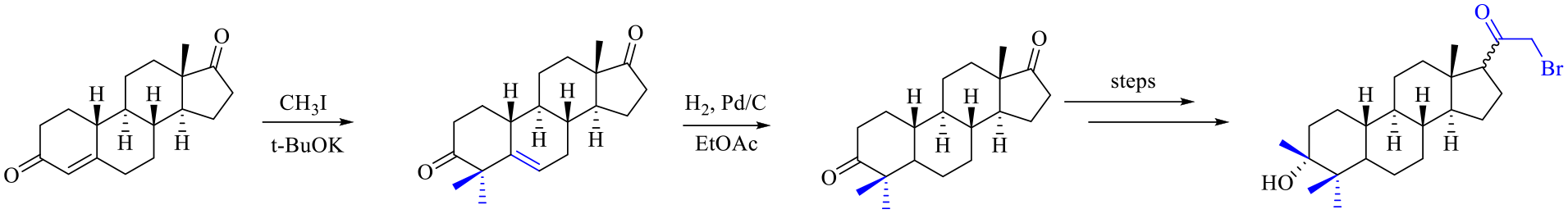
Herein, we disclose a new method for steroid bis-C-4 alkylation via an α,β-unsaturated ketone alkylation and an efficient reduction protocol. The obtained novel compound, 4,4-dimethyl-3α-hydroxy-3β-methyl-5α-21-bromo-19-nor-pregnan-20-one (
Results and discussion
Alkylation of a dienolate followed by reduction of the resulting alkene can be used for preparing α-alkyl-substituted ketone derivatives. As shown in Scheme 3, retrosynthetic analysis suggests that the novel compound

Retrosynthetic analysis of compound
Based on the retrosynthetic analysis of compound

The synthetic strategy toward compound
In this new synthetic route, compound
For the alkylation step, the side product of 4-monomethyl testosterone
Optimization of the alkylation conditions for the preparation of

Molar ratio.
HPLC peak area ratio: compound
Isolated yield based on
First, the standard alkylating agent (CH₃O)2SO2 was selected and different bases and solvents were screened (Table 1, entries 1–3). The results showed that when NaOCH3 was used as the base, no reaction occurred, even at 100 °C for 12 h. Using n-BuLi in diethyl ether solvent, the alkylating agent (CH₃O)2SO2 gave unsatisfactory reaction selectivity (68%) and poor a product yield (36%). Next, the active alkylating agent CH3I was investigated with different bases including K2CO3, NaOCH3, and t-BuOK, with t-BuOK being the best (Table 1, entries 4–12). The reaction selectivity and yield of
For the reduction step, several methods for the direct reduction of the alkene to an alkyl group are known in the literature. High-pressure hydrogenation with the Raney Ni and Pd/C is the most efficient method for alkene reduction. Thus, experiments were carried out to explore suitable reduction conditions for the preparation of compound
Optimization of reduction conditions for preparation of

w/w: catalyst dry weight/compound
HPLC peak area ratio: compound
Isolated yield based on compound
Reduction with 5% Raney-Ni in methanol was initially tested. The results demonstrated that a low H2 pressure gave a moderate yield of 61% (Table 2, entry 1), while a higher pressure gave a lower yield of 40% due to poor reaction selectivity (Table 2, entry 2). Reductions with 5% Pd/C as the catalyst were also evaluated. The effects of different solvents were studied first (Table 2, entries 3–5). Ethyl acetate was found to be the most suitable solvent giving a higher yield of 73% (Table 2, entry 5). By further optimizing the reaction pressure (entries 5–8), the yield of compound
Conclusion
In conclusion, we have developed an α,β-unsaturated ketone alkylation and an efficient reduction protocol for the synthesis of 4,4-dimethyl-3α-hydroxy-3β-5α-methyl-21-bromo-19-nor-pregnan-20-one without the need for hazardous alkyllithium reagents at extremely low reaction temperatures. The protocol involves methylation of a dienolate, alkene reduction, a Grignard reaction on a carbonyl group, a Wittig reaction, reduction with borane, and oxidation and bromination with a 49% overall yield. This work provides a new method for the synthesis of novel steroid derivatives modified at C-4 with the advantages of readily available reagents, relatively mild reaction conditions and moderate-to-good yields.
Experimental
All analytical reagents are commercially available (Macklin, J&K Scientific, and Aladdin) and were used directly without further purification. Reactions were monitored by thin-layer chromatography (TLC) on silica gel plates (GF-254), and the products were purified by silica gel column chromatography (200~300 mesh). Melting points were measured on a YRT-3 melting point apparatus using the capillary method without correction. 1H NMR and 13C NMR spectra were recorded on a Bruker Avance DPX-spectrometer at 300 or 400 MHz in CDCl3 or DMSO-d6 with tetramethylsilane (TMS) as an internal standard. Chemical shifts (δ) are reported in parts per million (ppm), coupling constants are expressed in Hz. High-resolution mass spectra (HRMS-ESI) were obtained using Agilent 1100 LC/MS Spectrometry Services.
4,4-dimethyl-5-ene-norandrostenedione (8 )
Potassium t-BuOK (164.9 g, 1.47 mol) was added in portions to a solution of 100 g (367 mmol) of 19-nor-testosterone (
The side product 4-methyl-4-ene-norandrostenedione (
4,4-Dimethyl-5α-19-norandrostanedione (7 )
5% Pd/C (5.0 g, dry weight, 0.05 w/w) was added to a solution of compound
3α-Hydroxy-4,4-dimethyl-5α-19-norandrostane-17-one (13 )
As the byproduct, the total reduction product
3α-Hydroxy-3β-methyl-4,4-dimethyl-5α-19-norandrostane-17-one (6 )
A solution of compound 7 (25.0 g, 82.7 mmol) in 500 mL of anhydrous toluene was added slowly to the MAD solution (620 mL, 248.1 mmol, 0.4 M in toluene) under a nitrogen atmosphere at −78 ℃. After the addition was complete, the mixture was stirred for 1 h at −78 ℃ and then MeMgBr (82.7 mL, 248.1 mmol, 3.0 M in THF) was added dropwise at a temperature below −75 ℃. The mixture was then stirred for a further 12 h at the same temperature after dropping finished. The reaction was quenched by adding 150 mL of saturated aqueous ammonium chloride solution. After the addition was complete, the mixture was stirred vigorously and then filtrated. Next, 300 mL of water was added to the filtrate, the organic phase was separated, and the aqueous phase was extracted with EtOAc (3 × 300 mL). The combined organic phase was and washed with water (3 × 200 mL) and brine (2 × 200 mL), dried over anhydrous Na2SO4 and concentrated. The residue was purified by silica gel column chromatography (petroleum ether/EtOAc = 5/1 v/v) to give compound
3α-Hydroxy-3β-methyl-4,4-dimethyl-5α-17-vinyl-19-norandrostane (11 )
A solution of 500 mL of anhydrous THF and ethyl triphenylphosphine bromide (97.6 g, 262.8 mmol) was added to 200 mL of toluene and potassium t-butanol (29.6 g, 262.9 mmol) dropwise under an N2 atmosphere at −10 to −5 ℃ with stirring for 20 min. After the addition was complete, the reaction mixture was stirred for 30 min at the same temperature. A solution of compound
3α-Hydroxy-3β-methyl-4,4-dimethyl-5α-19-nor-pregnan-20-ol (10 )
To a solution of 400 mL of anhydrous THF and compound
3α-Hydroxy-3β-methyl-4,4-dimethyl-5α-19-nor-pregnan-20-one (4 )
To a solution of 200 mL of dried dichloromethane and compound
3α-Hydroxy-3β-methyl-4,4-dimethyl-5α-21-bromo-19-nor-pregnan-20-one (3 )
To a solution of 100 mL of methanol, compound
Supplemental Material
sj-docx-1-chl-10.1177_17475198231204213 – Supplemental material for An α, β-unsaturated ketone alkylation and efficient reduction protocol for the synthesis of 3α-hydroxy-3β-methyl-4,4-dimethyl-5α-21-bromo-19-nor-pregnan-20-one
Supplemental material, sj-docx-1-chl-10.1177_17475198231204213 for An α, β-unsaturated ketone alkylation and efficient reduction protocol for the synthesis of 3α-hydroxy-3β-methyl-4,4-dimethyl-5α-21-bromo-19-nor-pregnan-20-one by Mingguang Zhang, Wenlong Wang, Chen Guo and Chunhuan Jiang in Journal of Chemical Research
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The authors are grateful to the Integration of Production and Research Project of Jiangsu Province (no. BY20221083).
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.


