Abstract
Large accumulations of copper (Cu) ions in the human body may cause damage, including organ and brain damage. In recent years, studies have proven that a large accumulation of Cu ions can lead to Parkinson’s disease and Alzheimer’s disease; therefore, it is great important to develop novel strategies for detecting trace Cu in environmental and biological samples. In this work, we designed two new coumarin-based colorimetric and fluorescent probes, HQ1 and HQ2. These two probes could selectively respond to Cu2+ with obvious color and fluorescence changes, and the presence of other metal ions had no effect on these changes. The two probes also exhibited high sensitivity for Cu2+, with a detection limit as low as 1.81 × 10–8 M/1.57 × 10–8 M. Notably, the two probes showed potential practical applications and were successfully used for detecting Cu2+ in a test strip, A549 cells, and living zebrafish larvae.

Introduction
Copper (Cu) is an essential trace element in the human body, and its biochemical function in the body is mainly as a catalyst. Cu is a component of Cu-containing enzymes and Cu-binding proteins, which can help maintain normal hematopoietic function and participate in iron metabolism and erythrogenesis. Cu plays an important role in maintaining the health of the central nervous system (CNS) and in the pathogenesis of some hereditary and incidental nervous disorders of the CNS, and also has certain effects on the immune system and hormone secretion.1–3
Because of mine wastewater and air pollution, Cu content in the living environment, drinking water, and crops is increasing. Cu continues to accumulate throughout the food chain, causing serious effects on biological systems and reproduction.4–10 Cu can also accumulate in the human body through the food chain from excessive eating leading to poisoning from Cu ions, adding to the burden on the body’s organs, especially the liver and gallbladder, where metabolic problems may arise, causing a series of health problems, such as cirrhosis of the liver, cancer, and other diseases. In recent years, studies have also shown that excessive accumulation of Cu ions can lead to Parkinson’s and Alzheimer’s disease.11–13 Therefore, it is of great importance to develop analytical methods for the rapid detection of Cu ions.
Fluorescent probes have attracted much attention because of their high sensitivity, good selectivity, fast response time, simple operation, and good biological imaging performance. In recent years, many small molecular fluorescent probes for detecting Cu ions have been developed.14–21 However, most of them have shortcomings such as complex synthesis, low selectivity, and slow response time. It is well known that the key element of probe design is the ability of an excellently chosen fluorophore to combine with the detection group for specific recognition. Coumarin dyes are widely used in fluorescent probes because of their low biotoxicity, high fluorescence sensitivity, good photostability, and easy modification of the main chain.22–32 Coumarin derivatives can also be used as fluorescence markers or imaging agents to identify tumor lesions and study the subcellular localization of drugs.33–44 The development of a sensor with high selectivity and sensitivity to copper ions remains an important challenge. In this paper, two novel fluorescent probes were synthesized using coumarin with an aldehyde (CHO group) as the fluorophore. Due to their stable hydrazone structure, they can have a good complexing effect on Cu ions, to realize the specific detection of Cu ions. Moreover, due to their low biological toxicity, they can detect Cu ions in cells and zebrafish, and have good biological imaging properties.
Results and discussion
Optical response to Cu2+
The optical behavior of probes HQ1 and HQ2 toward Cu2+ in DMSO: H2O (1:1, v/v) media was investigated by the absorption and emission method. As shown in Figure 1, the two probes have an absorption peak at 457 nm, and the absorption peak has a red shift after the addition of Cu2+. The absorption peaks of HQ1 and HQ2 shifted from 457 to 492 and 490 nm, respectively; addition of other metal ions (Ag+, Al3+, Ba2+, Cd2+, Cr3+, Fe3+, Fe2+, Hg2+, K+, Na+, Pb2+, Sr2+, and Zn2+) showed no effect on the absorption spectra of the probes at the same concentration and under similar conditions. As shown in Figure 2, both the probes emitted intense fluorescence at 525 nm, which was quenched by the addition of Cu2+; in contrast, addition of other metal ions (Ag+, Al3+, Ba2+, Cd2+, Cr3+, Fe3+, Fe2+, Hg2+, K+, Na+, Pb2+, Sr2+, and Zn2+) showed no effect on the absorption spectra of the probes at the same concentration and under similar conditions.

The absorption spectra of probes HQ1 and HQ2 upon addition of various metal ions in DMSO/H2O (1:1, v/v) solution (1.0 × 10−5 M).
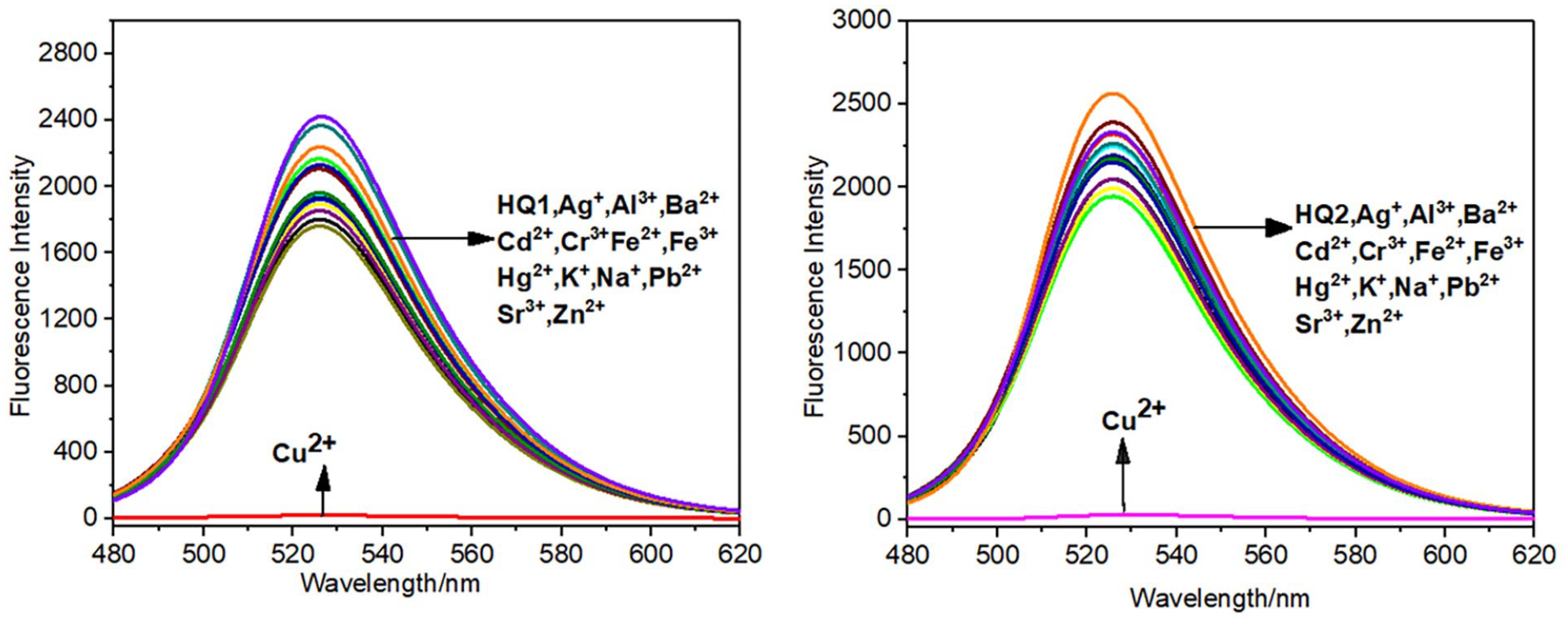
The fluorescence spectra of probes HQ1 and HQ2 upon addition of various metal ions in DMSO/H2O (1:1, v/v) solution (1.0 × 10−5 M). Excitation wavelength = 450 nm.
Also, the competition experiment was conducted to examine the binding ability of the probes toward other metal ions in DMSO/H2O solution (10−5 M 1:1, v/v). As shown in Figure 3, there were no obvious changes on the fluorescence intensity of the probes after adding various metal ions; however, the fluorescence was quenched rapidly after the addition of Cu2+, which proved again the high selectivity of the probes to Cu2+ ions.

Competitive tests for Cu2+ detection by probes HQ1 and HQ2. DMSO/H2O solution (10−5 M, 1:1, v/v). Excitation wavelength = 450 nm.
Quantitative analysis of probes HQ1 and HQ2 toward Cu2+ was studied in detail by a fluorescence titration experiment. During titration, the concentration of the control probe remained unchanged, and the concentration of Cu2+ was 0.5 μM and gradually increased. As depicted in Figure 4, with the addition of Cu2+, the fluorescence intensity gradually decreased until the fluorescence quenching was complete when 5 μM Cu2+ ions were added, and while the concentration of Cu ion continued to increase, the fluorescence in the quenching state did not change. Moreover, there was a good linear relation between fluorescence intensity and Cu2+ concentration. According to the formula DL = 3σ/k, the detection limit of HQ1/HQ2 for Cu2+ was calculated to be 18.1 nM/15.7 nM, which proved the high sensitivity of the synthetic probes and their potential application in the detection of Cu2+ ions.

Fluorescence titration spectra of probe HQ1 and HQ2 with increasing concentration of Cu2+ from 0 to 10 μM and the corresponding linear fitting. DMSO/H2O solution (10−5 M, 1:1, v/v). Excitation wavelength = 450 nm.
The response time of HQ1/HQ2 to Cu2+ in DMSO/H2O solution (10−5 M, 1:1, v/v) was investigated. After the addition of Cu2+, the fluorescence was completely quenched within 40 s/25 s and did not recover, indicating that the probes could respond quickly to Cu2+(Supplementary Figure S7).
pH effects
Next, we studied the detection effect of Cu2+ under different pH conditions. Fluorescence intensity of the probes in the presence and absence of Cu2+ is shown in Supplementary Figure S8. In the absence of Cu2+, the probe solution showed strong fluorescence intensity over a wide pH range (5–9), which indicated that the probes had high stability under acidic and alkaline conditions, while under strong acid and alkali conditions, the structure of the probes would be destroyed and it would be difficult to detect Cu ions. After adding Cu2+, the fluorescence intensity of the probe changed significantly, the fluorescence was quenched, and the fluorescence intensity was close to 0. It can be seen from the figure that the probes can detect Cu ions in the pH range of 5–9. Moreover, this detection result lays the foundation for the application of the probe in organisms.
“Naked-eye” detection
The visual detection of Cu ions by probe HQ1 was studied through a colorimetric experiment, and the results are shown in Figure 5. When different metal ions were added to the probe solution, it was observed that only the solution added with Cu2+ ions turned light brown in a short time, while the addition of other metal ions did not cause a color change, and the color was still as yellow as the probe solution, indicating that probe HQ1 can be used as a high-sensitivity sensor for naked eye recognition of Cu ions. Under the UV lamp at 365 nm, it was observed that the fluorescence of the solution added with Cu ions was completely quenched, while the solution added with other metal ions showed green fluorescence as for the probe solution, and the fluorescence color contrast was obvious. Since probe HQ2 showed the same color change as probe HQ1, its image is no longer displayed separately.

The selectivity of the metal ions was tested with a filter paper (a) in sunlight and (b) in UV light at 365 nm.
Detection mechanism
When the concentration of added Cu ions exceeds 0.5 equivalence, the fluorescence was completely quenched. Therefore, it is speculated that the probe responded to Cu ions in a ratio of 2:1, in DMSO/H2O (10−5 M, 1:1, v/v). The working curve of the Cu2+ probe HQ1 was further studied. The ratio of probe/[probe + Cu2+] (1/10, 2/10, 3/10, 4/10, 5/10, 6/10, 7/10, 8/10, and 9/10) was measured successively. The obtained data were analyzed and Job’s curve was drawn according to the results obtained as shown in Figure 6, which proves that the stoichiometric ratio of the reaction between probes and Cu2+ is 2:1.

Job’s plot of the probes with Cu2+ ([probe] + [Hg2+])=10 M, DMSO/H2O (1:1, v/v, 10−5 M) solution. Excitation wavelength = 450 nm.
There are unpaired single electrons and empty orbitals in the structure of the probe, so the probe can form a complex with coordination number 6 with Cu ions in a 2:1 ratio. Cu2+ coordinates with an O on the acyl hydrazone bond C = O, N on C = N bond, and O on C = O bond on the six-membered ring. The stable structure of a five-membered ring and a six-membered ring is formed between the complexes, as shown in Figure 7 and Supplementary Figure S9. After coordination with Cu2+, intermolecular charge transfer (ICT) between the probe and Cu2+ occurs. The paramagnetic Cu2+ quenches the fluorescence of the probe rapidly.

Complexation process diagram of probe HQ1 and Cu2+.
The structure and molecular orbital energy distribution of the synthetic probe and complex under B3LYP/6-31G (d) were calculated by the Gaussian 09 program and density functional theory. Supplementary Figure S10 shows the structure of the fluorescent probe and the [HQ1 + Cu2+] and [HQ2 + Cu2+] complex. Figure 8 shows the orbital energy distribution of the probe and complexes. Figure 8 shows that the energy gap band energies of [HQ1 + Cu2+] and [HQ2 + Cu2+] are lower than those of the original probe, and the structure is more stable, which is also in line with the phenomenon of red shift of the maximum absorption peak after adding Cu2+ to the UV–Vis absorption spectrum.

Corresponding orbital electron distribution of the probe and complex.
Paper test of the probe
Because the solution configuration still needs some cumbersome steps, the probe solution was made into a convenient filter paper strip to simplify the operational process. First, the filter paper was immersed in the fluorescent probe solution, and then dried naturally in air to obtain the Cu ion test paper. As shown in Figure 9, HQ1 test paper was observed to be yellow in sunlight. After dropping Cu ion solution onto the paper, the test paper turned brown, while dropping other metal ion solutions onto the paper did not cause a color change. Under the 365 nm UV lamp, it was observed that the test paper dripping Cu ions had no fluorescence, while the test paper dripping other metal ions showed green fluorescence as the HQ1 test paper. The experimental results show that the probe HQ1 can be made into a test paper for detecting Cu ions, which is more convenient for use. Since probe HQ2 shows the same color change as probe HQ1, its image is not displayed.

The selectivity of the metal ions was tested with a filter paper (a) in sunlight and (b) in UV light at 365 nm.
Analytical applications in living cells
To investigate the biological applications of probes HQ1 and HQ2, fluorescent imaging experiments were conducted in A549 cells in the presence of 10 μM probes and Cu2+ ions. First, the cytotoxicity of the probe to varied concentration of A549 cells (0, 5, 10, 15, and 20 μM) was evaluated by the 3-(4,5-dimethylthiazol-2-yi)-2,5-diphenyltetrazolium bromide (MTT) method. According to the results, the probes had low cytotoxicity to living cells. Even under high concentrations, the cell viability was close to 85% within 24 h (Supplementary Figure S11). Next, a concentration of 10 μM probe was used for the A549 cell imaging experiment. As shown in Figure 10, after 30 min of culturing the probe alone, the relevant region produced a green fluorescence. Then, Cu2+ was added to the probe solution and cultured for 10 min, and green fluorescence quenching was observed. These results prove that HQ1 and HQ2 are promising probes that can be used to detect Cu ions in living cells.

Fluorescence imaging of probes HQ1 and HQ2 in A549 cells.
Analytical applications in zebrafish
We further investigated the biological applications of these probes in zebrafish before and after addition of Cu2+. The 5-day zebrafish larvae were incubated in water containing 10 μM probe and Cu2+, then the fluorescence imaging was measured on a laser confocal microscope. As shown in Figure 11, the 5-day zebrafish larvae treated with water containing 10 μM of the probe for 30 min emitted a green fluorescence generated by the probe itself. After incubation with 10 μM Cu2+ for 10 min, the zebrafish larva exhibited fluorescence quenching. These results indicated that probes HQ1 and HQ2 could be used for monitoring low concentrations of Cu2++ in the living zebrafish model.

Fluorescence imaging of probes HQ1 and HQ2 in zebrafish.
Comparison with other probes
The designed probes were compared with other reported Cu2+ fluorescence probes, and the results are shown in Table 1. The distinctive merits of these probes include low detection limit, fast response time, wide pH range, living cells imaging, and zebrafish imaging.
Comparison with other probes.

Conclusion
In conclusion, two probes for detecting Cu ions were synthesized, which can react with Cu ions to form stable complexes. The two probes have good selectivity and can detect Cu ions in the presence of other metal ions; the response time is fast, with a reaction time to Cu ions of 1 min. They have a wide range of application. The probes can be made into test strips, which is convenient for practical use and the biological toxicity of the probes are low, so they have applications in cells and zebrafish. Therefore, the two probes explored in this paper can be considered as Cu ion sensors with prospects for wide-ranging applications in the future.
Experimental
Materials and measurements
All chemical reagents were obtained commercially and used without further purification. 1H NMR and 13C NMR spectra were recorded on a Bruker AV-400 spectrometer using TMS as internal standard. The high-resolution mass spectra (HRMS) were measured using a Q-TOF6510 mass spectrograph (Agilent, USA). The UV−Vis absorption spectra were analyzed using a UV-2600 PC spectrophotometer (Shimadzu, Japan). The fluorescence spectra were recorded on an F-7000 fluorescence spectrophotometer (Hitachi, Japan).
Synthesis of HQ1 and HQ2
The synthetic procedure for compound 1 has been described in previous literature.
50
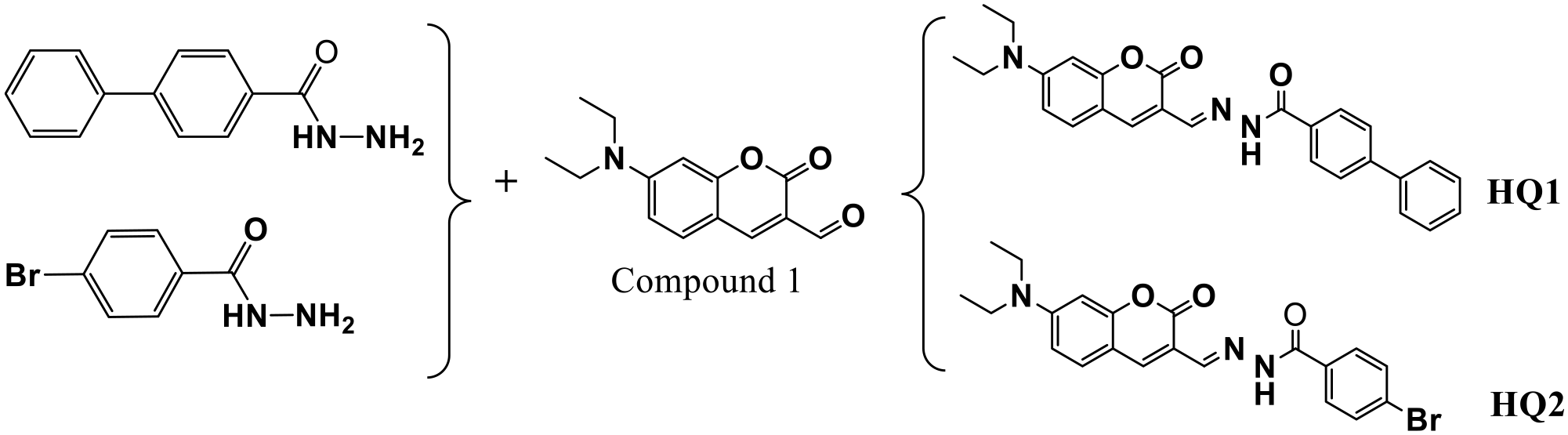
Synthesis step of probes HQ1 and HQ2.
Optical response measurements
HQ1 and HQ2 were dissolved in DMSO and the volume was made up to 100 ml to give the probe stock solution (1.0 × 10−3 M). The stock solutions of the metal ions with a concentration of 1.0 × 10−3 M were prepared by dissolving 1.0 mmol of their corresponding nitrate salts (AgNO3, AlNO3, Ba(NO3)2, Cd(NO3)2·4H2O, Cu(NO3)2·3H2O, FeSO4·7H2O, Fe(NO3)3·9H2O, Hg(NO3)2·H2O, KNO3, NaNO3, Pb(NO3)2, Sr(NO3)2, and Zn(NO3)2·6H2O) in water and the volume was made up to 100 mL. The probe stock solution and the metal ion solution were diluted to 10−5, and the corresponding absorption and fluorescence spectra were measured.
Cell culture and fluorescence imaging
A549 cells were grown in Dulbecco’s Modified Eagle Medium (DMEM) containing 10% fetal bovine serum (FBS) and 1% penicillin-streptomycin. After incubating at 37 °C in a humidified atmosphere with 5% CO2 for 24 h, cells were washed with phosphate buffered saline (PBS buffer, pH = 7.2) and incubated with fresh medium containing 10 μM probe HQ1 and HQ2 for 30 min. Then the cells were treated with different concentrations of Cu2+ (0, 10, 20, and 50 μM) for 10 min and the fluorescence imaging was measured on a laser confocal microscope (Olympus, Japan).
Zebrafish maintenance and fluorescence imaging
The wild type zebrafish eggs were grown in 6-well plates with water. After incubating in a light incubator at 28 ± 0.5 °C for 5 days, the zebrafish larvae were treated with new medium containing 10 μM probes HQ1 and HQ2 for 30 min. Then, 10 μM of Cu2+ was added to the medium, and the fluorescence imaging was measured on a laser confocal microscope after 10 min incubation.
Supplemental Material
sj-docx-1-chl-10.1177_17475198231199438 – Supplemental material for Two novel coumarin-based fluorescent probes for the detection of Cu2+ and biological applications
Supplemental material, sj-docx-1-chl-10.1177_17475198231199438 for Two novel coumarin-based fluorescent probes for the detection of Cu2+ and biological applications by Canxin Zhang, Li Huang, Lizhen Wang, Xia Meng, Jianhua Zhou and Hongdong Duan in Journal of Chemical Research
Footnotes
Author contributions
Availability of data and materials
The datasets used or analyzed during the current study are available from the corresponding author on reasonable request.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Jinan Talent Project for Universities (No. 2019GXRC044) and the Shandong High-end Foreign Experts Recruitment Program (No. WST2019006).
Ethical approval
Animal procedures were conducted in compliance with the NIH Guide for the Care and Use of Laboratory Animals (No. 8023, amended in 1996), and approved by the Animal Care and Use Committee of Qilu University of Technology, which also followed the guideline for the Care and Use of Laboratory Animals in China.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
